- 1School of BioSciences, University of Melbourne, Parkville, VIC, Australia
- 2Biosciences, College of Life and Environmental Sciences, University of Exeter, Exeter, UK
In higher plants the L-galactose pathway represents the major route for ascorbate biosynthesis. The first committed step of this pathway is catalyzed by the enzyme GDP-L-galactose phosphorylase and is encoded by two paralogs in Arabidopsis – VITAMIN C2 (VTC2) and VTC5. The first mutant of this enzyme, vtc2-1, isolated via an EMS mutagenesis screen, has approximately 20–30% of wildtype ascorbate levels and has been reported to have decreased growth under standard laboratory conditions. Here, we show that a T-DNA insertion into the VTC2 causes a similar reduction in ascorbate levels, but does not greatly affect plant growth. Subsequent segregation analysis revealed the growth defects of vtc2-1 mutants segregate independently of the vtc2-1 mutation. These observations suggest that it is the presence of an independent cryptic mutation that affects growth of vtc2-1 mutants, and not the 70–80% decrease in ascorbate levels that has been assumed in past studies.
Introduction
Ascorbate (vitamin C) is an important multifunctional antioxidant compound involved in stress tolerance (Conklin et al., 1996; Pastori et al., 2003; Sanmartin et al., 2003; Barth et al., 2004; Filkowski et al., 2004; Müller-Moulé et al., 2004; Chen and Gallie, 2005; Larkindale et al., 2005; Pavet et al., 2005) and redox signaling (Noctor and Foyer, 2011; Page et al., 2012). While several possible biosynthetic pathways for ascorbate have been proposed, it was demonstrated that the L-galactose pathway is the dominant route in Arabidopsis (Dowdle et al., 2007). In this pathway, GDP-D-mannose, formed from D-mannose 1-phosphate, is sequentially converted to GDP-L-galactose, L-galactose 1-phosphate, L-galactose, L-galactono-1,4-lactone, and finally to L-ascorbate. The first committed step of this pathway is catalyzed by GDP-L-galactose phosphorylase, which is encoded by the paralogous genes VITAMIN C2 (VTC2) and VTC5 and is most likely an important control point in the pathway (Yoshimura et al., 2014).
The first ascorbate-deficient mutants of Arabidopsis, vtc1-1 and vtc2-1, were identified via an ethyl-methanesulfonate (EMS) mutagenesis screen for ozone sensitive mutants (Conklin et al., 1996). Later, using a high-throughput nitroblue tetrazolium assay several more EMS-induced ascorbate-deficient mutants (vtc1-2, vtc2-2, vtc2-3, vtc3-1, vtc4-1) were identified (Conklin et al., 2000). Of these mutants, vtc1-1 and vtc2-1 have been extensively studied owing to their low levels of ascorbate (Veljovic-Jovanovic et al., 2001; Jander et al., 2002; Pastori et al., 2003; Müller-Moulé et al., 2004; Pavet et al., 2005; Dowdle et al., 2007; Mukherjee et al., 2010; Kempinski et al., 2011; Kerchev et al., 2011; Lee et al., 2011; Luna et al., 2011; Talla et al., 2011; Zechmann, 2011; Botanga et al., 2012; Brosché and Kangasjärvi, 2012; Page et al., 2012; Zhang et al., 2012; Wang et al., 2013). The vtc1-1 mutant has a Pro22Ser substitution in the active site of GDP-mannose pyrophosphorylase (Conklin et al., 1999) resulting in a 70% decrease in ascorbate levels (Pavet et al., 2005; Mukherjee et al., 2010; Kerchev et al., 2011; Zechmann, 2011; Zhang et al., 2012). vtc2-1 has a single base substitution (G to A) at the predicted 3′ splice site of the fifth intron, resulting in an 80–90% reduction in transcript levels and less than 2% of GDP-L-galactose phosphorylase activity in leaves (Dowdle et al., 2007). As a consequence, this mutant has 70–80% reduction in ascorbate levels (Müller-Moulé et al., 2004; Pavet et al., 2005; Dowdle et al., 2007; Kerchev et al., 2011). Both mutant lines display reduced growth suggesting that ascorbate plays an important role in the control of plant growth (Veljovic-Jovanovic et al., 2001; Pastori et al., 2003; Müller-Moulé et al., 2004; Pavet et al., 2005; Kerchev et al., 2011). The other vtc2 mutants, vtc2-2 and vtc2-3, have mis-sense mutations leading to Gly223Asp and Ser290Phe substitutions, respectively (Jander et al., 2002; Dowdle et al., 2007) and while the amount of ascorbate in these lines varies according to growth conditions, values typically range from 30–80% and 40–50% of wildtype levels (Conklin et al., 2000; Colville and Smirnoff, 2008). Interestingly, neither vtc2-2 nor vtc2-3 display noticeable growth defects (Conklin et al., 2000).
Because vtc2-1 has detectable levels of full-length VTC2 transcript (Dowdle et al., 2007) and is thus not a complete loss-of-function mutant, we obtained a T-DNA insertion mutant from the Arabidopsis stock center. We demonstrate that this mutant, here named vtc2-4, lacks wildtype VTC2 transcripts and, although it has a level of ascorbate similar to vtc2-1, exhibits a near wildtype growth phenotype under short-day and continuous light conditions. We suggest that the decreased growth phenotype of vtc2-1 is likely due to an independent cryptic mutation and is not due to ascorbate deficiency as widely assumed in the literature.
Materials and Methods
Plant Materials and Growth Conditions
Mutant alleles were all in the Col-0 background and were sourced from the ABRC stock center. For growth assays under short days, seeds were sown on soil and placed in a growth chamber and exposed to an 8 h light (150–250 μmol photons m-2 s-1)/16 h dark cycle at 21°C for 6 weeks. Plant weight was recorded and leaf surface area measurements taken by imagining freshly collected leaves and using ImageJ software to determine the surface area of leaf silhouettes. For growth under continuous light conditions, surface-sterilized seeds were germinated on agar media containing mineral salts (MM) with 2% sucrose (MMS) and 0.8% Bacto agar as described by Scholl et al. (1998). Seedlings were grown at 20°C in a climate chamber with a 16 h light (150–250 μmol photons m-2 s-1)/8 h dark cycle for 7 days before transfer to soil in a climate chamber with 24 h continuous light (150–250 μmol photons m-2 s-1).
For comparison of leaf area and chlorophyll fluorescence under short and long day conditions, plants were grown in pots (5 cm square and 5 cm deep) containing four parts Levington F2 compost (Scotts, Maryville, OH, USA) plus one part vermiculite. They were grown in short days (8 h) for 22 days after sowing in a controlled environment room at 23°C and 65% relative humidity with a light intensity of 200 μmol photons m-2 s-1. After 22 days, half the plants were transferred to long days (14 h) under otherwise identical conditions and the leaf area and chlorophyll fluorescence measured over 10 days. The plants were imaged with a CF Imager (Technologica Ltd., Colchester, UK). They were dark adapted for 30 min before measuring basal fluorescence (F0). This was followed by measuring maximum dark adapted fluorescence (Fm) resulting from a saturating light flash (6, 349 μmol photons m-2 s-1 for 0.8 s). Dark-adapted quantum efficiency of photosystem II was calculated as Fv/Fm (Fv = Fm-Fo) (Baker, 2008). Leaf area of the imaged rosettes was extracted from the chlorophyll fluorescence images and the instrument was calibrated with leaf discs of known area.
Measurement of Leaf Ascorbate Content in Seedlings
Two-week old seedlings were harvested for ascorbate assay. Three seedlings from each control line (Col-0, single mutant lines) and 15–20 vtc2-4;vtc5 seedlings were pooled for each biological replicate. Total ascorbate (ascorbate and dehydroascorbate) was measured by the iron (III) reduction assay (Kampfenkel et al., 1995) in an 8x scaled-down protocol.
RNA Isolation and RT-PCR Analysis
Total RNA from 3-week old shoot tissues was isolated using the Qiagen RNeasy Plant Mini Kit1 according to the manufacturer’s protocol. The extracted RNA was further treated to remove any contaminating DNA using the DNA-freeTM kit2 according to the manufacturer’s protocol. First strand cDNA was synthesized using the SuperScript® III First Strand Synthesis system2 according to the manufacturer’s protocol. The full length VTC2 coding DNA sequence was amplified with the following primers: forward (5′-CAAAAGAGTTCCGACCGTTG-3′) and reverse (5′-ACTGAAGGACAAGGCACTCG-3′). The transcript of the constitutively expressed ACTIN2 (ACT2) was used as an internal control with the following primers: forward (5′-GGTAACATTGTGCTCAGTGGTGG-3′) and reverse (5′-CTCGGCCTTGCAGATCCACATC-3′). The PCR was carried out using Promega GoTaq® Green Master Mix3 with the following conditions: initial denaturation 5 min at 94°C was followed by 30 cycles of 30 s denaturation step at 94°C, 30 s annealing step at 60°C and 60 s extension step at 72°C. The final extension step was for 5 min at 72°C.
Genotyping the vtc Mutants
Plants were genotyped by PCR using DNA extracted with the preparation method described in Edwards et al. (1991). The vtc2-1, vtc5-1, and vtc5-2 mutants were genotyped as described by Dowdle et al. (2007). The SAIL line of vtc2-4 was genotyped by performing PCR using a triplet of primers; two primers complementary to genomic DNA sequences situated on either side of the insertion site and a third primer complementary to the left border of the vector pDAP101. Primer sequences were as follows: forward (5′-TGATAATGGTTTCTGTAGCTTGGA-3′), reverse (5′-AAAACCAAGCTCTCTGCACAA-3′) and LB1 (5′-GCCTTTTCAGAAATGGATAAATAGCCTTGCTTCC - 3′). Accession Numbers vtc2-4 and vtc2-1W (line#4) have been deposited with ABRC and given the stock numbers CS69540 and CS69541, respectively.
Results
Genetic Characterization of a vtc2 T-DNA Insertion Mutant
A line predicted to have a T-DNA insertion in VTC2 (SAIL_769_H05/CS876707) was obtained from the Arabidopsis stock center (ABRC). PCR genotyping and sequencing of genomic DNA adjacent to the T-DNA left border confirmed the presence of the T-DNA within the fourth exon of VTC2 at position +620 downstream of the start codon (+1). Lines homozygous for this insertion mutation, hereafter called vtc2-4, were backcrossed to wildtype (Col-0) and the resulting F2 progeny PCR genotyped for presence of the T-DNA allele. Numbers of VTC2/VTC2:vtc2-4/VTC2:vtc2-4/vtc2-4 plants were consistent with an expected 1:2:1 segregation ratio (48:112:56; χ2 = 0.89; p > 0.6). Subsequent RT-PCR analysis failed to amplify a full-length VTC2 transcript from lines homozygous for vtc2-4 confirming presence of the T-DNA within this gene (Supplementary Figure S1). Measurement of total ascorbate in leaves of 4-week-old wildtype, vtc2-1 and vtc2-4 plants revealed that both vtc2-4 and vtc2-1 had approximately 30% the levels of ascorbate that are found in wildtype, showing that the T-DNA present in the vtc2-4 line conditions a similar decrease in ascorbate (Figure 1A).
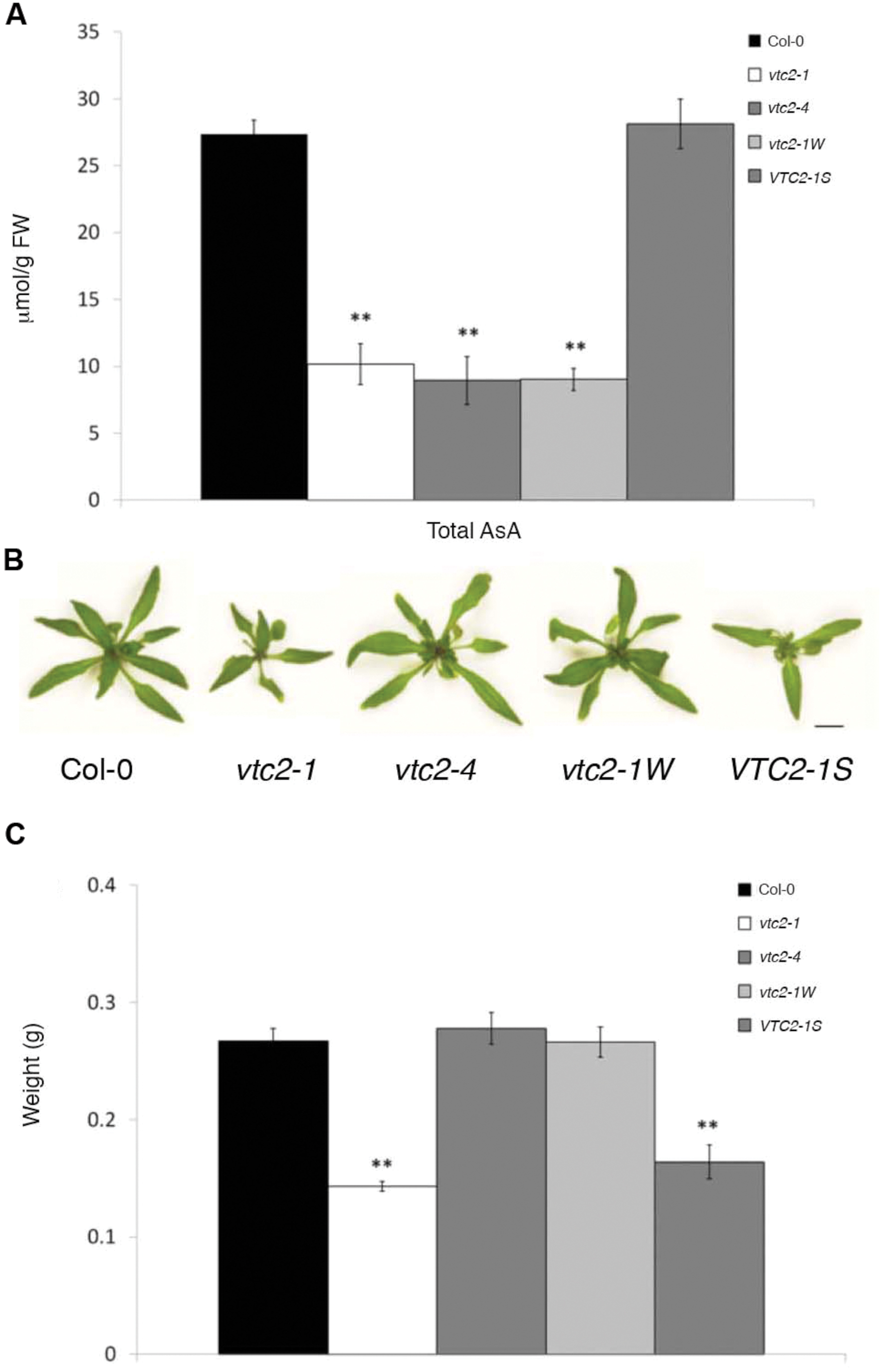
FIGURE 1. (A–C) Leaf ascorbate concentration and fresh weights of Arabidopsis thaliana vtc2 mutants. (A) Total ascorbate (ascorbate and dehydroascorbate) concentration in leaves of 4-week-old plants. Error bars indicate SE (n = 3). Significant differences from Col-0 wildtype were determined with Student’s t-test (∗∗p < 0.01). Significance values were adjusted for multiple comparisons using the Bonferroni correction. (B) Three-week-old Col-0 wildtype, vtc2-1, vtc2-4, vtc2-1W, VTC2-1S grown under continuous light. Scale bar = 10 mm. (C) Shoot fresh weight measurements of 3-week-old plants grown under continuous light conditions. Error bars indicate SE (n = 6). Significant differences from Col-0 wildtype were determined with Student’s t-test (∗∗p < 0.01). Significance values were adjusted for multiple comparisons using the Bonferroni correction.
Previous studies have shown that the EMS-induced mutant vtc2-1 has a growth defect (Müller-Moulé et al., 2004; Pavet et al., 2005; Kerchev et al., 2011). However, when vtc2-4 mutants were propagated on soil under continuous light, they had a wildtype appearance (Figure 1B) and fresh weight measurements differed little from wildtype (Figure 1C). In contrast, vtc2-1 mutants were significantly smaller than vtc2-4 mutants, with approximately half the fresh weight of wildtype. To ensure that the observed growth difference between vtc2-1 and vtc2-4 mutants are a general feature of these mutants and not just induced by long-days, leaf surface area measurements were assessed over a 10-day growth period under both short and long-days conditions. Regardless of day length, vtc2-4 mutants had a marginally smaller leaf surface area when compared to wildtype, whereas vtc2-1 mutants were considerably smaller than wildtype and vtc2-4 mutants (Figures 2A,B). A multi-factor ANOVA test confirmed that differences in leaf size between vtc mutant lines and wildtype under either growth condition was significant (p < 0.0001; Table 1). To determine whether the observed difference in leaf surface area arises from altered rates of growth, relative leaf expansion rate of rosettes under long and short day conditions were calculated. As expected, the growth rate was largely influenced by day length as each line displayed significantly higher growth under long days (Figure 2C). In contrast, there was no significant difference in the growth rate between wildtype and the vtc2 mutants under long days. While this was also true for wildtype and vtc2-4 mutants under short days, vtc2-1 mutants had a significantly reduced growth rate when compared to wildtype (Figure 2C). Finding a smaller rosette leaf area for both vtc2-1 and vtc2-4 in long days (Figure 2A), but similar relative expansion rate to wildtype (Figure 2C), implies that the small size results from initially smaller seedlings rather than from differences in intrinsic leaf expansion rate. However, under short-day conditions, vtc2-1 relative leaf expansion rate is compromised as well. Measuring the surface area of individual leaves collected from 6-week old short-day grown plants revealed that the size differences between wildtype and vtc2-1 mutants is mostly confined to the fifth and subsequent leaves (Figures 2D,E). Taken together these data show that the growth defects of vtc2-4 mutants are not of the same magnitude as those seen in vtc2-1 and hence may not be directly attributable to ascorbate deficiency.
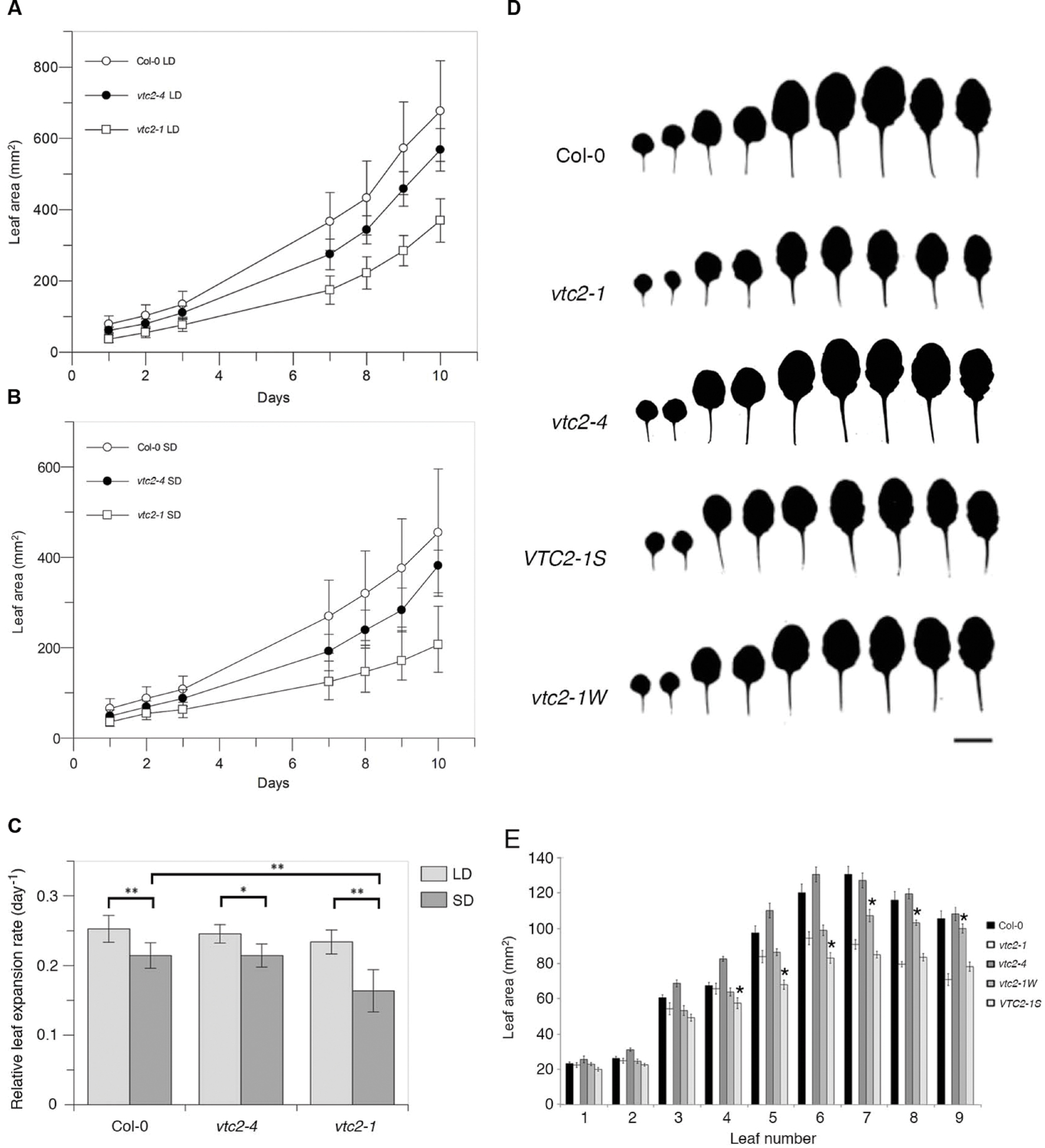
FIGURE 2. (A–C) Rosette and leaf area measurements of Arabidopsis thaliana vtc2 mutants. (A) Surface area measurements (mm2) of leaves taken over a 10-day period under long-day (14 h) conditions. Values are mean and error bars indicate SD (n = 12). (B) Surface area measurements (mm2) of leaves taken over a 10-day period under short-day (8 h) conditions. Values are mean and error bars indicate SD (n = 12). (C) Relative leaf expansion rate (RLER, day-1) was calculated as the slope of a linear fit to a graph of natural log leaf area (mm) versus time (days). RLER was calculated from data in Figures 2A,B. Values are mean and error bars indicate SD (n = 10–12). Significant differences were determined with a one-way ANOVA and Tukey HSD post hoc test (∗p = 0.005, ∗∗p < 0.001). (D) Silhouettes of leaves from 6-week-old plants grown under short-day conditions. Scale bar = 1 cm. (E) Surface area measurements (mm2) of leaves taken from 6-week-old plants grown under short-day conditions. Values are mean and error bars indicate SD (n = 7–10). Differences between leaf areas of vtc2-1, vtc2-1W and VTC2-1S were assessed using a Student’s t-test (∗p < 0.05).

TABLE 1. A comparison of the rosette leaf areas of wildtype (Col-0) and vtc2 mutants of Arabidopsis thaliana using multivariate ANOVA.
The Small Size Phenotype of vtc2-1 Is Not Linked to Ascorbate Deficiency
Since vtc2-1 and vtc2-4 have contrasting growth phenotypes, we undertook a genetic characterization of vtc2-1. From a cross between wildtype and vtc2-1 F1 individuals, heterozygous for vtc2-1, exhibited wildtype growth in soil under continuous light condition (data not shown). The presence of the vtc2-1 allele can be detected by CAPS (cleaved amplified polymorphic sequence) PCR (Dowdle et al., 2007). In a segregating F2 population grown in soil, genotyping confirmed that the vtc2-1 allele segregated in a Mendelian ratio (Table 2; χ2 = 2.23; p > 0.3). Four-week-old individual F2 plants were also visually scored for size as “Wildtype” or “Small.” While wildtype and vtc2-1 mutants grown in parallel showed a clear difference in size as previously reported (Veljovic-Jovanovic et al., 2001; Müller-Moulé et al., 2004; Pavet et al., 2005; Kerchev et al., 2011), some individual wildtype plants exhibited a similar growth size to vtc2-1 mutant plants. However, an excess of small individuals over the expected number suggested it was likely that some wildtype individuals were misclassified as “Small.” A number of individuals with a small growth phenotype were homozygous for the wildtype VTC2 allele and, conversely, some vtc2-1 homozygous individuals had a wildtype appearance (Table 2).

TABLE 2. Phenotypic scoring of 4-week-old F2 individuals from a cross between wildtype (Col-0) and vtc2-1 Arabidopsis thaliana mutants.
To score the growth phenotype more accurately a number of F2 lines derived from the Col-0/vtc2-1 cross were allowed to self-fertilize to produce individual F3 families, which were then observed as populations after 4-week-growth in soil under continuous light conditions (Table 3). Of seven F2 individuals wildtype at the VTC2 locus with an apparent small growth phenotype, five F3 families were uniformly small in size (called VTC2-1S; Figure 1B). Conversely, of 24 F2 individuals homozygous for vtc2-1 and apparently wildtype in size, 18 F3 families were uniformly wildtype in size (called vtc2-1W; Figure 1B). Measuring fresh weight and total ascorbate levels under continuous light conditions revealed that vtc2-1W plants had a similar weight to wildtype, but with ascorbate levels decreased by approximately 70% compared to wildtype. Likewise, fresh weight of a representative VTC2-1S line was similar to vtc2-1, but with ascorbate approaching wildtype levels (Figure 1C).
To better characterize the growth characteristics of these F3 lines, fresh weight and leaf surface area measurements of representative vtc2-1W and VTC2-1S plants grown under short-day were made (Figures 2D,E, Supplementary Figure S2). In agreement with the measurements made under continuous light conditions, the fresh weight of the vtc2-1W line was statistically different from vtc2-1 mutant controls (Supplementary Figure S2). When compared to vtc2-1 mutants, late arising leaves of vtc2-1W plants were larger (leaf 7 to leaf 9; increase ranged from 18 to 41%). In contrast, the fresh weight of a representative VTC2-1S line was not significantly different from vtc2-1 mutants (Supplementary Figure S2). This is despite finding that a few VTC2-1S leaves were slightly smaller than those of vtc2-1 (leaf 5–6; decrease ranged from 13 to 23%). In summary, these analyses demonstrate that ascorbate-deficiency is genetically separable from growth defects associated with the vtc2-1 mutant line.
Leaf Senescence in vtc2 Mutants
Given that ascorbate deficiency in vtc2-4 mutants is not associated with a strong growth defect, we next considered whether this line displays other ascorbate-deficient characte ristics. Previous studies have shown that reduced ascorbate is associated with faster leaf senescence (Barth et al., 2004). Senescence can be measured as a loss of leaf photosynthetic efficiency. Thus to assess the extent of leaf senescence in vtc2 mutants, we imaged dark-adapted quantum efficiency of photosystem II (Fv/Fm). Examining the Fv/Fm images of wildtype, vtc2-4 and vtc2-1 grown under both short and long days suggested bigger decreases in older leaves of vtc2 mutants compared to wildtype (Figure 3A). To quantify the different rates of senescence in each line, the % of leaf area below a fixed Fv/Fm value (0.75) over days 7–10 was calculated. This revealed a slight elevation in the rate of senescence under short days compared to long days for all lines tested which was only significant for vtc2-4 (p < 0.05) (Figure 3B). However, under both growth conditions, vtc2-1 mutants displayed a noticeable and statistically significant (p < 0.001) increase in the proportion of Fv/Fm values below 0.75 when compared to wildtype, whereas vtc2-4 mutants only exhibit a marginal and statistically non-significant increase (Figure 3B). These observations are consistent with vtc2-1 mutants displaying increased leaf senescence, which is not apparent in vtc2-4 under these growth conditions.
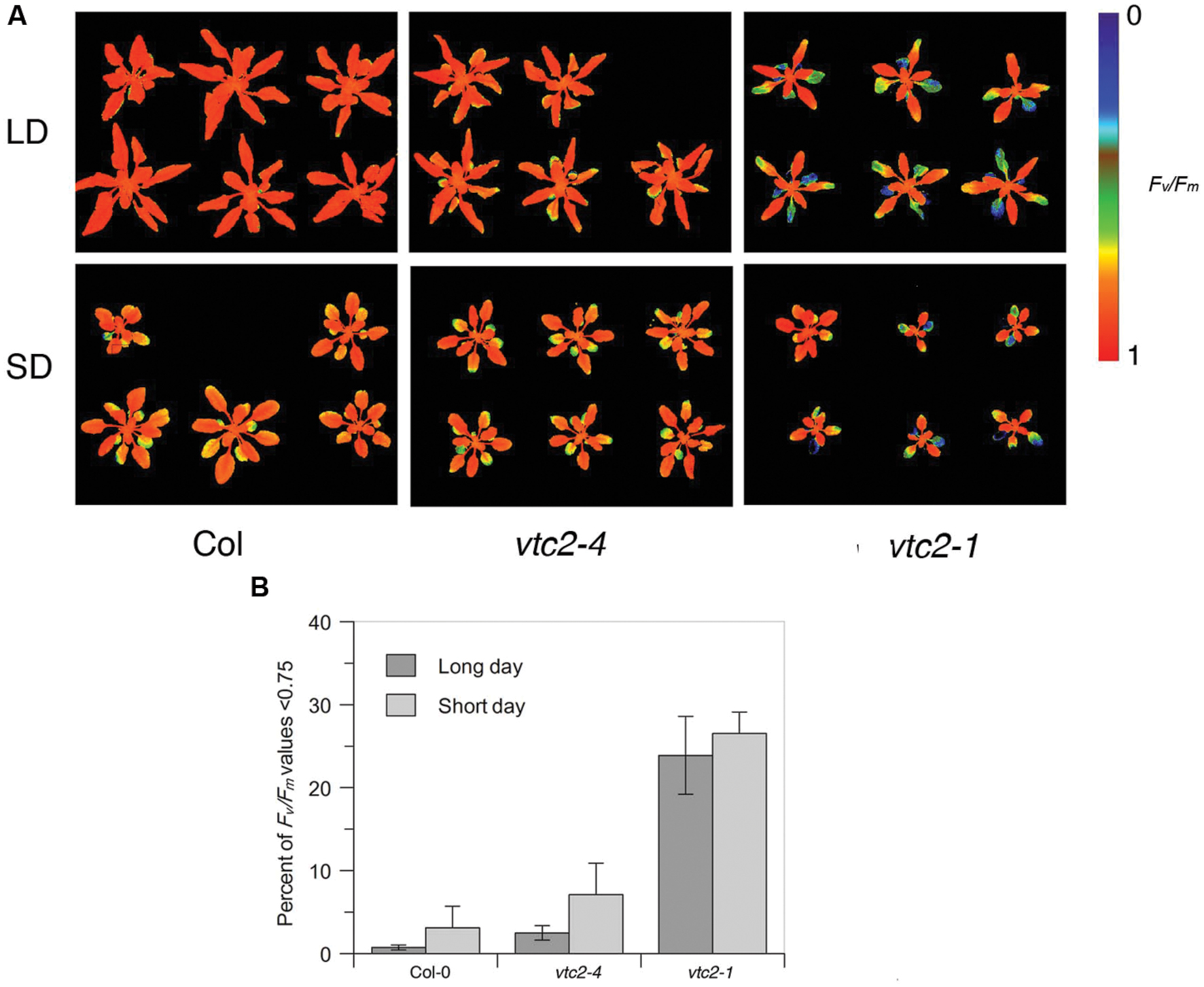
FIGURE 3. (A,B) Fv/Fm measurements of Arabidopsis thaliana vtc2 mutants. (A) Chlorophyll fluorescence images of short-day (SD) and long-day (LD) grown plants colored according to their Fv/Fm ratio on day 10 (see Figures 2A,B). (B) Percentage of leaf surface area with Fv/Fm values below 0.75 in short-day and long-day grown plants. Data are pooled from days 7–10 (see Figures 2A,B). Error bars indicate SD (n = 6–12). ANOVA shows significant differences (p < 0.001) between Col-0 and the vtc2-1 mutant, whereas daylength only has a significant effect on vtc2-4 mutant plants (p < 0.05).
Arabidopsis Lacking Functional GDP-L-galactose Phosphorylase has Undetectable Ascorbate and Exhibits a Seedling-Lethal Phenotype
The importance of the L-ascorbate biosynthetic pathway for Arabidopsis growth and development was demonstrated by Dowdle et al. (2007) when they showed that vtc2-1;vtc5 double mutants displayed a growth arrest following germination; a phenotype that was also associated with cotyledon bleaching. Given that vtc2-1 is not a complete loss-of-function allele and also apparently harbors a cryptic mutation/s that affect growth, we reconstituted the vtc2;vtc5 double mutant line by combining the vtc2-4 mutant with two different vtc5 mutant alleles (vtc5-1 and vtc5-2; Dowdle et al., 2007). The resulting vtc2-4;vtc5-1 and vtc2-4;vtc5-2 double mutants also exhibited a seedling-lethal phenotype, and cotyledons were subsequently bleached upon germination (Figure 4A). These were phenotypically indistinguishable from the vtc2-1;vtc5 double mutants previously described (Dowdle et al., 2007). Consistent with the previous study, both double mutants had undetectable levels of ascorbate (Figure 4B). The double mutants could be rescued to complete their life cycles by supplementation with ascorbate (Figure 4C). Thus these experiments show that the phenotype of the double mutants between vtc2-1 and vtc5 produced by Dowdle et al. (2007) is not caused by additional mutations in vtc2-1 and confirm the importance of the L-ascorbate biosynthetic pathway for seedling viability.
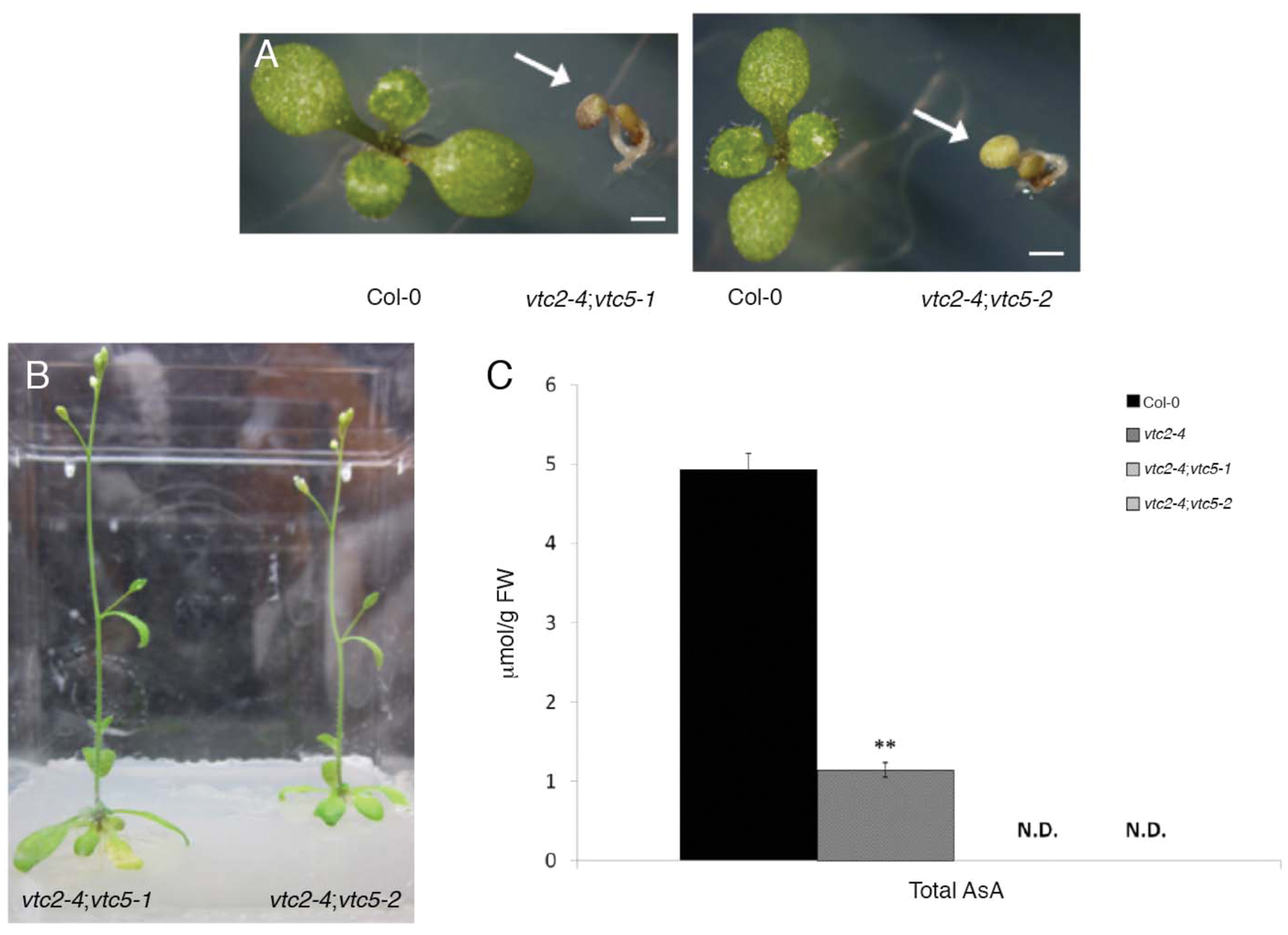
FIGURE 4. (A–C) Growth and leaf ascorbate concentration of Arabidopsis thaliana vtc2-4;vtc5 double mutants. (A) One-week old Col-0 and vtc2-4;vtc5-1 double mutants (arrow) grown on agar media. Bar = 1 mm. (B) Three-week-old vtc2-4;vtc5-1 double mutants grown on agar media supplemented with 0.5 mM L-ascorbic acid. (C) Total ascorbate (ascorbate and dehydroascorbate) concentration in leaves of 2-week-old seedlings grown on agar medium. Three seedlings from Col-0 and vtc2-4 and 15–20 vtc2-4;vtc5 seedlings were pooled for each sample. Error bars indicate SE (n = 3). Significant differences from Col-0 wildtype were determined with Student’s t-test with significance values adjusted for multiple comparisons using the Bonferroni correction (∗∗p < 0.01).
Discussion
It has been widely reported that Arabidopsis mutants with severe ascorbate deficiency exhibit a small growth phenotype and that this decreased growth is thus casually linked to ascorbate deficiency (Jander et al., 2002; Müller-Moulé et al., 2004; Pavet et al., 2005; Dowdle et al., 2007; Giacomelli et al., 2007; Kerchev et al., 2011). Here we show that the decreased growth phenotype of the vtc2-1 mutant line is not linked to ascorbate deficiency using two approaches; (1) the vtc2-4 mutant, an apparent null mutant which has comparable levels of ascorbate to vtc2-1, shows a different growth profile when compared to vtc2-1 mutants; (2) after backcrossing vtc2-1 to wildtype plants we readily isolated vtc2-1 ascorbate-deficient lines with weight and leaf sizes that are closer to wildtype than vtc2-1 mutants, as well as lines with a small growth phenotype that were homozygous for VTC2. It appears that the decreased growth phenotype of the vtc2-1 line is caused by an additional mutation/s that have likely arisen during the original EMS mutagenesis process (Conklin et al., 1996). Because of the difficulty of accurately scoring the size of individual F2 plants it is not clear from our analysis whether the vtc2 mutation and those associated with the growth defect are linked. However, it is unlikely that they are closely linked because it was relatively easy to identify F2 individuals homozygous for the wildtype allele at one locus but homozygous mutant at the other.
The vtc1-1 mutant (Conklin et al., 1996) also exhibits a decreased growth phenotype (Veljovic-Jovanovic et al., 2001; Pavet et al., 2005; Kerchev et al., 2011; Talla et al., 2011). VTC1 produces GDP-mannose, which has important functions in cell wall carbohydrate biosynthesis and protein glycosylation in addition to ascorbate biosynthesis (Conklin et al., 1999; Lukowitz et al., 2001). It is therefore possible that deficiency of GDP-mannose results in a number of different biochemical defects leading to a small growth phenotype that is not specifically due to ascorbate deficiency.
The vtc2-1 mutant line has been extensively analyzed (Müller-Moulé et al., 2003; Mukherjee et al., 2010; Gao et al., 2011; Zechmann, 2011; Botanga et al., 2012; Page et al., 2012) and has also been used to generate different mutant combinations in a number of studies. For example, interactions between redox metabolism and abscisic acid signaling were investigated by crossing vtc2-1 with an abscisic acid insensitive mutant (abi4). The vtc2-1;abi4 double mutant that was subsequently characterized, exhibited wildtype growth leading to the suggestion that abi4 suppressed the growth phenotype of vtc2-1 (Kerchev et al., 2011). It is plausible that from the cross, the vtc2-1;abi4 double mutant selected for analysis lacked the cryptic mutation(s) conferring the small growth phenotype. Furthermore, the use of whole genome microarray analysis to characterize differences between vtc2-1 and vtc2-1 abi4 mutant lines is likely to have been complicated by the possible absence of the cryptic mutation(s) in the double mutant background (Kerchev et al., 2011). The decreased growth phenotype of vtc2-1 has also been described in combination with mutations affecting chronic photo-oxidative stress (Müller-Moulé et al., 2004), chloroplastic ascorbate peroxidase (Giacomelli et al., 2007) and the autoimmune response (Zhu et al., 2013).
Our data indicate that the ascorbate deficiency of vtc2 mutants does not cause a large decrease in growth under laboratory conditions. While the vtc2-4 has a slightly smaller rosette area than wildtype, relative leaf expansion rate is identical. It is likely that vtc2-4 seedlings have smaller initial seedling size or vigor resulting in a magnifying size difference as the plants grow. Since the fully ascorbate deficient seedlings of the vtc2;vtc5 double mutant are not viable, it is apparent that a reduction greater than the ∼80% occurring in the vtc2-4 mutant is needed to impact growth. Ascorbate is amongst the most abundant of primary metabolites in Arabidopsis leaves (compare the widely reported values of 2–20 μmol g-1 fresh weight for ascorbate with the most abundant sugars, organic acids and amino acids reported in Szecowka et al., 2013). The presence of ascorbate concentrations in leaves that are much greater than needed to support growth in laboratory conditions emphasizes the importance of its protective functions for plants growing in stressful or fluctuating natural environments (Munné-Bosch et al., 2013).
Conclusion
In summary, we suggest that the interpretation of some studies of the commonly used vtc2-1 line or its derivatives may need to be re-evaluated. It is also important to take into consideration that the apparent cryptic mutation(s) affecting growth of vtc2-1 may have a significant impact on physiological status and gene expression unrelated to ascorbate deficiency, so although it was critical in identifying its biosynthetic pathway, it is not suitable for investigating the wider functions of ascorbate.
Author Contributions
BL, NS, and JG designed and performed the experiments, analyzed the data and drafted the manuscript. CC conceived the study and its design and coordination, and assisted with revisions of the manuscript. All authors read and consented to the final version of the manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
This research is supported by a grant from the Australian Research Council (BL, CSC, JFG) and a Biotechnology and Biological Sciences Research Council grant (NS; BB/G021678/1).
Supplementary Material
The Supplementary Material for this article can be found online at: http://journal.frontiersin.org/article/10.3389/fpls.2016.01025
FIGURE S1 | Reverse transcription polymerase chain reaction analysis of the vtc2-4 mutant. Total RNA was extracted from 10-day old seedlings and RT-PCR was performed using VTC2 specific primers (top panel) that flank the full-length transcript. ACTIN2 (ACT2) specific primers were used as an internal control (bottom panel).
FIGURE S2 | Fresh weight measurements of short-day grown plants. Shoot fresh weights of indicated lines were assessed after 6-week growth under short-day conditions. Error bars indicate SE (n = 7–10). Significant differences were determined using a Student’s t-test and statistical differences (p < 0.01) from Col-0 wildtype indicated with “a,” whereas “b” denotes differences from vtc2-1 mutants.
Abbreviations
ABRC, Arabidopsis Biological Resource Centre; EMS, Ethyl-Methanesulfonate; RT-PCR, Reverse Transcription Polymerase Chain Reaction; SAIL, Syngenta Arabidopsis Insertion Library.
Footnotes
References
Baker, N. R. (2008). Chlorophyll fluorescence: a probe of photosynthesis in vivo. Ann. Rev. Plant Biol. 59, 89–113. doi: 10.1146/annurev.arplant.59.032607.092759
Barth, C., Moeder, W., Klessig, D. F., and Conklin, P. L. (2004). The timing of senescence and response to pathogens is altered in the ascorbate-deficient Arabidopsis mutant vitamin c-1. Plant Physiol. 134, 1784–1792. doi: 10.1104/pp.103.032185
Botanga, C. J., Bethke, G., Chen, Z., Gallie, D. R., Feihn, O., and Glazebrook, J. (2012). Metabolite profiling of Arabidopsis inoculated with Alternaria brassicicola reveals that ascorbate reduces disease severity. Mol. Plant Microbe Interact. 25, 1628–1638. doi: 10.1094/MPMI-07-12-0179-R
Brosché, M., and Kangasjärvi, J. (2012). Low antioxidant concentrations impact on multiple signalling pathways in Arabidopsis thaliana partly through NPR1. J. Exp. Bot. 63, 1849–1861. doi: 10.1093/jxb/err358
Chen, Z., and Gallie, D. R. (2005). Increasing tolerance to ozone by elevating foliar ascorbic acid confers greater protection against ozone than increasing avoidance. Plant Physiol. 138, 1673–1689. doi: 10.1104/pp.105.062000
Colville, L., and Smirnoff, N. (2008). Antioxidant status, peroxidase activity, and PR protein transcript levels in ascorbate-deficient Arabidopsis thaliana vtc mutants. J. Exp. Bot. 59, 3857–3868. doi: 10.1093/jxb/ern229
Conklin, P. L., Norris, S. R., Wheeler, G. L., Williams, E. H., Smirnoff, N., and Last, R. L. (1999). Genetic evidence for the role of GDP-mannose in plant ascorbic acid (vitamin C) biosynthesis. Proc. Natl. Acad. Sci. U.S.A. 96, 4198–4203. doi: 10.1073/pnas.96.7.4198
Conklin, P. L., Saracco, S. A., Norris, S. R., and Last, R. L. (2000). Identification of ascorbic acid-deficient Arabidopsis thaliana mutants. Genetics 154, 847–856.
Conklin, P. L., Williams, E. H., and Last, R. L. (1996). Environmental stress sensitivity of an ascorbic acid-deficient Arabidopsis mutant. Proc. Natl. Acad. Sci. U.S.A. 93, 9970–9974. doi: 10.1073/pnas.93.18.9970
Dowdle, J., Ishikawa, T., Gatzek, S., Rolinski, S., and Smirnoff, N. (2007). Two genes in Arabidopsis thaliana encoding GDP-L-galactose phosphorylase are required for ascorbate biosynthesis and seedling viability. Plant J. 52, 673–689. doi: 10.1111/j.1365-313X.2007.03266.x
Edwards, K., Johnstone, C., and Thompson, C. (1991). A simple and rapid method for the preparation of plant genomic DNA for PCR analysis. Nucleic Acids Res. 19:1349.
Filkowski, J., Kovalchuk, O., and Kovalchuk, I. (2004). Genome stability of vtc1, tt4, and tt5 Arabidopsis thaliana mutants impaired in protection against oxidative stress. Plant J. 38, 60–69. doi: 10.1111/j.1365-313X.2004.02020.x
Gao, Y., Badejo, A. A., Shibata, H., Sawa, Y., Maruta, T., Shigeoka, S., et al. (2011). Expression analysis of the VTC2 and VTC5 genes encoding GDP-L-galactose phosphorylase, an enzyme involved in ascorbate biosynthesis, in Arabidopsis thaliana. Biosci. Biotechnol. Biochem. 75, 1783–1788. doi: 10.1271/bbb.110320
Giacomelli, L., Masi, A., Ripoll, D. R., Lee, M. J., and van Wijk, K. J. (2007). Arabidopsis thaliana deficient in two chloroplast ascorbate peroxidases shows accelerated light-induced necrosis when levels of cellular ascorbate are low. Plant Mol. Biol. 65, 627–644. doi: 10.1007/s11103-007-9227-y
Jander, G., Norris, S. R., Rounsley, S. D., Bush, D. F., Levin, I. M., and Last, R. L. (2002). Arabidopsis map-based cloning in the post-genome era. Plant Physiol. 129, 440–450. doi: 10.1104/pp.003533
Kampfenkel, K., Kushnir, S., Babiychuk, E., Inzé, D., and Van Montagu, M. (1995). Molecular characterization of a putative Arabidopsis thaliana copper transporter and its yeast homologue. J. Biol. Chem. 270, 28479–28486. doi: 10.1074/jbc.270.47.28479
Kempinski, C. F., Haffar, R., and Barth, C. (2011). Toward the mechanism of NH4+ sensitivity mediated by Arabidopsis GDP-mannose pyrophosphorylase. Plant Cell Environ. 34, 847–858. doi: 10.1111/j.1365-3040.2011.02290.x
Kerchev, P. I., Pellny, T. K., Vivancos, P. D., Kiddle, G., Hedden, P., Driscoll, S., et al. (2011). The transcription factor ABI4 is required for the ascorbic acid-dependent regulation of growth and regulation of jasmonate-dependent defense signaling pathways in Arabidopsis. Plant Cell 23, 3319–3334. doi: 10.1105/tpc.111.090100
Larkindale, J., Hall, J. D., Knight, M. R., and Vierling, E. (2005). Heat stress phenotypes of Arabidopsis mutants implicate multiple signaling pathways in the acquisition of thermotolerance. Plant Physiol. 138, 882–897. doi: 10.1104/pp.105.062257
Lee, Y., Park, C. H., Kim, A. R., Chang, S. C., Kim, S.-H., Lee, W. S., et al. (2011). The effect of ascorbic acid and dehydroascorbic acid on the root gravitropic response in Arabidopsis thaliana. Plant Physiol. Biochem. 49, 909–916. doi: 10.1016/j.plaphy.2011.05.013
Lukowitz, W., Nickle, T. C., Meinke, D. W., Last, R. L., Conklin, P. L., and Somerville, C. R. (2001). Arabidopsis cyt1 mutants are deficient in a mannose-1-phosphate guanylyltransferase and point to a requirement of N-linked glycosylation for cellulose biosynthesis. Proc. Natl. Acad. Sci. U.S.A. 98, 2262–2267. doi: 10.1073/pnas.051625798
Luna, E., Pastor, V., Robert, J., Flors, V., Mauch-Mani, B., and Ton, J. (2011). Callose deposition: a multifaceted plant defense response. Mol. Plant Microbe Interact. 24, 183–193. doi: 10.1094/MPMI-07-10-0149
Mukherjee, M., Larrimore, K. E., Ahmed, N. J., Bedick, T. S., Barghouthi, N. T., Traw, M. B., et al. (2010). Ascorbic acid deficiency in Arabidopsis induces constitutive priming that is dependent on hydrogen peroxide, salicylic acid, and the NPR1 gene. Mol. Plant Microbe Interact. 23, 340–351. doi: 10.1094/MPMI-23-3-0340
Müller-Moulé, P., Golan, T., and Niyogi, K. K. (2004). Ascorbate-deficient mutants of Arabidopsis grow in high light despite chronic photooxidative stress. Plant Physiol. 134, 1163–1172. doi: 10.1104/pp.103.032375
Müller-Moulé, P., Havaux, M., and Niyogi, K. K. (2003). Zeaxanthin deficiency enhances the high light sensitivity of an ascorbate deficient mutant of Arabidopsis. Plant Physiol. 133, 748–760. doi: 10.1104/pp.103.026252
Munné-Bosch, S., Queval, G., and Foyer, C. H. (2013). The impact of global change factors on redox signaling underpinning stress tolerance. Plant Physiol. 161, 5–19. doi: 10.1104/pp.112.205690
Noctor, G., and Foyer, C. H. (2011). Ascorbate and Glutathione: the heart of the redox hub. Plant Physiol. 555, 2–18. doi: 10.1104/pp.110.167569
Page, M., Sultana, N., Paszkiewicz, K., Florance, H., and Smirnoff, S. (2012). The influence of ascorbate on anthocyanin accumulation during high light acclimation in Arabidopsis thaliana: further evidence for redox control of anthocyanin synthesis. Plant Cell Environ. 35, 388–404. doi: 10.1111/j.1365-3040.2011.02369.x
Pastori, G. M., Kiddle, G., Antoniw, J., Bernard, S., Veljovic- Jovanovic, S., Verrier, P. J., et al. (2003). Leaf vitamin C contents modulate plant defense transcripts and regulate genes that control development through hormone signaling. Plant Cell 15, 939–951. doi: 10.1105/tpc.010538
Pavet, V., Olmos, E., Kiddle, G., Mowla, S., Kumar, S., Antoniw, J., et al. (2005). Ascorbic acid deficiency activates cell death and disease resistance responses in Arabidopsis. Plant Physiol. 139, 1291–1303. doi: 10.1104/pp.105.067686
Sanmartin, M., Drogoudi, P. D., Lyons, T., Pateraki, I., Barnes, J., and Kanellis, A. K. (2003). Over-expression of ascorbate oxidase in the apoplast of transgenic tobacco results in altered ascorbate and glutathione redox states and increased sensitivity to ozone. Planta 216, 918–928.
Scholl, R., Rivero-Lepinckas, L., and Crist, D. (1998). Growth of plants and preservation of seeds. Methods Mol. Biol. 82, 1–12. doi: 10.1385/0-89603-391-0:1
Szecowka, M., Heise, R., Tohge, T., Nunes-Nesi, A., Vosloh, D., Huege, J., et al. (2013). Metabolic fluxes in an illuminated Arabidopsis rosette. Plant Cell 25, 694–714. doi: 10.1105/tpc.112.106989
Talla, S., Riazunnisa, K., Padmavathi, L., Sunil, B., Rajsheel, P., and Raghavendra, A. S. (2011). Ascorbic acid is a key participant during the interactions between chloroplasts and mitochondria to optimize photosynthesis and protect against photoinhibition. J. Biosci. 36, 163–173. doi: 10.1007/s12038-011-9000-x
Veljovic-Jovanovic, S. D., Pignocchi, C., Noctor, G., and Foyer, C. H. (2001). Low ascorbic acid in the vtc-1 mutant of Arabidopsis is associated with decreased growth and intracellular redistribution of the antioxidant system. Plant Physiol. 127, 426–435. doi: 10.1104/pp.010141
Wang, J., Yu, Y., Zhang, Z., Quan, R., Zhang, H., Ma, L., et al. (2013). Arabidopsis CSN5B interacts with VTC1 and modulates ascorbic acid synthesis. Plant Cell 25, 625–636. doi: 10.1105/tpc.112.106880
Yoshimura, K., Nakane, T., Kume, S., Shiomi, Y., Maruta, T., Ishikawa, T., et al. (2014). Transient expression analysis revealed the importance of VTC2 expression level in light/dark regulation of ascorbate biosynthesis in Arabidopsis. Biosci. Biotechnol. Biochem. 78, 60–66. doi: 10.1080/09168451.2014.877831
Zechmann, B. (2011). Subcellular distribution of ascorbate in plants. Plant Signal. Behav. 6, 360–363. doi: 10.4161/psb.6.3.14342
Zhang, Z., Wang, J., Zhang, R., and Huang, R. (2012). The ethylene response factor AtERF98 enhances tolerance to salt through the transcriptional activation of ascorbic acid synthesis in Arabidopsis. Plant J. 71, 273–287. doi: 10.1111/j.1365-313X.2012.04996.x
Keywords: Arabidopsis, ascorbate, VTC2, VTC5, vitamin C, GDP-L-galactose phosphorylase
Citation: Lim B, Smirnoff N, Cobbett CS and Golz JF (2016) Ascorbate-Deficient vtc2 Mutants in Arabidopsis Do Not Exhibit Decreased Growth. Front. Plant Sci. 7:1025. doi: 10.3389/fpls.2016.01025
Received: 22 April 2016; Accepted: 28 June 2016;
Published: 13 July 2016.
Edited by:
Irene Murgia, Università degli Studi di Milano, ItalyReviewed by:
Christine Helen Foyer, University of Leeds, UKRobert L. Last, Michigan State University, USA
Copyright © 2016 Lim, Smirnoff, Cobbett and Golz. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: John F. Golz, amdvbHpAdW5pbWVsYi5lZHUuYXU= orcid.org/0000-0001-9478-5459
†Present address: Benson Lim, Genome Institute of Singapore, Genome, Singapore
 Benson Lim
Benson Lim Nicholas Smirnoff
Nicholas Smirnoff Christopher S. Cobbett1
Christopher S. Cobbett1 John F. Golz
John F. Golz