Abstract
Plants are sessile and as such their reactions to environmental challenges differ from those of mobile organisms. Many adaptions involve growth responses and hence, growth regulation is one of the most crucial biological processes for plant survival and fitness. The plant-specific TEOSINTE BRANCHED 1, CYCLOIDEA, PCF1 (TCP) transcription factor family is involved in plant development from cradle to grave, i.e., from seed germination throughout vegetative development until the formation of flowers and fruits. TCP transcription factors have an evolutionary conserved role as regulators in a variety of plant species, including orchids, tomatoes, peas, poplar, cotton, rice and the model plant Arabidopsis. Early TCP research focused on the regulatory functions of TCPs in the development of diverse organs via the cell cycle. Later research uncovered that TCP transcription factors are not static developmental regulators but crucial growth regulators that translate diverse endogenous and environmental signals into growth responses best fitted to ensure plant fitness and health. I will recapitulate the research on TCPs in this review focusing on two topics: the discovery of TCPs and the elucidation of their evolutionarily conserved roles across the plant kingdom, and the variety of signals, both endogenous (circadian clock, plant hormones) and environmental (pathogens, light, nutrients), TCPs respond to in the course of their developmental roles.
Discovery of TCPs – of Peloria and Other Mutants
Developmental plasticity is important for plant survival because plants are sessile organisms that have to adapt to suboptimal environmental conditions. It is crucial that these developmental adaptions are balanced, which means that multiple environmental stimuli have to be perceived and weighed against each other before a plant adjusts its growth. Hence, a plethora of regulatory proteins is involved in governing developmental responses to the environment. One family of transcription factors that is involved in multiple developmental processes are the TEOSINTE BRANCHED 1, CYCLOIDEA, PCF1 (TCP) proteins.
The common toadflax (Linaria vulgaris) is a perennial plant with bilateral, zygomorphic flowers that is native to Europe and large parts of northern Asia. When Carl Linnaeus was presented with a common toadflax that did not exhibit zygomorphic but radially symmetric flowers, he called it peloria from the Old Greek πέλωρ (pelór), which means monster. Linnaeus speculated that this monster was a hybrid between the common toadflax and a thitherto unknown plant and he was surprised to see that this hybrid was nevertheless able to propagate through seeds (Linnaeus and Rudberg, 1744). Whereas his hybrid hypothesis proved to be wrong, he used this case as evidence against immutability, the belief that all species are created at the beginning of the world and are unchanging (Smith, 1821). Peloria is a natural variation that occurs in toadflax, snapdragons (Antirrhinum majus) (Darwin, 1868) and in foxgloves (Digitalis purpurea) (Keeble et al., 1910), amongst other species.
About 250 years later, Luo et al. (1996) isolated the CYCLOIDEA (CYC) gene which is only expressed in the dorsal parts of the snapdragon flower and which is responsible for the regulation of zygomorphic flowers. A double mutant of CYC and its close homolog DICHOTOMA leads to radially symmetric snapdragon flowers (Luo et al., 1996). Cubas et al. (1999b) found that a homolog of the CYC gene was also responsible for floral symmetry in the common toadflax. Here, they could show that the CYC gene in peloric mutants was extensively methylated and silenced (Cubas et al., 1999b). At about the same time, Doebley et al. (1995) analyzed two quantitative trait loci that control morphological differences between domesticated maize (Zea mays) and its wild progenitor teosinte. They found the teosinte branched 1 (tb1) mutation, which leads to increased side shoot outgrowth, and showed that the difference between the maize and the teosinte variant of TB1 lies mainly in the regulatory regions of the gene, i.e., whereas the function remains the same, the expression pattern is different between domesticated maize and teosinte (Wang et al., 1999).
Kosugi et al. (1995) found that two promoter motifs that are important for the transcriptional regulation of the proliferating cell nuclear antigen (PCNA) gene in rice (Oryza sativa) were bound by two transcription factors that were designated PCF1 and PCF2 (Kosugi and Ohashi, 1997). Finally, Cubas et al. (1999a) determined that the above described proteins TB1, CYC and PCF1 and PCF2 share a conserved non-canonical bHLH region, the eponymous TCP domain (Kosugi and Ohashi, 1997).
Form and Function of TCP Transcription Factors
Whereas, this review will mainly focus on the evolutionarily conserved roles of TCPs in the regulation of plant development and their interactions with endogenous and environmental signals, it is crucial to understand how they function. TCP transcription factors are divided into two classes, class I and class II TCPs. These classes differ in the composition of their respective NLSs, the length of the second helix in the bHLH domain, and the presence of an arginine-rich domain of unknown functionality outside the bHLH domain (Cubas et al., 1999a). This so-called R domain is not found in class I TCPs and was predicted to form a hydrophilic α-helix or a coiled-coil structure that mediates protein–protein interactions (Lupas et al., 1991; Cubas et al., 1999a).
The basic region of the TCP domain is essential for DNA binding. Replacement of a conserved glycine–proline pair in the basic region by two lysines completely abolished DNA binding activity of TCP4 in electrophoretic mobility shift studies (Aggarwal et al., 2010). Addition of the major groove binding dye methyl green reduced TCP4 binding to DNA, indicating that TCP4 binds to the major groove in double stranded DNA (Aggarwal et al., 2010).
In various experimental approaches, class I and class II TCP proteins have been shown to recognize GC-rich sequences in target gene promoters (Kosugi and Ohashi, 1997; Li et al., 2005; Viola et al., 2011; Danisman et al., 2012). The differences between class I and class II binding preferences are dependent on the presence of glycine or aspartic acid at positions 11 or 15, respectively (Viola et al., 2012). Interestingly, the class I and class II consensus binding site sequences are not mutually exclusive, indicating that at least a subset of potential target genes are targeted by both class I and class II TCP proteins. This led to speculations about a possible antagonistic relation between class I and class II TCPs, where these proteins compete for common target genes and inhibit or activate gene expression depending on which class dominates the target gene promoter (Li et al., 2005). So far, this was shown in one case only, where the Arabidopsis class I TCP transcription factor TCP20 binds to the same promoter as the class II TCP4 and regulates the target gene LIPOXYGENASE2(LOX2) in the opposite direction to TCP4 (Danisman et al., 2012). It is likely though that more cases of class I-class II TCP antagonisms will be discovered in the future, as the two classes are frequently discovered to be involved in the same biological processes.
Similar to many transcription factor families, TCPs require dimerization to bind to DNA, as addition of deoxycholate, an inhibitor of protein–protein interactions, to electrophoretic mobility shift assays leads to a reduction of TCP binding to target sequences (Trémousaygue et al., 2003). Dimerization between TCP transcription factors first has been described between PCF1 and PCF2 in rice, which form homo- and heterodimers (Kosugi and Ohashi, 1997). Whereas the homodimer of TCP20 for example does not bind to the promoter of the iron homeostasis regulator BHLH39 in yeast one-hybrid experiments, the TCP20 heterodimer with TCP8 or TCP21 does (Andriankaja et al., 2014). A systematic yeast two-hybrid approach between Arabidopsis TCPs found that many protein–protein interactions are possible between TCPs and that there is a preference to bind to TCPs of the own class, i.e., class I TCPs preferably interact with class I TCPs and class II TCPs preferably interact with class II TCPs (Danisman et al., 2013). Dimerization of TCPs are facilitated by IDR (Valsecchi et al., 2013). These are characterized by low compactness, low globularity and higher structural flexibility and are typically present extensively in eukaryotic transcription factors (Liu et al., 2006). The C-terminal IDR of TCP8 is needed for self-assembly of TCP8 in dimers and higher order complexes. These IDRs potentially facilitate the flexibility of TCPs in the choice of interacting partners and thus increases the number of potential functions TCP transcription factors can be involved in Thieulin-Pardo et al. (2015). TCPs not only interact with TCPs: protein–protein interactions with a plethora of other proteins has been described, including negative regulators of effector-triggered immunity (Kim et al., 2014), components of the circadian clock (Pruneda-Paz et al., 2009, 2014; Giraud et al., 2010), and others (Trémousaygue et al., 2003; Tao et al., 2013).
Evolutionary Conserved Roles of TCPs
The three eponymous TCP proteins were characterized as regulators of branching, floral symmetry, and the cell cycle (Doebley et al., 1995; Luo et al., 1996). Later, both CYC-like and the PCF-like TCPs were shown to be involved in leaf development (Kosugi and Ohashi, 1997; Palatnik et al., 2003). TCP research since then has focused on these three developmental processes, mainly identifying evolutionarily conserved processes in a wide array of plant species and the role of cell cycle regulation in the observed phenotypes. Recently it became clear however that TCPs are not limited to branching, floral symmetry and leaf development, and neither are they limited to cell cycle mediated regulation of growth. Both will be discussed further below.
TEOSINTE BRANCHED 1, CYCLOIDEA, PCF1 transcription factors belong to an evolutionary conserved family that first appears in fresh water algae of the Charophyta family (Navaud et al., 2007). In the bryophyte Physcomitrella patens, knockout of the TCP transcription factor PpTCP5 leads to increased numbers of sporangia that are attached to a single seta, reminiscent of branching phenotypes of tcp mutants in higher land plants (Ortiz-Ramírez et al., 2016). Hence, control of meristematic activity of axillary meristems with a subsequent effect on branching patterns seems to be an ancient role of TCP transcription factors (Ortiz-Ramírez et al., 2016). Consistent with this finding, branching phenotypes are apparent both in monocot and dicot plant species. Overexpression of the rice OsTB1, an ortholog of maize TB1, led to a strong decrease in tiller number. The number of axillary buds was not affected in these plants but their outgrowth was Takeda et al. (2003). This fits to the observation that it is not the formation of axillary meristems but the outgrowth of these that is affected by TCPs (Braun et al., 2012). This has been shown in peas (Braun et al., 2012), poplar (Muhr et al., 2016), Arabidopsis (Aguilar-Martínez et al., 2007; Poza-Carrión et al., 2007) and potato (Nicolas et al., 2015).
TCP effect on floral development was shown in a wide range of plant species, including Arabidopsis, Antirrhinum, annual candytuft (Iberis amara) (Busch and Zachgo, 2007; Busch et al., 2012), angiosperms like Aristolochia arborea and Saruma henryi (Horn et al., 2015), Gerbera species (Broholm et al., 2008), rice (Yuan et al., 2009), sunflowers (Fambrini et al., 2012), peas (Wang et al., 2008), ragworts (Kim et al., 2008), Morrow’s honeysuckle (Lonicera morrowii) (Howarth and Donoghue, 2006), Knautia macedonica (Berger et al., 2016), and orchids (De Paolo et al., 2015).
Phylogenetic analysis revealed that the CYCLOIDEA-like TCPs underwent two major duplication events that both predate the formation of core eudicots (Howarth and Donoghue, 2006). In Arabidopsis, all three CYC clades are represented by TCP12, TCP1 and TCP18, respectively (Howarth and Donoghue, 2006). Especially the CYC2 clade, represented by TCP1 in Arabidopsis, underwent multiple additional duplications and has been studied for its effect on floral symmetry, as it contains the original CYC gene of Antirrhinum (Howarth and Donoghue, 2006). An interesting side note is that the duplication of the CYCLOIDEA-like TCPs nearly coincides with the major duplication events of the homeotic MADS-box transcription factors APETALA3, AGAMOUS and SEPALLATA, all three of them important factors for the definition of organ identity in flowering plants (Howarth and Donoghue, 2006). This suggests that the genetic components that are important for the definition of floral organs diversified at a similar time as the components that are important for the growth regulation of these. TCP transcription factors have been identified as targets of Arabidopsis APETALA1 and SEPALLATA3 (Kaufmann et al., 2009, 2010), highlighting a possible link between organ identity formation and growth regulation between MADS-box transcription factors and TCPs (Dornelas et al., 2011).
In Antirrhinum, CYC regulates symmetry via the Myb-domain transcription factor RADIALIS (Corley et al., 2005). Overexpression of CYC in Arabidopsis leads to larger petals containing enlarged petal cells (Costa et al., 2005). Regulation of floral growth is not restricted to the CYC-like class II TCPs. In the jaw-D mutant, petal development is different from wild type Arabidopsis (Palatnik et al., 2003). Nag et al. (2009) showed that this depends on miR319 regulation of TCP4. A microRNA-resistant form of TCP4 under the control of an APETALA3 promoter is expressed in floral organs only and leads to dramatically smaller flowers that only consist of carpels and sepals, missing any petals or stamens, whereas the seedlings of these plants look normal (Nag et al., 2009).
The zinc-finger transcriptional repressor RABBIT EARS controls the expression of the TCPs TCP5, TCP13, and TCP17 and misexpression of both RABBIT EARS and these TCPs leads to aberrant petal development in Arabidopsis (Huang and Irish, 2015). Repression of these TCPs leads to an early stop of mitotic activity during petal development (Huang and Irish, 2015). Interestingly the opposite occurs upon downregulation of TCP5, TCP13, and TCP17 in leaves, where leaf cells continue with mitotic divisions for a longer time than in wild type plants (Efroni et al., 2008). Here, the effect of TCP transcription factors on organ development is dependent on the organ-context. This underlines the importance of the regulatory interplay between TCPs and organ identity regulators. While there are hints at this interplay between TCPs and MADS box transcription factors during flower development, such an interplay remains to be shown during the development of other organs (Dornelas et al., 2011).
First indications for a role of TCPs in leaf development comes from work in Antirrhinum (Nath et al., 2003). The Antirrhinum class II TCP mutant cin displays crinkly leaves, which are the result of a change in the regulation of the cell cycle during leaf development (Nath et al., 2003). Essentially, mitotic divisions of developing leaf cells in the leaf tip are arrested first and those at the leaf base are arrested last. The result of this successive arresting behavior is a so called arrest front that moves from the leaf tip to the leaf base. The form of this arrest front is different in cin leaves than in wild type leaves, leading to a modified leaf curvature (Nath et al., 2003). In Arabidopsis, similar behavior is observed in the jaw-D mutant (Palatnik et al., 2003). Jaw-D is an overexpressor of the microRNA miR319a in which the CIN-like class II TCPs TCP2, TCP3, TCP4, TCP10, and TCP24 are downregulated (Palatnik et al., 2003). Jaw-D mutants display serrated leaves, abnormal petals and delayed leaf development and senescence (Palatnik et al., 2003). This phenotype derives from delayed leaf development, in which the mitotic arrest front starts later than in wild type plants (Efroni et al., 2008). Recently, it was shown that miR319a-regulated TCP transcription factors act redundantly with NGATHA transcription factors to limit meristematic activity of leaf meristems during leaf development (Alvarez et al., 2016). This phenotype was also apparent in plants expressing an artificial microRNA against the class II TCPs TCP5, TCP13, and TCP17 and the phenotype was extremely strong when these plants were crossed with jaw-D plants (Efroni et al., 2008).
Class II TCPs also regulate leaf development in tomato compound leaves. An ortholog of the Arabidopsis miR319-sensitive TCPs in tomato is LA and it is under the control of the tomato miR319 (Ori et al., 2007). La mutants exhibit simple leaves, whereas overexpression of miR319 without LA insensitivity to the microRNA leads to increased partitioning of the compound leaves. Also, miR319 overexpressing tomato leaves grow 3 months longer than wild type leaves and show the marks of late differentiation, which is a behavior that is identical to Arabidopsis jaw-D plants (Ori et al., 2007; Efroni et al., 2008). Overexpression of miR319 in the monocot Agrostis stolonifera (creeping bentgrass) leads to downregulation of class II TCPs and to the formation of wider and thicker leaves that are different from the wild type (Zhou et al., 2013). This phenotype stems from an increased number of cells in the transgenic bentgrass, similar to jaw-D in Arabidopsis (Efroni et al., 2008; Zhou et al., 2013). In general, expression of CIN-like genes is closely correlated with leaf shapes both in Solanaceae species and in the desert poplar (Populus euphratica) (Shleizer-Burko et al., 2011; Ma et al., 2016).
Expression of TCP3 with a dominant repressor domain led to severe disturbance of Arabidopsis development in all organs (Koyama et al., 2007), involving ectopic shoot formation, serrated leaves, modified sepals and petals, and wavy silique formation. This was due to misexpression of boundary specific genes, i.e., CUC and LATERAL ORGAN BOUNDARIES (Koyama et al., 2007). Also in Antirrhinum, an ortholog of Arabidopsis TCP15 was found to interact with CUPULIFORMIS, a protein that is related to Arabidopsis CUC proteins (Weir et al., 2004). Furthermore, the two Arabidopsis class I TCPs TCP14 and TCP15 were shown to be redundant in affecting cell proliferation during leaf development and in other tissues in Arabidopsis. The most obvious effect though was seen in internode length, which is reduced in tcp14 tcp15 mutants and leads to shorter plants (Kieffer et al., 2011).
Whereas TCP functions have thus been very well-characterized in these branching, flower and leaf development over a wide array of plant species (Figure 1), there are hints that this is just a subset of TCP roles in development. TCPs were shown to be upregulated upon imbibition of dry seeds and germination of tcp14 transposon insertion lines seemed to be lower than in wild type seeds (Tatematsu et al., 2008). Although here, expression of TCP14 in the transposon lines was not necessarily lower than in the wild type, indicating that TCP14 may not be the only cause of the reduced germination rate (Tatematsu et al., 2008). Downregulation of TCP expression in cotton led to reduced cotton hair fiber length as well as a higher of lateral shoots and a stunted growth indicative of a reduced apical dominance (Hao et al., 2012). Overexpression of miR319 in Chinese cabbage not only led to altered leaf development, also the cabbage heads were rounder than in cabbage with low miR319 expression and higher expression of its target gene BrpTCP4-1 (Mao et al., 2014). Heterologous expression of the rice OsTCP19 in Arabidopsis led to a lower number of lateral roots (Mukhopadhyay et al., 2015). In cucumber, mutation of a TCP gene led to a unique plant phenotype. The affected cucumber plants did not develop tendrils but shoots instead. The authors of this study hypothesize that here TCPs not only affect growth of an organ but also determine organ identity (Wang S.et al., 2015). A similar phenotype was found in melons where a single-nucleotide mutation in CmTCP1 led to the Chiba tendril-less mutation. Also here, the tendrils were converted to shoot and leaf-like structures (Mizuno et al., 2015). This would be the first indication that TCPs can act as organ identity regulators. Further research has yet to uncover whether the function of TCPs in organ identity regulation of tendrils is a unique and novel role or whether other plant organs also need TCPs to define their identity.
FIGURE 1
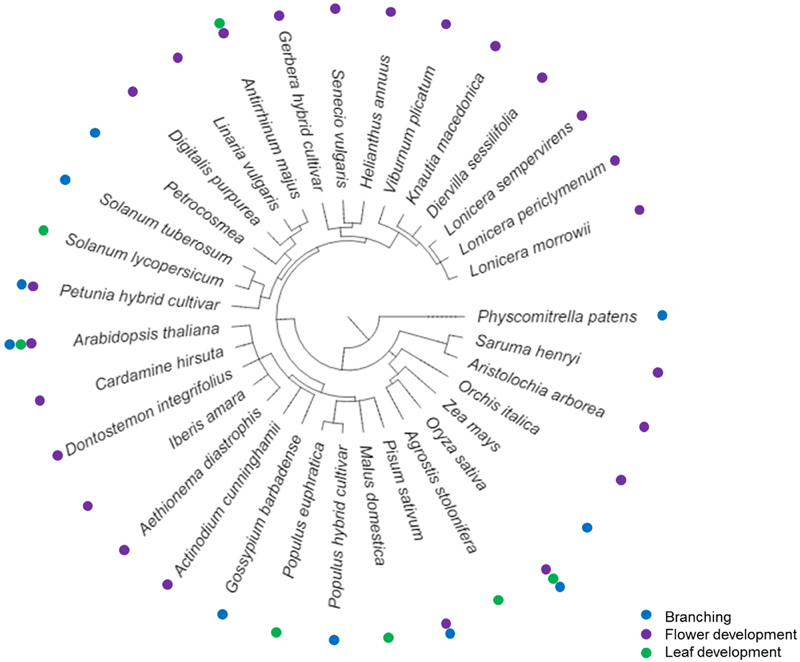
Phylogenetic tree of plant species in which TCP transcription factors are involved in branching (Takeda et al., 2003; Aguilar-Martínez et al., 2007; Poza-Carrión et al., 2007; Bai et al., 2012; Braun et al., 2012; Drummond et al., 2015; Nicolas et al., 2015; Muhr et al., 2016) (blue dots), flower development (Linnaeus and Rudberg, 1744; Keeble et al., 1910; Corley et al., 2005; Costa et al., 2005; Busch and Zachgo, 2007; Broholm et al., 2008; Kim et al., 2008; Nag et al., 2009; Yuan et al., 2009; Howarth et al., 2011; Busch et al., 2012; Tähtiharju et al., 2012; Claßen-Bockhoff et al., 2013; Juntheikki-Palovaara et al., 2014; De Paolo et al., 2015; Horn et al., 2015; Lucero et al., 2015; Wang et al., 2008; Wang X.et al., 2015; Yang et al., 2015; Berger et al., 2016) (purple dots) or leaf development (Kosugi and Ohashi, 1997; Nath et al., 2003; Palatnik et al., 2003; Koyama et al., 2007, 2010a,b; Ori et al., 2007; Efroni et al., 2008; Kieffer et al., 2011; Mimida et al., 2011; Sarvepalli and Nath, 2011; Danisman et al., 2012, 2013; Aguilar-Martínez and Sinha, 2013; Burko et al., 2013; Tao et al., 2013; Zhou et al., 2013; Ballester et al., 2015; Huang and Irish, 2015; Ma et al., 2016) (green dots), respectively. The phylogenetic tree was created using Phylotree and iTOL (Letunic and Bork, 2016).
TCP Functions Effect on the Cell Cycle – Direct or Indirect?
Early, TCP research focused on the cell cycle as main target of TCP regulation (Kosugi and Ohashi, 1997; Li et al., 2005). Whereas, binding to cell cycle genes has been shown in certain cases (Li et al., 2005; Davière et al., 2014), close analysis of cell division patterns and transcript changes during jaw-D leaf development indicated that the class II-TCP dependent regulation of the cell cycle may be indirect (Efroni et al., 2008). Also binding of the class I TCP TCP20 to cell cycle genes has been shown only once and in vitro (Li et al., 2005), whereas direct target gene analysis indicate that hormone synthesis, especially jasmonate synthesis, is rather directly targeted by TCP20 (Danisman et al., 2012). Both TCP4 and TCP20 affect leaf development via the synthesis of methyl jasmonate, a hormone that has multiple functions in plant development and response to wounding and pathogens (Schommer et al., 2008). Jasmonate, usually known for its role in wounding and pathogen response, does also affect the cell cycle (Świątek et al., 2002).
Jasmonate is not the only plant hormone that may mediate TCP regulation to the cell cycle. The evidence for hormone involvement in TCP-mediated growth regulation accumulated in the recent years (Nicolas and Cubas, 2016). TCP functions have been associated with abscisic acid (Tatematsu et al., 2008; González-Grandío et al., 2013; Mukhopadhyay et al., 2015), auxin (Kosugi et al., 1995; Ben-Gera and Ori, 2012; Uberti-Manassero et al., 2012; Das Gupta et al., 2014), brassinosteroid (Guo et al., 2010), cytokinin (Steiner et al., 2012; Efroni et al., 2013), GA (Yanai et al., 2011; Das Gupta et al., 2014; Davière et al., 2014), jasmonic acid (Schommer et al., 2008; Danisman et al., 2012), salicylic acid (Wang X.et al., 2015), and strigolactone signaling pathways (Dun et al., 2012; Hu et al., 2014) (Figure 2).
FIGURE 2
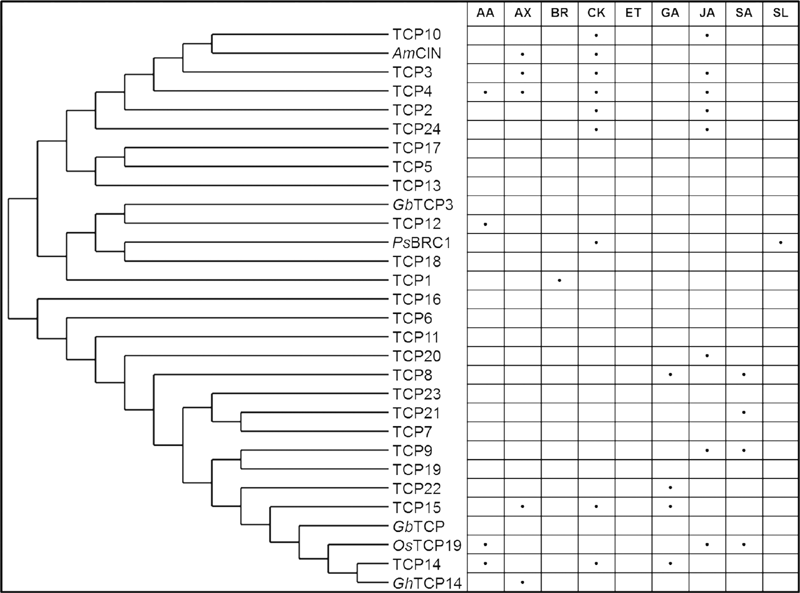
Hormonal pathways associated with Arabidopsis TCP transcription factors and orthologs. The proteins were plotted according to their phylogenetic similarity using PhyML and TreeDyne (Dereeper et al., 2008). AA, abscisic acid; AX, auxin; BR, brassinosteroids; CK, cytokinin; ET, ethylene; GA, gibberellic acid; JA, jasmonic acid; SA, salicylic acid; SL, strigolactones.
Apart from hormonal control of growth, TCP transcription factors are also involved in other biological processes that in turn affect growth. For example, binding sites of TCP transcription factors have been identified in the promoters of CYTOCHROME C1 and 103 genes that are encoding components of the mitochondrial oxidative phosphorylation machinery and protein biogenesis (Welchen and Gonzalez, 2006). The authors of this study proposed that the TCP transcription factors binding these sites coordinate mitochondria genesis and function with growth in new organs (Welchen and Gonzalez, 2006). Another study showed these genes contain a GGGC(C/T) element in their promoters which is important for diurnal regulation of their gene expression (Giraud et al., 2010). These promoters are bound by TCP transcription factors, implying a role in diurnal regulation of transcripts of the mitochondrial oxidative phosphorylation machinery (Giraud et al., 2010). Earlier TCP21 was found to bind to the promoter of the core clock gene CCA1 and regulate its expression (Pruneda-Paz et al., 2009). TCP21 serves as an inhibitor of CCA1 during the day and dimerization of TOC1 with TCP21 abolishes its binding to the CCA1 promoter. In a double mutant with the clock gene LHY, tcp21/lhy greatly reduces the period of CCA1 expression (Pruneda-Paz et al., 2009). Not only TCP21, other TCPs have also been found to bind to CCA1 in yeast based studies and co-immunoprecipitation experiments (Giraud et al., 2010; Pruneda-Paz et al., 2014). A recent study also showed that TCP20 and TCP22 act as activators of CCA1 in the morning, fulfilling an important role in the circuity of the circadian clock (Wu et al., 2016). This means that TCP proteins bind to the promoters of clock genes, regulate their expression, dimerize with clock proteins and bind to downstream targets of the clock (Pruneda-Paz et al., 2009, 2014; Giraud et al., 2010; Wu et al., 2016) (Figure 3). Altogether, it becomes clear that TCPs not only affect growth via the cell cycle. Instead, they act in different biological processes that directly or indirectly affect growth.
FIGURE 3
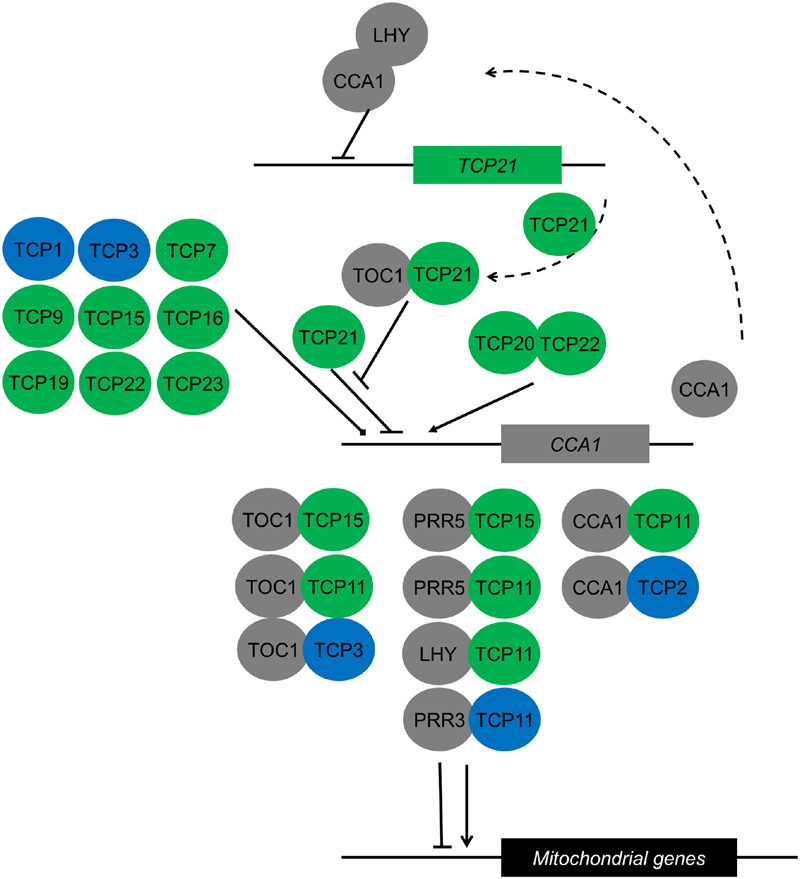
Interactions of TCP transcription factors with components of the circadian clock both within the central clock circuitry and in downstream processes. Class I and class II TCPs are depicted in green and blue, respectively. Known clock components are depicted in gray. Proteins are represented as circles, genes in squares. Dimers are depicted as overlapping circles. CCA1 inhibition by TCP21 is abolished by dimerization of TCP21 with TOC1. The CCA1/LHY dimer inhibits TCP21 expression (Pruneda-Paz et al., 2009). The effect of nine TCPs that bind to the CCA1 promoter in yeast one-hybrid studies is unknown (Pruneda-Paz et al., 2014). Downstream of the clock, TCP/clock component heterodimers regulate rhythmic expression of mitochondrial proteins depending on the number and arrangement of TCP binding sites in the mitochondrial gene promoters (Giraud et al., 2010).
Mediating Environmental Signals into Growth Responses
This picture becomes even more complex, as TCPs also mediate environmental signals into growth responses. TCPs were found to be involved in pathogen defense. First, an extensive study showed that both Pseudomonas syringae and Hyaloperonospora arabidopsidis infection led to reduction of TCP14 protein (Mukhtar et al., 2011). Secreted proteins from pathogenic bacteria transferred by the Aster leafhopper (Macrosteles quadrilineatus) to Arabidopsis were able to dimerize with and destabilize TCP2, TCP4, and TCP7 proteins, comprising both classes of TCP transcription factors (Sugio et al., 2011, 2014). Overexpression of the responsible phytoplasma protein SECRETED ASTER YELLOWS-WITCHES BROOM PROTEIN 11 in Arabidopsis destabilizes TCP2, TCP3, TCP4, TCP5, TCP10, TCP13, TCP17, and TCP24 and leads to jaw-D-like phenotypes (Sugio et al., 2011). Additionally, jasmonic acid levels in infected Arabidopsis leaves are significantly reduced in comparison with untreated leaves, indicating that the plant’s defense mechanisms are reduced upon infection by the pathogen. A similar effect has been found in apples, where the plant pathogen Candidatus Phytoplasma mali binds to two TCP transcription factors and induces morphogenetic changes that co-occur with reduction of jasmonic acid, salicylic acid, and abscisic acid levels (Janik et al., 2016). Further studies identified the class I TCPs TCP8 and TCP9 as important factors for the expression of ICS1, which encodes for a key enzyme in salicylic acid synthesis (Wang X.et al., 2015). In another study, TCP21 has been identified to bind to the promoter of ICS1 and induction of ICS1 expression by salicylic acid is blocked in tcp21 mutants (Zheng et al., 2015). Class I TCPs also interact with proteins known to regulate ICS1 expression, i.e., the transcription factors WRKY28, NAC019 and ETHYLENE INSENSITIVE 1 and the calmodulin binding protein SYSTEMIC ACQUIRED RESISTANCE DEFICIENT 1. Consequently, the tcp8 tcp9 double mutant shows increased sensitivity to infection with Pseudomonas syringae pv. maculicola ES4326 (Wang X.et al., 2015). TCP transcription factors partially control pathogen defense via a second pathway, i.e., by antagonizing the effect of SUPPRESSOR OF rps4-RLD1, a protein that negatively regulates effector-triggered immunity in Arabidopsis (Kim et al., 2014). Lack of TCPs in the triple mutant tcp8 tcp14 tcp15 leads to increased growth of Pseudomonas syringae DC3000 when compared to wild type plants (Kim et al., 2014).
Recent studies showed that TCP transcription factors regulate flowering time. A knockout of the class I TCP transcription factor TCP23 led to earlier flowering than the wild type, whereas TCP23 overexpressing lines showed delayed flowering behavior (Balsemão-Pires et al., 2013). The floral transition of axillary meristems in Arabidopsis is controlled by an interaction between the flowering time proteins FT and TWIN SISTER OF FT and BRC1 (Niwa et al., 2013). The protein–protein interactions between these transcription factors have been shown in yeast two-hybrid, bimolecular fluorescence complementation, and in vitro pull-down assays (Niwa et al., 2013). As brc1 mutants exhibit accelerated flowering and ft and twin sister of ft mutants exhibit slower flowering of axillary meristems, respectively, it seems that there is an antagonistic relationship between BRC1 and the flowering time proteins (Niwa et al., 2013). It is likely that dimerization of BRC1 with FT and TWIN SISTER OF FT represses their function in axillary meristems (Niwa et al., 2013). The apple FT orthologs MdFT1 and 2 were also found to interact with TCP transcription factors (Mimida et al., 2011). Overexpression of the tomato miR319 led to flowering with fewer leaves than in wild type tomato and it was shown that LA binds to the promoters of the tomato APETALA1 and FRUITFUL orthologs (Burko et al., 2013).
Perception of the red to far-red light ratio (R:FR) informs a plant of shading by neighboring vegetation and a lower R:FR ratio leads to suppressed axillary meristem outgrowth, allowing the plant to invest in a longer hypocotyl and eventually avoid the shading. In Arabidopsis, hypocotyl elongation is regulated via the bHLH transcription factor PHYTOCHROME INTERACTING FACTOR 4, which among others activates YUCCA8 expression to promote cell elongation (Sun et al., 2012). YUCCA2, 5, and 8 are also direct target genes of TCP4. In fact, induced overexpression of TCP4 leads to elongated hypocotyls and this effect is dependent on both auxin and brassinosteroid signaling (Challa et al., 2016). In potato, BRC1a regulation is dependent on the R:FR. BRC1a comes in two forms: the short form (BRC1aS) and the alternatively spliced long version (BRC1aL). Both result in proteins but the shorter form is cytoplasmatic and does not bind to target genes in the nucleus. The ratio between these two forms changes upon decapitation of potato shoots, exposure to darkness, and under low R:FR conditions (Nicolas et al., 2015). Whereas decapitation leads to a relative increase in BRC1aS, darkness and low R:FR treatments lead to a relative increase in BRC1aL content. The longer BRC1aL protein subsequently inhibits axillary branch elongation in potato shoots and stolons (Nicolas et al., 2015). Arabidopsis brc1 and brc2 (tcp12 and tcp18) show a reduced response to R:FR and the response is abolished in the brc1 brc2 double mutant (González-Grandío et al., 2013). TCP transcription factors are also involved in axillary bud outgrowth of Petunia. Here, GhTCP3 acts in conjunction with DECREASED APICAL DOMINANCE 2, a receptor protein that normally inactivates strigolactones in response to decreased R:FR (Drummond et al., 2015). Rice OsTCP15 is involved in the mesocotyl elongation in response to darkness and responds to strigolactone and cytokinin treatments, outlining the interplay between TCPs and different plant hormones in developmental regulation that is responsive to the environment (Hu et al., 2014).
Viola et al. (2013) showed that class I TCPs contain a conserved cysteine-20 which is sensitive to treatments by oxidants in a dose-dependent manner. This redox-dependent behavior of TCP15 is important for its effect in anthocyanin biosynthesis. A mutant in which the cysteine-20 of TCP15 was replaced by a serine accumulates less anthocyanin under high light stress than wild type plants (Viola et al., 2016). Plant extracts from TCP15 overexpressing plants showed that exposure to prolonged high light conditions leads to an abolishment of TCP15 DNA-binding activity in vivo, mirroring the in vitro phenotype (Viola et al., 2013, 2016). Thus, TCP15 function is reactive to high light input. While the anthocyanin response is not a direct developmental response, further analysis may show that there is a developmental effect.
Not only light affects TCPs, also other signals are perceived and lead to TCP-mediated growth regulation. For example, Guan et al. (2014) showed that TCP20 is involved in nutrient foraging of Arabidopsis roots. In split-root experiments wild type Arabidopsis develops an increased number of lateral roots in medium containing high nitrate concentrations (i.e., 5 mM NO3-) and close to no lateral roots in medium containing low nitrate concentrations (i.e., 0 mM NO3-). Tcp20 plants do not exhibit this behavior, indicating that the regulation of root foraging is under the control of TCP20 (Guan et al., 2014). Interestingly, TCP20 transcript levels are not under the control of nitrate levels, indicating that TCP20 is regulated on protein level, either by forming specific protein–protein dimers in the case of nitrate deficiency or via another regulatory mechanism. In rice, the transcript of the class I TCP OsTCP19 is upregulated during salt stress and water-deficit treatments (Mukhopadhyay et al., 2015). Heterologous OsTCP19 overexpression in Arabidopsis leads to reduced numbers of lateral roots but increased abiotic stress tolerance, i.e., plants grew better on Mannitol-containing medium and recovered better after water-deficit treatments. Here, LOX2 expression was reduced in the OsTCP19 overexpressors and ABA signaling genes were upregulated (Mukhopadhyay et al., 2015). Recent experiments revealed up- and down-regulation of several TCPs in Arabidopsis under osmotic stress, although a functional analysis of their role in response to osmotic stress has not been done yet (Kumar et al., 2016). In summary, these few results are first indications that TCPs are no mere static regulators of development, but that they do directly translate environmental signals into growth regulation (Figure 4).
FIGURE 4
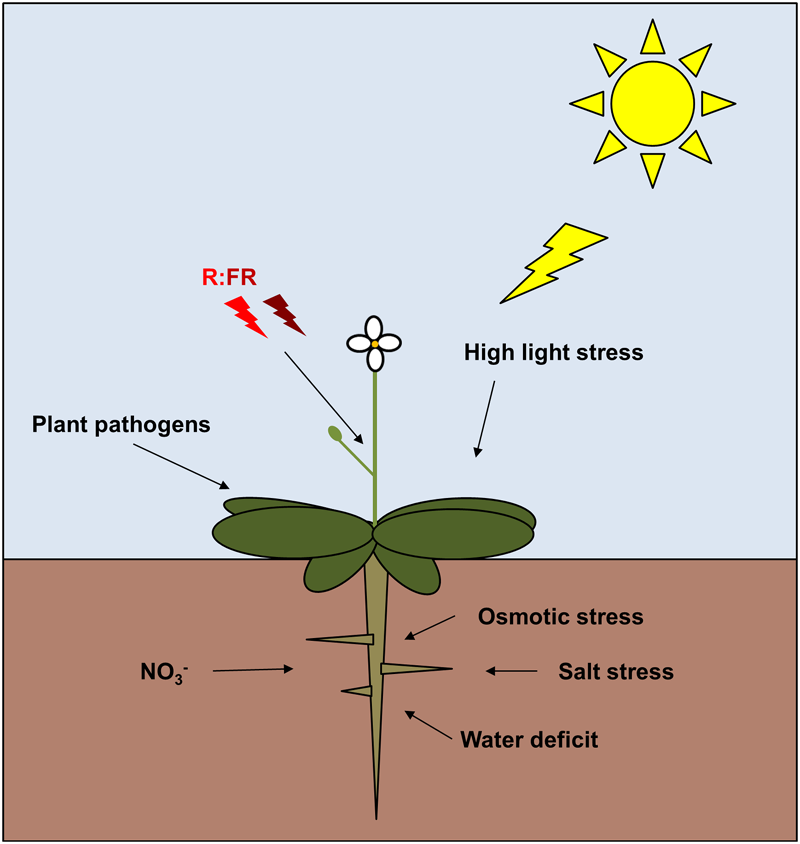
Schematic figure depicting the diversity of environmental signals that affect TCP functions in plants (Mukhtar et al., 2011; Sugio et al., 2011; Balsemão-Pires et al., 2013; González-Grandío et al., 2013; Niwa et al., 2013; Viola et al., 2013, 2016; Guan et al., 2014; Hu et al., 2014; Kim et al., 2014; Mukhopadhyay et al., 2015; Nicolas et al., 2015; Kumar et al., 2016).
Conclusion and Outlook
TCP transcription factors play a role in a multitude of growth processes over a wide range of plant species (Figure 1). They affect growth directly via the cell cycle and indirectly via influencing plant hormonal signaling and the circadian clock (Figures 2 and 3). Additionally, recent discoveries link TCP-controlled growth responses with environmental signals such as R:FR, high light stress, salt stress or the presence or absence of nutrients.
TCP transcription factors are involved in so many important developmental processes and interact with so many plant hormones that it is likely that future plant research will also uncover a lot more signals that TCPs react to. This will also mean that future TCP research will have to more closely elucidate how the interaction of TCPs with different signaling networks is regulated to ensure a measured response to environmental challenges. This research will have to uncover the roles of dimerization, transcriptional and post-transcriptional regulation as well as post-translational modifications in controlling and ensuring specific TCP functions in plant development.
Plant pathogens are targeting TCP transcription factors to manipulate plant architecture in their favor. If plant pathogens use TCPs in their best interests, maybe so should we. TCP transcription factors will be valuable tools in optimizing plant architecture and hardening plants in response to environmental challenges.
Statements
Author contributions
SD drafted, wrote and critically revised the article.
Funding
Work in the author’s lab is supported by the Bielefeld Young Researcher’s Fund and the core grant of Bielefeld University to D. Staiger.
Acknowledgments
We acknowledge support for the Article Processing Charge by the Deutsche Forschungsgemeinschaft and the Open Access Publication Fund of Bielefeld University.
Conflict of interest
The author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Abbreviations
- bHLH
basic helix-loop-helix
- BRC
BRANCHED
- CCA1
CIRCADIAN CLOCK ASSOCIATED 1
- CIN
CINCINNATA
- CUC
CUP-SHAPED COTYLEDON
- CYC
CYCLOIDEA
- FT
FLOWERING LOCUS T
- GA
gibberellic acid
- ICS1
ISOCHORISMATE SYNTHASE 1
- IDR
intrinsically disordered region
- jaw-D
JAGGED AND WAVY-D
- LA
LANCEOLATE
- LHY
LATE ELONGATED HYPOCOTYL
- LOX
LIPOXYGENASE
- NLS
nuclear localization signal
- PRR
PSEUDO RESPONSE REGULATOR
- TB1
TEOSINTHE BRANCHED 1
- TCP
TEOSINTE BRANCHED 1, CYCLOIDEA, PCF1
- TOC1
TIMING OF CAB EXPRESSION1
References
1
Aggarwal P. Gupta M. D. Joseph A. P. Chatterjee N. Srinivasan N. Nath U. (2010). Identification of specific DNA binding residues in the TCP family of transcription factors in Arabidopsis.Plant Cell221174–1189. 10.1105/tpc.109.066647
2
Aguilar-Martínez J. A. Poza-Carrión C. Cubas P. (2007). Arabidopsis BRANCHED1 acts as an integrator of branching signals within axillary buds.Plant Cell19458–472. 10.1105/tpc.106.048934
3
Aguilar-Martínez J. A. Sinha N. (2013). Analysis of the role of Arabidopsis class I TCP genes AtTCP7, AtTCP8, AtTCP22, and AtTCP23 in leaf development.Front. Plant Sci.4:406. 10.3389/fpls.2013.00406
4
Alvarez J. P. Furumizu C. Efroni I. Eshed Y. Bowman J. L. (2016). Active suppression of a leaf meristem orchestrates determinate leaf growth.eLife5:e15023. 10.7554/eLife.15023
5
Andriankaja M. E. Danisman S. Mignolet-Spruyt L. F. Claeys H. Kochanke I. Vermeersch M. et al (2014). Transcriptional coordination between leaf cell differentiation and chloroplast development established by TCP20 and the subgroup Ib bHLH transcription factors.Plant Mol. Biol.85233–245. 10.1007/s11103-014-0180-2
6
Bai F. Reinheimer R. Durantini D. Kellogg E. A. Schmidt R. J. (2012). TCP transcription factor, BRANCH ANGLE DEFECTIVE 1 (BAD1), is required for normal tassel branch angle formation in maize.Proc. Natl. Acad. Sci. U.S.A.10912225–12230. 10.1073/pnas.1202439109
7
Ballester P. Navarrete-Gómez M. Carbonero P. Oñate-Sánchez L. Ferrándiz C. (2015). Leaf expansion in Arabidopsis is controlled by a TCP-NGA regulatory module likely conserved in distantly related species.Physiol. Plant.15521–32. 10.1111/ppl.12327
8
Balsemão-Pires E. Andrade L. R. Sachetto-Martins G. (2013). Functional study of TCP23 in Arabidopsis thaliana during plant development.Plant Physiol. Biochem.67120–125. 10.1016/j.plaphy.2013.03.009
9
Ben-Gera H. Ori N. (2012). Auxin and LANCEOLATE affect leaf shape in tomato via different developmental processes.Plant Signal. Behav.71255–1257. 10.4161/psb.21550
10
Berger B. A. Thompson V. Lim A. Ricigliano V. Howarth D. G. (2016). Elaboration of bilateral symmetry across Knautia macedonica capitula related to changes in ventral petal expression of CYCLOIDEA-like genes.EvoDevo78. 10.1186/s13227-016-0045-7
11
Braun N. Germain A. de S. Pillot J.-P. Boutet-Mercey S. Dalmais M. et al (2012). The Pea TCP transcription factor PsBRC1 acts downstream of strigolactones to control shoot branching.Plant Physiol.158225–238. 10.1104/pp.111.182725
12
Broholm S. K. Tähtiharju S. Laitinen R. A. E. Albert V. A. Teeri T. H. Elomaa P. (2008). A TCP domain transcription factor controls flower type specification along the radial axis of the Gerbera (Asteraceae) inflorescence.Proc. Natl. Acad. Sci.1059117–9122. 10.1073/pnas.0801359105
13
Burko Y. Shleizer-Burko S. Yanai O. Shwartz I. Zelnik I. D. Jacob-Hirsch J. et al (2013). A role for APETALA1/FRUITFULL transcription factors in tomato leaf development.Plant Cell252070–2083. 10.1105/tpc.113.113035
14
Busch A. Horn S. Mühlhausen A. Mummenhoff K. Zachgo S. (2012). Corolla monosymmetry: evolution of a morphological novelty in the brassicaceae family.Mol. Biol. Evol.291241–1254. 10.1093/molbev/msr297
15
Busch A. Zachgo S. (2007). Control of corolla monosymmetry in the Brassicaceae Iberis amara.Proc. Natl. Acad. Sci. U.S.A.10416714–16719. 10.1073/pnas.0705338104
16
Challa K. R. Aggarwal P. Nath U. (2016). Activation of YUCCA5 by the transcription factor TCP4 integrates developmental and environmental signals to promote hypocotyl elongation in Arabidopsis.Plant Cell282117–2130. 10.1105/tpc.16.00360
17
Claßen-Bockhoff R. Ruonala R. Bull-Hereñu K. Marchant N. Albert V. A. (2013). The unique pseudanthium of Actinodium (Myrtaceae) - morphological reinvestigation and possible regulation by CYCLOIDEA-like genes.EvoDevo48. 10.1186/2041-9139-4-8
18
Corley S. B. Carpenter R. Copsey L. Coen E. (2005). Floral asymmetry involves an interplay between TCP and MYB transcription factors in Antirrhinum.Proc. Natl. Acad. Sci. U.S.A.1025068–5073. 10.1073/pnas.0501340102
19
Costa M. M. R. Fox S. Hanna A. I. Baxter C. Coen E. (2005). Evolution of regulatory interactions controlling floral asymmetry.Development1325093–5101. 10.1242/dev.02085
20
Cubas P. Lauter N. Doebley J. Coen E. (1999a). The TCP domain: a motif found in proteins regulating plant growth and development.Plant J.18215–222. 10.1046/j.1365-313X.1999.00444.x
21
Cubas P. Vincent C. Coen E. (1999b). An epigenetic mutation responsible for natural variation in floral symmetry.Nature401157–161. 10.1038/43657
22
Danisman S. Dijk A. D. J. van Bimbo A. Wal F. van der Hennig L. Folter S. et al (2013). Analysis of functional redundancies within the Arabidopsis TCP transcription factor family.J. Exp. Bot.645673–5685. 10.1093/jxb/ert337
23
Danisman S. Wal F. van der Dhondt S. Waites R. Folter S. de Bimbo A. et al (2012). Arabidopsis Class I and Class II TCP transcription factors regulate jasmonic acid metabolism and leaf development antagonistically.Plant Physiol.1591511–1523. 10.1104/pp.112.200303
24
Darwin C. R. (1868). The Variation of Animals and Plants Under Domestication.London: John Murray.
25
Das Gupta M. Aggarwal P. Nath U. (2014). CINCINNATA in Antirrhinum majus directly modulates genes involved in cytokinin and auxin signaling.New Phytol.204901–912. 10.1111/nph.12963
26
Davière J.-M. Wild M. Regnault T. Baumberger N. Eisler H. Genschik P. et al (2014). Class I TCP-DELLA interactions in inflorescence shoot apex determine plant height.Curr. Biol.241923–1928. 10.1016/j.cub.2014.07.012
27
De Paolo S. Gaudio L. Aceto S. (2015). Analysis of the TCP genes expressed in the inflorescence of the orchid Orchis italica.Sci. Rep.5:16265. 10.1038/srep16265
28
Dereeper A. Guignon V. Blanc G. Audic S. Buffet S. Chevenet F. et al (2008). Phylogeny.fr: robust phylogenetic analysis for the non-specialist.Nucleic Acids Res.36W465–W469. 10.1093/nar/gkn180
29
Doebley J. Stec A. Gustus C. (1995). teosinte branched1 and the origin of maize: evidence for epistasis and the evolution of dominance.Genetics141333–346.
30
Dornelas M. C. Patreze C. M. Angenent G. C. Immink R. G. H. (2011). MADS: the missing link between identity and growth?Trends Plant Sci.1689–97. 10.1016/j.tplants.2010.11.003
31
Drummond R. S. M. Janssen B. J. Luo Z. Oplaat C. Ledger S. E. Wohlers M. W. et al (2015). Environmental control of branching in Petunia.Plant Physiol.168735–751. 10.1104/pp.15.00486
32
Dun E. A. Germain A. de S. Rameau C. Beveridge C. A. (2012). Antagonistic action of strigolactone and cytokinin in bud outgrowth control.Plant Physiol.158487–498. 10.1104/pp.111.186783
33
Efroni I. Blum E. Goldshmidt A. Eshed Y. (2008). A protracted and dynamic maturation schedule underlies Arabidopsis leaf development.Plant Cell202293–2306. 10.1105/tpc.107.057521
34
Efroni I. Han S.-K. Kim H. J. Wu M.-F. Steiner E. Birnbaum K. D. et al (2013). Regulation of leaf maturation by chromatin-mediated modulation of cytokinin responses.Dev. Cell24438–445. 10.1016/j.devcel.2013.01.019
35
Fambrini M. Salvini M. Pugliesi C. (2012). A transposon-mediate inactivation of a CYCLOIDEA-like gene originates polysymmetric and androgynous ray flowers in Helianthus annuus.Genetica1391521–1529. 10.1007/s10709-012-9652-y
36
Giraud E. Ng S. Carrie C. Duncan O. Low J. Lee C. P. et al (2010). TCP transcription factors link the regulation of genes encoding mitochondrial proteins with the circadian clock in Arabidopsis thaliana.Plant Cell223921–3934. 10.1105/tpc.110.074518
37
González-Grandío E. Poza-Carrión C. Sorzano C. O. S. Cubas P. (2013). BRANCHED1 promotes axillary bud dormancy in response to shade in Arabidopsis.Plant Cell25834–850. 10.1105/tpc.112.108480
38
Guan P. Wang R. Nacry P. Breton G. Kay S. A. Pruneda-Paz J. L. et al (2014). Nitrate foraging by Arabidopsis roots is mediated by the transcription factor TCP20 through the systemic signaling pathway.Proc. Natl. Acad. Sci. U.S.A.11115267–15272. 10.1073/pnas.1411375111
39
Guo Z. Fujioka S. Blancaflor E. B. Miao S. Gou X. Li J. (2010). TCP1 Modulates brassinosteroid biosynthesis by regulating the expression of the key biosynthetic gene DWARF4 in Arabidopsis thaliana.Plant Cell221161–1173. 10.1105/tpc.109.069203
40
Hao J. Tu L. Hu H. Tan J. Deng F. Tang W. et al (2012). GbTCP, a cotton TCP transcription factor, confers fibre elongation and root hair development by a complex regulating system.J. Exp. Bot.636267–6281. 10.1093/jxb/ers278
41
Horn S. Pabón-Mora N. Theuß V. S. Busch A. Zachgo S. (2015). Analysis of the CYC/TB1 class of TCP transcription factors in basal angiosperms and magnoliids.Plant J.81559–571. 10.1111/tpj.12750
42
Howarth D. G. Donoghue M. J. (2006). Phylogenetic analysis of the “ECE” (CYC/TB1) clade reveals duplications predating the core eudicots.Proc. Natl. Acad. Sci. U.S.A.1039101–9106. 10.1073/pnas.0602827103
43
Howarth D. G. Martins T. Chimney E. Donoghue M. J. (2011). Diversification of CYCLOIDEA expression in the evolution of bilateral flower symmetry in Caprifoliaceae and Lonicera (Dipsacales).Ann. Bot.1071521–1532. 10.1093/aob/mcr049
44
Hu Z. Yamauchi T. Yang J. Jikumaru Y. Tsuchida-Mayama T. Ichikawa H. et al (2014). Strigolactone and cytokinin act antagonistically in regulating rice mesocotyl elongation in darkness.Plant Cell Physiol.5530–41. 10.1093/pcp/pct150
45
Huang T. Irish V. F. (2015). Temporal control of plant organ growth by TCP transcription factors.Curr. Biol.251765–1770. 10.1016/j.cub.2015.05.024
46
Janik K. Mithöfer A. Raffeiner M. Stellmach H. Hause B. Schlink K. (2016). An effector of apple proliferation phytoplasma targets TCP transcription factors - a generalized virulence strategy of phytoplasma?Mol. Plant Pathol.10.1111/mpp.12409[Epub ahead of print].
47
Juntheikki-Palovaara I. Tähtiharju S. Lan T. Broholm S. K. Rijpkema A. S. Ruonala R. et al (2014). Functional diversification of duplicated CYC2 clade genes in regulation of inflorescence development in Gerbera hybrida (Asteraceae).Plant J.79783–796. 10.1111/tpj.12583
48
Kaufmann K. Muiño J. M. Jauregui R. Airoldi C. A. Smaczniak C. Krajewski P. et al (2009). Target genes of the MADS transcription factor SEPALLATA3: integration of developmental and hormonal pathways in the Arabidopsis flower.PLoS Biol.7:e1000090. 10.1371/journal.pbio.1000090
49
Kaufmann K. Wellmer F. Muiño J. M. Ferrier T. Wuest S. E. Kumar V. et al (2010). Orchestration of floral initiation by APETALA1.Science32885–89. 10.1126/science.1185244
50
Keeble F. Pellew M. C. Jones W. N. (1910). The inheritance Op peloria and flowercolour in foxgloves (digitalis Purpurea).New Phytol.968–77. 10.1111/j.1469-8137.1910.tb05554.x
51
Kieffer M. Master V. Waites R. Davies B. (2011). TCP14 and TCP15 affect internode length and leaf shape in Arabidopsis.Plant J.68147–158. 10.1111/j.1365-313X.2011.04674.x
52
Kim M. Cui M.-L. Cubas P. Gillies A. Lee K. Chapman M. A. et al (2008). Regulatory genes control a key morphological and ecological trait transferred between species.Science3221116–1119. 10.1126/science.1164371
53
Kim S. H. Son G. H. Bhattacharjee S. Kim H. J. Nam J. C. Nguyen P. D. T. et al (2014). The Arabidopsis immune adaptor SRFR1 interacts with TCP transcription factors that redundantly contribute to effector-triggered immunity.Plant J.78978–989. 10.1111/tpj.12527
54
Kosugi S. Ohashi Y. (1997). PCF1 and PCF2 specifically bind to cis elements in the rice proliferating cell nuclear antigen gene.Plant Cell91607–1619. 10.1105/tpc.9.9.1607
55
Kosugi S. Suzuka I. Ohashi Y. (1995). Two of three promoter elements identified in a rice gene for proliferating cell nuclear antigen are essential for meristematic tissue-specific expression.Plant J.7877–886. 10.1046/j.1365-313X.1995.07060877.x
56
Koyama T. Furutani M. Tasaka M. Ohme-Takagi M. (2007). TCP Transcription factors control the morphology of shoot lateral organs via negative regulation of the expression of boundary-specific genes in Arabidopsis.Plant Cell19473–484. 10.1105/tpc.106.044792
57
Koyama T. Mitsuda N. Seki M. Shinozaki K. Ohme-Takagi M. (2010a). TCP transcription factors regulate the activities of ASYMMETRIC LEAVES1 and miR164, as Well as the auxin response, during differentiation of leaves in Arabidopsis.Plant Cell223574–3588. 10.1105/tpc.110.075598
58
Koyama T. Sato F. Ohme-Takagi M. (2010b). A role of TCP1 in the longitudinal elongation of leaves in Arabidopsis.Biosci. Biotechnol. Biochem.742145–2147. 10.1271/bbb.100442
59
Kumar D. Hazra S. Datta R. Chattopadhyay S. (2016). Transcriptome analysis of Arabidopsis mutants suggests a crosstalk between ABA, ethylene and GSH against combined cold and osmotic stress.Sci. Rep.636867. 10.1038/srep36867
60
Letunic I. Bork P. (2016). Interactive tree of life (iTOL) v3: an online tool for the display and annotation of phylogenetic and other trees.Nucleic Acids Res.44W242–W245. 10.1093/nar/gkw290
61
Li C. Potuschak T. Colón-Carmona A. Gutiérrez R. A. Doerner P. (2005). Arabidopsis TCP20 links regulation of growth and cell division control pathways.Proc. Natl. Acad. Sci. U.S.A.10212978–12983. 10.1073/pnas.0504039102
62
Linnaeus C. Rudberg D. (1744). Dissertatio Botanica de Peloria.Uppsala: Q-Med AB.
63
Liu J. Perumal N. B. Oldfield C. J. Su E. W. Uversky V. N. Dunker A. K. (2006). Intrinsic disorder in transcription factors.Biochemistry (Mosc.)456873–6888. 10.1021/bi0602718
64
Lucero L. E. Uberti-Manassero N. G. Arce A. L. Colombatti F. Alemano S. G. Gonzalez D. H. (2015). TCP15 modulates cytokinin and auxin responses during gynoecium development in Arabidopsis.Plant J.84267–282. 10.1111/tpj.12992
65
Luo D. Carpenter R. Vincent C. Copsey L. Coen E. (1996). Origin of floral asymmetry in Antirrhinum.Nature383794–799. 10.1038/383794a0
66
Lupas A. Dyke M. V. Stock J. (1991). Predicting coiled coils from protein sequences.Science2521162–1164. 10.1126/science.252.5009.1162
67
Ma X. Ma J. Fan D. Li C. Jiang Y. Luo K. (2016). Genome-wide Identification of TCP family transcription factors from populus euphratica and their involvement in leaf shape regulation.Sci. Rep.6:32795. 10.1038/srep32795
68
Mao Y. Wu F. Yu X. Bai J. Zhong W. He Y. (2014). microRNA319a-targeted Brassica rapa ssp. pekinensis TCP genes modulate head shape in chinese cabbage by differential cell division arrest in leaf regions.Plant Physiol.164710–720. 10.1104/pp.113.228007
69
Mimida N. Kidou S.-I. Iwanami H. Moriya S. Abe K. Voogd C. et al (2011). Apple FLOWERING LOCUS T proteins interact with transcription factors implicated in cell growth and organ development.Tree Physiol.31555–566. 10.1093/treephys/tpr028
70
Mizuno S. Sonoda M. Tamura Y. Nishino E. Suzuki H. Sato T. et al (2015). Chiba Tendril-Less locus determines tendril organ identity in melon (Cucumis melo L.) and potentially encodes a tendril-specific TCP homolog.J. Plant Res.128941–951. 10.1007/s10265-015-0747-2
71
Muhr M. Prüfer N. Paulat M. Teichmann T. (2016). Knockdown of strigolactone biosynthesis genes in Populus affects BRANCHED1 expression and shoot architecture.New Phytol.212613–626. 10.1111/nph.14076
72
Mukhopadhyay P. Tyagi A. K. Tyagi A. K. (2015). OsTCP19 influences developmental and abiotic stress signaling by modulating ABI4-mediated pathways.Sci. Rep.5:9998. 10.1038/srep09998
73
Mukhtar M. S. Carvunis A.-R. Dreze M. Epple P. Steinbrenner J. Moore J. et al (2011). Independently evolved virulence effectors converge onto hubs in a plant immune system network.Science333596–601. 10.1126/science.1203659
74
Nag A. King S. Jack T. (2009). miR319a targeting of TCP4 is critical for petal growth and development in Arabidopsis.Proc. Natl. Acad. Sci. U.S.A.10622534–22539. 10.1073/pnas.0908718106
75
Nath U. Crawford B. C. W. Carpenter R. Coen E. (2003). Genetic control of surface curvature.Science2991404–1407. 10.1126/science.1079354
76
Navaud O. Dabos P. Carnus E. Tremousaygue D. Hervé C. (2007). TCP transcription factors predate the emergence of land plants.J. Mol. Evol.6523–33. 10.1007/s00239-006-0174-z
77
Nicolas M. Cubas P. (2016). TCP factors: new kids on the signaling block.Curr. Opin. Plant Biol.3333–41. 10.1016/j.pbi.2016.05.006
78
Nicolas M. Rodríguez-Buey M. L. Franco-Zorrilla J. M. Cubas P. (2015). A recently evolved alternative splice site in the BRANCHED1a gene controls potato plant architecture.Curr. Biol.251799–1809. 10.1016/j.cub.2015.05.053
79
Niwa M. Daimon Y. Kurotani K. Higo A. Pruneda-Paz J. L. Breton G. et al (2013). BRANCHED1 interacts with FLOWERING LOCUS T to repress the floral transition of the axillary meristems in Arabidopsis.Plant Cell251228–1242. 10.1105/tpc.112.109090
80
Ori N. Cohen A. R. Etzioni A. Brand A. Yanai O. Shleizer S. et al (2007). Regulation of LANCEOLATE by miR319 is required for compound-leaf development in tomato.Nat. Genet.39787–791. 10.1038/ng2036
81
Ortiz-Ramírez C. Hernandez-Coronado M. Thamm A. Catarino B. Wang M. Dolan L. et al (2016). A transcriptome atlas of physcomitrella patens provides insights into the evolution and development of land plants.Mol. Plant.9205–220. 10.1016/j.molp.2015.12.002
82
Palatnik J. F. Allen E. Wu X. Schommer C. Schwab R. Carrington J. C. et al (2003). Control of leaf morphogenesis by microRNAs.Nature425257–263. 10.1038/nature01958
83
Poza-Carrión C. Aguilar-Martínez J. A. Cubas P. (2007). Role of TCP Gene BRANCHED1 in the control of shoot branching in Arabidopsis.Plant Signal. Behav.2551–552. 10.4161/psb.2.6.4811
84
Pruneda-Paz J. L. Breton G. Nagel D. H. Kang S. E. Bonaldi K. Doherty C. J. et al (2014). A genome-scale resource for the functional characterization of Arabidopsis transcription factors.Cell Rep.8622–632. 10.1016/j.celrep.2014.06.033
85
Pruneda-Paz J. L. Breton G. Para A. Kay S. A. (2009). A functional genomics approach reveals CHE as a component of the Arabidopsis circadian clock.Science3231481–1485. 10.1126/science.1167206
86
Sarvepalli K. Nath U. (2011). Interaction of TCP4-mediated growth module with phytohormones.Plant Signal. Behav.61440–1443. 10.4161/psb.6.10.17097
87
Schommer C. Palatnik J. F. Aggarwal P. Chételat A. Cubas P. Farmer E. E. et al (2008). Control of jasmonate biosynthesis and senescence by miR319 targets.PLoS Biol.6:e230. 10.1371/journal.pbio.0060230
88
Shleizer-Burko S. Burko Y. Ben-Herzel O. Ori N. (2011). Dynamic growth program regulated by LANCEOLATE enables flexible leaf patterning.Development138695–704. 10.1242/dev.056770
89
Smith S. J. E. (1821). A Selection of the Correspondence of Linnaeus, and other Naturalists: From the Original Manuscripts.Harlow: Longman.
90
Steiner E. Efroni I. Gopalraj M. Saathoff K. Tseng T.-S. Kieffer M. et al (2012). The Arabidopsis O-Linked N-Acetylglucosamine transferase SPINDLY interacts with class I TCPs to facilitate cytokinin responses in leaves and flowers.Plant Cell2496–108. 10.1105/tpc.111.093518
91
Sugio A. Kingdom H. N. MacLean A. M. Grieve V. M. Hogenhout S. A. (2011). Phytoplasma protein effector SAP11 enhances insect vector reproduction by manipulating plant development and defense hormone biosynthesis.Proc. Natl. Acad. Sci. U.S.A.108E1254–E1263. 10.1073/pnas.1105664108
92
Sugio A. MacLean A. M. Hogenhout S. A. (2014). The small phytoplasma virulence effector SAP11 contains distinct domains required for nuclear targeting and CIN-TCP binding and destabilization.New Phytol.202838–848. 10.1111/nph.12721
93
Sun J. Qi L. Li Y. Chu J. Li C. (2012). PIF4–mediated activation of YUCCA8 expression integrates temperature into the auxin pathway in regulating Arabidopsis hypocotyl growth.PLOS Genet8:e1002594. 10.1371/journal.pgen.1002594
94
Świątek A. Lenjou M. Van Bockstaele D. Inzé D. Van Onckelen H. (2002). Differential effect of jasmonic acid and abscisic acid on cell cycle progression in tobacco BY-2 cells.Plant Physiol.128201–211. 10.1104/pp.010592
95
Tähtiharju S. Rijpkema A. S. Vetterli A. Albert V. A. Teeri T. H. Elomaa P. (2012). Evolution and diversification of the CYC/TB1 gene family in asteraceae—a comparative study in Gerbera (Mutisieae) and sunflower (Heliantheae).Mol. Biol. Evol.291155–1166. 10.1093/molbev/msr283
96
Takeda T. Suwa Y. Suzuki M. Kitano H. Ueguchi-Tanaka M. Ashikari M. et al (2003). The OsTB1 gene negatively regulates lateral branching in rice.Plant J.33513–520. 10.1046/j.1365-313X.2003.01648.x
97
Tao Q. Guo D. Wei B. Zhang F. Pang C. Jiang H. et al (2013). The TIE1 transcriptional repressor links TCP transcription factors with TOPLESS/TOPLESS-RELATED corepressors and modulates leaf development in Arabidopsis.Plant Cell25421–437. 10.1105/tpc.113.109223
98
Tatematsu K. Nakabayashi K. Kamiya Y. Nambara E. (2008). Transcription factor AtTCP14 regulates embryonic growth potential during seed germination in Arabidopsis thaliana.Plant J.5342–52. 10.1111/j.1365-313X.2007.03308.x
99
Thieulin-Pardo G. Avilan L. Kojadinovic M. Gontero B. (2015). Fairy “tails”: flexibility and function of intrinsically disordered extensions in the photosynthetic world.Front. Mol. Biosci.2:23. 10.3389/fmolb.2015.00023
100
Trémousaygue D. Garnier L. Bardet C. Dabos P. Hervé C. Lescure B. (2003). Internal telomeric repeats and “TCP domain” protein-binding sites co-operate to regulate gene expression in Arabidopsis thaliana cycling cells.Plant J.33957–966. 10.1046/j.1365-313X.2003.01682.x
101
Uberti-Manassero N. G. Lucero L. E. Viola I. L. Vegetti A. C. Gonzalez D. H. (2012). The class I protein AtTCP15 modulates plant development through a pathway that overlaps with the one affected by CIN-like TCP proteins.J. Exp. Bot.63809–823. 10.1093/jxb/err305
102
Valsecchi I. Guittard-Crilat E. Maldiney R. Habricot Y. Lignon S. Lebrun R. et al (2013). The intrinsically disordered C-terminal region of Arabidopsis thaliana TCP8 transcription factor acts both as a transactivation and self-assembly domain.Mol. Biosyst.92282–2295. 10.1039/c3mb70128j
103
Viola I. L. Camoirano A. Gonzalez D. H. (2016). Redox-dependent modulation of anthocyanin biosynthesis by the TCP transcription factor TCP15 during exposure to high light intensity conditions in Arabidopsis.Plant Physiol.17074–85. 10.1104/pp.15.01016
104
Viola I. L. Güttlein L. N. Gonzalez D. H. (2013). Redox modulation of plant developmental regulators from the class I TCP transcription factor family.Plant Physiol.1621434–1447. 10.1104/pp.113.216416
105
Viola I. L. Reinheimer R. Ripoll R. Manassero N. G. U. Gonzalez D. H. (2012). Determinants of the DNA binding specificity of class I and Class II TCP transcription factors.J. Biol. Chem.287347–356. 10.1074/jbc.M111.256271
106
Viola I. L. Uberti Manassero N. G. Ripoll R. Gonzalez D. H. (2011). The Arabidopsis class I TCP transcription factor AtTCP11 is a developmental regulator with distinct DNA-binding properties due to the presence of a threonine residue at position 15 of the TCP domain.Biochem. J.435143–155. 10.1042/BJ20101019
107
Wang R.-L. Stec A. Hey J. Lukens L. Doebley J. (1999). The limits of selection during maize domestication.Nature398236–239. 10.1038/18435
108
Wang S. Yang X. Xu M. Lin X. Lin T. Qi J. et al (2015). A rare SNP identified a TCP transcription factor essential for tendril development in cucumber.Mol. Plant81795–1808. 10.1016/j.molp.2015.10.005
109
Wang X. Gao J. Zhu Z. Dong X. Wang X. Ren G. et al (2015). TCP transcription factors are critical for the coordinated regulation of ISOCHORISMATE SYNTHASE 1 expression in Arabidopsis thaliana.Plant J.82151–162. 10.1111/tpj.12803
110
Wang Z. Luo Y. Li X. Wang L. Xu S. Yang J. et al (2008). Genetic control of floral zygomorphy in pea (Pisum sativum L.).Proc. Natl. Acad. Sci. U.S.A.10510414–10419. 10.1073/pnas.0803291105
111
Weir I. Lu J. Cook H. Causier B. Schwarz-Sommer Z. Davies B. (2004). CUPULIFORMIS establishes lateral organ boundaries in Antirrhinum.Development131915–922. 10.1242/dev.00993
112
Welchen E. Gonzalez D. H. (2006). Overrepresentation of elements recognized by TCP-domain transcription factors in the upstream regions of nuclear genes encoding components of the mitochondrial oxidative phosphorylation machinery.Plant Physiol.141540–545. 10.1104/pp.105.075366
113
Wu J.-F. Tsai H.-L. Joanito I. Wu Y.-C. Chang C.-W. Li Y.-H. et al (2016). LWD–TCP complex activates the morning gene CCA1 in Arabidopsis.Nat. Commun.7:13181. 10.1038/ncomms13181
114
Yanai O. Shani E. Russ D. Ori N. (2011). Gibberellin partly mediates LANCEOLATE activity in tomato.Plant J.68571–582. 10.1111/j.1365-313X.2011.04716.x
115
Yang X. Zhao X.-G. Li C.-Q. Liu J. Qiu Z.-J. Dong Y. et al (2015). Distinct regulatory changes underlying differential expression of TEOSINTE BRANCHED1-CYCLOIDEA-PROLIFERATING CELL FACTOR genes associated with petal variations in zygomorphic flowers of Petrocosmea spp. of the family Gesneriaceae.Plant Physiol.1692138–2151. 10.1104/pp.15.01181
116
Yuan Z. Gao S. Xue D.-W. Luo D. Li L.-T. Ding S.-Y. et al (2009). RETARDED PALEA1 controls palea development and floral zygomorphy in rice.Plant Physiol.149235–244. 10.1104/pp.108.128231
117
Zheng X. Zhou M. Yoo H. Pruneda-Paz J. L. Spivey N. W. Kay S. A. et al (2015). Spatial and temporal regulation of biosynthesis of the plant immune signal salicylic acid.Proc. Natl. Acad. Sci. U.S.A.1129166–9173. 10.1073/pnas.1511182112
118
Zhou M. Li D. Li Z. Hu Q. Yang C. Zhu L. et al (2013). Constitutive expression of a miR319 gene alters plant development and enhances salt and drought tolerance in transgenic creeping bentgrass.Plant Physiol.1611375–1391. 10.1104/pp.112.208702
Summary
Keywords
transcription factor, TCP, development, evolution, plant hormones, signaling
Citation
Danisman S (2016) TCP Transcription Factors at the Interface between Environmental Challenges and the Plant’s Growth Responses. Front. Plant Sci. 7:1930. doi: 10.3389/fpls.2016.01930
Received
10 October 2016
Accepted
05 December 2016
Published
21 December 2016
Volume
7 - 2016
Edited by
José M. Romero, University of Seville, Spain
Reviewed by
Cristina Ferrandiz, Consejo Superior de Investigaciones Científicas – Instituto de Biologia Molecular y Celular de Plantas, Spain; Nobutaka Mitsuda, National Institute of Advanced Industrial Science and Technology, Japan; Daniel H. Gonzalez, National University of the Littoral, Argentina
Updates
Copyright
© 2016 Danisman.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Selahattin Danisman, selahattin.danisman@uni-bielefeld.de
This article was submitted to Plant Evolution and Development, a section of the journal Frontiers in Plant Science
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.