- 1College of Life Science, Fujian Agriculture and Forestry University, Fuzhou, China
- 2Key Laboratory of Crop Genetics and Breeding and Comprehensive Utilization, Ministry of Education/Fujian Agriculture and Forestry University, Fuzhou, China
- 3College of Crop Science, Fujian Agriculture and Forestry University, Fuzhou, China
In plants, Rho-related GTPases (Rops) are versatile molecular switches that regulate various biological processes, although their exact roles are not fully understood. Herein, we provide evidence that the ectopic expression of a Rop derived from Capsicum annuum, designated CaRop1, in tobacco plants modulates the response of these plants to Ralstonia solanacearum or aphid attack. The deduced amino acid sequence of CaRop1 harbors a conserved Rho domain and is highly homologous to Rops of other plant species. Transient expression of a CaRop1-GFP fusion protein in Nicotiana benthamiana leaf epidermal cells revealed localization of the GFP signal to the plasma membrane, cytoplasm, and nucleus. Overexpression (OE) of the wild-type CaRop1 or its dominant-negative mutant (DN-CaRop1) conferred substantial resistance to R. solanacearum infection and aphid attack, and this effect was accompanied by enhanced transcriptional expression of the hypersensitive-reaction marker gene HSR201; the jasmonic acid (JA)-responsive PR1b and LOX1; the insect resistance-associated NtPI-I, NtPI-II, and NtTPI; the ethylene (ET) production-associated NtACS1; and NPK1, a mitogen-activated protein kinase kinase kinase (MAPKKK) that interferes with N-, Bs2-, and Rx-mediated disease resistance. In contrast, OE of the constitutively active mutant of CaRop1(CA-CaRop1) enhanced susceptibility of the transgenic tobacco plants to R. solanacearum infection and aphid attack and downregulated or sustained the expression of HSR201, PR1b, NPK1, NtACS1, NtPI-I, NtPI-II, and NtTPI. These results collectively suggest that CaRop1 acts as a signaling switch in the crosstalk between Solanaceaes’s response to R. solanacearum infection and aphid attack possibly via JA/ET-mediated signaling machinery.
Introduction
In their natural habitats, plants are frequently exposed to attack by a wide variety of biological stresses, such as microbial pathogens and herbivorous predators. The evolutionary arms race between plants and their enemies has provided plants with a highly sophisticated defense system to perceive and discriminate invaders and to tailor their defense responses against specific invaders, thereby attaining higher fitness and survival rates. This defense system is regulated by complex signaling networks. Examples include calcium signaling; G-protein signaling; stimulation by phytohormones, specifically salicylic acid (SA), jasmonic acid (JA), and ethylene (ET); phosphorylation mediated by various kinases; and a burst of reactive oxygen species (ROS). Various transcription factors, such as WRKY and MYC2, have been frequently found to be shared by response pathways directed against plant pathogens/insects (Bodenhausen and Reymond, 2007; Dombrecht et al., 2007; Maffei et al., 2007; Pozo et al., 2008; Nascimento and Fett-Neto, 2010; Atamian et al., 2012; Kutyniok and Muller, 2012; Cheng et al., 2013; Dang et al., 2013, 2014; Kazan and Manners, 2013; Wang et al., 2013). The signaling pathways mediated by distinct components may interact with each other differently depending on the conditions. For example, SA- and JA-dependent signaling pathways may interact either synergistically or antagonistically, depending on the relative concentration of activating hormones (Mur et al., 2006). Components may act as key nodes in cross-communication signaling networks between plant responses and pathogen infection and insect attack (Bostock, 2005). In addition, the kinetics of the signaling may vary greatly in both quantity and timing, either in different plants or in the same plants attacked by different pathogens or insects in order to optimize the response against a single attacker (De Vos et al., 2005; Xie et al., 2012). Although plant responses to herbivores and pathogens has been the focus of much work over the past decades, the signaling pathways of plant responses to these challenges and their molecular and pathway crosstalk is still not well understood, especially with respect to early signaling cascades.
The Rho-related GTPases of plants (Rops) family, also known as the Rac family, belongs to the Ras superfamily of small GTPases (Zheng and Yang, 2000). Like other small GTPases, Rops act as versatile molecular switches by cycling between GDP-bound inactive and GTP-bound active forms in cells and are regulated by a set of guanine nucleotide exchange factors, GTPase-activating proteins (GAPs), and guanine nucleotide dissociation inhibitors (GDIs; Eklund et al., 2010). Loss-of-function mutation of these proteins are likely to induce no clear phenotypic effects due to additional redundant proteins. For this reason, Rops GTPase mutants that are constitutively active (CA) or dominant-negative (DN) due to the presence of conserved point mutations have become important tools for studying Rop proteins. In fact, Rop proteins in CA- and DN-transgenic lines of Arabidopsis and rice have been studied with these tools (Zheng and Yang, 2000; Ono et al., 2001; Fu et al., 2002; Tao et al., 2002; Zheng et al., 2002; Poraty-Gavra et al., 2013). Rops are important in a wide variety of physiological plant processes, such as polar morphogenesis of tip-growing cells in pollen tubes and root hairs (Kost, 2008; Lee and Yang, 2008), polar morphogenesis of leaf epidermal cells (Yang, 2008; Singh et al., 2013), branching of trichomes and root hairs (Duan et al., 2010; Singh et al., 2013), lignin and secondary cell wall synthesis (Kawasaki et al., 2006; Foucart et al., 2009), asymmetric cell division (Humphries et al., 2011), regulation of cytoskeletal dynamics (Mucha et al., 2011), cell expansion and stomata development (Pathuri et al., 2009), and abscisic acid (ABA)-mediated stomatal closure (Li and Liu, 2012). These proteins have also been implicated in adaptation of plants to various environmental cues, including pathogen infection (Agrawal et al., 2003; Thao et al., 2007; Chen et al., 2010a; Huesmann et al., 2011, 2012; Kim et al., 2012; Poraty-Gavra et al., 2013), hypoxia (Steffens and Sauter, 2010), salt-stress signaling (Luo et al., 2006), and drought stress (Li et al., 2012). Moreover, several biological processes have been found to involve specific Rops. For example, Atrop6 (DN) plants are small and have multiple inflorescence stems, twisted leaves, deformed leaf epidermis pavement cells, and differentially organized cytoskeleton. A SA-mediated defense response in these plants was conferred by overexpression (OE) of DN-rop6 (Poraty-Gavra et al., 2013). In addition, ABA-mediated responses affected by Rop11 include seed germination, seedling growth, stomatal closure, induction of ABA-responsive genes, and plant response to drought stress (Li et al., 2012); however, the functions of Rop family members in different plant species have not been full characterized.
Pepper (Capsicum annuum) is an important agricultural crop worldwide. As with typical Solanaceae sp., pepper plants are susceptible to many soil-borne pathogens and are, therefore, sensitive to continuous cropping. Of particular note is the susceptibility of this species to Phytophthora capsici and Ralstonia solanacearum, the two most important causal agents of pepper diseases worldwide. Aphids are notorious pests in pepper and cause heavy losses, either by direct attack or indirect damage as a vector for cytopathic viruses, including tobacco mosaic virus (TMV). Studies of interaction between pepper and pathogen/pest may contribute to development of new pepper cultivars with improved resistance to these biotic stresses and offer effective means for long-lasting control of pepper diseases and/or pests. Until now, no Rop/Rac gene has been cloned and functionally characterized in pepper disease or pest resistance. In the present study, we demonstrate that ectopic expression of the DN or CA mutants of CaRop1, a Rop from C. annuum, modulated the response of tobacco plants to R. solanacearum and aphid attack possibly via JA/ET-mediated signaling machinery.
Materials and Methods
Plant Materials and Growth Conditions
Pepper plants (C. annuum) from the genetically stable inbred line p120 were provided by the pepper breeding group at the Fujian Agriculture and Forestry University (FAFU). The seeds of Nicotiana benthamiana and the Nicotiana tabacum cultivar K326 (including its transgenic lines) were provided by Professor Fang of the College of Plant Protection at FAFU. C. annuum or N. benthamiana seeds were sown in a soil mix of peat moss and Perlite (2:1, v/v) in plastic pots. Tobacco seeds were surface sterilized in 75% alcohol for 30 s, incubated in 10% H2O2 for 10 min, and then washed five times with sterile ddH2O. Transgenic tobacco seeds were placed on Murashige and Skoog medium (MS; Murashige and Skoog, 1962) supplemented with 75 mg L-1 kanamycin for 2–3 weeks, while wild-type tobacco seeds were placed on MS medium without supplement. Surviving tobacco seedlings were transferred into soil mix (peat moss:perlite [2:1, v/v]) in plastic pots. All seedlings were grown in a greenhouse under white fluorescent light (70 μmol photons m-2s-1, OSRAM, China) for 16 h d-1 at 25°C and 70% relative humidity.
Isolation and Sequence Analysis of CaRop1 cDNA
Full-length cDNAs of Rop GTPase genes from pepper were obtained by searching expressed sequence tags (ESTs) of C. annuum (taxid: 4072) with the sequence of AtRac1 as a query using TBLASTN1. Matching ESTs were assembled into contigs using DNAMAN software. Specific PCR primers were designed from a contig with high sequence similarity to AtRac1. Corresponding cDNA clones were screened from a cDNA library by a PCR-based 96-well screening method (Munroe et al., 1995). Positive clones (λTriplEx2) were converted to pTriplEx2 by excision in vivo according to the manufacturer’s protocol (Clontech, Mountain View, CA, USA). The pTriplEx2 samples were sent to TaKaRa (Dalian, China) for sequencing. The sequence data were analyzed using the BLAST program from NCBI1. Homology and conserved domains were compared against sequences of other plant species in GenBank using DNAMAN software as described previously (Zheng and Yang, 2000; Agrawal et al., 2003). A phylogenetic tree was constructed with MEGA5.0 software using the neighbor-joining method.
Construction of CaRop1 Mutants
The CA and DN CaRop1 mutants were produced as described in previous studies by changing the glycine (G) and threonine (T) residues (marked by arrows in Figure 1) in the domain I to valine (V) and asparagine (N), respectively (Agrawal et al., 2003; Schallhart et al., 2012). An overlap-extension PCR protocol for site-directed mutagenesis was performed, and sequences around the point to be mutated (Figure 1) were designed as forward primers (CA-CaRop1-F, DN-CaRop1-F) and reverse primers (CA-CaRop1-R, DN-CaRop1-R). To produce CA-CaRop1, three rounds of PCR were performed. The first and second rounds of PCR were performed with the primer combinations CaRop1-F/CA-CaRop1-R and CA-CaRop1-F/CaRop1-R, respectively. The amplified products were mixed and used as the template for the third round of PCR, with the primer combination CaRop1-F/CaRop1-R. Similarly, to produce DN-CaRop1, the first and second rounds of PCR were performed with the primer combinations CaRop1-F/DN-CaRop1-R and DN-CaRop1-F/CaRop1-R, respectively, and the third round was carried out with the primer combination CaRop1-F/CaRop1-R (Table 1). The two amplified mutants were subcloned into pDONR207 entry vectors, according to the supplier’s instructions (Invitrogen, Carlsbad, CA, USA). The two mutants were confirmed by DNA sequencing.
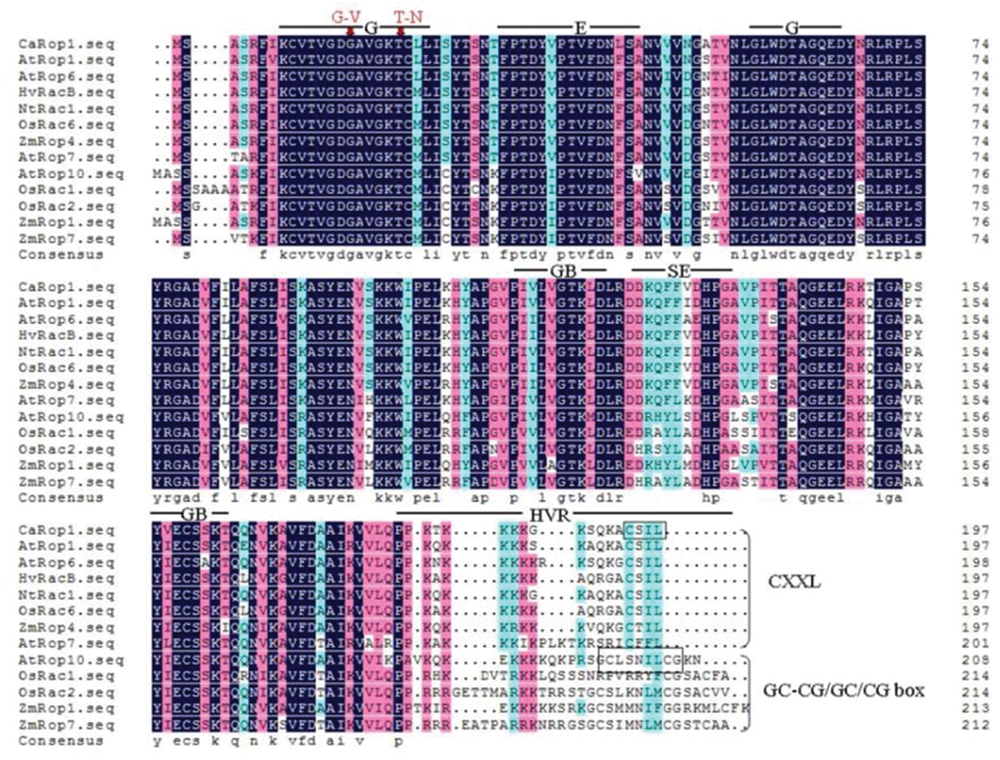
FIGURE 1. Analysis of the deduced amino acid sequence of CaRop1 and comparison with homologous sequences from other monocot and dicotyledonous (dicot) species using DNAMAN5.0. The high-, low-, and neutral-consensus amino acid residues are depicted in black, blue, and red colors, respectively. Bars indicate conserved functional domains of Rop/Rac family proteins. Abbreviations: G, GTPase domain; E, effector domain; GB, GDP/GTP-binding domain; SE, special effector loop (Rho insert region); HVR, C-terminal hypervariable region. To produce constitutively active (CA) and dominant-negative (DN) forms of CaRop1, the glycine (G) and threonine (T) residues marked with red arrows were changed to valine (V) and asparagine (N), respectively. The two types of Rops, based on the C-terminal motif, are indicated with a black frame and brace: the type-I Rops are characterized by the presence of a CXXL motif, and the type-II Rops lack this motif but contain a GC-CG/GC or CG box.
Construction of Vectors
For OE and transient expression, the full open reading frames (ORFs) of CaRop1, CA-CaRop1, or DN-CaRop1 in the entry vector pDONR207 were transferred into various gateway-compatible destination vectors. For OE vector construction, the ORFs of CaRop1, CA-CaRop1, or DN-CaRop1 were transferred into the pK7WG2 destination vector containing the CaMV35S promoter by LR reaction (Invitrogen, Carlsbad, CA, USA) to yield pK7WG2-CaRop1, pK7WG2-CA-CaRop1, and pK7WG2-DN-CaRop1. For construction of the CaRop1–GFP chimeric gene, a gateway compatible vector pMDC83 was employed. The ORF of CaRop1 without the termination codon was cloned at the N-terminus of intact GFP and in-frame to the GFP-coding sequences (Invitrogen).
Expression of CaRop1-GFP in N. benthamiana and Subcellular Localization
The recombinant plasmid p35S::CaRop1-GFP and the control plasmid p35S::GFP(pMDC83) were transformed into Agrobacterium tumefaciens GV3101. The subcellular localization of CaRop1 was determined as reported in our previous work with CaWRKY40 (Dang et al., 2013). A. tumefaciens cells carrying the different constructs were centrifuged and resuspended in an infiltration buffer (10 mM MgCl2, 10 mM MES, and 100 μM acetosyringone. pH 5.7) at a final OD600 of 0.4. Samples were infiltrated into 4–5-week-old N. benthamiana leaves. GFP was visualized using a laser scanning confocal microscope (Leica TCS SP8; Mannheim, Germany) with an excitation wavelength of 488 nm and a 505–530-nm band-pass emission filter.
Construction of OE Transgenic CaRop1, CA-CaRop1, and DN-CaRop1 Tobacco Lines
Because pepper plants are recalcitrant to genetic transformation, tobacco, which belongs to the same Solanaceae family as pepper, was used as a suitable transformation system for OE of CaRop1, CA-CaRop1, and DN-CaRop1 to investigate the function of CaRop1. The OE vectors pK7WG2-CaRop1, pK7WG2-CA-CaRop1, or pK7WG2-DN-CaRop1 was introduced into A. tumefaciens GV3101 via electroporation and then transformed into N. tabacum K326 using a conventional leaf-disk transformation method. The transformants were regenerated in MS medium containing 75 mg L-1 kanamycin, and the regenerated T0 plants, which were confirmed by PCR with kanamycin-specific primers, were cultivated in a soil mix of peat moss and Perlite (2:1, v/v) in plastic pots in the growth room. The T0 plants were self-pollinated to generate the seeds for the T1 lines. Similarly, the seedlings of the T1 lines were selected by kanamycin, and the seeds of the T2 lines were developed by self-pollination of the plants of the T1 lines. Expression levels of CaRop1, CA-CaRop1, and DN-CaRop1 in the tobacco transformants were measured using semi-quantitative RT-PCR compared to the K326 control plants. Plants of the T2 lines that exhibited high expression of CaRop1 and their CA and DN mutants were used for further experiments.
Pathogen Cultivation, Inoculation, and Plant Immunity Assay
Growth and inoculation of R. solanacearum FJC100301 were performed as described previously (Shang et al., 2012). Cells of FJC100301 were cultured in potato sucrose agar (PSA) medium (200 g potato, 20 g sucrose, 3 g beef extract, 5 g tryptone, 1 L water) and shaken at 200 rpm in an incubator at 28°C for 36 h. Then, samples were homogenized in sterile 10 mM MgCl2, and the cell density was diluted to 108 colony-forming units (CFU)/mL (OD600 = 0.8). For the disease resistance assay, 2-month-old seedlings of the transgenic lines and the K326 line were inoculated by infiltration of 10 μL of R. solanacearum suspension (OD600 = 0.8) into the third leaves from the top using a syringe with a needle. To test the effect of OE of the CA and DN mutants on the growth of R. solanacearum, inoculated leaves were harvested 72 h post-inoculation (hpi), and leaf disks 3 cm from the inoculation sites were collected and homogenized in 10 mM MgCl2 (three disks per sample, 7-mm diameter). The homogenate was plated on PSA medium at appropriate dilutions. After incubation at 28°C for 2 days, the colonies were counted (Moeder et al., 2005). For analysis of the hypersensitive response (HR), the inoculated leaves were harvested at 72 hpi, whole leaves were stained with trypan blue (Shang et al., 2012), and the typical phenotypes were photographed. The phenotypic effects of the OE of CaRop1 in the CA and DN mutants on the resistance of plants to pathogen inoculation were detected and photographed at 14 days post-inoculation (dpi). To analyze the transcriptional expression of defense-related genes in response to pathogen infection, the R. solanacearum inoculated third leaves from the top were harvested at 36 hpi for preparation of total RNA.
Aphid Culture and Bioassays
Aphids used in this study were harvested in a tobacco field at an experiment station at FAFU. An aphid population (Myzus persicae) was maintained on K326 plants in a pesticide-free greenhouse. The 5-month-old plants from the T2 tobacco lines and the control line were artificially inoculated with 10 aphid larvae on the abaxial surface of the third leaf from the top and were placed in a random arrangement in the pesticide-free greenhouse. The aphid population on each plant was photographed and counted 15 dpi. At least three plants of each line were examined.
Total RNA Isolation and Quantitative Real-Time RT-PCR
Total RNA was isolated with the TRIzol reagent (Invitrogen) following the manufacturer’s instructions. The RNA sample was then reverse transcribed using the PrimeScriptTM RT-PCR kit in a 10-μl volume. The resulting cDNA was diluted 10-fold and then amplified using SYBR® Premix Ex TaqTM II (TaKaRa Perfect Real Time; Dalian, China) using an Applied Biosystems 7500 Real-Time PCR system. Reactions were conducted in 10-μL volumes under the following conditions: 95°C for 30 s; 40 cycles of 95°C for 5 s followed by 60°C for 34 s; 95°C for 15 s; 60°C for 1 min; 95°C for 15 s; and 60°C for 15 s. Amplification of the target genes was monitored each cycle with SYBR-green fluorescence. The Ct (threshold cycle), which is defined as the real-time PCR cycle at which a statistically significant increase of reporter fluorescence is first detected, was used as a measure of the starting copy number of the target gene. Five replicates of each experiment were performed, and normalized transcript level data for the target genes were analyzed by qPCR and the Livak method (Livak and Schmittgen, 2001). Relative expression levels of pepper target genes were normalized to the expression of CaActin, and the relative expression levels of target genes in transgenic tobacco were normalized to the expression of NtEF1α.
Histochemical Staining
Staining with trypan blue was performed according to the previously published method of Cai et al. (2015).
Results
Cloning and Sequence Analysis of CaRop1 cDNA
To acquire full-length cDNAs of Rop family members in C. annuum, ESTs of C. annuum that are homologous to AtRac1 were searched using BLASTN with AtRac1 as a query sequence, and the acquired ESTs were assembled into contigs. A contig with high sequence similarity to AtRac1 was selected, and a specific pair of primers was designed for use in a PCR-based 96-well screen of a cDNA library of pepper leaves. A positive clone was acquired after three rounds of screening, and sequencing revealed that this clone was a full-length cDNA, which was designated as CaRop1 (GenBank Accession Number ABB71820). CaRop1 is 1,149 bp in length and harbors an ORF that encodes a 197-amino acid polypeptide with a conserved Rho domain with a predicted molecular mass of 21,443.7 kD and a pI of 9.3. Multiple alignment analysis showed that CaRop1 has a high sequence similarity to Rops of other plant species and contains all the structural features of a type-I plant Rop. This conclusion was based on the presence of the C-terminal canonical CaaL motif, which is required for prenylation and membrane attachment (Agrawal et al., 2003; Wu et al., 2011). A phylogenetic tree constructed from CaRop1 and Rops from other plant species revealed that CaRop1 belongs to a subgroup that also contains HvRacB, OsRacB, AtRop1, and AtRop6. All of these Rop proteins have been implicated in plant responses to attack by pathogens (Figure 2) (Schultheiss et al., 2002, 2003; Poraty-Gavra et al., 2013). Based on the tenet of phylogeny of function, that is, that genes of similar function are likely to be grouped in a phylogenetic tree (Pereira-Leal and Seabra, 2001), we speculate that CaRop1 participates in pathogen-response signaling.
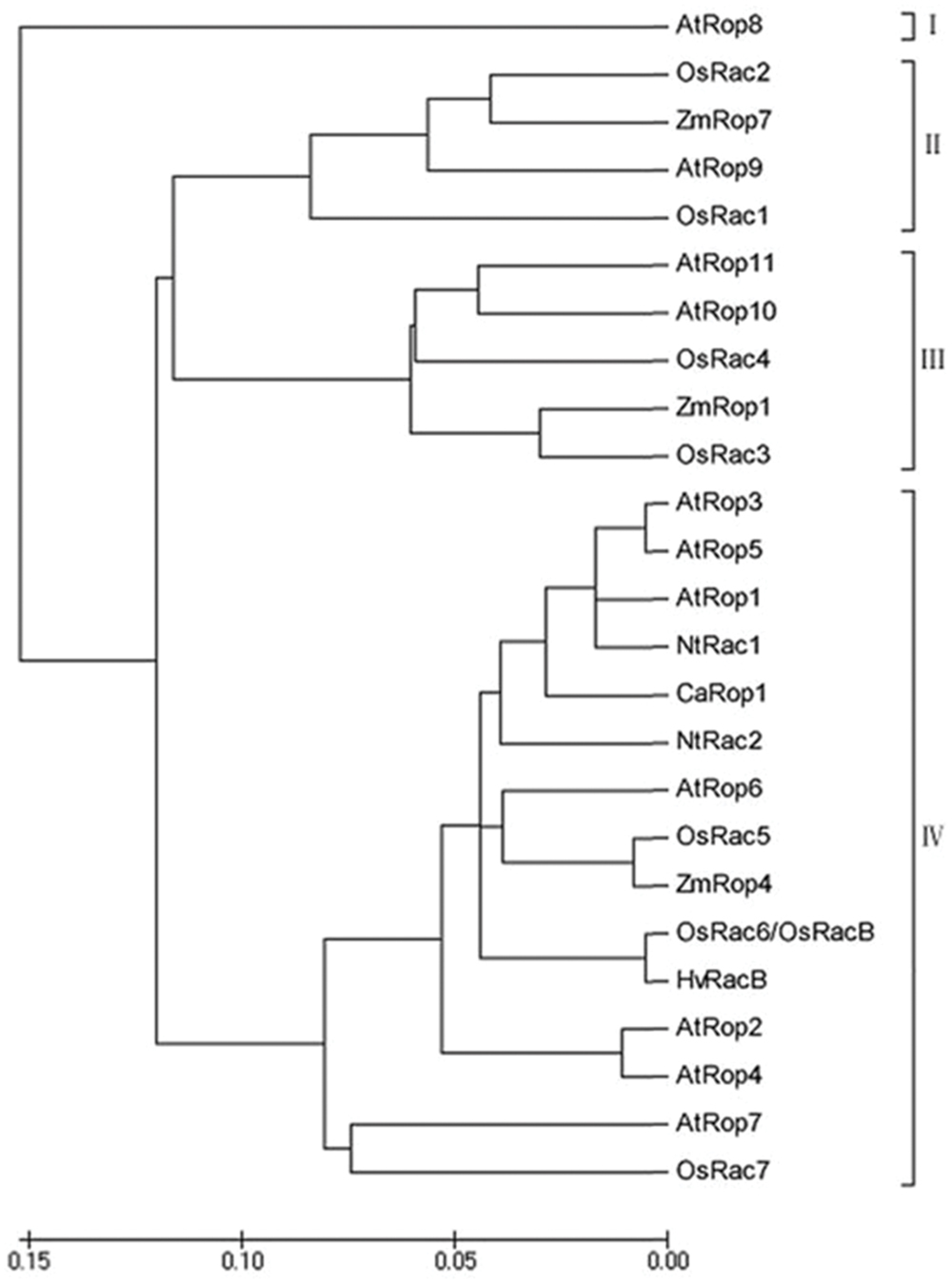
FIGURE 2. Phylogenetic Analysis of Rop/Rac GTPases. Phylogenetic tree for the deduced amino acid sequences of Rop GTPases from Arabidopsis, rice, and pepper was constructed using NJplot. Abbreviations for species: At, Arabidopsis thaliana; Os, Oryza sativa; Ca, Capsicum annuum; Zm, Zea mays; Hv, Hordeum vulgare; Nt, Nicotiana tabacum. Accession numbers: AtRop1-AtRop11 are At3g51300, At1g20090, At2g17800, At1g75840, At4g35950, At4g35020, At5g45970, At2g44690, At4g28950, At3g48040; and At5g62880. OsRac1-OsRac7 are Q9SSX0, Q68Y52, Q6Z808, Q67VP4, Q6EP31, Q6ZHA3, and Q6Z7L8; ZmRop1, ZmRop4, and ZmRop7 are NP_001104929, NP_001105719, and NP_001105523; HvRacB is AJ344223; NtRac1and NtRac2 are AAK31299 and AAD00118; and CaRop1 is DQ257288. Roman numerals I-IV refer to the four subgroups of Rops.
Subcellular Localization of CaRop1
To determine the subcellular localization of CaRop1 in vivo, the CaRop1 ORF without its termination codon was fused to intact GFP-coding sequences at the N-terminus. The localization pattern of CaRop1-GFP was analyzed in transient expression experiments using N. benthamiana leaf epidermis. Generally, GFP signals were observed throughout the N. benthamiana leaf epidermal cells in the plasma membrane, cytoplasm, and nucleus (Figure 3) in agreement with a previous observation that type I Rops in rice were present throughout the cell (Chen et al., 2010b).
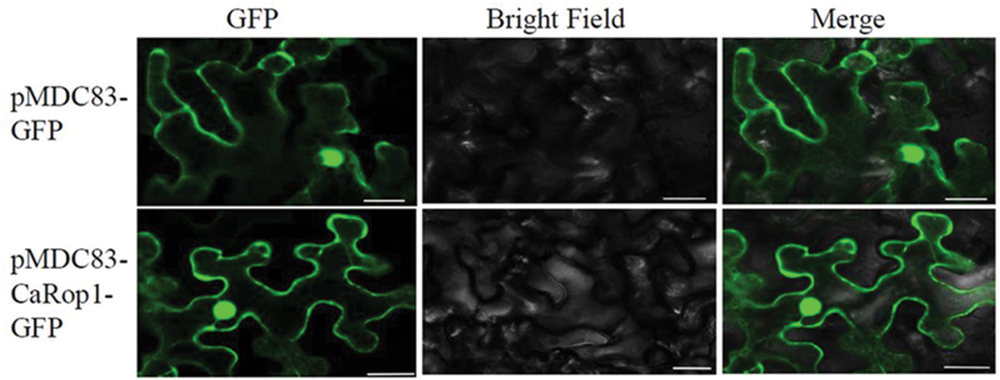
FIGURE 3. Subcellular localization of CaRop1 in Nicotiana benthamiana leaf epidermal cells. The CaRop1-GFP fusion protein localized at the plasma membrane, cytoplasm, and nucleus of N. benthamiana leaf epidermal cells. GFP alone was found throughout the cells. Images of GFP were obtained using a laser scanning confocal microscope with an excitation wavelength of 488 nm and a 505–530-nm band-pass emission filter.
Susceptibility of Tobacco to R. solanacearum Infection Was Reduced by OE of CaRop1 and DN-CaRop1 but Enhanced by OE of CA-CaRop1
To assay the possible function of CaRop1 in plant immunity, we generated transgenic tobacco lines that overexpressed CaRop1 or its CA or DN mutant. At least ten independent transgenic T2 tobacco lines were obtained for each genotype, and none of these lines exhibited any significant phenotypic difference or growth retardation compared to the wild-type K326 plants. Two T2 lines of each genotype were randomly chosen for further assay. These lines were challenged by R. solanacearum, a soil-borne bacterium that causes lethal disease by inducing wilting symptoms in more than 200 plant species, including economically important crops, such as pepper and tobacco. A highly virulent strain of R. solanacearum FJC100301 (Dang et al., 2013) was used to inoculate the plants of transgenic tobacco and the control. At 7 dpi, obvious wilting of leaves of K326 and CA-CaRop1-OE plants was observed, whereas only faint wilting was found in leaves of CaRop1- and DN-CaRop1-OE lines. At 14 dpi, extremely severe decaying symptoms were found in CA-CaRop1 and K326 plants, but the CaRop1- and DN-CaRop1-OE lines exhibited only limited lesions (Figure 4A). Consistently, higher CFU were detected in R. solanacearum-inoculated CA-CaRop1 lines and K326 plants compared to those in R. solanacearum-inoculated CaRop1 and DN-CaRop1 tobacco lines (Figure 4B). As HR cell death has been previously found to be a hallmark in plant immunity, especially in effector-triggered immunity (Jones and Dangl, 2006), HR cells in different tobacco transgenic plant lines were investigated. As evidenced by trypan blue staining, consistent HR cell death was observed in inoculated leaves of CaRop1- and DN-CaRop1-OE lines at 3 dpi, while little or no HR cell death was noted in the inoculated leaves of the control or CA-lines, respectively (Figure 4C).
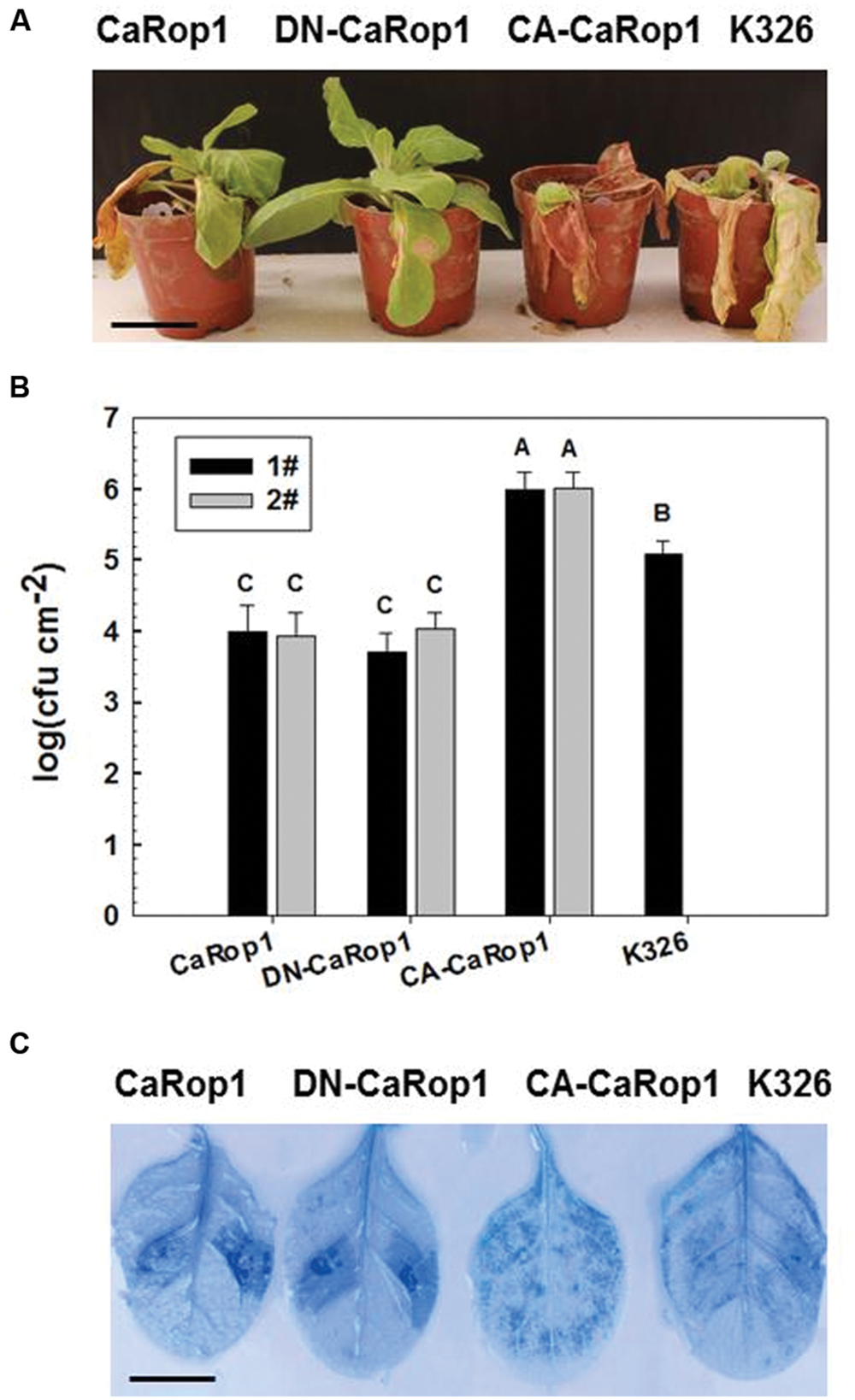
FIGURE 4. HR-associated cell death was triggered by overexpression of CA-CaRop1, DN-CaRop1, and CaRop1 in plants. (A) The phenotype of the CA-CaRop1, DN-CaRop1, and CaRop1 OE in tobacco plants after inoculation of R. solanacearum at 14 dpi is shown. Tobacco plants were equivalently inoculated with a concentration of 1 × 108 CFU/mL of bacteria in the lateral veins of the third leaves from the top. Three plants for each line were tested, and the phenotypes of the plants were photographed at 14 dpi. (B) The effect of overexpression of CA-CaRop1, DN-CaRop1, and CaRop1 on the growth of R. solanacearum. Leaf disks 3 cm away from the inoculated site were cut at 72 hpi, and the CFU/mL of R. solanacearum per sample (each from three leaf disks) were measured. The values are shown as mean ± SD of samples from three independent plants. The experiments were repeated three times with two lines for each construct. Different letters indicate statistically significant differences (Student–Newman–Kuels test; P < 0.01). (C) Trypan blue staining of the leaves of tobacco plants was performed 3 dpi of the third leaves from the top with 10 μL of R. solanacearum suspension (OD600 = 0.8) using a syringe with a needle. Scale bars = 1 cm.
To further elucidate the possible mode of action of CaRop1-mediated defense, transcriptional responses of known defense-associated marker genes were investigated in two independent CA-, DN- CaRop1-, and CaRop1-OE transgenic lines using qPCR. Defense-associated genes examined in this study include the HR-associated gene NtHSR201 (Takahashi et al., 2004), the SA-responsive genes NtPR2 and NtNPR1 (Spoel et al., 2003; Sohn et al., 2007), the ET production-associated gene NtACS1 (Sohn et al., 2007), the JA-responsive gene NtPR1b (Sohn et al., 2007), the JA biosynthesis-associated gene NtLOX (Halitschke and Baldwin, 2003), and NPK1, a MAPKKK gene involved in plant immunity (Jin et al., 2002). All of these genes were previously identified to be upregulated in response to R. solanacearum infection in tobacco (Dang et al., 2013). The transcript abundances of NtHSR201, NtACS1, NtPR1b, NtLOX, and NtNPK1 were higher at 36 hpi n R. solanacearum-inoculated plants of CaRop1 and DN-CaRop1 lines than in CA-CaRop1 lines and K326 plants, while transcriptional levels of NtNPR1 and NtPR2 did not differ notably among the tested genotypes (Figure 5).
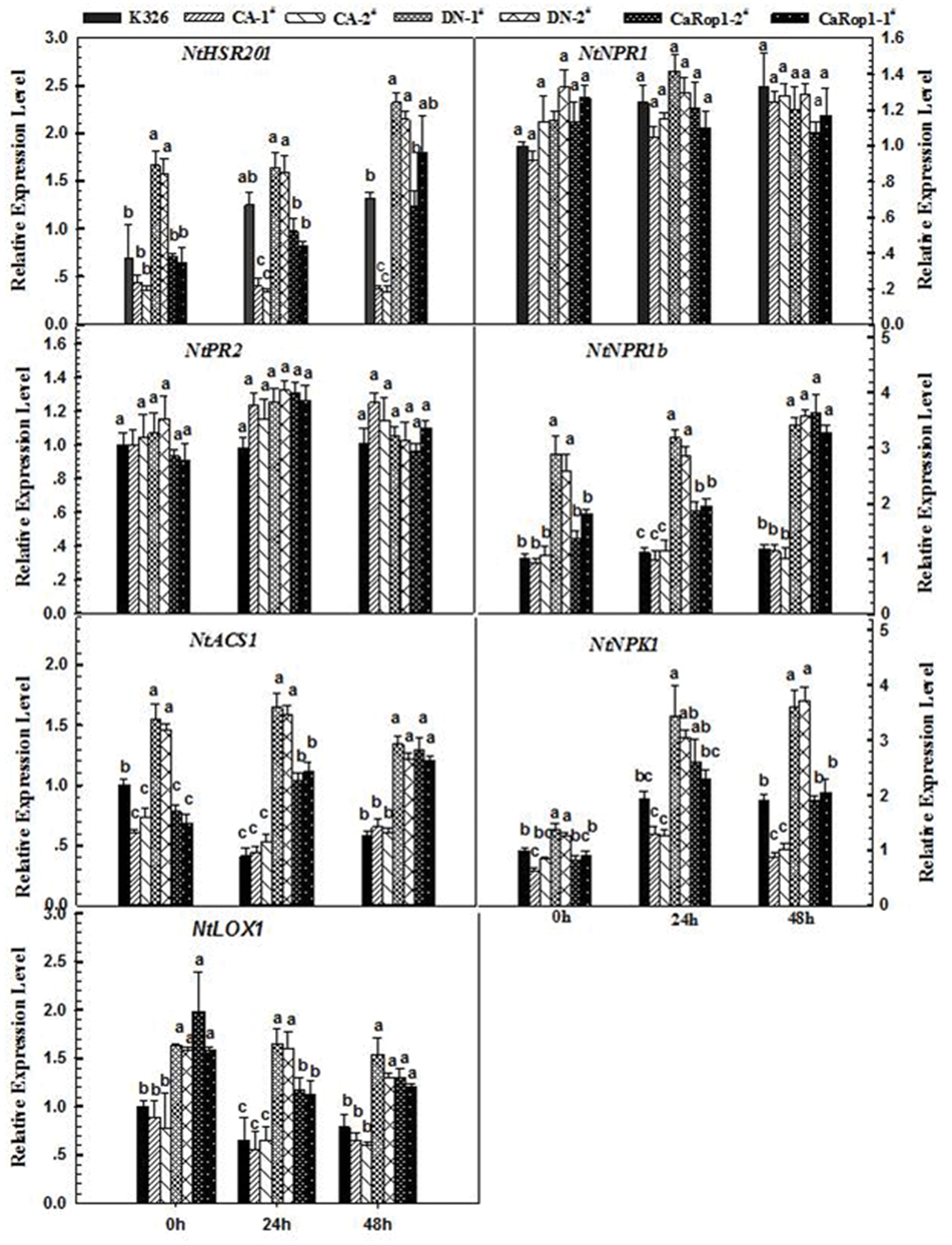
FIGURE 5. Immunity-associated marker genes were upregulated by OE of CaRop1 and DN-CaRop1 but were downregulated by overexpression of CA-CaRop1. Tobacco plants were equivalently inoculated with a concentration of 1 × 108 CFU/mL of bacteria in the lateral vein of the third leaves from the top, which were harvested at 36 hpi for preparation of RNA to be used as a template for real-time RT-PCR with specific primer pairs of immunity-associated marker genes, including NtHSR201, NtNPR1, NtPR2, NtPR1b, NtLOX1, NtACS1, and NtNPK1. Defense-related gene transcript levels of wild-type tobacco K326 were used as a reference, which was set as “1.” Each value is given as the average of three replicate experiments ±SD. Different letters indicate statistically significant differences (Student–Newman–Kuels test; P < 0.05).
Susceptibility of Tobacco to Aphid Attack Was Reduced by OE of DN-CaRop1 or CaRop1 but Was Enhanced by OE of CA-CaRop1
Plants of T2 tobacco lines and K326 tobacco were exposed to aphids in a greenhouse. The plants were attacked by the aphids, and large numbers colonized the abaxial surface of the leaves. Interestingly, the aphid population success varied considerably in different plant genotypes. CA-CaRop1 obviously facilitated the colonization and reproduction of aphid larva on the leaves, while DN-CaRop1 and CaRop1 strongly lowered aphid colonization. To confirm the defensive role of CaRop1 in aphid response, we repeated the aphid infestation experiment by artificial inoculation. Briefly, aphid adults (10 days) were transferred from leaves of nursery K326 seedlings to experimental plants with a fine toothpick. Fifteen days later, the number of aphids on each inoculated plant was assessed, and the typical phenotypes were photographed. Consistently, large numbers of aphids were found in the control plants and CA-CaRop1-OE plants, whereas, no aphids or only small numbers of aphids were found in the DN-CaRop1- and CaRop1-OE plants (Figures 6A,B).
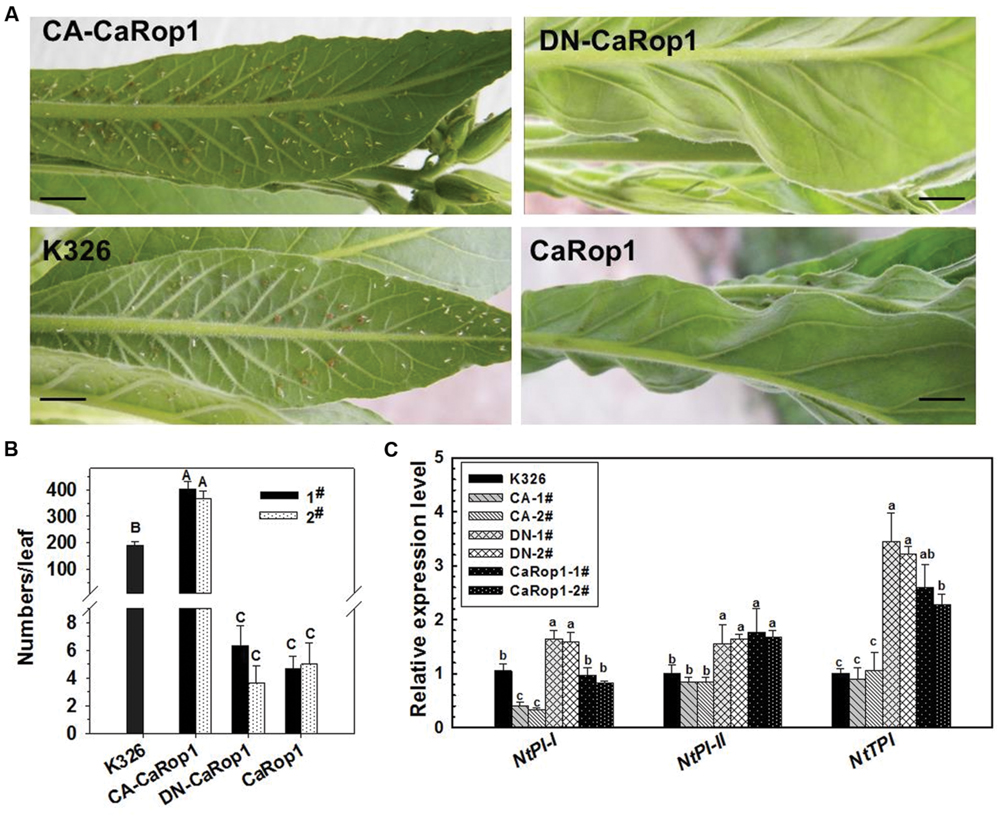
FIGURE 6. The effect of overexpression of DN-CaRop1, CA-CaRop1, and CaRop1 on the resistance of tobacco plants to aphids and on the transcriptional expression of NtPI-I, NtPI-II and NtTPI. (A) The aphid population on the artificially inoculated leaves of 5-month-old seedlings of CaRop1, DN-CaRop1, CA-CaRop1 overexpressing transgenic lines or the wild type K326 at 15 dpi. (B) The number of aphids on each artificially inoculated leaf of 5-month-old tobacco seedlings of different genotypes at 15 dpi. (C) Transcriptional expression of NtPI-I, NtPI-II, and NtTPI in 5-month-old DN-CaRop1, CA-CaRop1 and CaRop1 OE tobacco seedlings was determined by real-time RT-PCR. Transcript levels of the marker genes in K326 were used as a reference and were set as “1.” Each value is the average of three replicate experiments ±SD, and different letters indicate statistically significant differences (Student–Newman–Kuels test; P < 0.05). Scale bars = 1 cm.
To further investigate the role of CaRop1 in the response of tobacco plants to aphid attack and to elucidate the possible mechanism, we examined the effect of OE of CaRop1 on the transcript levels of NtPI-I, NtPI-II, and NtTPI in two independent transgenic lines (Figure 6C). Results of qPCR showed that the transcriptional expression of NtPI-I was significantly triggered by OE of CaRop1 or DN-CaRop1 but that this expression was downregulated or remained unchanged by OE of CA-CaRop1 in tobacco plants compared to that in the wild-type plants. On the other hand, the transcriptional expression of NtPI-II and NtTPI were significantly upregulated in CaRop1- or DN-CaRop1-OE tobacco plants but remained unchanged in CA-CaRop1-OE tobacco plants compared to that in the wild-type plants.
Discussion
The Rop/Rac family is highly conserved in the plant kingdom. To date, seven Rop/Rac family members have been identified in rice (Miki et al., 2005), and 11 have been identified in Arabidopsis (Zheng and Yang, 2000). The induced amino acid sequence of our newly identified CaRop1 contains a conserved Rho domain and has high sequence similarity to type I Rops, such as AtRop1 and AtRop6 in Arabidopsis and OsRacB in rice. Some type I Rops have been reported to localize to the plasma membrane, the cytoplasm, and the nucleus, due to their C-terminal regions (Bischoff et al., 2000; Schultheiss et al., 2003; Xiao et al., 2009; Chen et al., 2010b). This localization has been reported for OsRac5, OsRacB/OsRac6, and OsRac7 in rice; AtRop4 and AtRop6 in Arabidopsis; and HvRacB in barley (Bischoff et al., 2000; Schultheiss et al., 2003; Xiao et al., 2009; Chen et al., 2010b). Not surprisingly then, CaRop1, which also contains the conserved C-terminal canonical CaaL motif found in other type I Rops, was also localized to the nucleus, the cytoplasm, and plasma membrane. This similarity with other type 1 Rops suggests that CaRop1 is a type I Rop of C. annuum. Among the Rop/Rac family of proteins in plant species, differences with respect to structure, function, and expression have been described. The 11 Arabidopsis Rops are divided into four phylogenetic groups with distinct functions (Gu et al., 2004; Hou et al., 2011), and the seven members of the Rop family in rice differ in their expression patterns (Xiao et al., 2009).
In our study, OE of CaRop1 and DN-CaRop1 enhanced resistance of the transgenic tobacco plants to R. solanacearum inoculation, while OE of CA-CaRop1 actually enhanced susceptibility to this pathogen. The altered resistance of these transgenic plants was accompanied by transcriptional modification of NtHSR201, NtACS1, NtPR1b, and NtLOX1, which are genes that have been implicated previously in plant immunity (Czernic et al., 1996; Jin et al., 2002; Sohn et al., 2007; Gao et al., 2008; Borges et al., 2012; Dang et al., 2013; Nalam et al., 2015). These changes are consistent with our finding that immunity against R. solanacearum was altered by the ectopic expression of CaRop1 as well as by expression of DN and CA mutant CaRop1. Similarly, plants expressing a DN form of AtRop6 exhibit a gene expression profile associated with constitutive SA-mediated defense responses and enhanced pre-invasive defense responses to a host-adapted virulent powdery mildew fungus (Poraty-Gavra et al., 2013). In addition, ectopic expression of CA forms of HvRACB, HvRAC3, and HvRop6 in barley enhance susceptibility of transgenic plants to powdery mildew (Schultheiss et al., 2003, 2005). Since these Rops are grouped together in a phylogenetic tree (Figure 2), conservation of their function may also correlate with their structurally similarity. Furthermore, our results are also consistent with findings that RanGAP2, a GAP that mediates the transformation of GTP-bound Rop to GDP-bound Rop, acts as positive regulator of resistance of potato to virus X via interaction with the resistance protein Rx (Sacco et al., 2007; Tameling and Baulcombe, 2007; Rairdan et al., 2008; Tameling et al., 2010; Sturbois et al., 2012). In contrast, OE of a CA-OsRac1 induces cell death, and OE of a DN-OsRac1 blocks hydrogen peroxide production and cell death in transgenic lesion-mimic mutants (Fujiwara et al., 2006). Together, these results suggest that members of the Rop family in different plant species play important roles in plant immunity via different modes of action.
We also found that OE of CA-CaRop1 enhances the population size of aphid nymphs on leaves of inoculated transgenic tobacco, whereas OE of DN-CaRop1 or CaRop1 decreases the population size. Transcriptional expression of NtPI-I, NtPI-II, and NtTPI, which encode tobacco protease inhibitors, were previously found to be upregulated by methyl jasmonate (MeJA) vapors, wounding, and attack by Manduca sexta larvae in tobacco plants and are probably involved in insect resistance (Anderson et al., 1997; Rocha-Granados Cdel et al., 2005; Maheswaran et al., 2007; Srinivasan et al., 2009). Expression of these genes was triggered in transgenic tobacco by OE of DN-CaROP1; although, these levels were decreased or remained unchanged by OE of CA-CaRop1, suggesting that CaRop1 acts as a regulator in the response of plants to aphid attack. Partial overlap between defense signaling pathways against herbivores and microbial pathogens has been reported previously (De Vos et al., 2005, 2007). For example, responses to aphid attack at the proteome level are broadly similar to basal non-specific defense and stress responses in wheat (Ferry et al., 2011). Defense-associated pathogenesis-related responses and calcium-dependent signaling in wheat are also induced by the Russian wheat aphid 2 (Botha et al., 2010). Treating seeds with activators of plant defense, such as JA or β-aminobutyric acid, generate long-lasting priming of resistance to aphids as well as to the necrotrophic fungal pathogen Botrytis cinerea (Worrall et al., 2012). Additionally, signaling cascades, such as those related to alpha-DIOXYGENASE1, Hsp90, Sgt1, PAD4, SlSERK1, and MAPK, and to phytohormones, such as SA, JA, and ET, have also been implicated in plant responses to both pathogen and aphid attack (Zhou et al., 1998; Hamberg et al., 2003; Mantelin et al., 2011).
Together, these results suggest extensive crosstalk between the plant response to a pathogen and herbivore attack, possibly implicating the JA and ET pathways, which are shared in plant responses to pathogens and herbivores (Glazebrook, 2005; Zavala and Baldwin, 2006; Chehab et al., 2008; Howe and Jander, 2008; Simons et al., 2008; Demkura et al., 2010). In the present study of the functional characterization of CaRop1 using transgenic plants, CA-CaRop1, DN-CaRop1, and CaRop1 all significantly modulated the response of pepper to R. solanacearum and aphid attack, accompanied with transcriptional modulation of JA-dependent PR1b and ET-dependent ACS1. On the other hand, however, transcription of SA-dependent PR2 and NPR1 were not altered by CaRop1and its CA or DN mutant. These findings strongly suggest that the modulation of defense against R. solanacearum attack by CaRop1 is at least partially associated with JA and ET-dependent signaling machinery.
Overexpression of CaRop1 also decreased susceptibility of transgenic tobacco plants to R. solanacearum and aphid attack in a manner similar to that of DN-CaRop1. Upon inoculation with R. solanacearum, the mRNA levels of NtHSR201, NtPR1b, and NtACS1 in plants of the DN-CaRop1 lines were significantly higher than those in the K326 plants at 36 hpi. Similarly, upon aphid attack, NtPI-I, NtPI-II, and NtTPI were significantly enhanced by OE of CaRop1. We suggest that some components, perhaps GAPs, are induced by pathogen infection, and this induction, in turn, regulates the conversion of GTP-bound CaRop1 to GDP-bound CaRop1. Another important component that may be involved in CaRop1-mediated crosstalk between responses in pepper to R. solanacearum and to aphid attack is NPK1, a MAPKKK that was previously found to interfere with the function of the disease-resistance genes N, Bs2, and Rx (Jin et al., 2002). Our data show that NPK1 is transcriptionally induced by OE of DN-CaRop1 and by CaRop1 but that this transcription is decreased by OE of CA-CaRop1, suggesting that NPK1 acts as a downstream component in the CaRop1-mediated defense signaling pathway.
Collectively, our data demonstrate that the ectopic expression of CaRop1 modulates the response of tobacco plants to R. solanacearum and to aphid attack, possibly via the JA and ET signaling pathways. Furthermore, we speculate that CaRop1 may play a role in the crosstalk between the Solanaceae response to pathogen and insect attack. Further confirmation of the role of CaRop1 in pepper plants and identification of the effectors of CaRop1 or its direct downstream signaling components will likely provide new insight into the molecular mechanisms underlying CaRop1-mediated synergistic resistance to R. solanacearum and aphid attack.
Author Contributions
AQ and SH conceived and designed research. AQ and ZL conducted primary experiments. JL, YC, and DG performed replication genotyping with the transgenic plants. AQ and ZL contributed comments during manuscript preparation. DG contributed new reagents or analytical tools. SH and AQ performed expression analysis and editing of the manuscript. All authors read and approved the manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The Gateway destination vectors used in this study were kindly provided by Mark D. Curtis Sprenger-Haussels and B. Weisshaar. This work was supported by grants from the National Natural Science Foundation of China (31401890, 31572136, and 31501767).
Footnotes
References
Agrawal, G. K., Iwahashi, H., and Rakwal, R. (2003). Small GTPase ‘Rop’: molecular switch for plant defense responses. FEBS Lett. 546, 173–180. doi: 10.1016/S0014-5793(03)00646-X
Anderson, M. A., Van Heeswijck, R., West, J., Bateman, K., Lee, M., Christeller, J. T., et al. (1997). Proteinase inhibitors from Nicotiana alata enhance plant resistance to insect pests. J. Insect Physiol. 43, 833–842. doi: 10.1016/S0022-1910(97)00026-7
Atamian, H. S., Eulgem, T., and Kaloshian, I. (2012). SlWRKY70 is required for Mi-1-mediated resistance to aphids and nematodes in tomato. Planta 235, 299–309. doi: 10.1007/s00425-011-1509-6
Bischoff, F., Vahlkamp, L., Molendijk, A., and Palme, K. (2000). Localization of AtROP4 and AtROP6 and interaction with the guanine nucleotide dissociation inhibitor AtRhoGDI1 from Arabidopsis. Plant Mol. Biol. 42, 515–530. doi: 10.1023/A:1006341210147
Bodenhausen, N., and Reymond, P. (2007). Signaling pathways controlling induced resistance to insect herbivores in Arabidopsis. Mol. Plant Microbe Interact. 20, 1406–1420. doi: 10.1094/MPMI-20-11-1406
Borges, A., Melotto, M., Tsai, S. M., and Gomes Caldas, D. G. (2012). Changes in spatial and temporal gene expression during incompatible interaction between common bean and anthracnose pathogen. J. Plant Physiol. 169, 1216–1220. doi: 10.1016/j.jplph.2012.04.003
Bostock, R. M. (2005). Signal crosstalk and induced resistance: straddling the line between cost and benefit. Annu. Rev. Phytopathol. 43, 545–580. doi: 10.1146/annurev.phyto.41.052002.095505
Botha, A. M., Swanevelder, Z. H., and Lapitan, N. L. (2010). Transcript profiling of wheat genes expressed during feeding by two different biotypes of Diuraphis noxia. Environ. Entomol. 39, 1206–1231. doi: 10.1603/EN09248
Cai, H., Yang, S., Yan, Y., Xiao, Z., Cheng, J., Wu, J., et al. (2015). CaWRKY6 transcriptionally activates CaWRKY40, regulates Ralstonia solanacearum resistance, and confers high-temperature and high-humidity tolerance in pepper. J. Exp. Bot. 66, 3163–3174. doi: 10.1093/jxb/erv125
Chehab, E. W., Kaspi, R., Savchenko, T., Rowe, H., Negre-Zakharov, F., Kliebenstein, D., et al. (2008). Distinct roles of jasmonates and aldehydes in plant-defense responses. PLoS ONE 3:e1904. doi: 10.1371/journal.pone.0001904
Chen, L., Hamada, S., Fujiwara, M., Zhu, T., Thao, N. P., Wong, H. L., et al. (2010a). The Hop/Sti1-Hsp90 chaperone complex facilitates the maturation and transport of a PAMP receptor in rice innate immunity. Cell Host Microbe 7, 185–196. doi: 10.1016/j.chom.2010.02.008
Chen, L., Shiotani, K., Togashi, T., Miki, D., Aoyama, M., Wong, H. L., et al. (2010b). Analysis of the Rac/Rop small GTPase family in rice: expression, subcellular localization and role in disease resistance. Plant Cell Physiol. 51, 585–595. doi: 10.1093/pcp/pcq024
Cheng, X., Zhu, L., and He, G. (2013). Towards understanding of molecular interactions between rice and the brown planthopper. Mol. Plant. 6, 621–634. doi: 10.1093/mp/sst030
Czernic, P., Huang, H. C., and Marco, Y. (1996). Characterization of hsr201 and hsr515, two tobacco genes preferentially expressed during the hypersensitive reaction provoked by phytopathogenic bacteria. Plant Mol. Biol. 31, 255–265. doi: 10.1007/BF00021788
Dang, F., Wang, Y., She, J., Lei, Y., Liu, Z., Eulgem, T., et al. (2014). Overexpression of CaWRKY27, a subgroup IIe WRKY transcription factor of Capsicum annuum, positively regulates tobacco resistance to Ralstonia solanacearum infection. Physiol. Plant. 150, 397–411. doi: 10.1111/ppl.12093
Dang, F. F., Wang, Y. N., Yu, L., Eulgem, T., Lai, Y., Liu, Z. Q., et al. (2013). CaWRKY40, a WRKY protein of pepper, plays an important role in the regulation of tolerance to heat stress and resistance to Ralstonia solanacearum infection. Plant Cell Environ. 36, 757–774. doi: 10.1111/pce.12011
De Vos, M., Van Oosten, V. R., Jander, G., Dicke, M., and Pieterse, C. M. (2007). Plants under attack: multiple interactions with insects and microbes. Plant Signal. Behav. 2, 527–529. doi: 10.4161/psb.2.6.4663
De Vos, M., Van Oosten, V. R., Van Poecke, R. M., Van Pelt, J. A., Pozo, M. J., Mueller, M. J., et al. (2005). Signal signature and transcriptome changes of Arabidopsis during pathogen and insect attack. Mol. Plant Microbe Interact. 18, 923–937. doi: 10.1094/MPMI-18-0923
Demkura, P. V., Abdala, G., Baldwin, I. T., and Ballare, C. L. (2010). Jasmonate-dependent and -independent pathways mediate specific effects of solar ultraviolet B radiation on leaf phenolics and antiherbivore defense. Plant Physiol. 152, 1084–1095. doi: 10.1104/pp.109.148999
Dombrecht, B., Xue, G. P., Sprague, S. J., Kirkegaard, J. A., Ross, J. J., Reid, J. B., et al. (2007). MYC2 differentially modulates diverse jasmonate-dependent functions in Arabidopsis. Plant Cell 19, 2225–2245. doi: 10.1105/tpc.106.048017
Duan, Q., Kita, D., Li, C., Cheung, A. Y., and Wu, H. M. (2010). FERONIA receptor-like kinase regulates RHO GTPase signaling of root hair development. Proc. Natl. Acad. Sci. U.S.A. 107, 17821–17826. doi: 10.1073/pnas.1005366107
Eklund, D. M., Svensson, E. M., and Kost, B. (2010). Physcomitrella patens: a model to investigate the role of RAC/ROP GTPase signalling in tip growth. J. Exp. Bot. 61, 1917–1937. doi: 10.1093/jxb/erq080
Ferry, N., Stavroulakis, S., Guan, W., Davison, G. M., Bell, H. A., Weaver, R. J., et al. (2011). Molecular interactions between wheat and cereal aphid (Sitobion avenae): analysis of changes to the wheat proteome. Proteomics 11, 1985–2002. doi: 10.1002/pmic.200900801
Foucart, C., Jauneau, A., Gion, J. M., Amelot, N., Martinez, Y., Panegos, P., et al. (2009). Overexpression of EgROP1, a Eucalyptus vascular-expressed Rac-like small GTPase, affects secondary xylem formation in Arabidopsis thaliana. New Phytol. 183, 1014–1029. doi: 10.1111/j.1469-8137.2009.02910.x
Fu, Y., Li, H., and Yang, Z. (2002). The ROP2 GTPase controls the formation of cortical fine F-actin and the early phase of directional cell expansion during Arabidopsis organogenesis. Plant Cell 14, 777–794. doi: 10.1105/tpc.001537
Fujiwara, M., Umemura, K., Kawasaki, T., and Shimamoto, K. (2006). Proteomics of Rac GTPase signaling reveals its predominant role in elicitor-induced defense response of cultured rice cells. Plant Physiol. 140, 734–745. doi: 10.1104/pp.105.068395
Gao, X., Starr, J., Gobel, C., Engelberth, J., Feussner, I., Tumlinson, J., et al. (2008). Maize 9-lipoxygenase ZmLOX3 controls development, root-specific expression of defense genes, and resistance to root-knot nematodes. Mol. Plant Microbe Interact. 21, 98–109. doi: 10.1094/MPMI-21-1-0098
Glazebrook, J. (2005). Contrasting mechanisms of defense against biotrophic and necrotrophic pathogens. Annu. Rev. Phytopathol. 43, 205–227. doi: 10.1146/annurev.phyto.43.040204.135923
Gu, Y., Wang, Z., and Yang, Z. (2004). ROP/RAC GTPase: an old new master regulator for plant signaling. Curr. Opin. Plant Biol. 7, 527–536. doi: 10.1016/j.pbi.2004.07.006
Halitschke, R., and Baldwin, I. T. (2003). Antisense LOX expression increases herbivore performance by decreasing defense responses and inhibiting growth-related transcriptional reorganization in Nicotiana attenuata. Plant J. 36, 794–807. doi: 10.1046/j.1365-313X.2003.01921.x
Hamberg, M., Sanz, A., Rodriguez, M. J., Calvo, A. P., and Castresana, C. (2003). Activation of the fatty acid alpha-dioxygenase pathway during bacterial infection of tobacco leaves. Formation of oxylipins protecting against cell death. J. Biol. Chem. 278, 51796–51805.
Hou, S., Yang, Y., Wu, D., and Zhang, C. (2011). Plant immunity: evolutionary insights from PBS1, (Pto), and RIN4. Plant Signal. Behav. 6, 794–799.
Howe, G. A., and Jander, G. (2008). Plant immunity to insect herbivores. Annu. Rev. Plant Biol. 59, 41–66. doi: 10.1146/annurev.arplant.59.032607.092825
Huesmann, C., Hoefle, C., and Huckelhoven, R. (2011). ROPGAPs of Arabidopsis limit susceptibility to powdery mildew. Plant Signal. Behav. 6, 1691–1694. doi: 10.4161/psb.6.11.17943
Huesmann, C., Reiner, T., Hoefle, C., Preuss, J., Jurca, M. E., Domoki, M., et al. (2012). Barley ROP binding kinase1 is involved in microtubule organization and in basal penetration resistance to the barley powdery mildew fungus. Plant Physiol. 159, 311–320. doi: 10.1104/pp.111.191940
Humphries, J. A., Vejlupkova, Z., Luo, A., Meeley, R. B., Sylvester, A. W., Fowler, J. E., et al. (2011). ROP GTPases act with the receptor-like protein PAN1 to polarize asymmetric cell division in maize. Plant Cell 23, 2273–2284. doi: 10.1105/tpc.111.085597
Jin, H., Axtell, M. J., Dahlbeck, D., Ekwenna, O., Zhang, S., Staskawicz, B., et al. (2002). NPK1, an MEKK1-like mitogen-activated protein kinase kinase kinase, regulates innate immunity and development in plants. Dev. Cell 3, 291–297. doi: 10.1016/S1534-5807(02)00205-8
Jones, J. D., and Dangl, J. L. (2006). The plant immune system. Nature 444, 323–329. doi: 10.1038/nature05286
Kawasaki, T., Koita, H., Nakatsubo, T., Hasegawa, K., Wakabayashi, K., Takahashi, H., et al. (2006). Cinnamoyl-CoA reductase, a key enzyme in lignin biosynthesis, is an effector of small GTPase Rac in defense signaling in rice. Proc. Natl. Acad. Sci. U.S.A. 103, 230–235. doi: 10.1073/pnas.0509875103
Kazan, K., and Manners, J. M. (2013). MYC2: the master in action. Mol. Plant 6, 686–703. doi: 10.1093/mp/sss128
Kim, S. H., Oikawa, T., Kyozuka, J., Wong, H. L., Umemura, K., Kishi-Kaboshi, M., et al. (2012). The bHLH Rac Immunity1 (RAI1) is activated by OsRac1 via OsMAPK3 and OsMAPK6 in rice immunity. Plant Cell Physiol. 53, 740–754. doi: 10.1093/pcp/pcs033
Kost, B. (2008). Spatial control of Rho (Rac-Rop) signaling in tip-growing plant cells. Trends Cell Biol. 18, 119–127. doi: 10.1016/j.tcb.2008.01.003
Kutyniok, M., and Muller, C. (2012). Crosstalk between above- and belowground herbivores is mediated by minute metabolic responses of the host Arabidopsis thaliana. J. Exp. Bot. 63, 6199–6210. doi: 10.1093/jxb/ers274
Lee, Y. J., and Yang, Z. (2008). Tip growth: signaling in the apical dome. Curr. Opin. Plant Biol 11, 662–671. doi: 10.1016/j.pbi.2008.10.002
Li, Z., Kang, J., Sui, N., and Liu, D. (2012). ROP11 GTPase is a negative regulator of multiple ABA responses in Arabidopsis. J. Integr. Plant Biol. 54, 169–179. doi: 10.1111/j.1744-7909.2012.01100.x
Li, Z., and Liu, D. (2012). ROPGEF1 and ROPGEF4 are functional regulators of ROP11 GTPase in ABA-mediated stomatal closure in Arabidopsis. FEBS Lett. 586, 1253–1258. doi: 10.1016/j.febslet.2012.03.040
Livak, K. J., and Schmittgen, T. D. (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25, 402–408. doi: 10.1006/meth.2001.1262
Luo, M., Gu, S. H., Zhao, S. H., Zhang, F., and Wu, N. H. (2006). Rice GTPase OsRacB: potential accessory factor in plant salt-stress signaling. Acta Biochim. Biophys. Sin. (Shanghai) 38, 393–402. doi: 10.1111/j.1745-7270.2006.00172.x
Maffei, M. E., Mithofer, A., and Boland, W. (2007). Before gene expression: early events in plant-insect interaction. Trends Plant Sci. 12, 310–316. doi: 10.1016/j.tplants.2007.06.001
Maheswaran, G., Pridmore, L., Franz, P., and Anderson, M. A. (2007). A proteinase inhibitor from Nicotiana alata inhibits the normal development of light-brown apple moth, Epiphyas postvittana in transgenic apple plants. Plant Cell Rep. 26, 773–782. doi: 10.1007/s00299-006-0281-8
Mantelin, S., Peng, H. C., Li, B., Atamian, H. S., Takken, F. L., and Kaloshian, I. (2011). The receptor-like kinase SlSERK1 is required for Mi-1-mediated resistance to potato aphids in tomato. Plant J. 67, 459–471. doi: 10.1111/j.1365-313X.2011.04609.x
Miki, D., Itoh, R., and Shimamoto, K. (2005). RNA silencing of single and multiple members in a gene family of rice. Plant Physiol. 138, 1903–1913. doi: 10.1104/pp.105.063933
Moeder, W., Yoshioka, K., and Klessig, D. F. (2005). Involvement of the small GTPase Rac in the defense responses of tobacco to pathogens. Mol. Plant Microbe Interact. 18, 116–124. doi: 10.1094/MPMI-18-0116
Mucha, E., Fricke, I., Schaefer, A., Wittinghofer, A., and Berken, A. (2011). Rho proteins of plants–functional cycle and regulation of cytoskeletal dynamics. Eur. J. Cell Biol. 90, 934–943. doi: 10.1016/j.ejcb.2010.11.009
Munroe, D. J., Loebbert, R., Bric, E., Whitton, T., Prawitt, D., Vu, D., et al. (1995). Systematic screening of an arrayed cDNA library by PCR. Proc. Natl. Acad. Sci. U.S.A. 92, 2209–2213. doi: 10.1073/pnas.92.6.2209
Mur, L. A., Kenton, P., Atzorn, R., Miersch, O., and Wasternack, C. (2006). The outcomes of concentration-specific interactions between salicylate and jasmonate signaling include synergy, antagonism, and oxidative stress leading to cell death. Plant Physiol. 140, 249–262. doi: 10.1104/pp.105.072348
Murashige, T., and Skoog, F. (1962). A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant. 15, 473–497. doi: 10.1111/j.1399-3054.1962.tb08052.x
Nalam, V. J., Alam, S., Keereetaweep, J., Venables, B., Burdan, D., Lee, H., et al. (2015). Facilitation of Fusarium graminearum Infection by 9-Lipoxygenases in Arabidopsis and Wheat. Mol. Plant Microbe Interact. 28, 1142–1152. doi: 10.1094/MPMI-04-15-0096-R
Nascimento, N. C., and Fett-Neto, A. G. (2010). Plant secondary metabolism and challenges in modifying its operation: an overview. Methods Mol. Biol. 643, 1–13. doi: 10.1007/978-1-60761-723-5_1
Ono, E., Wong, H. L., Kawasaki, T., Hasegawa, M., Kodama, O., and Shimamoto, K. (2001). Essential role of the small GTPase Rac in disease resistance of rice. Proc. Natl. Acad. Sci. U.S.A. 98, 759–764. doi: 10.1073/pnas.98.2.759
Pathuri, I. P., Eichmann, R., and Huckelhoven, R. (2009). Plant small monomeric G-proteins (RAC/ROPs) of barley are common elements of susceptibility to fungal leaf pathogens, cell expansion and stomata development. Plant Signal. Behav. 4, 109–110. doi: 10.4161/psb.4.2.7477
Pereira-Leal, J. B., and Seabra, M. C. (2001). Evolution of the Rab family of small GTP-binding proteins. J. Mol. Biol. 313, 889–901. doi: 10.1006/jmbi.2001.5072
Poraty-Gavra, L., Zimmermann, P., Haigis, S., Bednarek, P., Hazak, O., Stelmakh, O. R., et al. (2013). The Arabidopsis Rho of plants GTPase AtROP6 functions in developmental and pathogen response pathways. Plant Physiol. 161, 1172–1188. doi: 10.1104/pp.112.213165
Pozo, M. J., Van Der Ent, S., Van Loon, L. C., and Pieterse, C. M. (2008). Transcription factor MYC2 is involved in priming for enhanced defense during rhizobacteria-induced systemic resistance in Arabidopsis thaliana. New Phytol. 180, 511–523. doi: 10.1111/j.1469-8137.2008.02578.x
Rairdan, G. J., Collier, S. M., Sacco, M. A., Baldwin, T. T., Boettrich, T., and Moffett, P. (2008). The coiled-coil and nucleotide binding domains of the Potato Rx disease resistance protein function in pathogen recognition and signaling. Plant Cell 20, 739–751. doi: 10.1105/tpc.107.056036
Rocha-Granados Cdel, M., Sanchez-Hernandez, C., Martinez-Gallardo, N. A., Ochoa-Alejo, N., and Delano-Frier, J. P. (2005). The expression of the hydroxyproline-rich glycopeptide systemin precursor A in response to (a)biotic stress and elicitors is indicative of its role in the regulation of the wound response in tobacco (Nicotiana tabacum L.). Planta 222, 794–810. doi: 10.1007/s00425-005-0024-z
Sacco, M. A., Mansoor, S., and Moffett, P. (2007). A RanGAP protein physically interacts with the NB-LRR protein Rx, and is required for Rx-mediated viral resistance. Plant J. 52, 82–93. doi: 10.1111/j.1365-313X.2007.03213.x
Schallhart, N., Tusch, M. J., Wallinger, C., Staudacher, K., and Traugott, M. (2012). Effects of plant identity and diversity on the dietary choice of a soil-living insect herbivore. Ecology 93, 2650–2657. doi: 10.1890/11-2067.1
Schultheiss, H., Dechert, C., Kogel, K. H., and Huckelhoven, R. (2002). A small GTP-binding host protein is required for entry of powdery mildew fungus into epidermal cells of barley. Plant Physiol. 128, 1447–1454. doi: 10.1104/pp.010805
Schultheiss, H., Dechert, C., Kogel, K. H., and Huckelhoven, R. (2003). Functional analysis of barley RAC/ROP G-protein family members in susceptibility to the powdery mildew fungus. Plant J. 36, 589–601. doi: 10.1046/j.1365-313X.2003.01905.x
Schultheiss, H., Hensel, G., Imani, J., Broeders, S., Sonnewald, U., Kogel, K. H., et al. (2005). Ectopic expression of constitutively activated RACB in barley enhances susceptibility to powdery mildew and abiotic stress. Plant Physiol. 139, 353–362. doi: 10.1104/pp.105.066613
Shang, W., Meng, C., Zheng, K., Ding, M., Zhang, Z., Hong, J., et al. (2012). [Expression and subcellular location of NSm protein of Tomato spotted wilt virus in plant and insect cells]. Wei Sheng Wu Xue Bao 52, 962–968.
Simons, L., Bultman, T. L., and Sullivan, T. J. (2008). Effects of methyl jasmonate and an endophytic fungus on plant resistance to insect herbivores. J. Chem. Ecol. 34, 1511–1517. doi: 10.1007/s10886-008-9551-y
Singh, M. K., Ren, F., Giesemann, T., Dal Bosco, C., Pasternak, T. P., Blein, T., et al. (2013). Modification of plant Rac/Rop GTPase signalling using bacterial toxin transgenes. Plant J. 73, 314–324. doi: 10.1111/tpj.12040
Sohn, S. I., Kim, Y. H., Kim, B. R., Lee, S. Y., Lim, C. K., Hur, J. H., et al. (2007). Transgenic tobacco expressing the hrpN(EP) gene from Erwinia pyrifoliae triggers defense responses against botrytis cinerea. Mol. Cells 24, 232–239.
Spoel, S. H., Koornneef, A., Claessens, S. M., Korzelius, J. P., Van Pelt, J. A., Mueller, M. J., et al. (2003). NPR1 modulates cross-talk between salicylate- and jasmonate-dependent defense pathways through a novel function in the cytosol. Plant Cell 15, 760–770. doi: 10.1105/tpc.009159
Srinivasan, T., Kumar, K. R., and Kirti, P. B. (2009). Constitutive expression of a trypsin protease inhibitor confers multiple stress tolerance in transgenic tobacco. Plant Cell Physiol. 50, 541–553. doi: 10.1093/pcp/pcp014
Steffens, B., and Sauter, M. (2010). G proteins as regulators in ethylene-mediated hypoxia signaling. Plant Signal. Behav. 5, 375–378. doi: 10.4161/psb.5.4.10910
Sturbois, B., Dubrana-Ourabah, M. P., Gombert, J., Lasseur, B., Macquet, A., Faure, C., et al. (2012). Identification and characterization of tomato mutants affected in the Rx-mediated resistance to PVX isolates. Mol. Plant Microbe Interact. 25, 341–354. doi: 10.1094/MPMI-07-11-0181
Takahashi, Y., Uehara, Y., Berberich, T., Ito, A., Saitoh, H., Miyazaki, A., et al. (2004). A subset of hypersensitive response marker genes, including HSR203J, is the downstream target of a spermine signal transduction pathway in tobacco. Plant J. 40, 586–595. doi: 10.1111/j.1365-313X.2004.02234.x
Tameling, W. I., and Baulcombe, D. C. (2007). Physical association of the NB-LRR resistance protein Rx with a Ran GTPase-activating protein is required for extreme resistance to Potato virus X. Plant Cell 19, 1682–1694. doi: 10.1105/tpc.107.050880
Tameling, W. I., Nooijen, C., Ludwig, N., Boter, M., Slootweg, E., Goverse, A., et al. (2010). RanGAP2 mediates nucleocytoplasmic partitioning of the NB-LRR immune receptor Rx in the Solanaceae, thereby dictating Rx function. Plant Cell 22, 4176–4194. doi: 10.1105/tpc.110.077461
Tao, L. Z., Cheung, A. Y., and Wu, H. M. (2002). Plant Rac-like GTPases are activated by auxin and mediate auxin-responsive gene expression. Plant Cell 14, 2745–2760. doi: 10.1105/tpc.006320
Thao, N. P., Chen, L., Nakashima, A., Hara, S., Umemura, K., Takahashi, A., et al. (2007). RAR1 and HSP90 form a complex with Rac/Rop GTPase and function in innate-immune responses in rice. Plant Cell 19, 4035–4045. doi: 10.1105/tpc.107.055517
Wang, Y., Dang, F., Liu, Z., Wang, X., Eulgem, T., Lai, Y., et al. (2013). CaWRKY58, encoding a group I WRKY transcription factor of Capsicum annuum, negatively regulates resistance to Ralstonia solanacearum infection. Mol. Plant Pathol. 14, 131–144. doi: 10.1111/j.1364-3703.2012.00836.x
Worrall, D., Holroyd, G. H., Moore, J. P., Glowacz, M., Croft, P., Taylor, J. E., et al. (2012). Treating seeds with activators of plant defence generates long-lasting priming of resistance to pests and pathogens. New Phytol. 193, 770–778. doi: 10.1111/j.1469-8137.2011.03987.x
Wu, H. M., Hazak, O., Cheung, A. Y., and Yalovsky, S. (2011). RAC/ROP GTPases and auxin signaling. Plant Cell 23, 1208–1218. doi: 10.1105/tpc.111.083907
Xiao, B. Z., Chen, X., Xiang, C. B., Tang, N., Zhang, Q. F., and Xiong, L. Z. (2009). Evaluation of seven function-known candidate genes for their effects on improving drought resistance of transgenic rice under field conditions. Mol. Plant. 2, 73–83. doi: 10.1093/mp/ssn068
Xie, W., Meng, Q. S., Wu, Q. J., Wang, S. L., Yang, X., Yang, N. N., et al. (2012). Pyrosequencing the Bemisia tabaci transcriptome reveals a highly diverse bacterial community and a robust system for insecticide resistance. PLoS ONE 7:e35181. doi: 10.1371/journal.pone.0035181
Yang, Z. (2008). Cell polarity signaling in Arabidopsis. Annu. Rev. Cell Dev. Biol. 24, 551–575. doi: 10.1146/annurev.cellbio.23.090506.123233
Zavala, J. A., and Baldwin, I. T. (2006). Jasmonic acid signalling and herbivore resistance traits constrain regrowth after herbivore attack in Nicotiana attenuata. Plant Cell Environ. 29, 1751–1760. doi: 10.1111/j.1365-3040.2006.01551.x
Zheng, Z. L., Nafisi, M., Tam, A., Li, H., Crowell, D. N., Chary, S. N., et al. (2002). Plasma membrane-associated ROP10 small GTPase is a specific negative regulator of abscisic acid responses in Arabidopsis. Plant Cell 14, 2787–2797. doi: 10.1105/tpc.005611
Zheng, Z. L., and Yang, Z. (2000). The Rop GTPase: an emerging signaling switch in plants. Plant Mol. Biol. 44, 1–9. doi: 10.1023/A:1006402628948
Keywords: CaRop1, Capsicum annuum, Ralstonia solanacearum, aphid, JA/ET-mediated signaling
Citation: Qiu A, Liu Z, Li J, Chen Y, Guan D and He S (2016) The Ectopic Expression of CaRop1 Modulates the Response of Tobacco Plants to Ralstonia solanacearum and Aphids. Front. Plant Sci. 7:1177. doi: 10.3389/fpls.2016.01177
Received: 27 April 2016; Accepted: 21 July 2016;
Published: 08 August 2016.
Edited by:
Agnieszka Ludwików, Adam Mickiewicz University in Poznań, PolandReviewed by:
Taras P. Pasternak, University of Freiburg, GermanyUpinder S. Gill, Samuel Roberts Noble Foundation, USA
Copyright © 2016 Qiu, Liu, Li, Chen, Guan and He. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Shuilin He, c2hsaGUyMDEzMDRAYWxpeXVuLmNvbQ==
†These authors have contributed equally to this work.
 Ailian Qiu1,2†
Ailian Qiu1,2† Zhiqin Liu
Zhiqin Liu Shuilin He
Shuilin He