- 1Department of Bioenergy Science and Technology, Chonnam National University, Gwangju, South Korea
- 2Department of Bioindustry and Bioresource Engineering, Plant Engineering Research Institute, Sejong University, Seoul, South Korea
Triacylglycerol (TAG) is an energy-rich reserve in plant seeds that is composed of glycerol esters with three fatty acids. Since TAG can be used as a feedstock for the production of biofuels and bio-chemicals, producing TAGs in vegetative tissue is an alternative way of meeting the increasing demand for its usage. The WRINKLED1 (WRI1) gene is a well-established key transcriptional regulator involved in the upregulation of fatty acid biosynthesis in developing seeds. WRI1s from Arabidopsis and several other crops have been previously employed for increasing TAGs in seed and vegetative tissues. In the present study, we first identified three functional CsWRI1 genes (CsWRI1A. B, and C) from the Camelina oil crop and tested their ability to induce TAG synthesis in leaves. The amino acid sequences of CsWRI1s exhibited more than 90% identity with those of Arabidopsis WRI1. The transcript levels of the three CsWRI1 genes showed higher expression levels in developing seeds than in vegetative and floral tissues. When the CsWRI1A. B, or C was introduced into Arabidopsis wri1-3 loss-of-function mutant, the fatty acid content was restored to near wild-type levels and percentages of the wrinkled seeds were remarkably reduced in the transgenic lines relative to wri1-3 mutant line. In addition, the fluorescent signals of the enhanced yellow fluorescent protein (eYFP) fused to the CsWRI1 genes were observed in the nuclei of Nicotiana benthamiana leaf epidermal cells. Nile red staining indicated that the transient expression of CsWRI1A. B, or C caused an enhanced accumulation of oil bodies in N. benthamiana leaves. The levels of TAGs was higher by approximately 2.5- to 4.0-fold in N. benthamiana fresh leaves expressing CsWRI1 genes than in the control leaves. These results suggest that the three Camelina WRI1s can be used as key transcriptional regulators to increase fatty acids in biomass.
Introduction
Camelina sativa L. is an underdeveloped oil crop in the family Brassicaceae, which has several advantages in the agronomic and environmental context over the current developed oil crops. Camelina has a relatively short growing period (85–100 days to maturity) and can be cultivated twice in 1 year (Putnam et al., 1993). In comparison with other oil crops, it requires lower amounts of fertilizer for growth and is more resistant to various stresses such as cold and drought (Putnam et al., 1993; Kim et al., 2013; Bansal and Durrett, 2016). Camelina seed oil is composed of 35–45% triacylglycerol (TAG), which has a high proportion of polyunsaturated fatty acids (PUFAs) (Lu and Kang, 2008; Bansal and Durrett, 2016). Approximately 40% of the total fatty acid (FA) content is composed of linolenic acid (18:3) and linoleic acid (18:2). The considerable amount of PUFAs confers considerable susceptibility to oxidation, which makes it less suitable for biodiesel production and domestic cooking, such as frying vegetables (Soriano and Narani, 2012). However, breeding and genetic engineering programs can generate new varieties of Camelina with a lower PUFA content for stable oxidation (Kang et al., 2011; Nguyen et al., 2013). Camelina can be transformed using the Agrobacterium-mediated flower-dip method, which is a relatively simple and rapid route to generating transgenic plants with superior agronomic traits (Lu and Kang, 2008; Liu et al., 2012). The whole genome sequence and seed transcriptome data present valuable resources for the understanding of the function of genes involved in oil biosynthesis in Camelina seeds (Hutcheon et al., 2010; Nguyen et al., 2013; Kagale et al., 2014).
Triacylglycerol is a neutral lipid molecule that stores carbons and hydrogens that are utilized for energy production in the life cycle of plants (Athenstaedt and Daum, 2006). TAGs are mainly observed in seeds, where they are used as energy stores for seed germination; they also occur in chloroplasts of senescent leaves where they aid in fatty acid sequestration and in pollen, where they have been shown to promote pollen germination in Arabidopsis (Kaup et al., 2002; Kim et al., 2002; Athenstaedt and Daum, 2006). The first step of fatty acid synthesis in seeds is the formation of malonyl-coenzyme A (CoA) from acetyl-CoA by acetyl-CoA carboxylase (ACCase). Malonyl-CoA-ACP malonyltransferase catalyzes the conversion of malonyl-CoA to malonyl-ACP. Then, malonyl-ACP is condensed with acetyl-CoA to make 3-ketoacyl-ACP. After that the series of sequential condensation reactions of malonyl-ACP to 3-ketoacyl-ACP or the growing of acyl-ACP by the fatty acid synthase complex results in 16- to 18-carbon fatty acyl-ACPs in the plastids (Chapman and Ohlrogge, 2012). The fatty acyl group is hydrolyzed by thioesterases (FATA/FATB). The fatty acyl chains are exported to the cytoplasm and activated to fatty acyl-CoAs by long-chain acyl-CoA synthetase (LACS). Fatty acyl-CoA pools are utilized for the esterification of fatty acids with glycerol-3-phosphate (G3P) sequentially by three acyltransferases called G3P acyltransferase (GPAT), lysophosphatidic acid acyltransferase (LPAT), and diacylglycerol acyltransferase (DGAT) in the endoplasmic reticulum (ER) to synthesize TAG. Oil bodies containing TAG are formed in the ER membrane budding from the TAG synthesizing site as a droplet (Li-Beisson et al., 2010).
The network for AFL master regulators, ABSCISIC ACID INSENSITIVE3 (ABI3), FUSCA3 (FUS3), LEAFY COTYLEDON1 (LEC1), and LEC2 have been studied with the aim of controlling the biosynthesis of Arabidopsis seed oil (North et al., 2010; Roscoe et al., 2015; Devic and Roscoe, 2016). This network can regulate the expression of genes encoding enzymes that synthesize the storage lipids and protein reserves in seeds. As a key transcription factor for lipid accumulation, WRINKLED1 (WRI1) is located downstream in this AFL network (Roscoe et al., 2015). Ectopic expression of FUS3 can regulate WRI1 expression (Yamamoto et al., 2010). LEC2 may regulate fatty acid biosynthesis genes through direct regulation of WRI1 (Baud et al., 2007). Although ABI3 is not known to directly regulate the expression of WRI1, it is known that ABI3, FUS3, LEC1, and LEC2 regulate the downstream genes through synergistic interactions with each other during seed maturation (Devic and Roscoe, 2016). The WRI1 gene was first identified in an Arabidopsis mutant exhibiting wrinkled seed morphology; it encodes an AP2/EREBP type transcription factor (Focks and Benning, 1998). WRI1 regulates the expression of genes encoding the enzymes involved in catalytic processes such as phosphoglycerate mutase, plastidic pyruvate kinase β subunit 1 (PI-PKβ1), and pyruvate dehydrogenase (PDHE1α) enzymes during glycolysis, wherein it has been observed to increase the amount of pyruvate during acetyl-CoA synthesis (Baud et al., 2007, 2009; Maeo et al., 2009; Fukuda et al., 2013). Furthermore, WRI1 directly binds to the promoters of genes encoding key enzymes for fatty acid biosynthesis during seed maturation, such as biotin carboxyl carrier protein isoform 2 (BCCP2), acyl carrier protein 1 (ACP1), and keto-ACP synthase 1 (KAS1) (Baud et al., 2009; Maeo et al., 2009).
Arabidopsis wri1 mutants have been shown to exhibit an up to 80% lower TAG content than the wild-type seeds, with correspondingly increased sucrose levels. This implies that WRI1 is a regulator for carbon allocation between sucrose and fatty acids in developing seeds (Focks and Benning, 1998; Ma et al., 2015). Following the discovery of Arabidopsis WRI1 (AtWRI1), its homologues have been identified from the seeds of rapeseed (Brassica napus L.), corn (Zea mays L.), and oil palm (Elaeis guineensis Jacq.) (Liu et al., 2010; Shen et al., 2010; Pouvreau et al., 2011; Ma et al., 2013; Wu et al., 2014). In addition to seeds, WRI1 homologs have been identified in non-seed tissues containing oil, such as the stem of poplar (Populus trichocarpa L.), tuber parenchyma of nutsedge (Cyperus esculentus L.), and leaf blades of Brachypodium distachyon L. Beauv. (Grimberg et al., 2015; Yang et al., 2015). Overexpression of the WRI1 genes contributed to increased TAG levels in the seeds and vegetative tissues. Seed-specific expression of AtWRI1 transgene increased TAG content to levels 14–30% higher than that in the wild-type seeds in Arabidopsis. B. napus, corn, and Camelina (Cernac and Benning, 2004; Liu et al., 2010; Shen et al., 2010; Pouvreau et al., 2011; Wu et al., 2014; An and Suh, 2015). Ectopic overexpression of WRI1 stimulated oil production in the vegetative tissues (Vanhercke et al., 2013, 2014; Nookaraju et al., 2014; Zale et al., 2016). Five WRI1 isoforms have been isolated from diverse species such as Arabidopsis, potato (Solanum tuberosum L.), oat (Avena sativa L.), and nutsedge. These genes were introduced into leaves using the agroinfiltration method, which resulted in an increase in TAG content from 0.05% to 2.20% in Nicotiana benthamiana Domin. leaves (Grimberg et al., 2015).
In the present study, we first identified three Camelina WRI1 isoforms, CsWRI1A, CsWRI1B, and CsWRI1C, that complemented the wrinkled-seed phenotype and partially restored TAG content in Arabidopsis wri1-3 mutants. We then studied the CsWRI1 genes to identify a potential candidate to act as a transcriptional regulator for inducing TAG production in vegetative tissues.
Materials and Methods
Plant Materials and Growth Conditions
Camelina sativa L. CAME, Arabidopsis thaliana accession Columbia-0, and N. benthamiana plants were grown in long-day growth conditions with 16 h/8 h light/dark photoperiod at 21–24°C, and 50–60% humidity in a sterilized soil mixture (peat moss enriched soil:vermiculite:perlite in 4:2:1 ratio) (An and Suh, 2015).
Gene Identification and Isolation
Total RNAs were isolated from developing Camelina seeds 20 days after flowering (DAF) based on the protocol for RNA isolation from seeds (Oñate-Sánchez and Vicente-Carbajosa, 2008). cDNAs were then synthesized by RT-PCR using primers listed in Supplementary Table 1. The PCR protocol was as follows: denaturation at 94°C for 30 s, annealing at 58–62°C for 20 s, and extension at 72°C for 120 s. PCR products were cloned into the pGEM T-easy vector (Promega) and their nucleotide sequences were determined.
Protein Sequence and Phylogenetic Tree Analysis
Amino acid sequences were aligned using CLUSTALW, and dendrograms for phylogenetic analysis were constructed using the MEGA 6.06 program1 with the maximum likelihood method and a bootstrap value of 500 replicates (Tamura et al., 2013).
RT-PCR Analysis
In Camelina, transcript expression was analyzed in various tissues of the aerial parts such as the flower buds and leaves of 5-week-old plants, stems from 4-week-old plants, roots from 2-week-old plants, open flowers from plants older than 6 weeks, and developing seeds 10, 20, and 30 DAF. To assess the expression level of the transgene in Arabidopsis complementation lines, total RNAs were extracted from developing seeds of transgenic Arabidopsis 6–8 DAF. Total RNAs were isolated from these tissues using QIAGEN RNeasy® plant mini kit (50), following the manufacturers protocols. In addition, RNAs from developing seeds of Camelina and Arabidopsis were isolated according to the method described in Oñate-Sánchez and Vicente-Carbajosa (2008). First, cDNAs were synthesized from these RNAs by the GoScriptTM reverse transcription system (Promega) followed by PCR by the Access QuickTM RT-PCR system (Promega) to analyze the transcript levels of each gene using gene-specific primers (Supplementary Table 1). The PCR protocol was as follows: denaturation at 94°C for 30 s, annealing at 58–62°C for 20 s, and extension at 72°C for 20 s. These cycles were performed 35 times. To control equal cDNA loading in RT-PCR, CsACTIN11 and EIF4A1(At3g13920) gene-specific primers listed in Supplementary Table 1 were used as controls for transcript levels in various tissues (Hutcheon et al., 2010).
Vector Construction
To express the CsWRI1 gene in transgenic plants, each cDNA containing an ORF was inserted under the sequences of CaMV 35S promoter by digestion at the BamHI (5′-terminus) and Sac1 (3′-terminus) restriction endonuclease sites in the pBA002 vector (Kim et al., 2006). This vector contains the herbicide resistance marker gene PAT (Phosphinothricin acetyltransferase), which enables selection of the transgenic plants. These constructs were transformed into Agrobacterium tumefaciens GV3101 using the freeze-thaw method (An, 1987), which were then used for the transformation of Arabidopsis by the Agrobacterium-mediated floral dip method (Zhang et al., 2006).
CsWRI1 cDNAs containing an ORF without the stop codon were inserted at the SacI (5′-terminus) and XmaI (3′-terminus) sites between the CaMV 35S promoter and enhanced yellow fluorescent protein (eYFP) to enable fusion of the eYFP with the in-frame stop codon, and allow expression under the control of the CaMV 35S promoter in the pPZP212 vector (GenBank U10462). These constructs were then used for the detection of fluorescence signals of CsWRI1 proteins in the epidermal cells of N. benthamiana infected by Agrobacterium tumefaciens GV3101.
Subcellular Localization
The vectors expressing CsWRI1s:eYFP under the control of the CaMV 35S promoter were transformed into Agrobacterium tumefaciens LBA4404, followed by the infection of the N. benthamiana leaf epidermis using the agroinfiltration method (Yoo et al., 2007). The N. benthamiana leaves were further grown for 12 h, and the fluorescence was observed using the TCS SP5 AOBS/Tandem laser scanning confocal microscope (Leica Microsystems, Germany). The emission wavelength was 571–617 nm and the excitation wavelengths were 520–554 nm for YFP and 572–618 nm for RFP.
Generation of Transgenic Arabidopsis
Progeny seeds harvested from the transformed Arabidopsis plants were germinated and grown on 1/2 MS medium (1% sucrose, 0.7% phytoagar) containing 5 μg/mL of phosphinothricin (PPT). PPT-resistant T1 plants were tested for the presence of the transgene in the genome by PCR analysis with primers listed in Supplementary Table 1. T2 seeds from T1 transgenic plants were selected for the observation of seed morphology and determination of the fatty acid content.
Seed Morphology Analysis
Seed morphology was observed using light and scanning electron microscopes. Dry seeds of the wild-type and the wri1-3 mutant were coated with platinum particles using a Hitachi E1030 coater on aluminum stubs. Their images were scanned using a field emission scanning electron microscope (FE-SEM, Hitachi S-4700, Tokyo, Japan). The ratio of seeds rescued from the wrinkled phenotype to normal in the transgenic lines was recorded from the photographs taken of the T2 seed siblings using an Axiocam, MRc 5 camera equipped with a light microscope (Carl Zeiss: SteREO Lumar. V12).
Nile Red Staining
The leaves of N. benthamiana were infiltrated with Agrobacterium containing an empty vector (pBA002) or the CsWRI1 vector. These leaves were stained with Nile red solution [10 μg/mL in 0.1 M Tris-HCl buffer (pH 8) Sigma-N3013] at room temperature for 30 min. The stained leaves were washed with 0.1 M Tris-HCl buffer (pH 8) for 10 min, followed by observation of red fluorescence of the oil bodies at wavelengths of 560 nm for excitation and 615 nm for emission using a tandem laser confocal scanning microscope (TCS SP5 AOBS, Leica Microsystems, Germany).
Thin Layer Chromatograph (TLC) Analysis
The fresh leaves of N. benthamiana were homogenized and immersed in isopropanol at 65°C for 15 min. Chloroform and water were mixed in same tube, then total lipids were extracted twice using a chloroform:methanol solution (2:1, v/v). The solvents of the lipid phase washed with 1 M KCl were collected and evaporated using nitrogen gas. Finally, total lipids were dissolved in chloroform containing tri 17:0-TAG (glyceryl triheptadecanoate; Sigma-T2151) as an internal standard and separated by TLC (Silica gel 60, MERCK) in hexane:diethylether:acetic acid (70:30:1, v/v/v). They were then visualized by spraying 0.01% primuline (Sigma-206865) in 80% acetone under UV light (Vanhercke et al., 2013). TAG bands scraped from TLC were dissolved in toluene and esterified with methanol and H2SO4 mixture at 95°C for 90 min. The esterified TAGs were analyzed for their fatty acid content using gas chromatography as described in the following section.
Fatty Acid Analysis
To measure total fatty acid profile and content, total lipids extracted from Arabidopsis seeds or N. benthamiana fresh leaves and tri 17:0-TAG in toluene were combined and esterified with methanol mixed with H2SO4 at 95°C for 90 min. After the reaction, 0.9% NaCl solution and hexane were added to extract the fatty acid methyl esters. The upper phase containing the fatty acid methyl esters was concentrated by N2 air and separated in a DB23 column (30 m × 0.25 mm, 0.25 μm film thickness; J&W Scientific, Folsom. CA, USA) using GC-2010 gas chromatography (Shimadzu, Japan). The temperature of the reaction ranged from 160°C to 220°C, increasing at a rate of 2.5°C per min. The peak area on the retention time for each fatty acid was characterized and measured by comparison with the known standard fatty acid profiles, and the concentration of each fatty acid was calculated by comparing its peak area with that of the internal standard.
Results
Camelina Possesses Three Copies of WRI1
In order to identify the factors involved in the accumulation of higher oil content in Camelina, we first focused on identifying the homologous genes of Arabidopsis WRINKLED1 (AtWRI1) in two different Camelina genome databases2,3. Three WRI1 scaffolds and three WRI1 loci were identified in different chromosomes of the hexaploid genome (Supplementary Table 2) (Kagale et al., 2014). Three WRI1 scaffolds showed overall 28–36% nucleotide sequence identity in intron and 5′- and 3′-nocoding regions (Supplementary Figure 1) (Kagale et al., 2014). Based on the differences in nucleotide sequences we can detect the corresponding three distinct WRI1 transcripts from developing seeds in Camelina by cloning their cDNAs. These three Camelina cDNAs were designated as CsWRI1A (GenBank KY129795), CsWRI1B (GenBank KY129796), and CsWRI1C (GenBank KY129797) based on the identity with WRI1 genomic DNA sequences in Camelina genome scaffolds and Arabidopsis WRI1. Excluding the non-coding regions and the intron, the three CsWRI1 genes exhibited a high identity in nucleotide sequences (Supplementary Figure 2). Compared with that of the AtWRI1, the three CsWRI1s exhibited a high identity in the amino acid (aa) sequence levels (∼95%) with 4, 1, and 3 amino acids that are longer than that of the 430 aa open reading frame (ORF) of Arabidopsis. An AP2/EREBP DNA binding motif and a VYL transcriptional activation motif are conserved between Camelina and Arabidopsis (Figure 1).
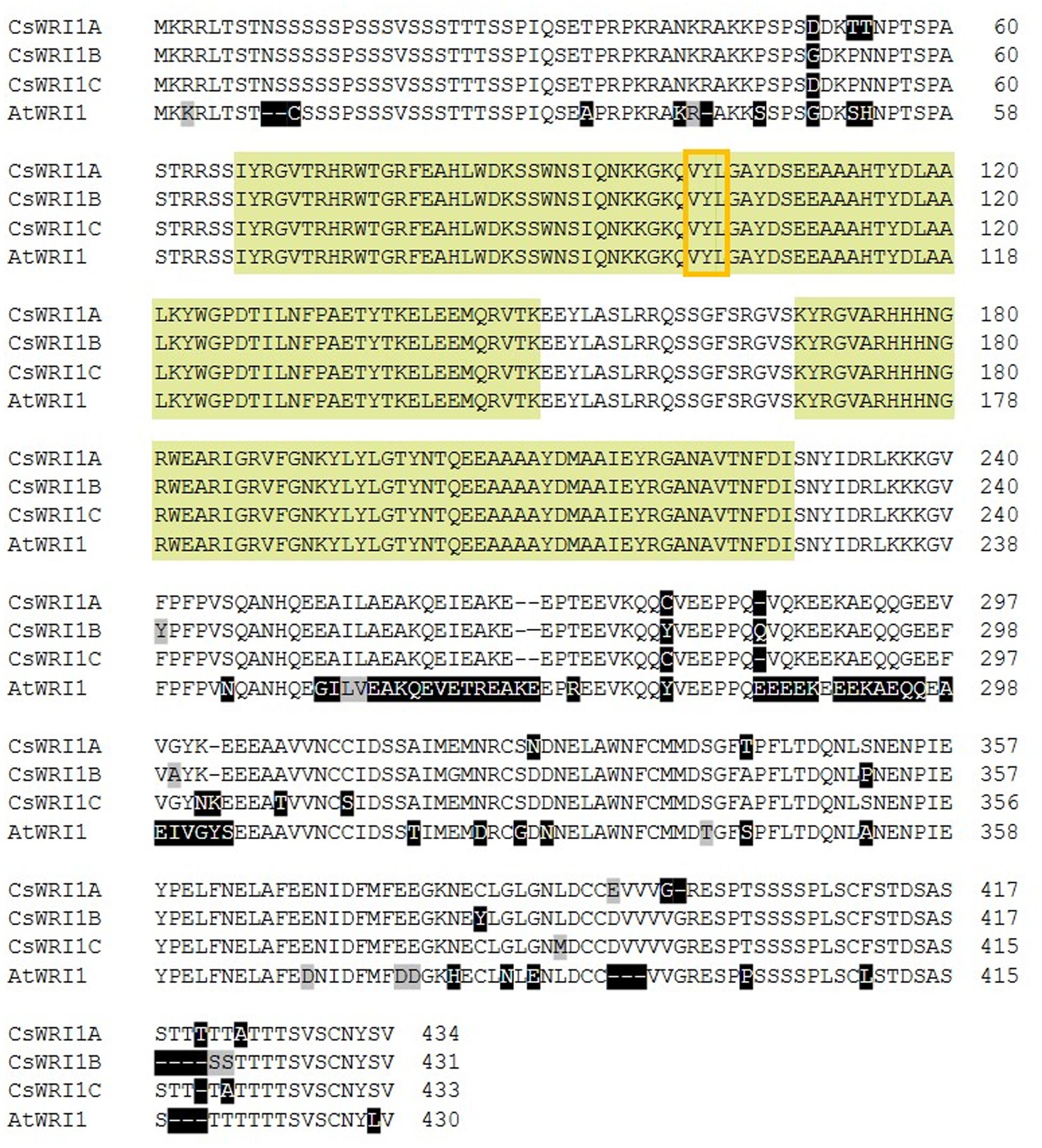
FIGURE 1. Alignment of the deduced amino acid sequences of WRI1 isoforms from C. sativa and A. thaliana. Non-conserved and conservatively changed amino acid residues are shaded in black and gray, respectively. Bright green and orange boxes indicate conserved AP2/EREBP DNA binding motifs and the ‘VYL’ motif for transcriptional activation of WRI1, respectively.
Several WRI1 genes were identified from crop plants following Arabidopsis (Figure 2) (Shen et al., 2010; Wu et al., 2014). The majority are single WRI1 genes identified from each species, except for maize, in which two isoforms, ZmWRI1a and ZmWRI1b, are present in the genome. Owing to its hexaploid genome, Camelina has three WRI1 isoforms, which is the highest number of copies discovered in any plant species to date. Phylogenetic analysis indicates that the CsWRI1s are grouped in the same clade as these of Arabidopsis and B. napus among the various WRI1s that are currently identified from plants (Figure 2).
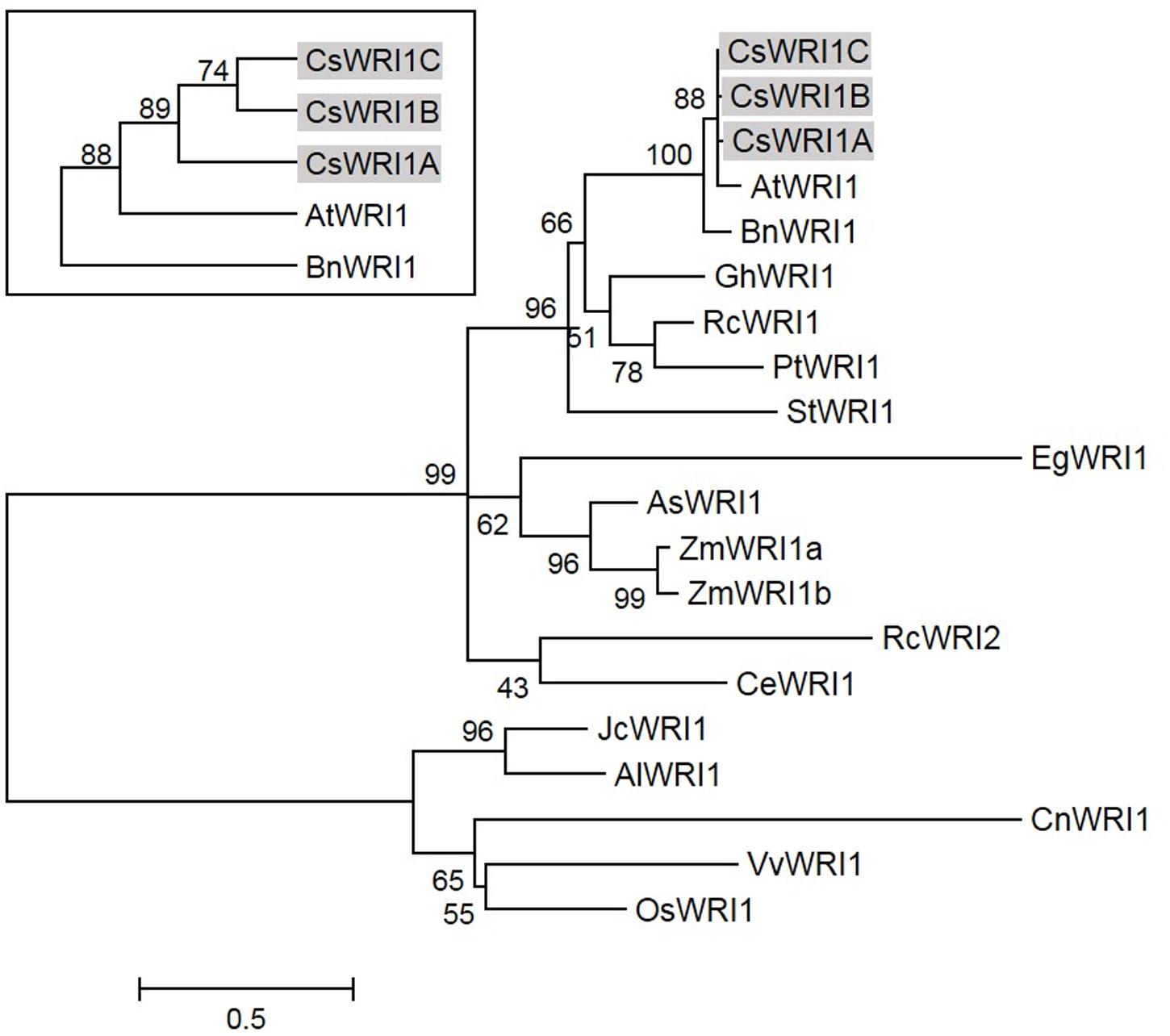
FIGURE 2. Phylogenetic tree of WRI1 from higher plants including C. sativa. Phylogenetic tree was generated using MEGA6.06 by the maximum likelihood method. Bootstrap value percentages of 500 replicates are shown at the branching points. AlWRI1, Arabidopsis lyrata (EFH52510.1); AsWRI1, Avena sativa (SRX1079426); BnWRI1, Brassica napus (ABD16282.1); CeWRI1, Cyperus esculentus (SRX1079431); CnWRI1, Cocos nucifera (JQ040545); EgWRI1, Elaeis guineensis; GhWRI1, Gossypium hirsutum (TC200263); JcWRI1, Jatropha curcas (AIA57945.1); OsWRI1, Oryza sativa (CAE00853.1); PtWRI1, Populus trichocarpa (SRX1079428); RcWRI1, Ricinus communis (AB774159.1, AB774160.1); StWRI1, Solanum tuberosum (SRX1079426); VvWRI1, Vitis vinifera (CBI32013.3); ZmWRI1, Zea mays (ACF83189.1, ACF80269.1).
Three WRI1s Are Predominantly Expressed in Developing Seeds
Since Arabidopsis WRI1 is known to express during seed development (Baud et al., 2007), the three CsWRI1s were analyzed for the expression pattern of their transcripts in developing seeds, which was then compared with that of the various tissues in Camelina. To distinguish the transcript of each CsWRI1 isoform from the three highly homologous genes, transcript-specific primers were designed and their specificity was confirmed by cDNA-PCR. Figure 3A shows each set of primers that were able to detect the specific transcript for CsWRI1 isoform. Using these gene-specific primers, RT-PCR analysis were performed in samples of the root, stems, leaves, flower buds, open flowers and three different stages of developing seeds. The expression levels of CsWRI1A, CsWRI1B, and CsWRI1C were predominantly higher in developing seeds than in other organs (Figure 3B). The Csa06g028810 transcript, showing 98% identity with CsWRI1B, represents high expression in developing seeds in the transcriptome analysis (Supplementary Table 3). These results indicate that all three CsWRI1 isoforms are actively transcribed during seed development.
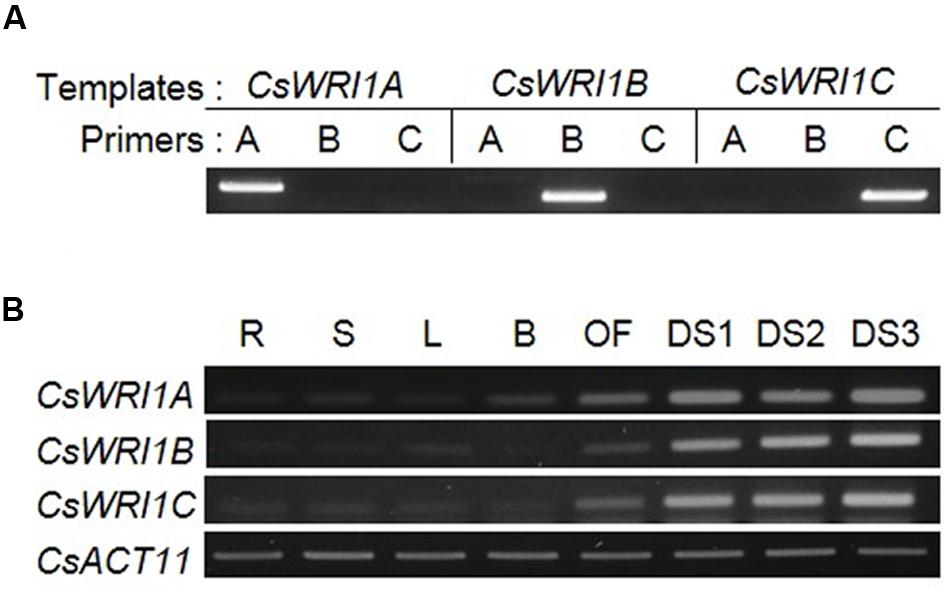
FIGURE 3. Expression of three CsWRI1 isoforms in various C. sativa organs. (A) The primer specificity of three CsWRI1 isoforms. Plasmids harboring CsWRI1A. CsWRI1B, or CsWRI1C were used in PCR analysis as templates. PCR products were analyzed on 1% agarose gels. (B) Total RNAs were isolated from the roots (R) of 2-week-old stems (S) of 4-week-old, leaves (L) and buds (B) from 5-week-old, and open flowers (OF) and developing seeds (DS1, DS2, and DS3; 10, 20, and 30 days after flowering, respectively) of 6-week-old C. sativa plants.
Three WRI1 Are Localized in the Nucleus
The amino acid sequences derived from the three CsWRI1 cDNAs revealed that the protein might be a transcription factor containing the AP2/EREBP DNA binding and VYL activation motifs (Figure 1). In order to detect the subcellular localization of Camelina WRI1, each CsWRI1 cDNA was fused in-frame with eYFP under the CaMV 35S promoter in the Agrobacterium binary expression vector (Figure 4A). Agrobacteria containing the CsWRI1s:eYFP genes were infiltrated into N. benthamiana leaves and the fluorescent signals were visualized under the confocal scanning microscope. The yellow fluorescent signals emitted by CsWRI1:eYFP were detected throughout the nucleus, except for the nucleolus, in all three WRI1 isoforms (Figures 4B,E,H). These signals identically overlapped with the blue fluorescent signal (Figures 4C,F,I) emitted by the nuclei stained with 4′,6-diamidino-2-phenylindole (DAPI) under UV light. The subcellular localization of CsWRI1A, CsWRI1B, and CsWRI1C indicate that all three isoforms localize in the nucleus (Figures 4D,G,J).
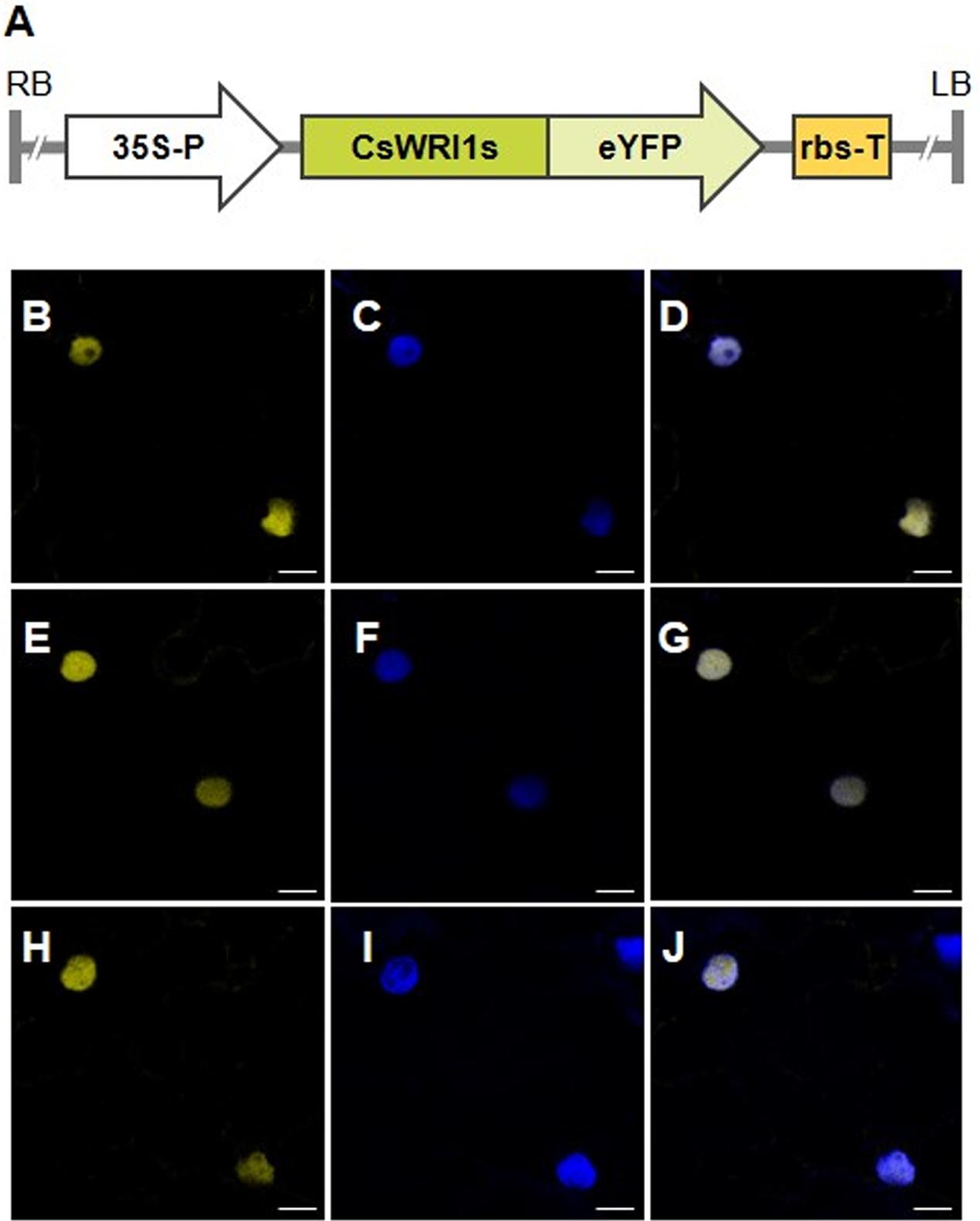
FIGURE 4. Subcellular localization of CsWRI1s:eYFP proteins in N. benthamiana epidermis. (A) Schematic diagram of CsWRI1A:eYFP. CsWRI1B:eYFP, and CsWRI1C:eYFP constructs. 35S-P, cauliflower mosaic virus 35S promoter; LB, left border; RB, right border; rbs-T, the terminator of ribulose-1,5-bisphosphate carboxylase and oxygenase small subunit from pea (Pisum sativum). (B–J) Agrobacterium harboring the CsWRI1A:eYFP (upper row), CsWRI1B:eYFP (middle row) or CsWRI1C:eYFP (bottom row) construct was infiltrated into N. benthamiana leaves and then the fluorescent signals were visualized under laser confocal scanning microscopy. YFP signals (B,E,H) from the CsWRI1s:eYFP constructs. The nucleus (C,F,I) was visualized by staining with DAPI under the UV filter. Merged image between signals of YFP and the nucleus (D,G,J). EV, empty vector (pBA002). Bars = 10 μm.
Three CsWRI1s Functionally Rescued the Arabidopsis wri1-3 Mutant Phenotype
Arabidopsis has a single WRI1 gene in its genome. Mutant seeds with T-DNA inserted in the fifth exon of AtWRI1 were obtained from the Arabidopsis Biological Resource Center (ABRC) and we identified the wri1-3 homozygous mutant (Figure 5A). This mutant exhibited the wrinkled phenotype in seeds, in contrast to the round phenotype of wild-type seeds (Figure 5B). To test if the three Camelina WRI1 genes can work functionally instead of AtWRI1 in the wri1-3 mutant, each of the CsWRI1 cDNAs was inserted between the CaMV 35S promoter and nos terminator in plant expression vectors (Figure 5C). These vectors were transformed into wri1-3, and the generated transgenic plants were selected based on the resistance to the herbicide Basta. Among the Basta resistant transgenic plants, 9–10 transgenic plants were analyzed for the presence of the transgene insert by the genomic DNA-PCR method with gene-specific primers for each CsWRI1. Transgenes were detected in genome DNA isolated from the leaves of T1 transformants, but not in that of wri1-3 and empty vector transgenic leaves (Figure 5D).
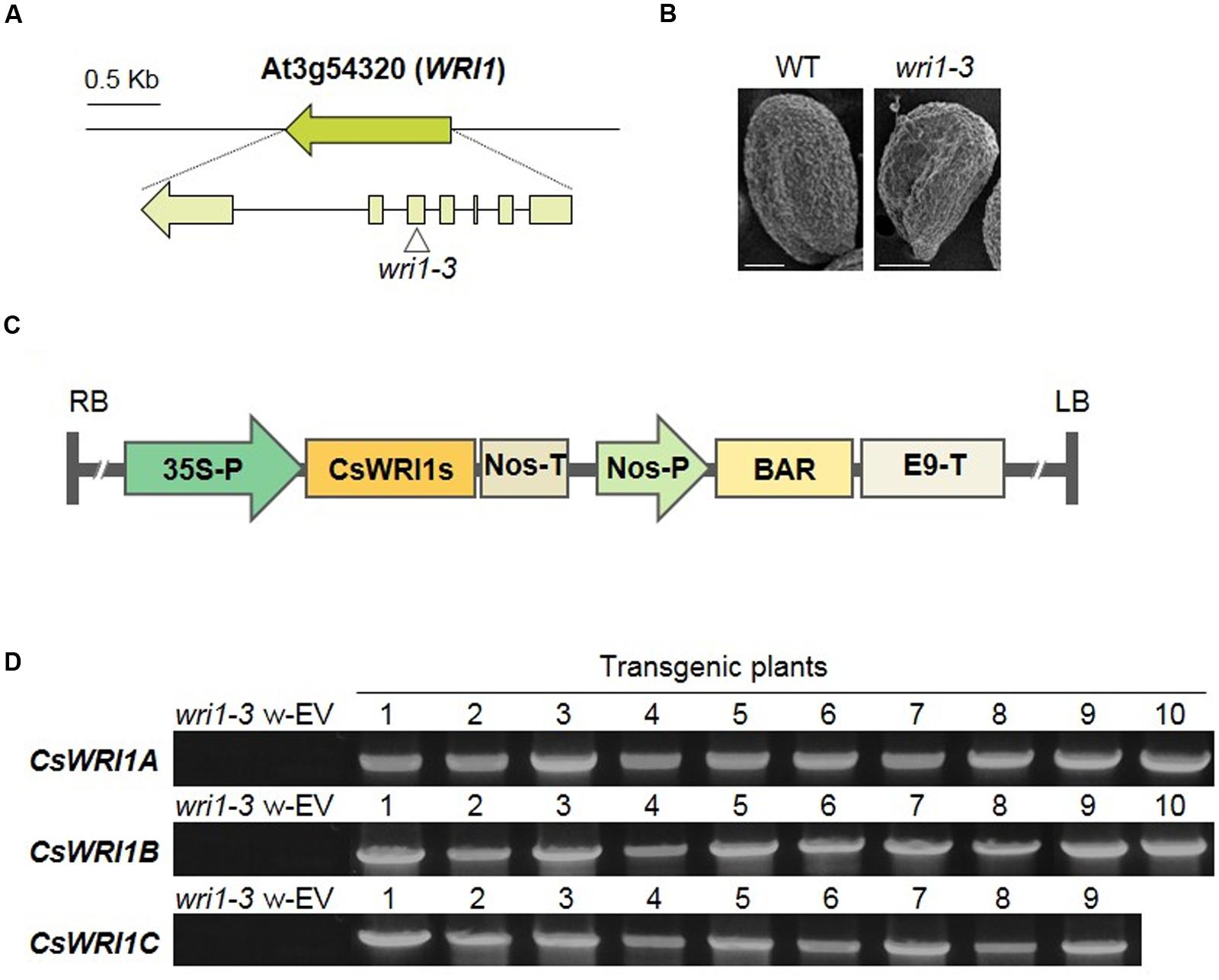
FIGURE 5. Isolation of T-DNA inserted wri1-3 knockout mutant and complementation of CsWRI1A. CsWRI1B, and CsWRI1C in wri1-3 mutants. (A) Genomic organization of the WRI1 gene inserted with T-DNA in wri1-3. (B) SEM image of the seed morphology of WT and wri1-3 mutant seeds. Bar = 10 μm. (C) Schematic diagram of the binary vector constructs for the expression of CsWRI1s in Arabidopsis wri1-3 mutant. (D) Genomic DNA-PCR of CsWRI1s transgenes of WT, wri1-3 mutant, and complementation transgenic lines. w-EV represents transgenic Arabidopsis wri1-3 mutant introducing empty vector. The numbers indicate independent transgenic lines (T1).
In the transgenic Arabidopsis wri1-3 plants expressing CsWRI1s, the fatty acid content of seeds in the T2 generation in nine to ten independent lines for each Camelina WRI1 isoform was measured and compared with those of the wri1-3, empty vector transformed wri1-3, and wild-type plants (Supplementary Table 4, Figure 6). To detect the fatty acid amount in seeds accurately, the dry weight of seeds and seed number were used for the measurement in three representative lines for each transgene. All three CsWRI1 cDNAs were able to partially complement the wri1-3 phenotype, as evidenced by a greater seed fatty acid content compared to the mutant and the vector-only transformant control irrespective of expression on the basis of seed weight or seed number (Figures 6A–C, Supplementary Table 5). These results suggest that CsWRI1A, B, and C variants are each potentially involved in regulating the level of fatty acid biosynthesis in developing seeds.
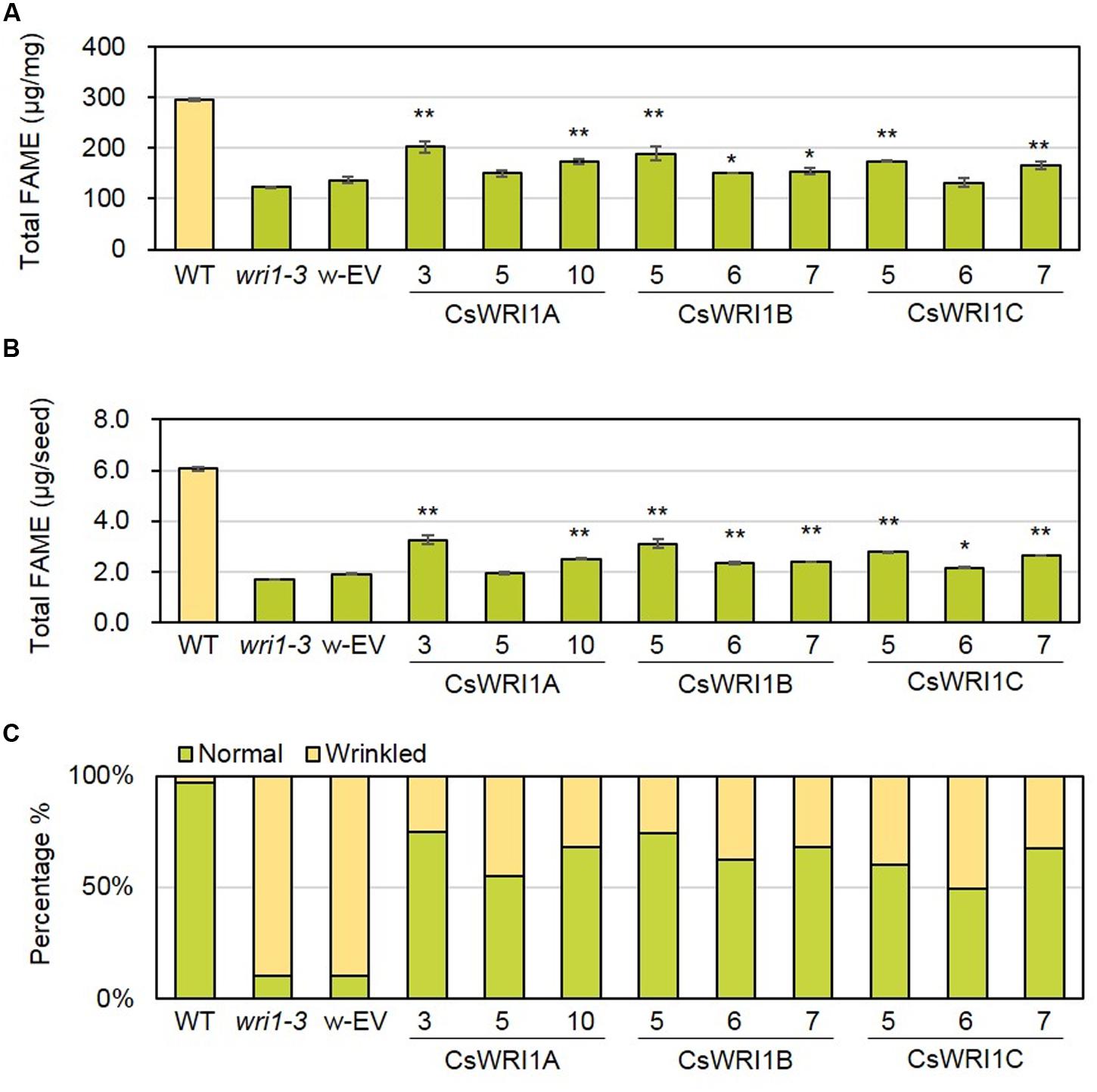
FIGURE 6. Fatty acid content and rescue of the wrinkled phenotype from seeds of WT, wri1-3, and transgenic plants. Fatty acids were extracted from dry seeds of WT, wri1-3, and transgenic plants (T2), transmethylated, and analyzed using gas chromatography. Fatty acid content was expressed on the basis of seed dry weight (A) or seed number (B). Error bar indicates SE of three independent measurements. (∗∗P < 0.01; ∗P < 0.05; Student’s t-test). (C) The graph showing morphology of dry seeds from WT, wri1-3, and transgenic lines (T2). w-EV represents transgenic Arabidopsis of wri1-3 mutant introducing empty vector. The numbers indicate independent transgenic lines.
We analyzed transcript levels of each CsWRI1 transgene in developing seeds with three best rescued plants for each CsWRI1 gene among all of the transgenic lines and compared them with the wri1-3 host line. RT-PCR analysis showed that each CsWRI1A. B, and C were expressed in developing seeds of transgenic plants and enhanced the expression level of genes involved in glycolysis and fatty acid synthesis, PI-PKβ1 and BCCP2 (Figure 7). These RT-PCR results showed correlation between the expression of CsWRI1 transgene, the partial complementation of seed fatty acid content, and seed morphology.
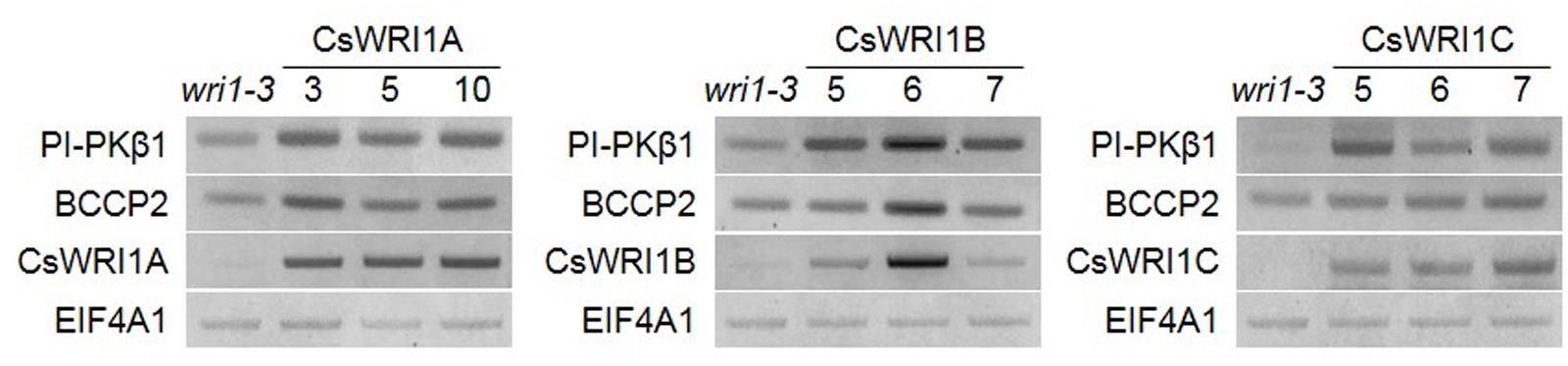
FIGURE 7. Expression of CsWRI1s and WRI1 downstream targets in developing Arabidopsis seeds. Total RNA was isolated from developing seeds 6–8 days after flowering of wri1-3 mutant and transgenic plants (T2). The isolated RNAs were subjected to RT-PCR analysis. The EIF4A1 gene was used to determine RNA quality and quantity. PI-PKβ1, At5g52920. BCCP2, At5g15530.
Ectopic Expression of Three CsWRI1s Induces Accumulation of Oil Bodies and Increases Fatty Acid Content in N. benthamiana Leaves
A limited number of WRI1 genes identified from model plants, such as Arabidopsis for dicots (Cernac and Benning, 2004), B. distachyon for temperate grasses (Yang et al., 2015), and B. napus (Liu et al., 2010) and maize (Shen et al., 2010) for crops, have been tested for their ability to enhance the oil content in developing seeds and vegetative tissues (Grimberg et al., 2015). We tested the potential of each Camelina WRI1 isoform to enhance the vegetable oil biosynthesis in N. benthamiana leaves. Agrobacteria containing the plant expression vector cloned with each CsWRI1 cDNA under the control of CaMV 35S promoter were infiltrated into N. benthamiana leaves (Figure 8A). The frequencies of oil bodies and fatty acid levels were examined and it was observed that leaf tissues expressing each of the CsWRI1 clearly indicated a greater number of oil bodies stained with Nile red than in the control leaves transformed with the empty vector (Figures 8B,C).
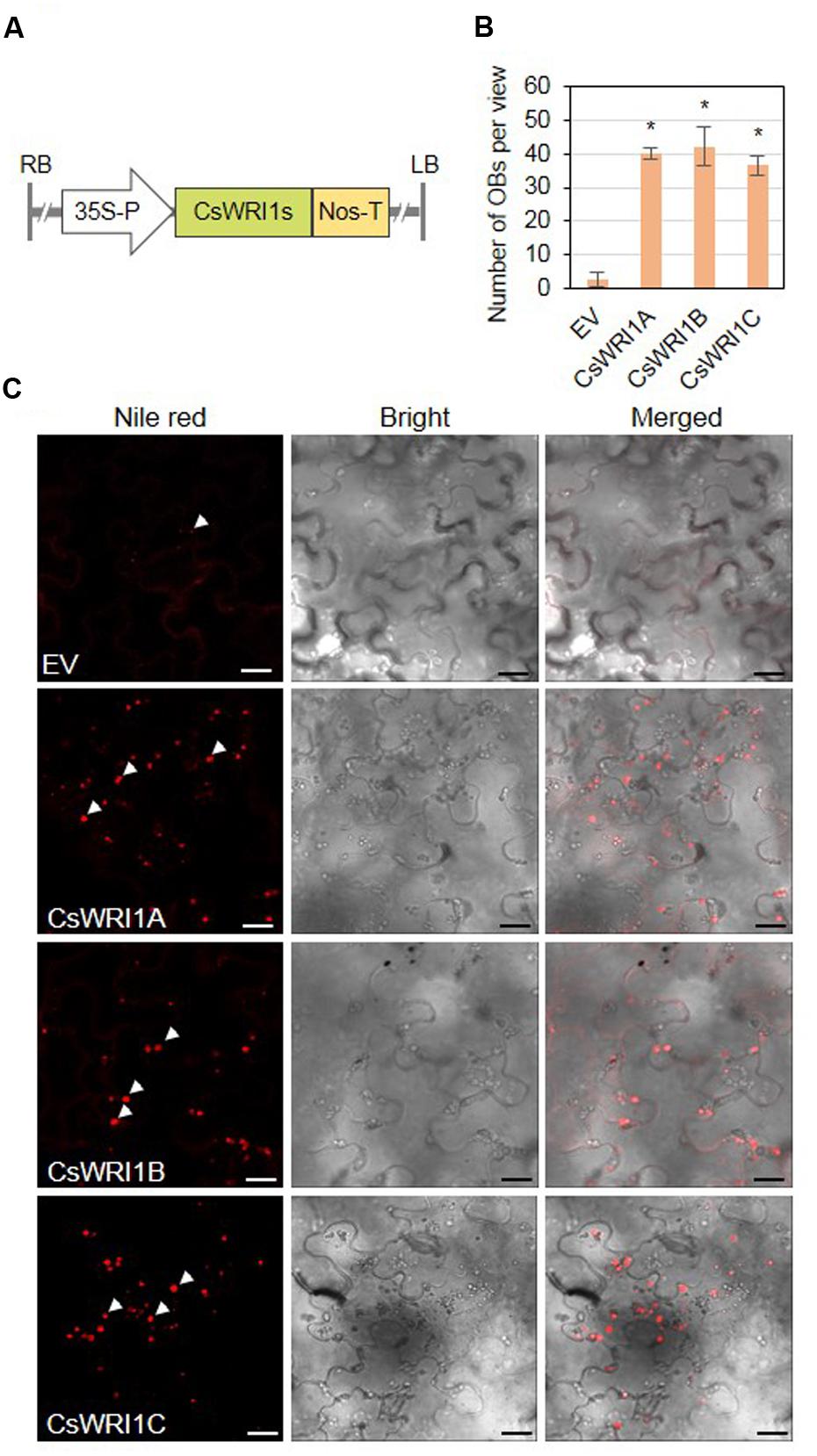
FIGURE 8. Transient expression of three CsWRI1 isoforms in N. benthamiana leaves. (A) Schematic diagram of the binary vector constructs for the transient expression of CsWRI1s in N. benthamiana leaves. (B) Oil body (OB) counts in N. benthaminana leaves expressing empty vector (pBA002), CsWRI1A. CsWRI1B, or CsWRI1C. Values are averages and SD of three individual images. Data were statistically analyzed using Student’s t-test (∗P < 0.01). (C) Agrobacterium harboring CsWRI1A. CsWRI1B, or CsWRI1C was infiltrated in N. benthamiana leaves and then the leaf disks were stained with Nile red solution. The fluorescent signals were visualized by laser confocal scanning microscopy. The white arrows indicate OBs. Bars = 20 μm.
In order to determine whether there is a correlation between the induced oil bodies in N. benthamiana leaves and an increased TAG content, N. benthamiana leaves infiltrated by CsWRI1s or the empty vector were measured for the fatty acid content in TAGs fractionated by TLC. Expression of each CsWRI1 enhanced TAG content 2.5- to 4.0-fold in fresh leaves compared to the empty-vector control (Figure 9A). Measurement of the total lipid amount in fresh leaves showed that CsWRI1s produced 17–41% more fatty acids in transformed leaves compared with the control leaves transformed with the empty vector (Figure 9B). The TAG and total lipid content in leaves transformed by CsWRI1s had a fatty acid composition somewhat different to that of the controls. Five fatty acids (18:3 > 16:0 > 18:2 > 18:1 > 18:0) accounted for 97–98% of all TAGs. Transformation with CsWRI1s also induced a subtle change in the fatty acid composition of TAGs, such as an increase in 18:1 and a decrease in 18:0 compared to the empty vector controls. In the total lipid analysis, five fatty acids (18:3 > 16:0 > 18:2 > 16:3 > 18:0) accounted for 94–95% of all lipids. Among major fatty acids in leaves, 18:3 was decreased and 18:2 increased in transformed leaves compared to the control leaves (Table 1). The enhanced leaf TAG content suggests that CsWRI1s can be employed to increase the vegetable oil content in biomass.
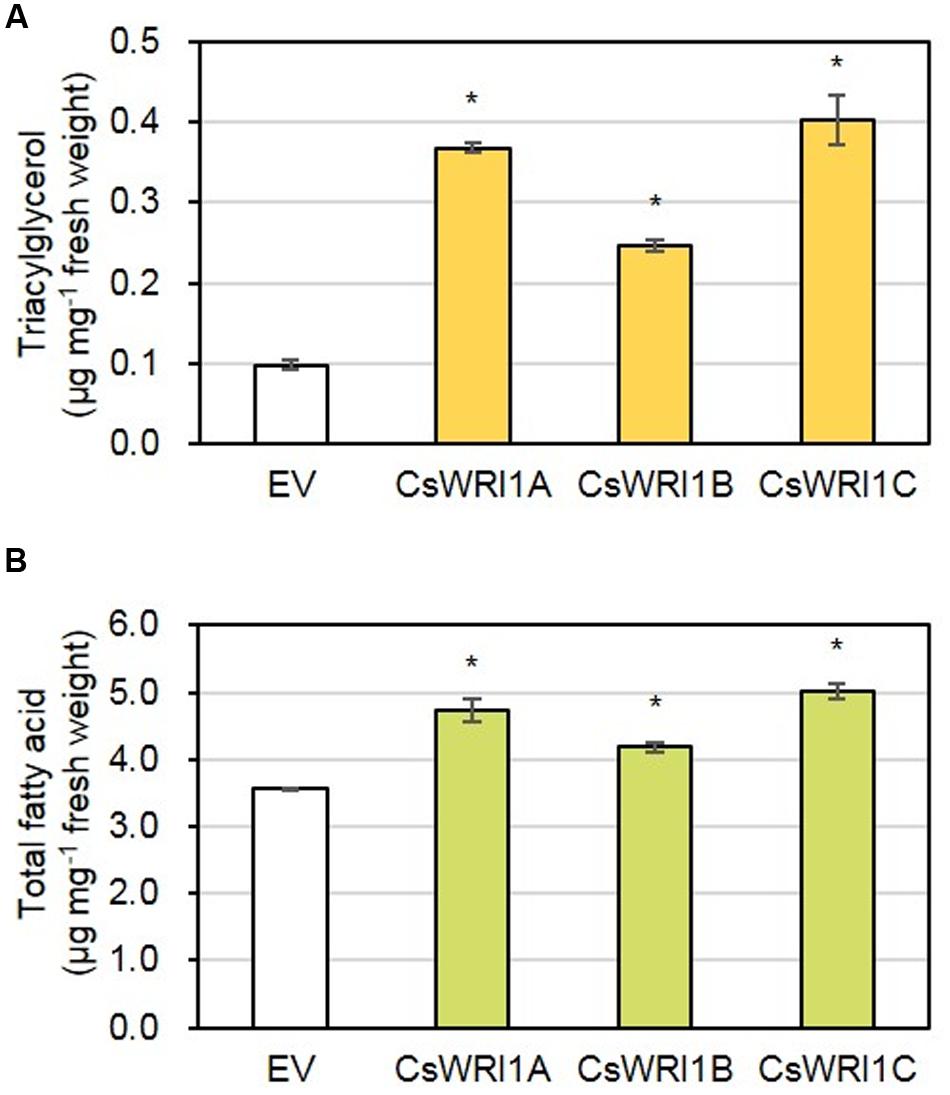
FIGURE 9. Fatty acid content present in triacylglycerols (A) and total lipids (B) of N. benthamiana leaves transiently overexpressing three CsWRI1 isoforms. Agrobacterium harboring empty vector (EV), CsWRI1A. CsWRI1B, or CsWRI1C was infiltrated to 5-week-old N. benthamiana leaves and the N. benthamiana plants were further incubated for 5 days. Total lipids were extracted from N. benthamiana leaf disks including the injection region and analyzed by gas chromatography or fractionated on thin layer chromatography. The eluted TAG fractions were transmethylated and the fatty acid methyl esters were analyzed by gas chromatography. Each value is the mean ± SE of three independent measurements. Data were statistically analyzed using Student’s t-test (∗P < 0.01).
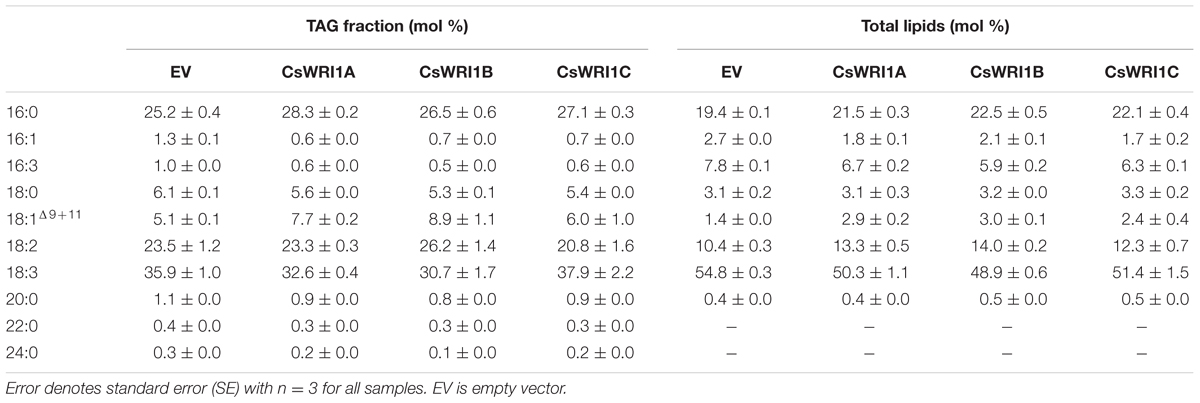
TABLE 1. Fatty acid profile of the TAG fraction and total lipids of leaf disks transiently expressing the three CsWRI1 genes.
Discussion
Camelina has an advantage as an oil crop as it is amenable to metabolic engineering for the manipulation of vegetable oil biosynthesis (Bansal and Durrett, 2016). Metabolic engineering in Camelina is underway to modify the fatty acid composition by blocking the endogenous fatty acid desaturase 2 (FAD) and fatty acid elongase (FAE) 1 pathways, which would make it more suitable for biofuel (Nguyen et al., 2013), and to incorporate new pathways to produce unusual fatty acids for industrial uses (Snapp et al., 2014; Liu et al., 2015; Nguyen et al., 2015), and omega-3 long-chain polyunsaturated fatty acids for human consumption (Petrie et al., 2014). Wax esters replaced 21% of the seed oil TAGs in Camelina transformed with two genes encoding a fatty alcohol-forming acyl-CoA reductase (FAR) and a wax ester synthase (WS) (Iven et al., 2016). Currently only two key genes that are involved in fatty acid and TAG biosynthesis metabolism, FAD2 (Kang et al., 2011) and DGAT1 (Kim et al., 2016), respectively, have been identified to exist in three copies in Camelina. In the present study, we show that the gene for WRI1, a regulator for fatty acid biosynthesis, also exists in three copies in the Camelina genome (Figure 1, Supplementary Table 2). The three copies (CsWRI1A, CsWRI1B, and CsWRI1C) were very similar in their amino acid sequences to those of A. thaliana (Figure 1). Phylogenetic tree analysis grouped the three CsWRI1s and those of Arabidopsis and Brassica to same clade (Figure 2). The AP2/EREBP DNA binding domain and a key ‘VYL’ motif for controlling the target gene’s transcription involved in fatty acid biosynthesis (Ma et al., 2013) were identically conserved but the N- and C-terminal amino acid sequences were moderately diverse in three Camelina and Arabidopsis WRI1s (Figure 1). A very high homology was observed among the three Camelina WRI1 genes and Arabidopsis WRI1, which may be construed as evidence for a whole-genome triplication event from the ancestral crucifer genome (Kagale et al., 2014). The nuclear localization of WRI1 has not been confirmed in any previous report, although it has been shown that WRI1 binds to the regulatory element of the promoters in fatty acid biosynthesis as a transcription factor (Baud et al., 2009; Maeo et al., 2009). In this report, we demonstrated that WRI1s are localized in the nucleus by using fluorescent fusion proteins (Figure 4).
All three Camelina WRI1s were abundantly expressed in developing seeds (Figure 3). CsWRI1A, the gene with the highest homology with Arabidopsis WRI1 exhibited higher expression levels than those of CsWRI1B and CsWRI1C (Figure 3). Ectopic expression of Camelina WRI1s partially replaced Arabidopsis WRI1 functionality by partially recovering the reduced amount of fatty acids and reversing the wrinkled phenotype in wri1-3 seeds as compared to those of the wild-type (Figures 5 and 6). Each of CsWRI1s transgenic lines upregulated PI-PKβ1 and BCCP2 downstream target genes in developing seeds compared with wri1-3 host plants (Figure 7). The reason that CsWRI1s could not fully recover the fatty acid amount and reverse the wrinkled phenotype in transgenic seeds may be that the use of the CaMV 35S promoter or the regulation of CsWRI1 by endogenous factors FUS3 and LEC2 may not be optimal for seed oil accumulation (Devic and Roscoe, 2016). The expression of phosphatidylcholine diacylglycerol cholinephosphotransferase (PDCT) of flax (Linum usitatissimum) under the control of CaMV 35S promoter in Arabidopsis rod1 mutant could not recover the PUFA content comparable with that observed in wild-type plants (Wickramarathna et al., 2015). In addition, the introduction of a vector combined with CaMV 35S promoter and B. rapa FAD2-1 gene into Arabidopsis fad2-2 loss-of-function mutant did not achieve the level of linoleic acid found in seeds of the wild-type plant (Jung et al., 2011). On the other hand, a recent study showed that the overexpression of WRI1 by FUS3 seed-specific promoter resulted in a stronger effect than that from CaMV 35S promoter alone for the induction of more oil production in seeds (Kanai et al., 2016). These similar results suggest that the CaMV 35S promoter might not be appropriate for metabolic engineering to enhance seed oil accumulation.
Oil production in leaves is an alternative way to increase the production of vegetable oils in limited cultivating lands and under water supply constraints to meet the increasing demand for vegetable oils as foods and biofuels. Oil accumulation in developing seed requires seed maturation controlled by four master regulators: ABI3, FUS3, LEC1, and LEC2. LEC2 is expressed early on in seed development, while FUS3 and ABI3 are more implicated in seed maturation processes (Wang et al., 2007; Wang and Perry, 2013; Roscoe et al., 2015; Zhang et al., 2016). WRI1 is a regulator of lipid accumulation downstream of the above four master regulators (Roscoe et al., 2015). Recently, researchers have attempted to produce vegetable oil (TAG or fatty acids) in leaves with the overexpression of a key regulator WRI1 for fatty acid synthesis as well as senescence-inducible or xylem-specific expression of LEC2, which is a transcription factor involved in the early stages of seed development (Kim et al., 2015). In most cases, since Arabidopsis WRI1 gene has not shown any negative effect on plant growth, it has been used mainly for the production of TAG in the vegetative tissues. Overexpression of Arabidopsis WRI1 alone elevated TAG levels to 2.8-fold in its seedlings (Sanjaya et al., 2011). Co-expression of Arabidopsis WRI1 and DGAT1, involved in the final step of TAG biosynthesis, and oleosin, responsible for the stability of TAG, resulted in an accumulation of 15% TAG of the estimated 17.7% total lipids in N. benthamiana leaves (Vanhercke et al., 2014). Co-expression of WRI1 and DGAT1-2 in a transgenic sugarcane plant, which also possessed suppressed endogenous genes for ADP-glucose pyrophosphorylase (AGPase) and peroximal ABC transport 1 (PXA1), resulted in increased total fatty acid content of up to 4.7 and 1.7% in leaves and stems, respectively (Zale et al., 2016). Since WRI1 is a regulator for lipid accumulation even in non-seed tissues (Kilaru et al., 2015), WRI1 paralogs distinct from seed-specific WRI1 were observed in the tallow layer, the non-seed tissue, in Chinese tallow (Triadica sebifera), which is a valuable oilseed-producing tree (Divi et al., 2016). In yet another study, the ectopic expression of B. distachyon WRI1 isolated from the vegetative tissues induced an increase in TAG content and simultaneously resulted in cell death in the leaf blades (Yang et al., 2015). Ectopic expression of WRI1s from diverse species and tissues in leaves of N. benthamiana has been shown to result in the production of TAG content ranging from 0.05 to 2.2%. (Grimberg et al., 2015). In the present study, the overexpression of three Camelina WRI1 isoforms resulted in the formation of oil bodies (Figure 8) and the production of approximately 0.025–0.04% TAG in fresh leaves, which represents a 2.5- to 4.0-fold increase from control leaves (Figure 9A). The amount of total lipids in fresh leaves was increased by a further 17–41% by introducing CsWRI1s (Figure 9B). The fatty acid composition of TAG and leaves was also found to be different (Table 1). Two major fatty acid levels were altered: 18:1 was increased and 18:0 was decreased in the TAG fraction, whereas 18:3 was decreased and 18:2 was increased in the total lipids of CsWRI1s transformants, compared with empty vector control leaves. This suggests CsWRI1s may drive 18:1 deposition for TAG synthesis. In conclusion, three WRI1 genes isolated from the hexaploid genome of Camelina, an emerging oil seed crop, can be used for the regulation of transcriptional factors to produce vegetable oils in engineered biomass.
Author Contributions
MCS and HUK designed the research; DA, HK, SJ, and YSG did experiments; and DA, HK, HUK, and MCS analyzed the data and wrote a manuscript.
Funding
This work was supported by Grants from the Korea Institute of Planning and Evaluation for Technology of the Ministry for Food, Agriculture, Forestry and Fisheries (312033-05), Republic of Korea, and the Next-Generation BioGreen 21 Program (No. PJ011052) of the Rural Development Administration, Republic of Korea.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We would like to thank Young-Woo Seo for performing the confocal laser microscope analysis at the Korea Basic Science Institute Gwangju Center.
Supplementary Material
The Supplementary Material for this article can be found online at: http://journal.frontiersin.org/article/10.3389/fpls.2017.00034/full#supplementary-material
Footnotes
References
An, D., and Suh, M. C. (2015). Overexpression of Arabidopsis WRI1 enhanced seed mass and storage oil content in Camelina sativa. Plant Biotechnol. Rep. 9, 137–148. doi: 10.1007/s11816-015-0351-x
An, G. (1987). Binary Ti vectors for plant transformation and promoter analysis. Methods Enzymol. 153, 292–305. doi: 10.1186/1472-6750-10-67
Athenstaedt, K., and Daum, G. (2006). The life cycle of neutral lipids: synthesis, storage and degradation. Cell. Mol. Life Sci. 63, 1355–1369. doi: 10.1007/s00018-006-6016-8
Bansal, S., and Durrett, T. P. (2016). Camelina sativa: an ideal platform for the metabolic engineering and field production of industrial lipids. Biochimie 120, 9–16. doi: 10.1016/j.biochi.2015.06.009
Baud, S., Mendoza, M. S., To, A., Harscoet, E., Lepiniec, L., and Dubreucq, B. (2007). WRINKLED1 specifies the regulatory action of LEAFY COTYLEDON2 towards fatty acid metabolism during seed maturation in Arabidopsis. Plant J. 50, 825–838. doi: 10.1111/j.1365-313X.2007.03092.x
Baud, S., Wuilleme, S., To, A., Rochat, C., and Lepiniec, L. (2009). Role of WRINKLED1 in the transcriptional regulation of glycolytic and fatty acid biosynthetic genes in Arabidopsis. Plant J. 60, 933–947. doi: 10.1111/j.1365-313X.2009.04011.x
Cernac, A., and Benning, C. (2004). WRINKLED1 encodes an AP2/EREB domain protein involved in the control of storage compound biosynthesis in Arabidopsis. Plant J. 40, 575–585. doi: 10.1111/j.1365-313X.2004.02235.x
Chapman, K. D., and Ohlrogge, J. B. (2012). Compartmentation of triacylglycerol accumulation in plants. J. Biol. Chem. 287, 2288–2294. doi: 10.1074/jbc.R111.290072
Devic, M., and Roscoe, T. (2016). Seed maturation: simplification of control networks in plants. Plant Sci. 252, 335–346. doi: 10.1016/j.plantsci.2016.08.012
Divi, U. K., Zhou, X. R., Wang, P., Butlin, J., Zhang, D. M., Liu, Q., et al. (2016). Deep sequencing of the fruit transcriptome and lipid accumulation in a non-seed tissue of chinese tallow, a potential biofuel crop. Plant Cell Physiol. 57, 125–137. doi: 10.1093/pcp/pcv181
Focks, N., and Benning, C. (1998). wrinkled1: A novel, low-seed-oil mutant of Arabidopsis with a deficiency in the seed-specific regulation of carbohydrate metabolism. Plant Physiol. 118, 91–101. doi: 10.1104/pp.118.1.91
Fukuda, N., Ikawa, Y., Aoyagi, T., and Kozaki, A. (2013). Expression of the genes coding for plastidic acetyl-CoA carboxylase subunits is regulated by a location-sensitive transcription factor binding site. Plant Mol. Biol. 82, 473–483. doi: 10.1007/s11103-013-0075-7
Grimberg, A., Carlsson, A. S., Marttila, S., Bhalerao, R., and Hofvander, P. (2015). Transcriptional transitions in Nicotiana benthamiana leaves upon induction of oil synthesis by WRINKLED1 homologs from diverse species and tissues. BMC Plant Biol. 15:192. doi: 10.1186/s12870-015-0579-1
Hutcheon, C., Ditt, R. F., Beilstein, M., Comai, L., Schroeder, J., Goldstein, E., et al. (2010). Polyploid genome of Camelina sativa revealed by isolation of fatty acid synthesis genes. BMC Plant Biol. 10:223. doi: 10.1186/1471-2229-10-233
Iven, T., Hornung, E., Heilmann, M., and Feussner, I. (2016). Synthesis of oleyl oleate wax esters in Arabidopsis thaliana and Camelina sativa seed oil. Plant Biotechnol. J. 14, 252–259. doi: 10.1111/pbi.12379
Jung, J. H., Kim, H., Go, Y. S., Lee, S. B., Hur, C.-G., Kim, H. U., et al. (2011). Identification of functional BrFAD2-1 gene encoding microsomal delta-12 fatty acid desaturase from Brassica rapa and development of Brassica napus containing high oleic acid contents. Plant Cell Rep. 30, 1881–1892. doi: 10.1007/s00299-011-1095-x
Kagale, S., Koh, C., Nixon, J., Bollina, V., Clarke, W. E., Tuteja, R., et al. (2014). The emerging biofuel crop Camelina sativa retains a highly undifferentiated hexaploid genome structure. Nat. Commun. 5:3706. doi: 10.1038/ncomms4706
Kanai, M., Mano, S., Kondo, M., Hayashi, M., and Nishimura, M. (2016). Extension of oil biosynthesis during the mid-phase of seed development enhances oil content in Arabidopsis seeds. Plant Biotechnol. J. 14, 1241–1250. doi: 10.1111/pbi.12489
Kang, J., Snapp, A. R., and Lu, C. (2011). Identification of three genes encoding microsomal oleate desaturases (FAD2) from the oilseed crop Camelina sativa. Plant Physiol. Biochem. 49, 223–229. doi: 10.1016/j.plaphy.2010.12.004
Kaup, M. T., Froese, C. D., and Thompson, J. E. (2002). A role for diacylglycerol acyltransferase during leaf senescence. Plant Physiol. 129, 1616–1626. doi: 10.1104/pp.003087
Kilaru, A., Cao, X., Dabbs, P. B., Sung, H. J., Rahman, M. M., Thrower, N., et al. (2015). Oil biosynthesis in a basal angiosperm: transcriptome analysis of Persea Americana mesocarp. BMC Plant Biol. 15:203. doi: 10.1186/s12870-015-0586-2
Kim, H., Park, J. H., Kim, D. J., Kim, A. Y., and Suh, M. C. (2016). Functional analysis of diacylglycerol acyltransferase1 genes from Camelina sativa and effects of CsDGAT1B overexpression on seed mass and storage oil content in C. sativa. Plant Biotechnol. Rep. 10, 141–153. doi: 10.1007/s11816-016-0394-7
Kim, H. S., Oh, J. M., Luan, S., Carlson, J. E., and Ahn, S. J. (2013). Cold stress causes rapid but differential changes in properties of plasma membrane H(+)-ATPase of camelina and rapeseed. J. Plant Physiol. 170, 828–837. doi: 10.1016/j.jplph.2013.01.007
Kim, H. U., Hsieh, K., Ratnayake, C., and Huang, A. H. C. (2002). A novel group of oleosins is present inside the pollen of Arabidopsis. J. Biol. Chem. 277, 22677–22684. doi: 10.1074/jbc.M109298200
Kim, H. U., Lee, K. R., Jung, S. J., Shin, H. A., Go, Y. S., Suh, M. C., et al. (2015). Senescence-inducible LEC2 enhances triacylglycerol accumulation in leaves without negatively affecting plant growth. Plant Biotechnol. J. 13, 1346–1359. doi: 10.1111/pbi.12354
Kim, Y. S., Kim, S. G., Park, J. E., Park, H. Y., Lim, M. H., Chua, N. H., et al. (2006). A membrane-bound NAC transcription factor regulates cell division in Arabidopsis. Plant Cell 18, 3132–3144. doi: 10.1105/tpc.106.043018
Li-Beisson, Y., Shorrosh, B., Beisson, F., Andersson, M. X., Arondel, V., Bates, P. D., et al. (2010). Acyl-lipid metabolism. Arabidopsis Book 8:e0133. doi: 10.1199/tab.0133
Liu, J., Hua, W., Zhan, G., Wei, F., Wang, X., Liu, G., et al. (2010). Increasing seed mass and oil content in transgenic Arabidopsis by the overexpression of wri1-like gene from Brassica napus. Plant Physiol. Biochem. 48, 9–15. doi: 10.1016/j.plaphy.2009.09.007
Liu, J., Rice, A., McGlew, K., Shaw, V., Park, H., Clemente, T., et al. (2015). Metabolic engineering of oilseed crops to produce high levels of novel acetyl glyceride oils with reduced viscosity, freezing point and calorific value. Plant Biotechnol. J. 13, 858–865. doi: 10.1111/pbi.12325
Liu, X., Brost, J., Hutcheon, C., Guilfoil, R., Wilson, A. K., Leung, S., et al. (2012). Transformation of the oilseed crop Camelina sativa by Agrobacterium-mediated floral dip and simple large-scale screening of transformants. In Vitro Cell Dev. Biol. 48, 462–468. doi: 10.1007/s11627-012-9459-7
Lu, C., and Kang, J. (2008). Generation of transgenic plants of a potential oilseed crop Camelina sativa by Agrobacterium-mediated transformation. Plant Cell Rep. 27, 273–278. doi: 10.1007/s00299-007-0454-0
Ma, W., Kong, Q., Arondel, V., Kilaru, A., Bates, P. D., Thrower, N. A., et al. (2013). Wrinkled1, a ubiquitous regulator in oil accumulating tissues from Arabidopsis embryos to oil palm mesocarp. PLoS ONE 8:e68887. doi: 10.1371/journal.pone.0068887
Ma, W., Kong, Q., Grix, M., Mantyla, J. J., Yang, Y., Benning, C., et al. (2015). Deletion of a C-terminal intrinsically disordered region of WRINKLED1 affects its stability and enhances oil accumulation in Arabidopsis. Plant J. 83, 864–874. doi: 10.1111/tpj.12933
Maeo, K., Tokuda, T., Ayame, A., Mitsui, N., Kawai, T., Tsukagoshi, H., et al. (2009). An AP2-type transcription factor, WRINKLED1, of Arabidopsis thaliana binds to the AW-box sequence conserved among proximal upstream regions of genes involved in fatty acid synthesis. Plant J. 60, 476–487. doi: 10.1111/j.1365-313X.2009.03967.x
Nguyen, H. T., Park, H., Koster, K. L., Cahoon, R. E., Nguyen, H. T., Shanklin, J., et al. (2015). Redirection of metabolic flux for high levels of omega-7 monounsaturated fatty acid accumulation in camelina seeds. Plant Biotechnol. J. 13, 38–50. doi: 10.1111/pbi.12233
Nguyen, H. T., Silva, J. E., Podicheti, R., Macrander, J., Yang, W., Nazarenus, T. J., et al. (2013). Camelina seed transcriptome: a tool for meal and oil improvement and translational research. Plant Biotechnol. J. 11, 759–769. doi: 10.1111/pbi.12068
Nookaraju, A., Pandey, S. K., Fujino, T., Kim, J. Y., Suh, M. C., and Joshi, C. P. (2014). Enhanced accumulation of fatty acids and triacylglycerols in transgenic tobacco stems for enhanced bioenergy production. Plant Cell Rep. 33, 1041–1052. doi: 10.1007/s00299-014-1582-y
North, H., Baud, S., Debeaujon, I., Dubos, C., Dubreucq, B., Grappin, P., et al. (2010). Arabidopsis seed secrets unravelled after a decade of genetic and omics-driven research. Plant J. 61, 971–981. doi: 10.1111/j.1365-313X.2009.04095.x
Oñate-Sánchez, L., and Vicente-Carbajosa, J. (2008). DNA-free RNA isolation protocols for Arabidopsis thaliana, including seeds and siliques. BMC Res. Notes 1:93. doi: 10.1186/1756-0500-1-93
Petrie, J. R., Shrestha, P., Belide, S., Kennedy, Y., Lester, G., Liu, Q., et al. (2014). Metabolic engineering Camelina sativa with fish oil-like levels of DHA. PLoS ONE 9:e85061. doi: 10.1371/journal.pone.0085061
Pouvreau, B., Baud, S., Vernoud, V., Morin, V., Py, C., Gendrot, G., et al. (2011). Duplicate maize wrinkled1 transcription factors activate target genes involved in seed oil biosynthesis. Plant Physiol. 156, 674–686. doi: 10.1104/pp.111.173641
Putnam, D. H., Budin, J. T., Field, L. A., and Breene, W. M. (1993). “Camelina: a promising low-input oilseed,” in New Crops, eds J. Janick and J. E. Simon (New York, NY: John Wiley & Sons).
Roscoe, T. T., Guilleminot, J., Bessoule, J. J., Berger, F., and Devic, M. (2015). Complementation of seed maturation phenotypes by ectopic expression of ABSCISIC ACID INSENSITIVE3, FUSCA3 and LEAFY COTYLEDON2 in Arabidopsis. Plant Cell Physiol. 56, 1215–1228. doi: 10.1093/pcp/pcv049
Sanjaya, Durrett, T. P., Weise, S. E., and Benning, C. (2011). Increasing the energy density of vegetative tissues by diverting carbon from starch to oil biosynthesis in transgenic Arabidopsis. Plant Biotechnol. J. 9, 874–883. doi: 10.1111/j.1467-7652.2011.00599.x
Shen, B., Allen, W. B., Zheng, P., Li, C., Glassman, K., Ranch, J., et al. (2010). Expression of ZmLEC1 and ZmWRI1 increases seed oil production in Maize. Plant Physiol. 153, 980–987. doi: 10.1104/pp.110.157537
Snapp, A. R., Kang, J., Qi, X., and Lu, C. (2014). A fatty acid condensing enzyme from Physaria fendleri increases hydroxy fatty acid accumulation in transgenic oilseeds of Camelina sativa. Planta 240, 599–610. doi: 10.1007/s00425-014-2122-2
Soriano, N. U., and Narani, A. (2012). Evaluation of biodiesel derived from Camelina sativa oil. J. Am. Oil Chem. Soc. 89, 917–923. doi: 10.1007/s11746-011-1970-1
Tamura, K., Stecher, G., Peterson, D., Filipski, A., and Kumar, S. (2013). MEGA6: molecular evolutionary genetics analysis version 6.0. Mol. Biol. Evol. 30,k2725–2729. doi: 10.1093/molbev/mst197
Vanhercke, T., El Tahchy, A., Liu, Q., Zhou, X. R., Shrestha, P., Divi, U. K., et al. (2014). Metabolic engineering of biomass for high energy density: oilseed-like triacylglycerol yields from plant leaves. Plant Biotechnol. J. 12, 231–239. doi: 10.1111/pbi.12131
Vanhercke, T., El Tahchy, A., Shrestha, P., Zhou, X. R., Singh, S. P., and Petrie, J. R. (2013). Synergistic effect of WRI1 and DGAT1 coexpression on triacylglycerol biosynthesis in plants. FEBS Lett. 587, 364–369. doi: 10.1016/j.febslet.2012.12.018
Wang, F., and Perry, S. E. (2013). Identification of direct targets of FUSCA3, a key regulator of Arabidopsis seed development. Plant Physiol. 161, 1251–1264. doi: 10.1104/pp.112.212282
Wang, H., Guo, J., Lambert, K. N., and Lin, Y. (2007). Developmental control of Arabidopsis seed oil biosynthesis. Planta 226, 773–783. doi: 10.1007/s00425-007-0524-0
Wickramarathna, A. D., Siloto, R. M. P., Mietkiewska, E., Singer, S. D., Pan, X., and Weselake, R. J. (2015). Heterologous expression of flax PHOSPHOLIPID:DIACYLGLYCEROL CHOLINEPHOSPHOTRANSFERASE (PDCT) increases polyunsaturated fatty acid content in yeast and Arabidopsis seeds. BMC Biotechnol. 15:63. doi: 10.1186/s12896-015-0156-6
Wu, X. L., Liu, Z. H., Hu, Z. H., and Huang, R. Z. (2014). BnWRI1 coordinates fatty acid biosynthesis and photosynthesis pathways during oil accumulation in rapeseed. J. Integr. Plant Biol. 56, 582–593. doi: 10.1111/jipb.12158
Yamamoto, A., Kagaya, Y., Usui, H., Hobo, T., Takeda, S., and Hattori, T. (2010). Diverse roles and mechanisms of gene regulation by the Arabidopsis seed maturation master regulator FUS3 revealed by microarray analysis. Plant Cell Physiol. 51, 2031–2046. doi: 10.1093/pcp/pcq162
Yang, Y., Munz, J., Cass, C., Zienkiewicz, A., Kong, Q., Ma, W., et al. (2015). Ectopic expression of WRINKLED1 affects fatty acid homeostasis in Brachypodium distachyon vegetative tissues. Plant Physiol. 169, 1836–1847. doi: 10.1104/pp.15.01236
Yoo, S.-D., Cho, Y.-H., and Sheen, J. (2007). Arabidopsis mesophyll protoplasts: a versatile cell system for transient gene expression analysis. Nat. Protocols 2, 1565–1572. doi: 10.1038/nprot.2007.199
Zale, J., Jung, J. H., Kim, J. Y., Pathak, B., Karan, R., Liu, H., et al. (2016). Metabolic engineering of sugarcane to accumulate energy-dense triacylglycerols in vegetative biomass. Plant Biotechnol. J. 14, 661–669. doi: 10.1111/pbi.12411
Zhang, M., Cao, X., Jia, Q., and Ohlrogge, J. (2016). FUSCA3 activates triacylglycerol accumulation in Arabidopsis seedlings and tobacco BY2 cells. Plant J. 88, 95–107. doi: 10.1111/tpj.13233
Keywords: Camelina sativa, fatty acid, leaves, oil, triacylglycerol, WRINKLED1
Citation: An D, Kim H, Ju S, Go YS, Kim HU and Suh MC (2017) Expression of Camelina WRINKLED1 Isoforms Rescue the Seed Phenotype of the Arabidopsis wri1 Mutant and Increase the Triacylglycerol Content in Tobacco Leaves. Front. Plant Sci. 8:34. doi: 10.3389/fpls.2017.00034
Received: 18 September 2016; Accepted: 06 January 2017;
Published: 24 January 2017.
Edited by:
Raúl Alvarez-Venegas, CINVESTAV, MexicoReviewed by:
Xue-Rong Zhou, CSIRO Agriculture, AustraliaThomas James Roscoe, Institut de Recherche Pour le Développement, France
Copyright © 2017 An, Kim, Ju, Go, Kim and Suh. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Hyun Uk Kim, aHVraW02NEBzZWpvbmcuYWMua3I= Mi Chung Suh, bWNzdWhAY2hvbm5hbS5hYy5rcg==
†Present address: Young Sam Go, Advanced Radiation Technology Institute, Korea Atomic Energy Research Institute, 29 Geumgu-gil, Jeongeup-si, South Korea
‡These authors have contributed equally to this work.
 Dahee An1‡
Dahee An1‡ Young Sam Go
Young Sam Go Hyun Uk Kim
Hyun Uk Kim Mi Chung Suh
Mi Chung Suh