- State Key Laboratory of Crop Stress Biology for Arid Areas, College of Horticulture, Northwest A&F University, Yangling, China
Phytocystatins (PhyCys) comprise a group of inhibitors for cysteine proteinases in plants. They play a wide range of important roles in regulating endogenous processes and protecting plants against various environmental stresses, but the underlying mechanisms remain largely unknown. Here, we detailed the biological functions of MpCYS4, a member of cystatin genes isolated from Malus prunifolia. This gene was activated under water deficit, heat (40°C), exogenous abscisic acid (ABA), or methyl viologen (MV) (Tan et al., 2014a). At cellular level, MpCYS4 protein was found to be localized in the nucleus, cytoplasm, and plasma membrane of onion epidermal cells. Recombinant MpCYS4 cystatin expressed in Escherichia coli was purified and it exhibited cysteine protease inhibitor activity. Transgenic overexpression of MpCYS4 in Arabidopsis (Arabidopsis thaliana) and apple (Malus domestica) led to ABA hypersensitivity and series of ABA-associated phenotypes, such as enhanced ABA-induced stomatal closing, altered expression of many ABA/stress-responsive genes, and enhanced drought tolerance. Taken together, our results demonstrate that MpCYS4 is involved in ABA-mediated stress signal transduction and confers drought tolerance at least in part by enhancing stomatal closure and up-regulating the transcriptional levels of ABA- and drought-related genes. These findings provide new insights into the molecular mechanisms by which phytocystatins influence plant growth, development, and tolerance to stress.
Introduction
Cysteine proteinase inhibitors (cystatins) have been identified in many eukaryotes and extensively studied in insects and mammals (Rawlings et al., 2008). Plant cystatins, or phytocystatins (PhyCys), cluster into an independent evolutionary branch on cystatin phylogenetic tree. They are devoid of both putative glycosylation sites and disulfide bridges, and can inactivate catalytic activity of papain-like C1A cysteine proteinases or legumain-like C13 peptidases due to a tight and irreversible interaction (Martinez and Diaz, 2008; Benchabane et al., 2010). Phytocystatins could function in regulating the activity of endogenous cysteine proteinases during a variety of physiological processes, including plant growth and development, the reserves mobilization in seeds and tubers, programmed cell death, and senescence (Belenghi et al., 2003; Martinez et al., 2009; Weeda et al., 2009; Díaz-Mendoza et al., 2014). In addition, they also could inhibit exogenous proteases in the digestive tracts of insects (Outchkourov et al., 2004; Alvarez-Alfageme et al., 2007), protect plants from attacks by pests (Carrillo et al., 2011), and help prevent pathogen invasions (Popovic et al., 2013).
Accumulating evidence suggests that phytocystatins play an important role in plants exposed to stressful environmental conditions (van der Vyver et al., 2003; Zhang et al., 2008; Benchabane et al., 2010; Wang et al., 2012; Sun et al., 2014; Kunert et al., 2015). For example, transgenic tobacco (Nicotiana tabacum) plants expressing the rice cystatin, oryzacystatin-I (OC-I), are more resistant to the negative impacts that chilling stress can have on photosynthesis (van der Vyver et al., 2003). Similarly, Zhang et al. (2008) have shown that overexpression of two cystatins, AtCYSa and AtCYSb, in transformed Arabidopsis (Arabidopsis thaliana) is associated with enhanced tolerance to high salt, water deficit, cold, and oxidative stresses, and the amounts of APXb and NADP-ME protein are much higher in transgenic lines. Quain et al. (2014) reported that OC-I-inhibited cysteine proteinases could affect plant growth and increase drought stress resistance associated with strigolactone pathways. An AtCYS4 (Arabidopsis phytocystatin 4) transcriptional activator, DREB2C (dehydration-responsive element-binding protein 2C), has been identified that can bind to the AtCYS4 promoter and regulate the expression of AtCYS4 in response to heat stress (Je et al., 2014). Although these studies have greatly improved our understanding on the biological roles of PhyCys during plant response to environmental stresses, the underlying mechanisms at the molecular level remain largely unknown.
Plants are sessile organisms capable to adapt diverse adverse growth conditions, including drought, high salinity, heat, extreme light levels, and various metals in the soil. Among them, drought is one of the most serious stresses that affect agricultural production. When plants encounter water deficits, cells trigger a network of signaling events to reprogram their processes. Hormone-mediated signaling pathways, particularly those associated with the synthesis and perception of abscisic acid (ABA), are highly responsive to drought (Knight and Knight, 2001; Bray, 2002). ABA contents in the cells are significantly increased under drought conditions, thereby inducing stomatal closure and gene expression as part of a coping mechanism (Xie et al., 2006; Finkelstein, 2013; Clauw et al., 2015).
Apple (Malus domestica), one of the most widely cultivated woody plants in temperate regions, is the fourth most economically important fruit tree after Citrus sp., Vitis vinifera, and Musa sp. (Hummer and Janick, 2009). While, water deficits have become a critical source of abiotic stress that affect apple growth, productivity, and geographic distribution. Therefore, identifying drought-related genes and defining their functions will enrich our knowledge about drought-signaling networks in apple and will be important for improving the adaptability of apple to water-deficit conditions.
We previously identified 26 putative PhyCys genes within the entire apple genome and monitored their transcription patterns in the roots, stems, leaves, flowers, and seeds as well as in response to drought, low temperature (4°C), heat (40°C), ABA, and oxidative stress (Tan et al., 2014a). However, their biological roles had not yet been systematically evaluated. Here, we report that MpCYS4, GenBank Accession No. KF477275, original name MdCYS21, a stress-responsive cystatin gene isolated from M. prunifolia, acts as a positive regulator in drought stress responses, and its function may be due, at least partly, to its interaction within stress-responsive ABA signaling transduction that mediates stomatal regulation and a variety of ABA/stress-related genes expression to cope with drought conditions.
Materials and Methods
Plant Materials and Growth Conditions
In vitro shoot cultures of rootstock M26 from M. domestica were sub-cultured at 4-week intervals on an Murashige and Skoog (MS) agar medium containing 0.5 mg L−1 indole butyric acid (IBA) and 1.0 mg L−1 6-benzylaminopurine (6-BA). Initial growing conditions were 25°C, 100 μmol photons m−2 s−1, and a 16-h light/8-h dark photoperiod. For root induction, 4-week-old shoots were shifted to MS medium supplemented with 0.3 mg L−1 indoleacetic acid (IAA).
Seeds of Arabidopsis ecotype “Columbia” (Col-0) were first incubated in dark at 4°C for 3 days, then surface-sterilized according to the protocol described by Weigel and Glazebrook (2002), and sown on medium containing ½-strength MS salts, sucrose (3%, w/v), and agar (0.75%, w/v). Seedlings were transferred to soil (rich soil:vermiculite, 2:1, v/v) after germination for 7–10 days and grown in a climate chamber at 22°C, 70% relative humidity, 100 μmol photons m−2 s−1, and 16-h light/8-h dark photoperiod.
Subcellular Localization Analysis
For subcellular localization, the coding sequence of MpCYS4, without the termination codon, was cloned into the XhoI/SacI site of pBI221-GFP vector (Clontech, Palo Alto, CA, USA). The specific primers are shown in Table S1. For transient expression in onion epidermal cells, pBI221-MpCYS4-GFP plasmids or blank vector pBI221 were inserted into the cells using a PDS-1000 biolistic helium gun device (Bio-Rad, Hercules, CA, USA), as the protocol described by Diaz et al. (2005). Then, the cells were cultured on MS medium in dark for 24 h before observation of the fluorescent proteins with a LSM510 confocal laser-scanning microscope (Carl Zeiss, Thornwood, NY, USA).
Cysteine-Protease Inhibition Assay
The coding region of MpCYS4 was cloned into the EcoRV/XhoI site of pET-32a expression vector (Novagen, Gibbstown, NJ, USA), introduced into Escherichia coli BL21 strain. The specific primers are shown in Table S1. Bacterial cells expressing the fusion protein were harvested after induction with isopropyl β-D-thiogalactopyranoside (IPTG). The recombinant protein was purified using a nickel column chromatography procedure (Novagen) and quantified by method of Bradford (1976). Testing the capacity of the recombinant MpCYS4 protein to inhibit cysteine proteases has been described previously (Gaddour et al., 2001), using papain (EC3.4.22.2; Sigma, St. Louis, MO, USA) and benzoyl-L-arginine-p-nitroanilide (BANA) as substrate. Control assays used the equivalent molar amount of purified recombinant protein isolated from E. coli that harbored the empty pET-32a vectors. Absorbance of the enzyme reaction was recorded at 405 nm with a benchmark microplate reader (Bio-Rad).
Transformation and Regeneration of Arabidopsis
The coding region of MpCYS4 was cloned into the pBI121 vector (Clontech) under the CaMV 35S promoter. The specific primers are shown in Table S1. Subsequently, the resultant vectors were genetically transformed into Arabidopsis ecotype “Col-0.” Transformation was accomplished via the floral dip method using Agrobacterium tumefaciens strain EHA105 harboring the recombinant plasmid, as described by Clough and Bent (1998). Putative transgenic Arabidopsis plants were selected on MS medium containing 50 mg L−1 kanamycin. After further selecting at a 3:1 segregation ratio, T3 homozygous transgenic lines were derived for the phenotypic analysis.
Seed Germination Assay
For germination assays, Arabidopsis seeds after surface-sterilized, were placed on the germination media containing various concentrations of mannitol (an osmotic agent; Wang et al., 2011) or ABA (A1049; Sigma). Germination was considered complete when radicles had emerged by 1 mm. The experiments were repeated for three times, and approximately 50 seeds per line were sown for each experiment.
Physiological Measurements
During drought period, net photosynthesis (Pn) was monitored using a Li-6400 portable photosynthesis system (LiCor, Huntington Beach, CA, USA), at a constant airflow rate of 500 μmol photons m−2 s−1, with vapor pressure deficit of 2.0–3.4 kPa and cuvette CO2 concentration of 400 μmol CO2 mol−1 air. Data were obtained from light exposed, fully-expanded leaves on each of five plants. Values for relative water content (RWC) were determined according to method described by Gaxiola et al. (2001). Electrolyte leakage (EL) was determined from leaves as described by Dionisio-Sese and Tobita (1998), with an electrical conductivity meter (DSS-307; SPSIC, Shanghai, China). Chlorophyll was extracted with 80% acetone, and the concentration was determined spectrophotometrically according to the method of Lichtenthaler and Wellburn (1983). ABA was extracted and measured as described by Li et al. (2015), with a high-performance liquid chromatograph (HPLC) (LC2010A; Shimadzu, Kyoto, Japan).
Stomatal Aperture Analysis
The ABA-induced stomatal closure was measured from leaves of 4-week-old plants that had been detached and floated abaxial side down on a stomatal opening solution containing 10 mM MES-KOH (pH 6.15), 20 mM KCl, and 1 mM CaCl2. After they were incubated for 2 h under lights, they were then treated for 2 h with various concentrations of ABA added to the solution. Obtained from epidermal peels, the stomata were photographed and counted with a JSM-6360LV scanning electronic microscope (JEOL Ltd., Tokyo, Japan) as previously described (Tan et al., 2014b). Their sizes were measured using Image J software, using at least 50 apertures from each treatment.
Stress Tolerance Assay in Arabidopsis
The rate of transpiration (water loss) was evaluated by placing detached, fresh Arabidopsis leaves on open Petri dishes with abaxial side up and weighing them at regular intervals. The treatment was conducted at room temperature. Third to fourth true rosette leaves were collected from 3-week-old soil-grown plants under standard growing conditions as described above. In each experiment, approximately 50 leaves per line were used and the entire test was repeated for three times.
To assess drought resistance of the soil-grown plants, 1-week-old seedlings were transplanted in soil for 3 weeks under standard growing conditions before being subjected to water deficit by withholding water for 14 days (severe stress). The leaf RWC and stomatal apertures were measured from tissues sampled after 7 days. Digital images and plant survival rates were recorded on Day 14. Experimental variations were minimized by growing the same number of plants on each tray. The entire test was repeated at least three times.
For ABA treatment, 100 μM (±)-ABA (Sigma; A1049) was sprayed onto 5-day-old seedlings grown on MS plates to ensure total foliar coverage. In parallel experiments, water-only was sprayed as the control. Samples were collected after 0, 3, 6, 12, or 24 h of treatment, then frozen in liquid nitrogen and stored at −80°C for RNA extractions.
Genetic Transformation and Stress Tolerance Assay in Apple
The MpCYS4 overexpression vector (pBI121-MpCYS4) was transferred into apple by Agrobacterium-mediated transformation, using leaf discs of apple rootstock M26, as described by Kotoda et al. (2010). After cultivation in dark for 2 weeks, the explants were moved under lights. Their shoots were excised and then propagated on multiplication medium containing 50 mg L−1 kanamycin. The rooted plantlets were transferred to plastic pots (4 × 5 × 5 cm) filled with a mixture of forest soil, sand, perlite, and vermiculite. Growth conditions were 24 ± 2°C, 70 ± 5% relative humidity, 100 μmol photons m−2 s−1, and a 16-h light/8-h dark photoperiod.
After 2 months, the plants were transplanted in plastic culture pots (30 × 26 × 22 cm) contained a mixture of forest soil:organic substrate:sand (5:1:1, v:v:v), and placed in the greenhouse under ambient light, with temperature ranging from 20 to 35°C and a relative humidity of 65–70% at Northwest A&F University, Yangling, China (34°20′N, 108°24′E). After 3 months of growth under this condition, healthy and uniformly sized plants (30 per line, for a total of 60) were subjected to a water deficit. Another 30 plants per line (Control) continued to receive irrigation daily to maintain the saturated soil water content. Soil water content (v/v) was determined with an HH2 Moisture Meter (Delta-T Devices, Cambridge, UK). Drought was induced by withholding water for 8 days. At 9:00 a.m. on alternate days, Pn was recorded on the ninth to twelfth leaves from the stem base of five plants with similar soil water content. Then, the leaves were rapidly frozen in liquid nitrogen and stored at −80°C for RNA extractions. At each time point, another set of samples was collected to determine leaf RWC. After 3 days of treatment, additional leaves were sampled for stomatal observation, ABA measurement, and RNA-Seq analysis. Leaf chlorophyll concentrations and EL values were measured at the final time point.
RNA Extraction, Quantification, and Gene Expression Analysis
Total RNA was isolated using E.Z.N.A.® Plant RNA Kit (Omega Biotek, Norcross, GA, USA) according to the manufacturer instructions. RNA integrity was checked on a 1.2% agarose gel and RNA quantity was assessed with a NanoDrop 1000 Spectrophotometer (Thermo Scientific, Wilmington, DE, USA). First-strand cDNAs were synthesized from 2 μg of total RNA with SYBR Prime Script RT-PCR Kit II (TaKaRa, Tokyo, Japan).
Semi-quantitative RT-PCRs were performed to examine the transcript levels of MpCYS4 in apple and Arabidopsis. Apple Actin and Arabidopsis Actin were used as respective loading controls. Quantitative real-time PCR (qRT-PCR) was conducted on an iQ5.0 detection instrument (Bio-Rad) using SYBR Green qPCR kits (TaKaRa). Reactions were performed in triplicate with a volume of 20 μL, and apple EF-1α or Arabidopsis Actin was amplified as the appropriate internal control. The relative quantity of target gene transcript was determined by applying the 2−ΔΔCT method (Livak and Schmittgen, 2001). Primers are listed in Tables S1, S2. Each value was expressed as mean and standard deviation (SD) calculated from the result of three independent replicates.
RNA-Seq Analysis
Transcriptional profiling of WT apple and transgenic line #4 was conducted by RNA-Seq. Three biological replicates were used for each genotype, with samples collected from plants grown under normal conditions as well as after drought induced by withholding irrigation for 3 days. Each biological replicate comprised samples from three to four plants that were mixed after collection to generate one independent pool. Total RNA (2 μg per sample) was prepared and used for both cDNA library construction (Zhong et al., 2011) and RNA-Seq by the Gene Denovo Biotechnology Corporation (Guangzhou, China), with an Illumina HiSeq™ 2500 (Illumina, San Diego, CA, USA). The files of raw fastq were checked by FastQC (http://www.bioinformatics.babraham.ac.uk/projects/fastqc). After the adaptor sequences were removed using fastqmcf (https://code.google.com/p/ea-utils/wiki/FastqMcf), the reads were aligned to the database for M. domestica genome (Velasco et al., 2010; http://www.nature.com/ng/journal/v42/n10/full/ng.654.html) using TopHat2 (Kim et al., 2013). All usable reads were then normalized into fragments per kilobase of transcript per million mapped reads (FPKM) (Mortazavi et al., 2008). The transcript abundance differences among samples were computed with the ratio of FPKM values. The significance of those differences was evaluated with a false discovery rate (FDR) as the threshold of P-value for multiple tests (Yekutieli and Benjamini, 2001). Genes with FDR <0.05 and fold-changes >2 were used in later analyses. Our methods for assessing the significance of differentially expressed genes (DEGs) followed those described by Anders and Huber (2010).
Statistical Analysis
All statistical analyses were conducted using SigmaPlot software (Systat Software). Data were analyzed via one-way ANOVA using the SPSS-11. Statistical differences were compared based on Student's t-tests. *, **, and *** indicates significance level at P < 0.05, P < 0.01, and P < 0.001, respectively.
Results
Molecular Characterization, Subcellular Localization, and Cysteine-Protease Inhibition of MpCYS4
MpCYS4 encoded a phytocystatin protein containing 248 amino acids with a predicted molecular weight of 27.72 kDa. Characteristic sequences and motifs of MpCYS4 are highlighted in Figure 1A, including the reactive site QVVAG in the central part, two glycine residues (GG) near the N-terminal, PW residues near the C-terminal, and a SNSL motif in the C-terminal extension. The typical sequence of PhyCys, LARFAV, was also localized at the N-terminus of the protein.
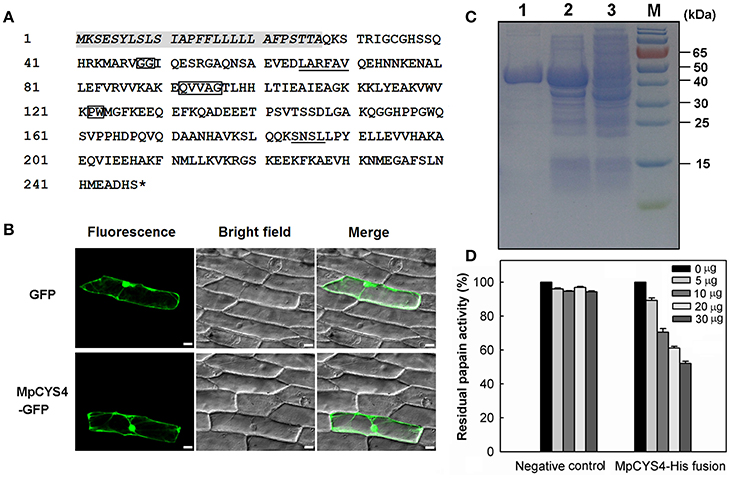
Figure 1. Amino acid sequence analysis, subcellular localization, and cysteine-protease inhibition of MpCYS4. (A) Deduced amino acid sequence of MpCYS4, with putative signal peptide italicized. Conserved signature sequences of PhyCys are enclosed with rectangles (N-terminal G; QXVXG; P/AW); LARFAV-like motif and SNSL motif are underlined. *Represents the termination codon; (B) Subcellular localization of MpCYS4–GFP fusion protein in onion epidermal cells. GFP alone or MpCYS4–GFP, corresponding bright field images, and overlay from merging of bright and fluorescent illumination are shown. GFP or MpCYS4–GFP fusion was driven by CaMV 35S promoter. Onion epidermal peels were bombarded with DNA-coated gold particles, and GFP expression was visualized 24 h later. Scale bar, 50 μm; (C) SDS-PAGE (12%) analysis of bacterial expression and purification of MpCYS4 representing (1) purified recombinant MpCYS4–His fusion protein, (2) total soluble protein fraction after 5 h of IPTG induction, and (3) total soluble protein fraction without IPTG induction; (D) In vitro inhibition of cysteine protease activity by recombinant MpCYS4 protein. Inhibition of recombinant MpCYS4 protein to papain expressed as residual enzyme activity in presence of increasing inhibitor concentrations. Purified induced protein of empty pET32a was used as negative control. Data are means ± SD from 3 independent experiments.
To investigate the subcellular localization of MpCYS4 protein, we fused it with GFP at the N-terminal and transiently expressed in onion epidermal cells. After 24 h, we detected the accumulation of MpCYS4-GFP fusion protein in the cell membrane, cytoplasm, and nucleus of those cells, which was the same as the subcellular localization of the GFP protein (Figure 1B). This result was consistent with previous reports for other cystatins, such as BvM14-cystatin from Beta vulgaris “M14” (Wang et al., 2012) and GsCPI14 from Glycine soja (Sun et al., 2014).
His-tagged fusion protein of MpCYS4 was expressed in E. coli cells and affinity-purified (Figure 1C). To confirm that this gene is a functional cysteine protease inhibitor, we used an in vitro assay with papain as substrate. Testing the behavior of the recombinant MpCYS4 protein against cysteine protease revealed that papain activity was blocked in a concentration-dependent manner (Figure 1D). Meanwhile, when the purified and induced protein of empty pET32a was added into the reaction as a negative control, this activity was not inhibited.
MpCYS4 Overexpression Enhanced the Drought Tolerance of Arabidopsis
We previously demonstrated that MpCYS4 is up-regulated by drought, heat, exogenous ABA, or MV treatments in leaves of M. prunifolia (Tan et al., 2014a). We further investigated its potential functioning during abiotic stress responses by analyzing the phenotypes of MpCYS4-overexpressing plants. The coding sequence of MpCYS4 was inserted into a transgenic vector and constitutively expressed in transgenic Arabidopsis plants. In all, we obtained 16 independent T3 homozygous lines and verified them via PCR analysis (data not shown). Two representative lines with the highest expression—overexpressing (OE)-4 and OE-13—were selected for further examination (Figures S1A,B). On a MS growth medium, seed germination and elongation of the primary roots from PCaMV35S:MpCYS4 was retarded when compared with the wild-type (WT) (Figures S1C–E), while no obvious difference was observed for soil-grown plants in their aerial parts.
Because MpCYS4 expression was strongly induced by water deficit in M. prunifolia (Tan et al., 2014a), our study focused on the drought tolerance of 35S:MpCYS4 transgenic Arabidopsis. Seeds from the 35S:MpCYS4 transgenic and WT plants were germinated and seedlings were then grown on a MS medium containing 400 mM mannitol. Both germination and post-germinative growth of all genotypes were repressed, albeit to varying degrees. For example, the seeds of transgenic lines (OE-4 and OE-13) germinated much faster compared to the WT. After 14 days, only 19% of the latter had germinated compared with 83% for OE-4 and 73% for OE-13 (Figure S1C). Seedlings of the transgenic lines also grew faster and had longer primary roots and higher fresh weights (FWs) than the WT (Figures S1D,E). The results suggested that MpCYS4 overexpression alleviates the adverse effects induced by osmotic stress on Arabidopsis growth.
The rate of water loss was slower for 35S:MpCYS4 plants than the WT, based on FW measurements from detached rosette leaves. As shown in Figure 2A, after 3 h on open Petri dishes, the FWs of detached rosette leaves were reduced to 69% of the original value for OE-4, 66.7% for OE-13, and 60.1% for the WT. In addition, the survival rates for 35S:MpCYS4 plants deprived of water for 14 days were higher (77.8% for OE-4, 73.9% for OE-13) than for similarly treated WT plants (10.7%) (Figure 2B). These results confirmed that overexpression of MpCYS4 could greatly improve drought resistance in transgenic Arabidopsis.
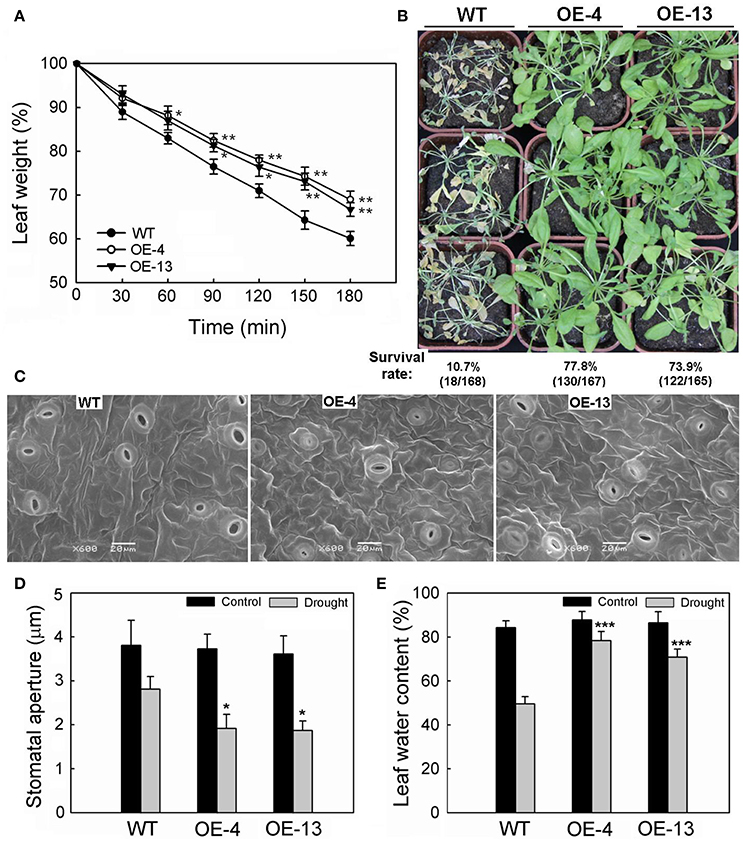
Figure 2. Responses to drought by 35S:MpCYS4 and wild-type (WT) Arabidopsis. (A) Transpiration rates. Leaves at same developmental stages were excised and weighed at various time points after detachment. Data are means ± SD from 3 independent experiments. Values were significantly different from WT at *P < 0.05 or **P < 0.01, based on Student's t-tests; (B) Drought tolerance test. Watering of 4-week-old plants was stopped for 14 days. Survival rates (% indicated below each line) were calculated as number of surviving plants divided by total number of plants tested in 3 independent experiments; (C) Stomatal apertures of WT (left), 35S:MpCYS4 transgenic plants OE-4 (center), and OE-13 (right). Stomatal guard cells were observed during middle of water-deficit period via scanning electron microscopy. Scale bar, 20 μm; (D) Measurement of stomatal aperture on WT and transgenic (OE-4 and OE-13) plants corresponding to (C). Data are mean ratios from 3 independent experiments. At least 50 stomatal apertures were measured per line per experiment. *Values were significantly different from WT at P < 0.05, based on Student's t-tests. (E) Relative water content in WT and transgenic lines OE-4 and OE-13 after 7 days of drought treatment. Data are means ± SD from 3 independent experiments. ***Values were significantly different from WT at P < 0.001, based on Student's t-tests.
MpCYS4 Affected Stomatal Behavior in Response to Drought Stress
The function of MpCYS4 in controlling water losses prompted our investigation into the operation of stomatal behavior, a major factor affecting water-holding capacity in plant leaves. The stomatal density did not show significant difference between 35S:MpCYS4 Arabidopsis and WT plants before and after 7 days of drought treatment (Figure S2), which suggested that the higher drought resistance of 35S:MpCYS4 Arabidopsis is unrelated to stomatal density. Whereas, compared with the WT, stomatal apertures were smaller on 35S:MpCYS4 leaves when subjected to water-deficit conditions (Figure 2C). As shown in Figure 2D, no obvious difference was detected among 35S:MpCYS4 and wild-type plants under normal conditions. However, drought treatment led to a higher occurrence of closure in the transgenic lines, with stomatal dimensions for the WT being reduced to approximately 74% of the size measured from untreated plants vs. reductions of 51.5 and 51.8% of the original, unstressed sizes for transgenic lines OE-4 and OE-13, respectively. Consistent with these results, the RWC of leaves in the WT was 49.6%, as opposed to 78.4% for OE-4 and 70.9% for OE-13 after 7 days of drought treatment (Figure 2E). These findings indicated that, during periods of dehydration, the stomata of the transgenic lines responded to water deficits better than did the WT plants.
Transgenic Arabidopsis Plants Over-Expressing MpCYS4 Were Hypersensitive to Abscisic Acid
Stomatal closure is one of the most important ABA-induced environmental responses. Then, we examined 35S:MpCYS4 plants to determine whether MpCYS4 overexpression affects ABA sensitivity. When germinated on an MS medium supplemented with ABA, both the germination and post-germinative developmental stages of the 35S:MpCYS4 proved hyposensitive to exogenous ABA compared with the WT (Figures 3A,B). In the presence of 0.25 μM ABA, 35S:MpCYS4 seedlings were dramatically restrained while the WT seedlings showed expanded, green cotyledons (Figure 3A). To determine whether MpCYS4 is involved in ABA-induced stomatal movement, we treated WT and 35S:MpCYS4 leaves with ABA and found that such treatment resulted in a higher degree of closure in the transgenic plants (Figure 3C). Thus, MpCYS4 seems to have an important role in ABA-mediated control of the guard cells.
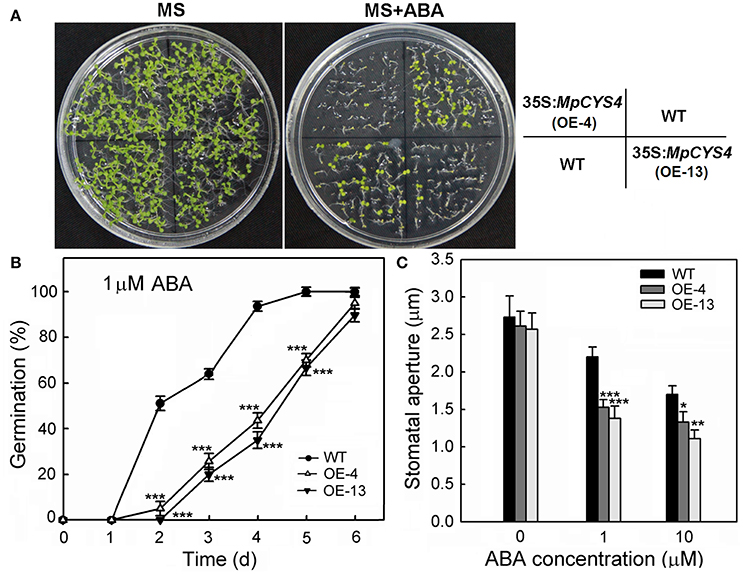
Figure 3. Responses of wild-type (WT) and 35S:MpCYS4 Arabidopsis upon ABA treatment. (A) ABA sensitivity in seedlings. Approximately 50 seeds from 4 independent seed lots of simultaneously grown WT and 35S:MpCYS4 (lines OE-4 and OE-13) were surface-sterilized and sown on 1/2 Murashige and Skoog (MS) agar plates supplemented with ABA (mixed isomers; Sigma A1049). Images were taken on Day 9 after germination on agar medium supplemented with 0 μM or 0.25 μM ABA; (B) Germination rates after 6 days. Seeds were surface-sterilized and sown on 1/2 MS agar plates with 1 μM ABA. Results are means ± SD from 3 independent experiments (approximately 50 seeds per line per experiment); (C) Size of stomatal apertures. Stomata were fully opened prior to ABA treatment. Rosette leaves of 4-week-old plants were detached and floated abaxial side down on opening solution for 2 h prior to ABA treatment. Leaves were then treated with 0, 1, or 10 μM ABA for 2 h before apertures were measured. Stomatal apertures in epidermal peels were observed under scanning electronic microscope. Data are mean ratios ± SD from 3 independent experiments. At least 50 stomatal apertures were measured per treatment. Values were significantly different from WT at *P < 0.05, **P < 0.01, or ***P < 0.001, based on Student's t-tests.
Constitutive Expression of MpCYS4 Affected Expression of ABA-Responsive Genes
Our germination and post-germination assays revealed an increased sensitivity by 35S:MpCYS4 plants under ABA exposure, while endogenous ABA levels did not show significant difference between 35S:MpCYS4 Arabidopsis and WT seedlings before and after subjected to 100 μM ABA for 24 h (Figure S3). The specific and dramatic induction of expression for stress genes represented a prominent response to ABA at the molecular level. Therefore, we used 35S:MpCYS4 and WT plants treated with 100 μM ABA to assess the expression patterns of several ABA-responsive genes by qRT-PCR.
Upon ABA treatment, three A-type protein phosphatase 2C (PP2C) family members—ABA-INSENSITIVE1 (ABI1), ABI2, and AtPP2C—were induced in the WT and even more strongly in 35S:MpCYS4 seedlings (Figure 4). Other genes in the molecular network of the ABA signaling pathway were also tested, e.g., OPEN STOMATA1 (OST1); the NADPH oxidase F (AtrbohF); the anion channel SLOW ANION CHANNEL-ASSOCIATED 1 (SLAC1); ABA-responsive promoter element (ABRE) binding transcription factors ABI5 and ABF3; and other downstream targets such as LOW TEMPERATURE-INDUCED 65-kDa PROTEIN (RD29B), RESPONSIVE TO ABA 18 (RAB18), RESPONSIVE TO DEHYDRATION 22 (RD22), and COLD-REGULATED PROTEIN COR6.6 (KIN2), which serve as marker genes for monitoring ABA and stress responses in plants. In our experiments, those genes were highly induced upon ABA treatment in the WT plants and even significantly more in the 35S:MpCYS4 plants. Although expression was detected for all of them, the extent and kinetics of this induction differed among markers. These findings suggested that MpCYS4 could interact with ABA-mediated signaling process and affect multiple ABA-responsive genes.
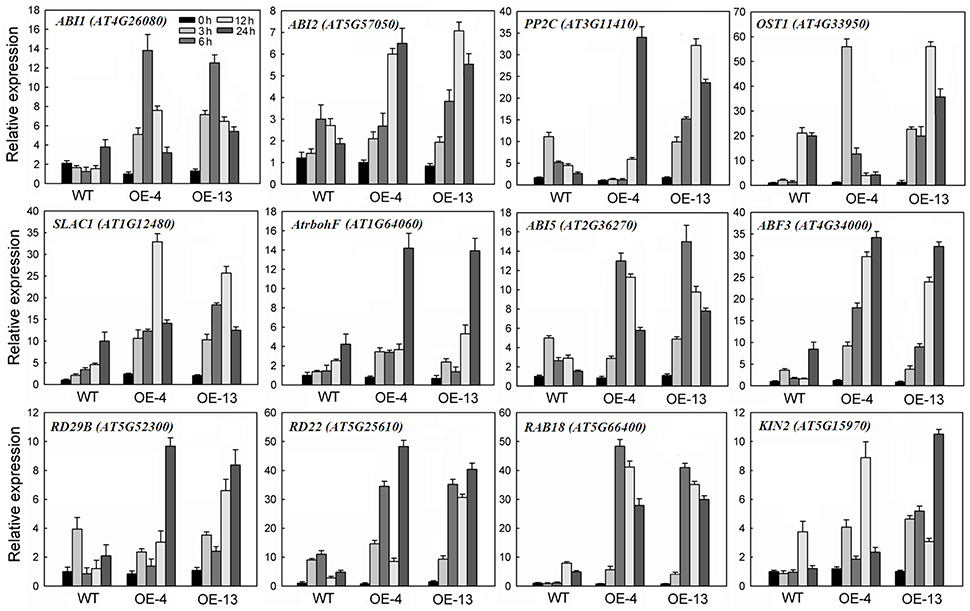
Figure 4. Quantitative real-time PCR analysis of expression of ABA- and stress-responsive genes in wild-type (WT) and 35S:MpCYS4 transgenic Arabidopsis, as induced by 100 μM ABA. All transcript levels were normalized relative to WT under non-treated control (0 h) conditions. AtActin served as reference gene. Data are means ± SD of 3 independent experiments.
MpCYS4 Overexpression Enhanced Drought Tolerance of Transgenic Apple and Promoted Stomatal Closure in Response to Drought Stress
To examine whether MpCYS4 confers tolerance to drought in apple, MpCYS4 transgenic apples were obtained. The three MpCYS4-overexpressing lines—#1, #3, and #4—which were detected with higher MpCYS4 transcript level were chosen for further investigation (Figures S4A,B). Under normal growing conditions, the aerial portions of those transgenic plants showed apparent dwarfing and slower development when compared with the traits of the WT (Figures S4C–F). However, the root systems did not show obviously different among the transgenic lines and the WT (data not shown). Drought stress was induced by withholding water from these plants for 8 days. No obvious difference was observed among the tested lines on their growth potential at the onset of the experiment. While, the transgenic lines grew much better than the WT as the stress period was prolonged. The rate of net photosynthesis decreased rapidly after drought treatment, with rates being significantly lower for the WT than for 35S:MpCYS4 plants (Figure 5A). In addition, the RWC calculated for leaves from each line was significantly reduced under drought conditions. However, overexpression of MpCYS4 alleviated this response obviously over time (Figure 5B). Consistently, this phenotype was also confirmed by other measurements, with the transgenic lines having much less EL and higher chlorophyll concentrations than the WT at the end of the treatment period (Figures 5C,D).
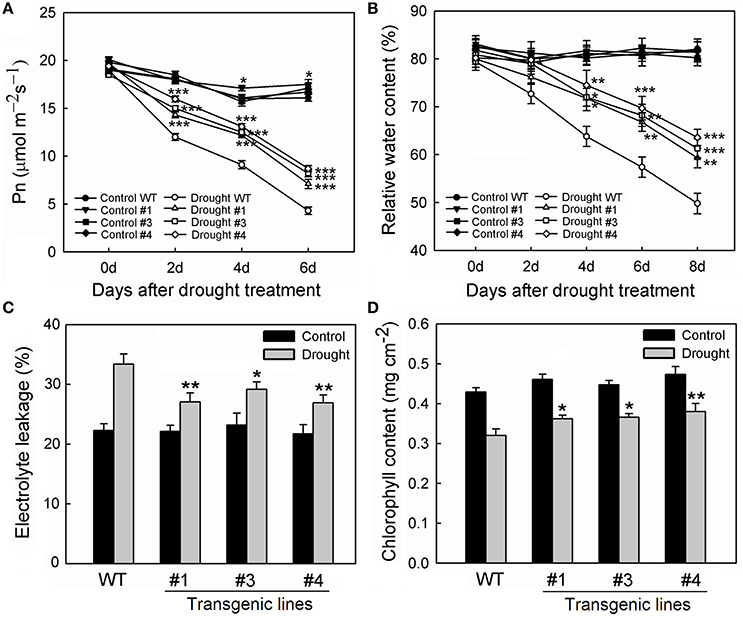
Figure 5. Comparison of drought tolerance among wild-type (WT) and 35S:MpCYS4 transgenic apple lines #1, #3, and #4. Plants of all genotypes were subjected to drought by withholding water for 8 days. Net photosynthetic rate (Pn) (A) and relative water content (B) were measured on alternate days; electrolyte leakage (C) and chlorophyll concentration (D) were measured on Day 8 of treatment. Results are means ± SD from 3 independent experiments. Values were significantly different from WT at *P < 0.05, **P < 0.01 or ***P < 0.001, based on Student's t-tests.
Meanwhile, the stomata characters of the 35S:MpCYS4 plants and WT plants were checked using scanning electron microscopy after water was withheld for 3 days. Under normal conditions, both the stomatal density and aperture sizes did not differ significantly between the two genotypes (Figure S5; Figures 6A,B). However, the transgenic lines showed much greater stomatal closure than the WT when drought stress was induced (Figure 6B). Stomatal apertures were also measured from overexpressing and WT plants in response to ABA treatment. In the absence of ABA, sizes were not obviously different among genotypes. However, after 2 h of exposure to 50 or 100 μM ABA, the degree of closure was greater in the 35S:MpCYS4 plants (Figure 6C), thereby indicating that they had a drought-insensitive phenotype associated with ABA-mediated stomatal closure under the water deficit. Then we examined 35S:MpCYS4 apple plants to determine whether MpCYS4 overexpression affects endogenous ABA levels. As shown in Figure S6, no obvious difference was detected among 35S:MpCYS4 and WT plants under normal conditions. Drought stress increased the levels of ABA in both WT and MpCYS4 transgenic lines, but no significant differences appeared among them. These data indicated that MpCYS4 could function in plants ABA sensitivity but not in the biosynthesis of ABA under drought stress conditions.
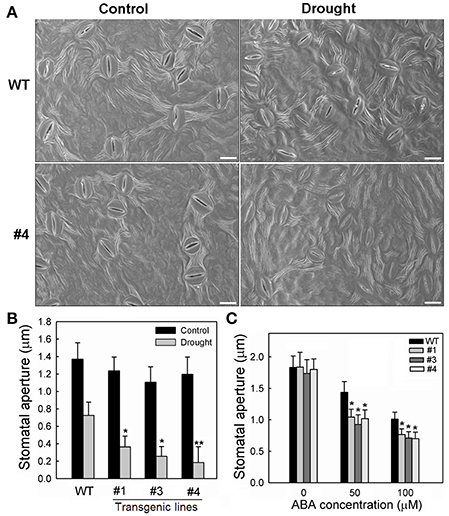
Figure 6. Stomatal responses by wild-type (WT) and 35S:MpCYS4 transgenic apple lines #1, #3, and #4 under drought or ABA treatment. (A) Stomatal guard cells of WT and transgenic lines were observed on Day 3 of water-deficit period via scanning electron microscopy. Representative photographs for stomata from WT and #4. Scale bar, 20 μm; (B) Quantitative data for stomatal apertures on leaves from tested plants; (C) Effects of ABA treatment on apertures of WT and MpCYS4-overexpressing lines #1, #3, and #4. Data are mean ratios ± SD of 3 independent experiments. At least 50 stomatal apertures were measured per line per experiment. Values were significantly different from WT at *P < 0.05 or **P < 0.01, based on Student's t-tests.
MpCYS4 Overexpression Led to Dramatic Transcriptomic Alterations in Apple Leaves
To obtain further insight into the molecular mechanisms by which MpCYS4 mediates tolerance to drought stress in apple, we performed a large-scale RNA sequencing (RNA-Seq) with RNA extracts from leaves of WT and #4 transgenic plants either grown under normal conditions or subjected to drought by withholding irrigation for 3 days. A total of 12 samples were used with three biological replicates for each genotype. Clean reads were obtained after removing rRNA contaminated reads, with mapping to the genome sequence made up approximately 70% (Table S3). To further evaluate the robustness of the RNA-Seq data, we calculated the correlation coefficients of the transcriptome profiles among the 12 samples. The value could reach 0.99 between each set of biological replicates (Table S4). Based on a two-fold criterion, 82 and 69 genes were up and down-regulated, respectively, in the transgenic line when compared with the WT in the absence of drought stress (Figure S7A). Under water deficit, even more genes exhibited differential expression in line #4 than in WT, with 1556 and 1327 genes being up and down-regulated, respectively (Figure S7B). Among the DEGs, 15 and 8 were commonly up or down-regulated, respectively, in #4 under control and after water was withheld for 3 days (Figure S7C).
It is worth mentioning that many DEGs involved in ABA signaling transduction and stress response were identified under water-deficit conditions (Table 1), including PYL4 (MDP0000228470) and PYL9 (MDP0000284624), which were down-regulated in stressed transgenic line. By contrast, genes encoding ABI1 (MDP0000437033), ABI2 (MDP0000231674), HAB1 (MDP0000265371 and MDP0000178692), OST1 (MDP0000224969), and the ABRE binding transcription factor ABF3 (MDP0000701734 and MDP0000248567) were up-regulated in those transgenic plants. Furthermore, transcript levels of several other regulatory proteins associated with stress signal transduction, such as mitogen-activated protein kinase, calcium-dependent protein kinase and transcription factors, including NAC, WRKY, MYB, MYC, MADS, DREB, basic helix-loop-helix (bHLH) transcription factor, heat stress transcription factor (HSF), and APETALA2/ethylene-responsive factor (AP2/ERF) were activated in the overexpression line. In addition, the genes encoding major stress-responsive functional proteins that have been demonstrated to play a direct role in stress response, such as ascorbate peroxidase (APX), catalase (CAT), glutathione S-transferase (GST), late embryogenesis abundant protein (LEA), RD29B, RD22, and KIN2 were also enriched in drought-treated 35S:MpCYS4 plants. To validate the results from this RNA-Seq analysis, we used qRT-PCR to evaluate the expression of 12 of those genes (Figure 7). It was evident that the expression patterns of those selected genes were largely consistent with what we found from our RNA-Seq data despite the difference in absolute fold-changes between the two methods. This demonstrated that our RNA-Seq results were reliable.
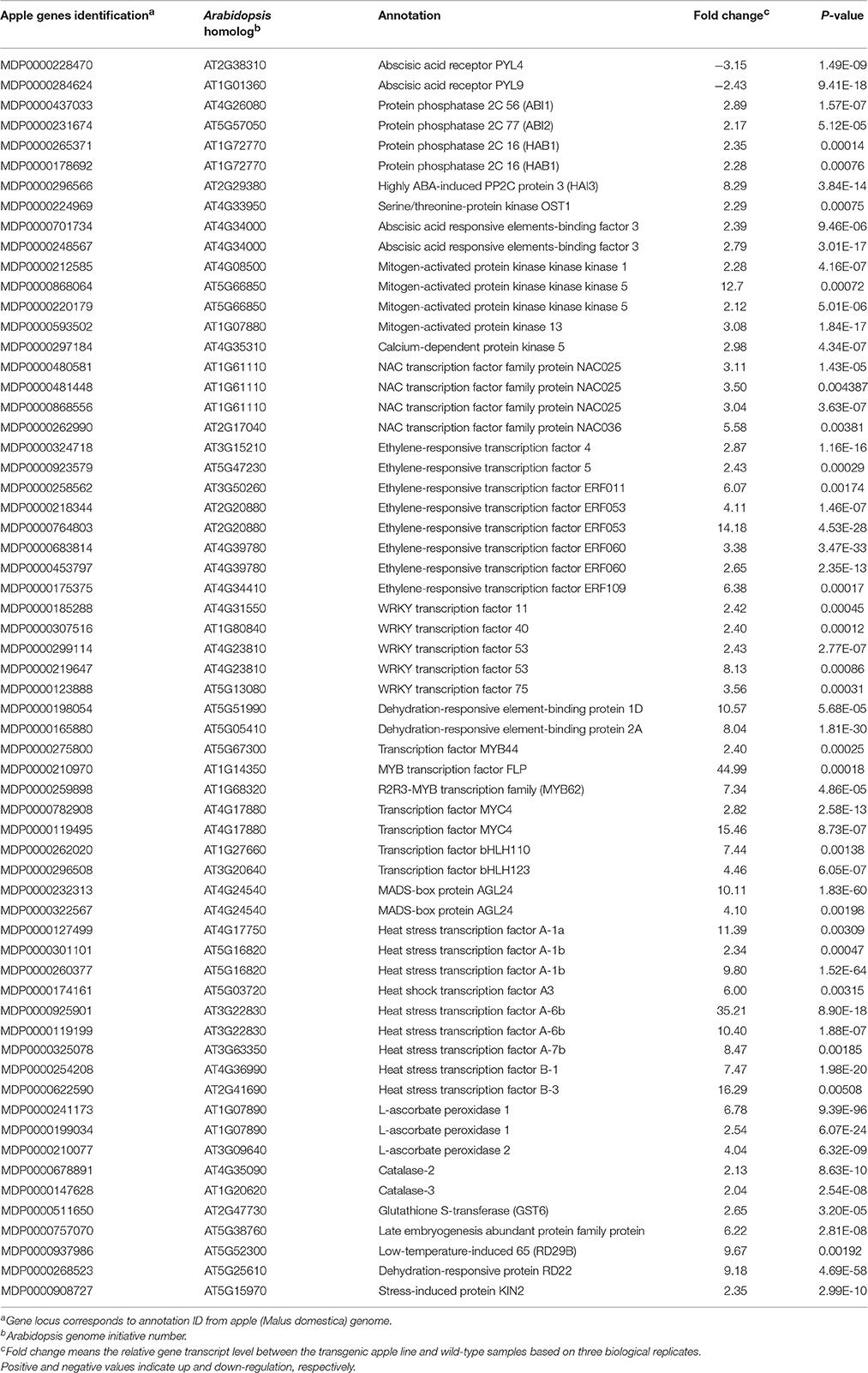
Table 1. Differentially expressed genes (DEGs) involved in ABA signaling transduction and stress response in the transgenic apple compared to wild-type under drought stress.
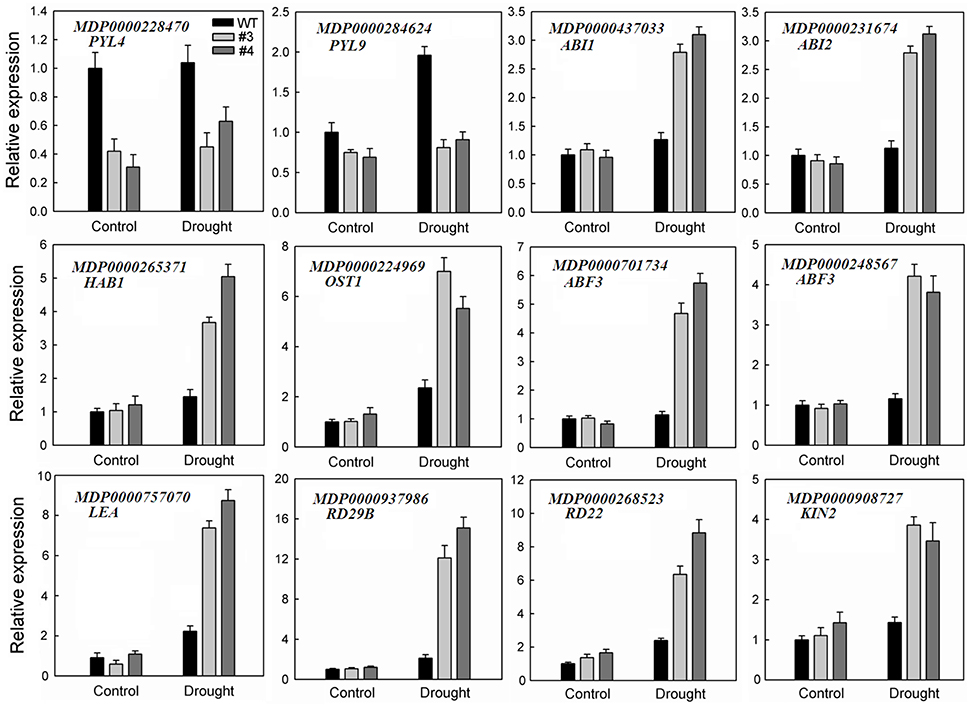
Figure 7. Validation via quantitative real-time RT-PCR analysis of selected transcriptome-based ABA and stress-responsive differentially expressed genes (DEGs) in 35S:MpCYS4 transgenic apple compared with wild-type (WT) under drought conditions. All transcript levels were normalized relative to WT for non-treated control. MdEF-1α served as reference gene. Data are means ± SD of 3 independent experiments.
Discussion
Environmental stresses, such as water deficits, high salt, or extreme temperatures, have adverse impacts on plant growth and development. Plants adapt and can become tolerant to these challenging conditions through various biochemical and physiological processes. In doing so, many stress-related genes are induced and responses are activated. The PhyCys are thought to function in regulating endogenous processes and guarding against various adverse growing conditions. Here, we focused on evaluating the biological roles of MpCYS4, a cystatin gene isolated from M. prunifolia.
MpCYS4, which contains one cystatin-like domain and characteristic motifs of phytocystatins, is most homologous to AtCYS-6 (At3g12490), HvCPI-4 (AJ748344), and OC-XII (Os01g16430) within Group C of the four cystatin groups (Tan et al., 2014a). After conducting the biochemical assay using purified MpCYS4 protein, we found that it activity to inhibit the catalytic activity of papain, consistent with earlier reports about cystatins isolated from Arabidopsis (Zhang et al., 2008), Hordeum vulgare (Gaddour et al., 2001), and Brassica oleracea (Eason et al., 2014). Expression analysis showed a pattern of MpCYS4 activation under water deficit, exposure to 40°C, or treatment with exogenous ABA or MV (Tan et al., 2014a), suggesting its involvement in the response to environmental stresses. Subsequently, we introduced MpCYS4 into Arabidopsis and apple to characterize its biological functions in detail.
Stomata are present on the leaf surface bordered by a pair of guard cells, and allow plants to regulate gas exchange and water loss by transpiration (Xie et al., 2012). Their opening and closing is affected by environmental and internal parameters that maintain the water balance and functioning of complex signal transduction pathways (Hetherington and Woodward, 2003; Clauw et al., 2015). Stomatal closure is a key ABA-mediated process for coping with water deficits (Cutler et al., 2010; Lee et al., 2013). ABA-insensitive mutants (i.e., abi1 and abi2) are very susceptible to drought because regulation of their stomatal apertures is impaired (Schroeder et al., 2001). We found that overexpression of MpCYS4 resulted in reduced water loss and enhanced drought resistance. Furthermore, the stomatal openings of 35S:MpCYS4 transgenic plants were smaller than those of WT under water-deficit conditions, and stomatal closure was more sensitive in 35S:MpCYS4 plants than in the WT after ABA treatment. These phenotypes were observed both in transgenic Arabidopsis and apple. Therefore, during water deficits, MpCYS4 could affect ABA-mediated stomatal closure that minimizing water loss through transpiration to cope with drought conditions.
ABA is also associated with the regulation of complex signal transduction that subsequently induces the expression of stress-responsive genes to confer plant resistance (Shinozaki and Yamaguchi-Shinozaki, 2007; Pierik and Testerink, 2014). In our experiments, upon ABA treatment, we could not find significant differences between 35S:MpCYS4 Arabidopsis and WT plants in endogenous ABA levels, while expression increased for genes that encode Ser/Thr PP2Cs, ABI1, ABI2, and AtPP2CA, in 35S:MpCYS4 Arabidopsis. These proteins belong to Group A PP2Cs and play negative roles in plant ABA signal transduction (Ma et al., 2009; Park et al., 2009).
Transcript levels of OST1, which encodes an SNF1-related protein kinase 2 (SnRK2)-type protein kinase participated in ABA-mediated stomatal closure (Xie et al., 2006), were also apparently higher in the 35S:MpCYS4 Arabidopsis. This gene activates the anion channel SLAC1 through phosphorylation, thereby regulating guard cell turgor and stomatal apertures (Geiger et al., 2009; Lee et al., 2009). Moreover, the plasma membrane-localized NADPH oxidase, AtrbohF, which contributes to H2O2 generation in guard cells, is also targeted by OST1 (Kwak et al., 2003; Sirichandra et al., 2009). Consistently, the enhanced expression of SLAC1 and AtrbohF was detected in our MpCYS4-overexpressing Arabidopsis, perhaps associated with the activating of OST1 protein kinase. ABA is considered fundamental for triggering the activity of channels or transporters on guard cell membrane, thereby decreasing turgor, inducing stomatal closure (Brodribb and McAdam, 2013), and, ultimately, elevating the synthesis of H2O2 in guard cells via NADPH oxidase, thus, increased levels of H2O2 modulate ABA-controlled closure (Pei et al., 2000; Xie et al., 2014). So, we might conclude the elevated expression of OST1, SLAC1, and AtrbohF contributes to the increased ABA-induced stomatal closure in 35S:MpCYS4 Arabidopsis. Furthermore, ABRE binding transcription factors ABI5 and ABF3, as well as other downstream targets RD29B, RD22, RAB18, and KIN2, showed enhanced transcription in ABA-treated 35S:MpCYS4 Arabidopsis. Thus, the altered expression of these genes may be responsible for ABA sensitivity in transgenic plants.
In addition, the RNA-Seq data revealed that MpCYS4 overexpression led to a comprehensive transcriptomic alteration in the transgenic apple. In particular, MpCYS4 not only induced but also suppressed the expression levels of many genes, indicating that it has both positive and negative impacts on the transcript atlas. We believe it is worth mentioning that the number of DEGs was markedly higher under drought (1556 up-regulated and 1327 down-regulated) than under normal conditions (82 and 69, respectively). One possible explanation is that MpCYS4 undergoes certain unidentified modifications when plants are exposed to drought, which then alters its regulatory mode in activating or suppressing genes involved in the stress signaling network. Among the DEGs in our transgenic line, many have been annotated or confirmed to be involved in stress tolerance, either regulatory genes or functional ones. Transcript accumulations of two ABA receptor genes, PYL4 and PYL9, were suppressed in 35S:MpCYS4 plants under water-deficit conditions. Meanwhile, the expression of genes encoding a group of Ser/Thr PP2Cs, such as ABI1, ABI2, and HAB1, as well as OST1 and ABRE binding transcription factors, was enhanced in those plants. Moreover, genes encoding major abiotic stress-responsive proteins, including DREB2A, LEA, RD29B, RD22, and KIN2, were also more strongly expressed in transgenic plants than in WT under drought stress. Thus, MpCYS4 appears to positively affect the ABA signaling pathway by the induction of multiple stress-responsive genes, consistent with the results obtained from preliminary assays with Arabidopsis.
In conclusion, we have demonstrated that MpCYS4, a cystatin gene isolated from M. prunifolia, functions in mediating stomatal regulation and many ABA- and stress-related genes expression in response to drought. These properties indicate MpCYS4 as a potentially appropriate candidate to manipulate plant drought response, improve our understanding of plant defense mechanisms against environmental challenges and provide a new tool for possible genetic alterations of phytocystatins. Such findings also can be used as a foundation for investigating the pivotal roles that these genes have in plant stress responses and in multiple signal transduction pathways. Further investigations are necessary to elucidate the molecular mechanism by which MpCYS4 interacts with stress-responsive ABA signaling transduction, and pursue its target proteins and regulatory mechanisms that support MpCYS4-mediated drought stress response in apple.
Availability of Supporting Data
The raw sequencing data of RNA-Seq analysis has been deposited in NBCI SRA under accession number SRP095318.
Author Contributions
YT performed most of the experiments and analyzed the data; ML, YY, XS, NW, and BL provided technical assistance to YT; FM provided the critical intellectual input in design of the study and preparation of the manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The reviewer HS and handling Editor declared their shared affiliation, and the handling Editor states that the process nevertheless met the standards of a fair and objective review.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (31572108), the State Key Program of the National Natural Science Foundation of China (31330068), and the earmarked fund for the China Agriculture Research System (CARS-28). We thank Mr. Zhengwei Ma for management of the potted apple plants.
Supplementary Material
The Supplementary Material for this article can be found online at: http://journal.frontiersin.org/article/10.3389/fpls.2017.00033/full#supplementary-material
References
Alvarez-Alfageme, F., Martínez, M., Pascual-Ruiz, S., Castañera, P., Diaz, I., and Ortego, F. (2007). Effects of potato plants expressing a barley cystatin on the predatory bug Podisus maculiventris via herbivorous prey feeding on the plant. Transgen. Res. 16, 1–13. doi: 10.1007/s11248-006-9022-6
Anders, S., and Huber, W. (2010). Differential expression analysis for sequence count data. Genome Biol. 11:R106. doi: 10.1186/gb-2010-11-10-r106
Belenghi, B., Acconcia, F., Trovato, M., Perazzolli, M., Bocedi, A., Polticelli, F., et al. (2003). AtCYS1, a cystatin from Arabidopsis thaliana, suppresses hypersensitive cell death. Eur. J. Biochem. 270, 2593–2604. doi: 10.1046/j.1432-1033.2003.03630.x
Benchabane, M., Schlüter, U., Vorster, J., Goulet, M. C., and Michaud, D. (2010). Plant cystatins. Biochimie 92, 1657–1666. doi: 10.1016/j.biochi.2010.06.006
Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254. doi: 10.1016/0003-2697(76)90527-3
Bray, E. A. (2002). Abscisic acid regulation of gene expression during water-deficit stress in the era of the Arabidopsis genome. Plant Cell Environ. 25, 153–161. doi: 10.1046/j.1365-3040.2002.00746.x
Brodribb, T. J., and McAdam, S. A. (2013). Abscisic acid mediates a divergence in the drought response of two conifers. Plant Physiol. 162, 1370–1377. doi: 10.1104/pp.113.217877
Carrillo, L., Martinez, M., Ramessar, K., Cambra, I., Castañera, P., Ortego, F., et al. (2011). Expression of a barley cystatin gene in maize enhances resistance against phytophagous mites by altering their cysteine-proteases. Plant Cell Rep. 30, 101–112. doi: 10.1007/s00299-010-0948-z
Clauw, P., Coppens, F., De Beuf, K., Dhondt, S., Van Daele, T., Maleux, K., et al. (2015). Leaf responses to mild drought stress in natural variants of Arabidopsis. Plant Physiol. 167, 800–816. doi: 10.1104/pp.114.254284
Clough, S. J., and Bent, A. F. (1998). Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 16, 735–743. doi: 10.1046/j.1365-313x.1998.00343.x
Cutler, S. R., Rodriguez, P. L., Finkelstein, R. R., and Abrams, S. R. (2010). Abscisic acid: emergence of a core signaling network. Annu. Rev. Plant Biol. 61, 651–679. doi: 10.1146/annurev-arplant-042809-112122
Diaz, I., Martinez, M., Isabel-LaMoneda, I., Rubio-Somoza, I., and Carbonero, P. (2005). The DOF protein, SAD, interacts with GAMYB in plant nuclei and activates transcription of endosperm-specific genes during barley seed development. Plant J. 42, 652–662. doi: 10.1111/j.1365-313X.2005.02402.x
Díaz-Mendoza, M., Velasco-Arroyo, B., Gonzalez-Melendi, P., Martínez, M., and Díaz, I. (2014). C1A cysteine protease–cystatin interactions in leaf senescence. J. Exp. Bot. 65, 3825–3833. doi: 10.1093/jxb/eru043
Dionisio-Sese, M. L., and Tobita, S. (1998). Antioxidant responses of rice seedlings to salinity stress. Plant Sci. 135, 1–9. doi: 10.1016/S0168-9452(98)00025-9
Eason, J. R., West, P. J., Brummell, D. A., Watson, L. M., Somerfield, S. D., and McLachlan, A. R. G. (2014). Overexpression of the protease inhibitor BoCPI-1 in broccoli delays chlorophyll loss after harvest and causes down-regulation of cysteine protease gene expression. Postharv. Biol. Technol. 97, 23–31. doi: 10.1016/j.postharvbio.2014.06.006
Finkelstein, R. (2013). Abscisic acid synthesis and response. Arabidopsis Book 11:e0166. doi: 10.1199/tab.0166
Gaddour, K., Vicente-Carbajosa, J., Lara, P., Isabel-Lamoneda, I., Díaz, I., and Carbonero, P. (2001). A constitutive cystatin-encoding gene from barley (Icy) responds differentially to abiotic stimuli. Plant Mol. Biol. 45, 599–608. doi: 10.1023/A:1010697204686
Gaxiola, R. A., Li, J., Undurraga, S., Dang, L. M., Allen, G. J., Alper, S. L., et al. (2001). Drought- and salt-tolerant plants result from overexpression of the AVP1 H+-pump. Proc. Natl. Acad. Sci. U.S.A. 98, 11444–11449. doi: 10.1073/pnas.191389398
Geiger, D., Scherzer, S., Mumm, P., Stange, A., Marten, I., Bauer, H., et al. (2009). Activity of guard cell anion channel SLAC1 is controlled by drought-stress signaling kinase-phosphatase pair. Proc. Natl. Acad. Sci. U.S.A. 106, 21425–21430. doi: 10.1073/pnas.0912021106
Hetherington, A. M., and Woodward, F. I. (2003). The role of stomata in sensing and driving environmental change. Nature 424, 901–908. doi: 10.1038/nature01843
Hummer, K. E., and Janick, J. (2009). “Rosaceae: taxonomy, economic importance, genomics,” in Genetics and Genomics of Rosaceae, eds K. M. Folta and S. E. Gardiner (New York, NY: Springer Press), 1–17.
Je, J., Song, C., Hwang, J. E., Chung, W. S., and Lim, C. O. (2014). DREB2C acts as a transcriptional activator of the thermo tolerance-related phytocystatin 4 (AtCYS4) gene. Transgen. Res. 23, 109–123. doi: 10.1007/s11248-013-9735-2
Kim, D., Pertea, G., Trapnell, C., Pimentel, H., Kelley, R., and Salzberg, S. L. (2013). TopHat2: accurate alignment of transcriptomes in the presence of insertions, deletions and gene fusions. Genome Biol. 14:R36. doi: 10.1186/gb-2013-14-4-r36
Knight, H., and Knight, M. R. (2001). Abiotic stress signalling pathways: specificity and cross-talk. Trends Plant Sci. 6, 262–267. doi: 10.1016/S1360-1385(01)01946-X
Kotoda, N., Hayashi, H., Suzuki, M., Igarashi, M., Hatsuyama, Y., Kidou, S., et al. (2010). Molecular characterization of FLOWERING LOCUS T-Like gene of apple (Malus × domestica Borkh.). Plant Cell Physiol. 51, 561–575. doi: 10.1093/pcp/pcq021
Kunert, K. J., van Wyk, S. G., Cullis, C. A., Vorster, B. J., and Foyer, C. H. (2015). Potential use of phytocystatins in crop improvement, with a particular focus on legumes. J. Exp. Bot. 66, 3559–3570. doi: 10.1093/jxb/erv211
Kwak, J. M., Mori, I. C., Pei, Z. M., Leonhardt, N., Angel Torres, M., Dangl, J. L., et al. (2003). NADPH oxidase AtrbohD and AtrbohF genes function in ROS-dependent ABA signaling in Arabidopsis. EMBO J. 22, 2623–2633. doi: 10.1093/emboj/cdg277
Lee, S. C., Lan, W., Buchanan, B. B., and Luan, S. (2009). A protein kinase-phosphatase pair interacts with an ion channel to regulate ABA signaling in plant guard cells. Proc. Natl. Acad. Sci. U.S.A. 106, 21419–21424. doi: 10.1073/pnas.0910601106
Lee, S. C., Lim, C. W., Lan, W., He, K., and Luan, S. (2013). ABA signaling in guard cells entails a dynamic protein-protein interaction relay from the PYL-RCAR family receptors to ion channels. Mol. Plant 6, 528–538. doi: 10.1093/mp/sss078
Li, C., Tan, D. X., Liang, D., Chang, C., Jia, D., and Ma, F. (2015). Melatonin mediates the regulation of ABA metabolism, free-radical scavenging, and stomatal behaviour in two Malus species under drought stress. J. Exp. Bot. 66, 669–680. doi: 10.1093/jxb/eru476
Lichtenthaler, K., and Wellburn, A. R. (1983). Determinations of total carotenoids and chlorophylls a and b of leaf extracts in different solvents. Biochem. Soc. Trans. 11, 591–592. doi: 10.1042/bst0110591
Livak, K. J., and Schmittgen, T. D. (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25, 402–408. doi: 10.1006/meth.2001.1262
Ma, Y., Szostkiewicz, I., Korte, A., Moes, D., Yang, Y., Christmann, A., et al. (2009). Regulators of PP2C phosphatase activity function as abscisic acid sensors. Science 324, 1064–1068. doi: 10.1126/science.1172408
Martinez, M., Cambra, I., Carrillo, L., Diaz-Mendoza, M., and Diaz, I. (2009). Characterization of the entire cystatin gene family in barley and their target cathepsin L-like cysteine-proteases, partners in the hordein mobilization during seed germination. Plant Physiol. 151, 1531–1545. doi: 10.1104/pp.109.146019
Martinez, M., and Diaz, I. (2008). The origin and evolution of plant cystatins and their target cysteine proteinases indicate a complex functional relationship. BMC Evol. Biol. 8:198. doi: 10.1186/1471-2148-8-198
Mortazavi, A., Williams, B. A., McCue, K., Schaeffer, L., and Wold, B. (2008). Mapping and quantifying mammalian transcriptomes by RNA-Seq. Nat. Methods 5, 621–628. doi: 10.1038/nmeth.1226
Outchkourov, N. S., de Kogel, W. J., Wiegers, G. L., Abrahamson, M., and Jongsma, M. A. (2004). Engineered multidomain cysteine protease inhibitors yield resistance against western flower thrips (Frankliniella occidentalis) in greenhouse trials. Plant Biotechnol. J. 2, 449–458. doi: 10.1111/j.1467-7652.2004.00089.x
Park, S. Y., Fung, P., Nishimura, N., Jensen, D. R., Fujii, H., Zhao, Y., et al. (2009). Abscisic acid inhibits type 2C protein phosphatases via the PYR/PYL family of START proteins. Science 324, 1068–1071. doi: 10.1126/science.1173041
Pei, Z. M., Murata, Y., Benning, G., Thomine, S., Klüsener, B., Allen, G. J., et al. (2000). Calcium channels activated by hydrogen peroxide mediate abscisic acid signalling in guard cells. Nature 406, 731–734. doi: 10.1038/35021067
Pierik, R., and Testerink, C. (2014). The art of being flexible: how to escape from shade, salt, and drought. Plant Physiol. 166, 5–22. doi: 10.1104/pp.114.239160
Popovic, M., Andjelkovic, U., Burazer, L., Lindner, B., Petersen, A., and Gavrovic-Jankulovic, M. (2013). Biochemical and immunological characterization of a recombinantly-produced antifungal cysteine proteinase inhibitor from green kiwifruit (Actinidia deliciosa). Phytochemistry 94, 53–59. doi: 10.1016/j.phytochem.2013.06.006
Quain, M. D., Makgopa, M. E., Márquez-García, B., Comadira, G., Fernandez-Garcia, N., Olmos, E., et al. (2014). Ectopic phytocystatin expression leads to enhanced drought stress tolerance in soybean (Glycine max) and Arabidopsis thaliana through effects on strigolactone pathways and can also result in improved seed traits. Plant Biotechnol. J. 12, 903–913. doi: 10.1111/pbi.12193
Rawlings, N. D., Morton, F. R., Yin Kok, C., Kong, J., and Barrett, A. J. (2008). MEROPS: the peptidase database. Nucleic Acids Res. 36, 320–325. doi: 10.1093/nar/gkm954
Schroeder, J. I., Allen, G. J., Hugouvieux, V., Kwak, J. M., and Waner, D. (2001). Guard cell signal transduction. Annu. Rev. Plant Biol. 52, 627–658. doi: 10.1146/annurev.arplant.52.1.627
Shinozaki, K., and Yamaguchi-Shinozaki, K. (2007). Gene networks involved in drought stress response and tolerance. J. Exp. Bot. 58, 221–227. doi: 10.1093/jxb/erl164
Sirichandra, C., Gu, D., Hu, H. C., Davanture, M., Lee, S., Djaoui, M., et al. (2009). Phosphorylation of the Arabidopsis AtrbohF NADPH oxidase by OST1 protein kinase. FEBS Lett. 583, 2982–2986. doi: 10.1016/j.febslet.2009.08.033
Sun, X. L., Yang, S. S., Sun, M. Z., Wang, S. T., Ding, X. D., Zhu, D., et al. (2014). A novel Glycine soja cysteine proteinase inhibitor GsCPI14, interacting with the calcium/calmodulin-binding receptor-like kinase GsCBRLK, regulated plant tolerance to alkali stress. Plant Mol. Biol. 85, 33–48. doi: 10.1007/s11103-013-0167-4
Tan, Y. X., Qin, Y., Li, Y. L., Li, M. J., and Ma, F. W. (2014b). Overexpression of MpGR-RBP1, a glycine-rich RNA-binding protein gene from Malus prunifolia (Willd.) Borkh., confers salt stress tolerance and protects against oxidative stress in Arabidopsis. Plant Cell Tiss. Organ. Cult. 119, 635–646. doi: 10.1007/s11240-014-0563-8
Tan, Y. X., Wang, S. C., Liang, D., Li, M. J., and Ma, F. W. (2014a). Genome-wide identification and expression profiling of the cystatin gene family in apple (Malus × domestica Borkh.). Plant Physiol. Biochem. 79, 88–97. doi: 10.1016/j.plaphy.2014.03.011
van der Vyver, C., Schneidereit, J., Driscoll, S., Turner, J., Kunert, K., and Foyer, C. H. (2003). Oryzacystatin-1 expression in transformed tobacco produces a conditional growth phenotype and enhances chilling tolerance. Plant Biotechnol. J. 1, 101–112. doi: 10.1046/j.1467-7652.2003.00010.x
Velasco, R., Zharkikh, A., Affourtit, J., Dhingra, A., Cestaro, A., Kalyanaraman, A., et al. (2010). The genome of the domesticated apple (Malus × domestica Borkh.). Nat. Gen. 42, 833–839. doi: 10.1038/ng.654
Wang, J., Sun, P. P., Chen, C. L., Wang, Y., Fu, X. Z., and Liu, J. H. (2011). An arginine decarboxylase gene PtADC from Poncirus trifoliata confers abiotic stress tolerance and promotes primary root growth in Arabidopsis. J. Exp. Bot. 62, 2899–2914. doi: 10.1093/jxb/erq463
Wang, Y. G., Zhan, Y. N., Wu, C., Gong, S. L., Zhu, N., Chen, S. X., et al. (2012). Cloning of a cystatin gene from sugar beet M14 that can enhance plant salt tolerance. Plant Sci. 191–192, 93–99. doi: 10.1016/j.plantsci.2012.05.001
Weeda, S. M., Kumar, G. N., and Richard Knowles, N. (2009). Developmentally linked changes in proteases and protease inhibitors suggest a role for potato multicystatin in regulating protein content of potato tubers. Planta 230, 73–84. doi: 10.1007/s00425-009-0928-0
Weigel, D., and Glazebrook, J. (2002). Arabidopsis: A Laboratory Manual. New York, NY: Cold Spring Harbor Laboratory Press.
Xie, C., Zhang, R., Qu, Y., Miao, Z., Zhang, Y., Shen, X., et al. (2012). Overexpression of MtCAS31 enhances drought tolerance in transgenic Arabidopsis by reducing stomatal density. New Phytol. 195, 124–135. doi: 10.1111/j.1469-8137.2012.04136.x
Xie, X., Wang, Y., Williamson, L., Holroyd, G. H., Tagliavia, C., Murchie, E., et al. (2006). The identification of genes involved in the stomatal response to reduced atmospheric relative humidity. Curr. Biol. 16, 882–887. doi: 10.1016/j.cub.2006.03.028
Xie, Y., Mao, Y., Zhang, W., Lai, D., Wang, Q., and Shen, W. (2014). Reactive oxygen species-dependent nitric oxide production contributes to hydrogen-promoted stomatal closure in Arabidopsis. Plant Physiol. 165, 759–773. doi: 10.1104/pp.114.237925
Yekutieli, D., and Benjamini, Y. (2001). The control of the false discovery rate in multiple testing under dependency. Ann. Stat. 29, 1165–1188. doi: 10.1214/aos/1013699998
Zhang, X., Liu, S., and Takano, T. (2008). Two cysteine proteinase inhibitors from Arabidopsis thaliana, AtCYSa and AtCYSb, increasing the salt, drought, oxidation and cold tolerance. Plant Mol. Biol. 68, 131–143. doi: 10.1007/s11103-008-9357-x
Keywords: ABA sensitivity, ABA/stress-responsive gene, apple, drought tolerance, plant cystatin, stomatal regulation
Citation: Tan Y, Li M, Yang Y, Sun X, Wang N, Liang B and Ma F (2017) Overexpression of MpCYS4, A Phytocystatin Gene from Malus prunifolia (Willd.) Borkh., Enhances Stomatal Closure to Confer Drought Tolerance in Transgenic Arabidopsis and Apple. Front. Plant Sci. 8:33. doi: 10.3389/fpls.2017.00033
Received: 30 October 2016; Accepted: 06 January 2017;
Published: 24 January 2017.
Edited by:
Rudy Dolferus, Commonwealth Scientific and Industrial Research Organisation, AustraliaReviewed by:
Yanmin Zhu, Agricultural Research Service (USDA), USAHarriet Sangma, Commonwealth Scientific and Industrial Research Organisation, Australia
Copyright © 2017 Tan, Li, Yang, Sun, Wang, Liang and Ma. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Fengwang Ma, ZndtNjRAbndzdWFmLmVkdS5jbg==; ZndtNjRAc2luYS5jb20=
 Yanxiao Tan
Yanxiao Tan Na Wang
Na Wang Fengwang Ma
Fengwang Ma