- 1South African Sugarcane Research Institute, Mount Edgecombe, South Africa
- 2School of Animal, Plant and Environmental Sciences, University of the Witwatersrand, Johannesburg, South Africa
Soils of the tropics and sub-tropics are typically acid and depleted of soluble sources of silicon (Si) due to weathering and leaching associated with high rainfall and temperatures. Together with intensive cropping, this leads to marginal or deficient plant Si levels in Si-accumulating crops such as rice and sugarcane. Although such deficiencies can be corrected with exogenous application of Si sources, there is controversy over the effectiveness of sources in relation to their total Si content, and their capacity to raise soil and plant Si concentrations. This study tested the hypothesis that the total Si content and provision of plant-available Si from six sources directly affects subsequent plant Si uptake as reflected in leaf Si concentration. Two trials with potted cane plants were established with the following Si sources as treatments: calcium silicate slag, fused magnesium (thermo) phosphate, volcanic rock dust, magnesium silicate, and granular potassium silicate. Silicon sources were applied at rates intended to achieve equivalent elemental soil Si concentrations; controls were untreated or lime-treated. Analyses were conducted to determine soil and leaf elemental concentrations. Among the sources, calcium silicate produced the highest leaf Si concentrations, yet lower plant-available soil Si concentrations than the thermophosphate. The latter, with slightly higher total Si than the slag, produced substantially greater increases in soil Si than all other products, yet did not significantly raise leaf Si above the controls. All other sources did not significantly increase soil or leaf Si concentrations, despite their high Si content. Hence, the total Si content of sources does not necessarily concur with a product's provision of soluble soil Si and subsequent plant uptake. Furthermore, even where soil pH was raised, plant uptake from thermophosphate was well below expectation, possibly due to its limited liming capacity. The ability of the calcium silicate to provide Si while simultaneously and significantly increasing soil pH, and thereby reducing reaction of Si with exchangeable Al3+, is proposed as a potential explanation for the greater Si uptake into the shoot from this source.
Introduction
Although silicon (Si) is abundant in the Earth's crust (28.8%) (Wedepohl, 1995), it is not considered an essential element for terrestrial plants other than the Equisitaceae (Epstein, 1994). However, there is now considerable evidence for its role in plant health and ecology (Cooke and Leishman, 2011, 2012), and specifically in mitigating numerous abiotic and biotic stresses, including water and salinity stress, metal toxicities, nutrient imbalance, fungal and bacterial pathogens, and insect herbivores (reviews by Ma, 2004; Datnoff et al., 2007; Liang et al., 2007; Epstein, 2009; Reynolds et al., 2009; Zhu and Gong, 2014; Adrees et al., 2015). Among crop species that accumulate Si to levels >1.0% shoot Si dry mass (Ma and Takahashi, 2002), rice (Oryza sativa L.) and sugarcane (Saccharum spp. hybrids) have been well-studied, and are capable of removing up to 470 and 700 kg Si ha−1 annum−1, respectively, on Si-rich soils (Ross et al., 1974; Savant et al., 1997b, 1999; Meena et al., 2014). Yield responses in rice and sugarcane to soil Si amendments have frequently been recorded on the weathered tropical or sub-tropical soils on which they are largely grown (e.g., Oxisols, Ultisols, and organic Histosols) (Cheong and Halais, 1969; Elawad et al., 1982; Yamauchi and Winslow, 1989; Savant et al., 1997b, 1999; Alvarez and Datnoff, 2001; Meyer and Keeping, 2001; Berthelsen et al., 2001b; Tsujimoto et al., 2014). Due to high rainfall and temperatures, such soils have typically been depleted (desilicated) of soluble sources of Si (McKeague and Cline, 1963c; Savant et al., 1997a; Epstein, 2001; Meena et al., 2014), leading to marginal or deficient levels of plant Si in these crops (Savant et al., 1997a, 1999; Meena et al., 2014). Besides being low in essential nutrients, these highly weathered soils are also acidic and may therefore be high in soluble forms of aluminum (Al) (where soil pHw < 5.5) (Sanchez, 1976; Fageria et al., 1988), which in turn can remove soluble Si through reaction to form insoluble hydroxyaluminosilicates (HASs) (Farmer et al., 1979; Doucet et al., 2001; Schneider et al., 2004).
Plants take up Si as monomeric silicic acid (H4SiO4), the dominant form of Si in soil solution (Epstein, 1994). The solubility of silicic acid in soil solution is strongly pH-dependent and related to its adsorption/desorption reactions on soil colloids. Solubility and concentration in soil solution is highest at low pH and decreases progressively up to a pH of 9.8, the pK1 of silicic acid, where the latter dissociates to form H3. At this pH, the silicate anion is maximally adsorbed to soil surfaces, especially Al and Fe hydrous oxides, causing the concentration of Si in soil solution to decrease (Beckwith and Reeve, 1963; Jones and Handreck, 1963; McKeague and Cline, 1963a,b; Haynes, 2014; Liang et al., 2015). This relationship between Si solubility and pH is one of the major factors accounting for the loss of Si in weathered, acidic soils, and is exacerbated through intensive, long-term cropping and resulting export of Si from the landscape (Berthelsen et al., 2001b; Sommer et al., 2006; Vandevenne et al., 2012; Haynes, 2014).
A further problem in weathered, acid soils is, as noted above, the occurrence of high levels of exchangeable Al3+. Soil acidity and associated Al3+ toxicity have long been recognized as significant, and increasing, constraints in sugarcane production in the South African sugar industry (Sumner, 1970; Meyer et al., 1971; Moberly and Meyer, 1975; Schroeder et al., 1994) and indeed wider agricultural production in South Africa (Barnard and du Preez, 2004). For soils under sugarcane production in South Africa, Miles et al. (2014b) showed convincingly that available Si is strongly limited under conditions of high exchangeable H++Al3+. Their results indicated that high levels (>40 mg kg−1) of soluble Si occurred only where exchangeable H++Al3+ levels were below approximately 0.5 cmolcL−1 and that Al3+ is probably a key factor in constraining plant-available Si in acid soils. Differences in available soil Si and pH across regions are strongly reflected in Si uptake by sugarcane, with leaf Si content consistently higher (10–25 g kg−1 dry matter) in less acid soils (pH > 6.5), but seldom exceeding the industry threshold (Miles and Rhodes, 2013) of 7.5 g kg−1 dry matter in the more weathered acid soils of the coastal and hinterland sugarcane production regions (pH ≤ 5.5) (Van der Laan and Miles, 2010; Miles et al., 2011).
Hence, in these soils there is an urgent need to replenish plant-available soil Si in order to sustain maximum crop production, reduce abiotic stresses (especially water stress and Al toxicity) (Meyer and Keeping, 2001), and as a means to augment plant resistance of more susceptible cultivars to the lepidopteran stalk borer Eldana saccharina Walker (Keeping and Meyer, 2006; Kvedaras and Keeping, 2007; Kvedaras et al., 2007; Keeping et al., 2014). Silicon amendment can also reduce infections of brown rust (Puccinia melanocephala H. and P. Sydow), which occurs in several rust-susceptible cultivars in South Africa (Ramouthar et al., 2016). With this in mind, recent research efforts on provision of Si for sugarcane production in South Africa have focussed on identifying sources with high plant-available Si, and which can simultaneously correct soil pH and reduce Al toxicity (Rhodes et al., 2013; Keeping et al., 2014, 2017). Calcium (magnesium) silicate, supplied in the form of metallurgical slags, has proven most effective in supplying plant-available Si for sugarcane (Gascho, 2001; Berthelsen et al., 2001a; Meyer and Keeping, 2001; Bokhtiar et al., 2012; McCray and Ji, 2013; Crusciol et al., 2014; Tubana et al., 2016). Keeping et al. (2017) found that alkaline Si sources, such as calcium (Ca) silicate slag, cement, and granulated ground blast furnace slag, produced significantly more plant-available Si and greater plant uptake than sources with little or no pH-corrective capacity, such as potassium (K) silicate, bagasse fly ash, and diatomaceous earth. In line with their ability to increase soil pH, slags and cement also significantly reduced Al saturation, and to an extent equivalent to that of dolomitic lime applied at the same rate (Keeping et al., 2017).
However, other Si sources, such as thermophosphates, sometimes referred to as fused magnesium (Mg) phosphate (see Ma and Takahashi, 2002, p. 18), have also shown significant potential in supplying Si (Korndörfer and Gascho, 1999; Gascho, 2001; Kingston, 2011), as has volcanic rock dust (crushed basalt) when applied to sugarcane on highly weathered soils in Mauritius (D'Hotman De Villiers, 1961, 1962). The latter amendment, applied at rates from 110 to 440 tons ha−1, produced cumulative yield responses of between 49 and 90 tons cane ha−1 over five crops. Subsequent studies confirmed that the soluble silicon in the basalt accounted for the favorable yield increases (Halais and Parish, 1964). However, previous work has shown that the total Si content of a source, its provision of plant-available Si (as determined by soil tests), and uptake of Si, especially in rice and sugarcane, did not necessarily concur (Gascho, 2001; Ma and Takahashi, 2002; Kingston, 2011; Haynes et al., 2013; McCray and Ji, 2013; Elephant et al., 2016; Keeping et al., 2017). This observation, together with the novel opportunity to investigate several new Si sources (volcanic rock dust, magnesium silicate and slow-release potassium silicate) for sugarcane in South Africa, prompted further study of the relationship between total source (or product) Si content, available (calcium chloride (CaCl2) extractable) soil Si following application, and subsequent plant Si accumulation, along with the potential of these sources to reduce acid saturation.
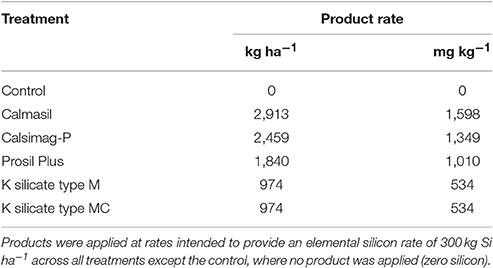
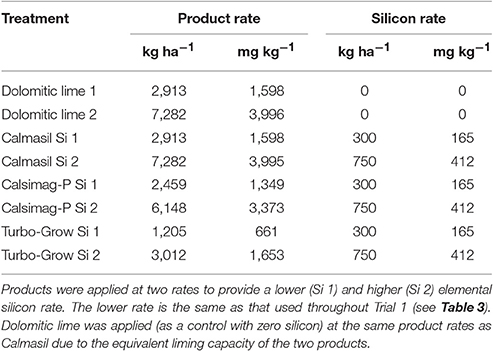
To this end, two trials were conducted using potted sugarcane grown in a low-Si soil, supplied with Si at a single (Trial 1) or two (Trial 2) elemental rates via applications of a Ca silicate slag (Calmasil®, http://www.pbd-lime.co.za/calmasil.htm), thermophosphate (Calsimag-P®), Mg silicate (Prosil Plus®), granular K silicate, volcanic rock (basalt) dust (Turbo-Grow®, www.turbo-grow.co.za), and, in Trial 2, a dolomitic lime control (Table 1). Calmasil is a slag by-product of the stainless industry, while Calsimag-P is manufactured by blending and fusing apatite and serpentine in a furnace, and the resulting amorphous Ca/Mg/P/Si complex is milled to a fine powder and granulated. The Mg silicate (Prosil Plus) is also a serpentine mineral source derived from crushed volcanic rock mined from Colombian batholiths. The K silicate consisted of two types: “Type M” slow release granules that contained compounds of Mg, which imparted a “free running” characteristic to the granules and their slow dissolution in water; “Type MC hardened” slow release granules that contained both Mg and Ca for the same reasons, and had been oven dried at 130–140°C to further reduce their solubility. The study tested the hypothesis that the Si content of these sources (as specified by the supplier) and available soil Si following application, directly affects subsequent plant Si uptake as reflected in leaf Si concentration. More specifically, the thermophosphate and especially volcanic rock dust, Mg silicate and K silicate, with higher total Si contents than the slag (>2-fold higher for the rock dust and 3-fold higher for the K silicate; Table 1), were predicted to produce significantly higher leaf Si concentrations than the latter. Furthermore, the sources were compared with respect to their ability to ameliorate soil acidity and Al toxicity, and to supply Ca and Mg to the soil and plant.

Table 1. Product name (in alphabetical order), supplier (all South Africa based except for Prosil Plus) and silicon content of products used in Trials 1 and 2.
Materials and Methods
The trials were established in a randomized design in a shadehouse with clear polycarbonate roofing and walls of 40% green shade cloth, over the period November 2013 to May 2014 (Trial 1; 27 weeks) and October 2014 to March 2015 (Trial 2; 22 weeks). Treatments were replicated 12 times in each trial, with one pot in each row of pots comprising a single replicate of each treatment. All pots (total of 84 in Trial 1 and 96 in Trial 2) were filled with soil collected from the same site within a sugarcane field (Field 380, Inanda Farm, 29°37′37″S, 30°56′58″E, KwaZulu-Natal (KZN), South Africa). The soil collected for Trial 1 was taken from an area immediately adjacent to that collected for Trial 2. The soil type was an Inceptisol (Soil Survey Staff, 2006), which in the USA is among the soil orders commonly found in humid and sub-humid regions, and known to have limiting plant-available Si (Tubana et al., 2016). In KZN, the soil consists of gray loamy sands, moderately to strongly acid, with a low level of fertility (Beater, 1970), and is typical of the weathered, acidic, low-Si soils of the rainfed regions of the South African sugar industry, as described earlier. The soil for each trial was collected from the top 15 cm layer within an area of ~400 m2, air dried, thoroughly mixed, and passed through a 1 mm sieve. Single samples for analysis were taken from the mixed and sieved bulk soil for each trial before it was placed into pots. The soil properties of each bulk collection are summarized in Table 2. Although the collection site was specifically chosen due to the acid nature of the soil, it was discovered after analysis that the soil for Trial 1 was of a higher pH, higher Ca and clay content, and much lower acid saturation than that for Trial 2 (Table 2). The most likely explanation is that the area from which the soil for Trial 1 had been collected had inadvertently been limed or used as a site for dumping lime by the grower some time—possibly several years —previously. Consequently, control treatments incorporating dolomitic lime were not included in Trial 1 (see below). Fortuitously, these differences between the soil in each trial provided an opportunity to compare Si uptake and effect on soil properties of two sources (Calmasil and Calsimag-P) common to both trials.
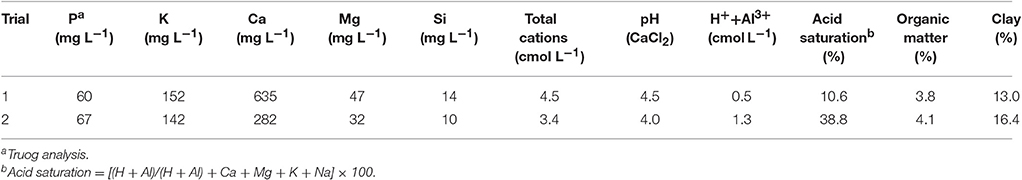
Table 2. Characteristics of soil from Inanda Farm (KwaZulu-Natal, South Africa) collected September 2013 from immediately adjacent areas in the same field for Trials 1 and 2.
Treatments and Fertilizer
Plastic pots (6.41 L) were filled with 6,700 g of soil and application rates for all Si treatments (products), lime and fertilizers were converted from kg ha−1 to g kg−1, based on the average (disturbed) soil density (1,215 g cm−3) and a top-soil depth of 15 cm; i.e., a soil mass of 1,822,500 kg ha−1. For Trial 1, the five Si sources (Calmasil, Calsimag-P, Prosil Plus, K silicate Type M and Type MC) were all applied at product rates (Table 3) intended to provide an elemental Si rate of 300 kg Si ha−1 across all treatments. The control was left untreated, as the soil collected for Trial 1 was of a slightly higher pH and lower acid saturation than that for Trial 2 (Table 2). Consequently, control treatments incorporating dolomitic lime were considered unnecessary for Trial 1.
For Trial 2, Calmasil, Calsimag-P, and Turbo-Grow, plus a dolomitic lime control with zero Si, were each applied at two rates to produce a total of eight treatments (Table 4). The Si sources were applied to provide a lower Si rate (Si 1) of 300 kg Si ha−1 and a higher rate (Si 2) of 750 kg Si ha−1 (Table 4). The rates for the dolomitic (Kulu) lime control were made equivalent to that of Calmasil, given the similar neutralizing capacity of the two materials (Calmasil = 102.8% of pure CaCO3).
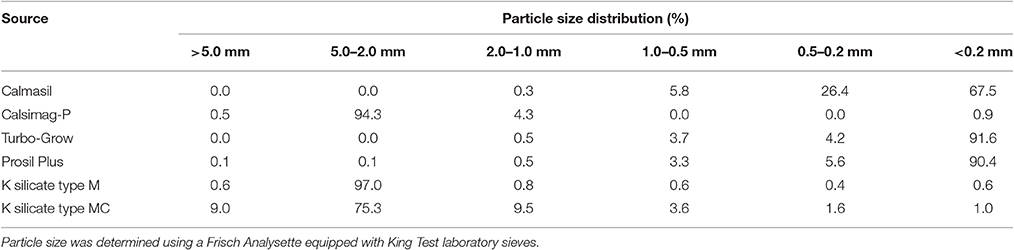
The particle size distributions of the Si sources used in Trials 1 and 2 (Table 5) were determined by shaking 100 mg samples through 8 sieve sizes for 5 min using a Fritsch (Germany) Pulverisette 03502 mechanical vibrator.
Treatments and fertilizer were simultaneously hand-incorporated into the entire volume of soil following initial moistening with 1,000 ml water pot−1. Basal fertilizers were applied as follows: 88 mg nitrogen kg−1 as limestone ammonium nitrate (LAN), 66 mg phosphorous (P) kg−1 as Ca di-orthophosphate, 2 mg copper kg−1 as copper sulfate, and 8 mg zinc kg−1 as zinc sulfate. In both trials, 173 mg K kg−1 as K chloride was supplied to all treatments except the K silicate treatments in Trial 1, where top-up K at 35 mg kg−1 was provided as K chloride. Top dressings of LAN (54 mg N kg−1) and K chloride (71 mg K kg−1, Trial 1) or K sulfate (Trial 2, 69 mg K kg−1 and 28 mg S kg−1) were supplied twice monthly after planting.
Plants
Sugarcane transplants of variety N12 (Anon, 2006) were produced from single-budded setts cut from mature stalks of field-grown cane of the same age and from the same field. Single 1-month old transplants were planted into each pot immediately after the first soil sample (see below). Pots were drip irrigated daily to weekly, depending on moisture demand.
Soil and Leaf Analysis
Soil samples were taken with an augur inserted to the base of all pots in each trial at 7 days after application of treatments and again at 7 days after the trials were harvested at age 27 weeks (Trial 1) or 21 weeks (Trial 2). The purpose of the 7-day interval before the first sample was to allow time for equilibration and any sorption of H4SiO4 to sesquioxides and soil surfaces that may affect measurement of its availability (Babu et al., 2016a). Samples from 3 (Trial 1) or 4 (Trial 2) adjacent replicates of the same treatment were composited to reduce analysis costs. Plant-available soil Si was determined using 0.01 M calcium chloride (CaCl2) extraction, a widely-accepted method that provides a close approximation of the soil environment (Berthelsen and Korndörfer, 2005; Sauer et al., 2006; Haynes et al., 2013; Miles et al., 2014b; Babu et al., 2016b). Soil analyses were performed by the South African Sugarcane Research Institute (SASRI) Fertilizer Advisory Service (FAS), with Si and P concentrations determined using the ammonium molybdate blue colourimetry (Liang et al., 2015) and Truog methods (Miles et al., 2014a), respectively. All soil analyses in the FAS are performed on a volumetric basis. Only results pertinent to the hypotheses tested in this study and for elements provided by the Si sources are reported, i.e., soil concentrations of Si, Ca, Mg, P, as well as pH and acid saturation.
Leaf sampling was conducted once, at harvest. The third fully unfurled or “top visible dewlap” leaf was removed from the major tillers in each pot; leaf blades were stripped from the midrib and the blades dried, ground and analyzed for their elemental nutrient content by the SASRI FAS. Samples from 3 adjacent replicates of the same treatment were composited to produce sufficient material for analysis. Leaf Si content was determined using the dry ashing and molybdenum blue colorimetry method (Liang et al., 2015). As for soil, only results pertinent to the hypotheses tested and for elements provided by the Si sources are reported, i.e., leaf concentrations of Si, Ca, Mg, P.
Data Analysis
All data were tested for univariate normality (Shapiro-Wilk test) and homogeneity of variance (Bartlett's test), and appropriate transformations (log or square root) applied when these conditions were not met, prior to analysis of variance. Where ANOVA yielded significant differences between treatments, planned comparisons of means were performed using the Holm-Sidak multiple-comparisons test. All analyses were carried out using Genstat 14th Edition.
Results
Effects of Si Sources on Soil Properties in Pre-plant and Post-harvest Soil Samples—Trial 1
The soil treatments significantly affected (P < 0.001; ANOVA) soil Si concentrations in the pre-plant and post-harvest soil samples. Of the five Si sources applied, only Calmasil and Calsimag-P significantly increased CaCl2-extractable soil Si above that of the untreated control in pre-plant and post-harvest samples (Figure 1A). Calsimag-P released greater quantities of Si than Calmasil, but the difference was significant only in the post-harvest soil sample (Figure 1A); this was despite application of Calsimag-P at a lower product rate (Table 3) to compensate for its higher total Si content. Concentrations of extractable Si diminished between the pre-plant and post-harvest soil samples by 59% (control), 77% (Calmasil), 44% (Calsimag-P), 52% (Prosil Plus), 69% (K silicate type M), and 67% (K silicate type MC) (Figure 1A).
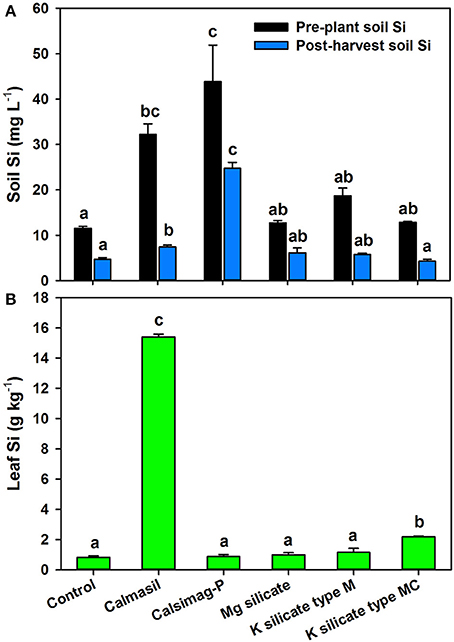
Figure 1. Silicon concentrations in soil (A) and third leaf (B) following application at rates specified in Table 3 of different silicon sources (represented on X-axis) in Trial 1. The control was untreated. In (A), black bars represent pre-plant soil and blue bars represent post-harvest soil; mean values for bars of the same color and with the same letter/s above them are not significantly different. In (B), mean values for bars with the same letter above them are not significantly different (Holm-Sidak test, P < 0.05; ANOVA, P < 0.001 for soil and leaf Si). Error bars are standard errors. Mg silicate = Prosil Plus
The soil treatments significantly affected soil Ca concentrations in the post-harvest sample and Mg and P concentrations in the pre-plant and post-harvest soil samples (Table 6). In the post-harvest treatment, Calmasil supplied significantly more Ca than all other treatments except Calsimag-P. None of the other treatments differed in this respect (Table 6). Calmasil and Calsimag-P both supplied significantly more Mg than the other treatments in pre-plant and post-harvest samples, and Prosil Plus significantly more Mg than the K silicate treatments, which did not differ from the control (Table 6). Calsimag-P increased P significantly compared with the Prosil Plus and K silicate treatments in the pre-plant sample and significantly above all other treatments in the post-harvest sample (Table 6).
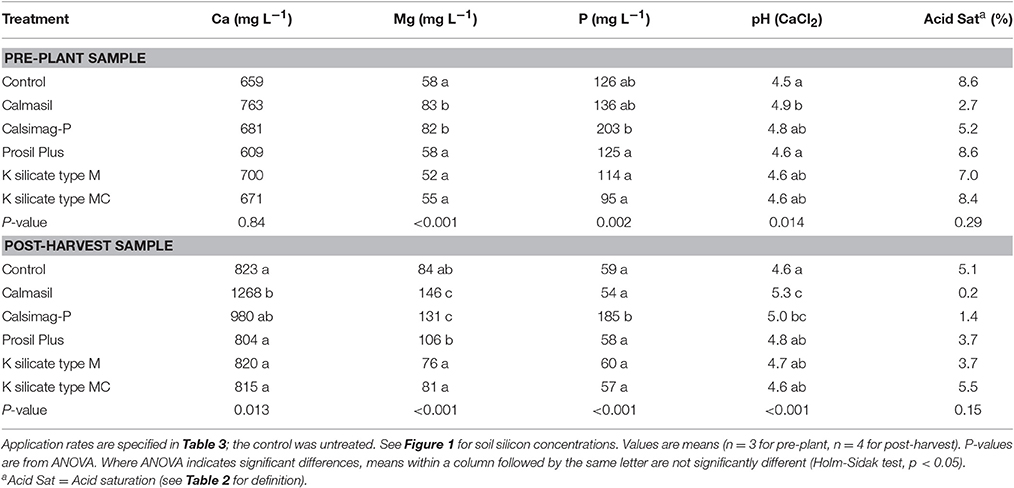
Table 6. Soil concentrations of elements (calcium, magnesium, phosphorus) provided by the silicon sources, soil pH and acid saturation in pre-plant and post-harvest samples from Trial 1.
Although the treatments significantly affected soil pH in pre-plant and post-harvest samples, they had no effect on acid saturation (Table 6). In the pre-plant sample, Calmasil raised pH significantly above the control and Prosil Plus, but not the other treatments (Table 6), while in the post-harvest sample, Calmasil increased pH significantly above that of all other treatments except Calsimag-P; the latter was also significantly higher than the control. Prosil Plus and K silicate did not differ from the control (Table 6).
Effects of Si Sources on Leaf Si, Ca, Mg, and P Concentrations—Trial 1
The soil treatments significantly affected leaf Si concentrations (P < 0.001; ANOVA); however, only Calmasil (by 19-fold) and to a much lesser extent K silicate type MC (by ~3-fold) increased leaf Si above the control (Figure 1B). It is clear from Figure 1 that for Calmasil and Calsimag-P leaf Si did not increase in direct relation to the concentration of extractable soil Si.
The treatments had no significant effects on leaf Ca, Mg, or P concentrations (Table 7).
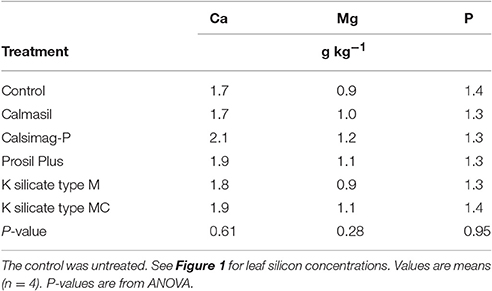
Table 7. Leaf concentrations of elements (calcium, magnesium, phosphorus) provided by the silicon sources applied in Trial 1 at the rates specified in Table 3.
Effects of Si Sources on Soil Properties in Pre-plant and Post-harvest Soil Samples—Trial 2
The soil treatments significantly affected (P < 0.001; ANOVA) soil Si concentrations in the pre-plant and post-harvest soil samples. Of the three Si sources applied, only Calmasil and Calsimag-P significantly elevated CaCl2-extractable soil Si above that of the dolomitic lime control (Figure 2A). Calsimag-P released significantly greater quantities of Si at the higher and lower elemental Si rates than Calmasil at each rate (Figure 2A), even though Calsimag-P was applied at a lower product rate (Table 4) to compensate for its higher total Si content. In the post-harvest sample, Calsimag-P at the lower rate (Si 1) produced significantly higher extractable Si than Calmasil at the higher rate (Si 2) (Figure 2A). Turbo-Grow did not raise extractable soil Si above that of the lime control in both soil samples (Figure 2A). Concentrations of extractable Si decreased between the pre-plant and post-harvest soil samples by 59% (Calmasil Si 1), 57% (Calmasil Si 2), 40% (Calsimag-P Si 1), and 35% (Calsimag-P Si 2), (Figure 2A). Soil Si also decreased over the course of the trial in the Lime 1 (29%), Lime 2 (21%), Turbo-Grow Si 1 (49%), and Turbo-Grow Si 2 (31%) treatments (Figure 2A).
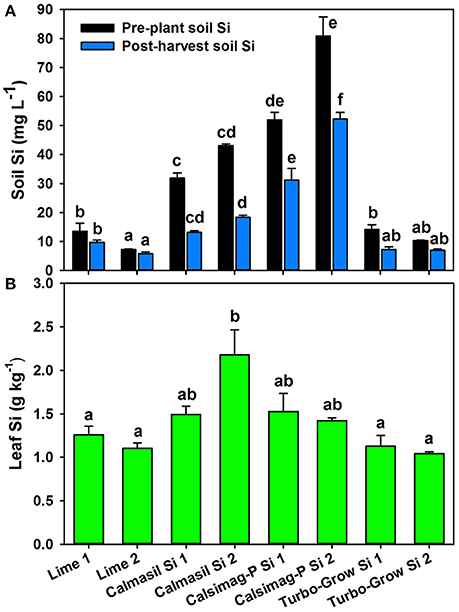
Figure 2. Silicon concentrations in soil (A) and third leaf (B) following application of different silicon sources (represented on X-axis) at lower and higher rates (Si 1 and Si 2; see Table 4) in Trial 2. Product and silicon application rates are specified in Table 4; the lime control was applied at the same product rates as Calmasil. In (A), black bars represent pre-plant soil and blue bars represent post-harvest soil; mean values for bars of the same color and with the same letter/s above them are not significantly different. In (B), mean values for bars with the same letter above them are not significantly different (Holm-Sidak test, P < 0.05; ANOVA, P < 0.001 for soil and leaf Si). Error bars are standard errors.
The soil treatments significantly affected soil Ca and Mg concentrations in the pre-plant and post-harvest soil samples, and P in the pre-plant sample (Table 8). In the pre-plant sample, Calmasil Si 2 supplied significantly more Ca than all other treatments and significantly more Mg than all other treatments except Turbo-Grow Si 2 (Table 8). Turbo-Grow Si 2 provided significantly more Ca than dolomitic lime 2 and Calsimag-P Si 2 (Table 8). Compared with lime (which was applied at the same product rates), Calmasil provided 48 and 65% more Ca, and 47 and 63% more Mg, at Si 1 and Si 2, respectively.
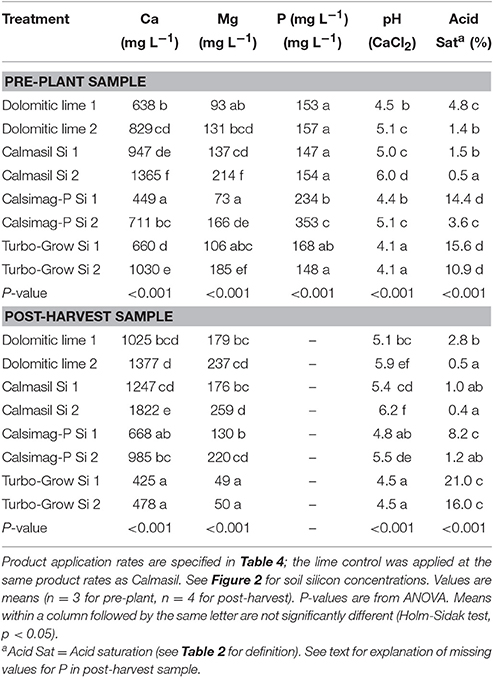
Table 8. Soil concentrations of elements (calcium, magnesium, phosphorus) provided by the silicon sources applied at lower and higher rates (Si 1 and Si 2), soil pH and acid saturation in pre-plant and post-harvest samples from Trial 2.
In the post-harvest sample, Calmasil Si 2 still supplied significantly more Ca than all other treatments; however, its supply of Mg was not significantly greater than that of lime 2 or Calsimag-P Si 2 (Table 8). The supply of Ca and Mg by Turbo-Grow at both treatment rates diminished to concentrations significantly lower than that of all other treatments, other than Ca provided by Calsimag-P Si 1 (Table 8). In the pre-plant sample, Calsimag-P Si 2 raised soil P significantly above all other treatments (and 2.3-fold above that of the control) and Calsimag-P Si 1 above all treatments except Turbo-Grow Si 1 (Table 8). Soil P is not presented for post-harvest samples in Table 8, because the FAS laboratory employs different soil P test methods depending on soil pH (Truog for pH ≤ 5.5 and resin for pH > 5.5; Miles et al., 2014a), thus rendering the data from different treatments non-comparable.
The soil treatments significantly affected soil pH and acid saturation in the pre-plant and post-harvest soil samples (Table 8). Calmasil Si 2 raised soil pH significantly above that of all other treatments in the pre-plant and post-harvest samples, with the exception of lime 2 in the post-harvest sample (Table 8). In the pre-plant sample, the lower rate of Calmasil (i.e., Si 1) was as effective in correcting pH as the higher rate of lime (i.e., lime 2) (Table 8). Although Calsimag-P was applied at a lower product rate than dolomitic lime (Table 4), it nonetheless produced pH levels that were comparable with those of lime at their respective low and high rates (Table 8). Turbo-Grow evidently had little effect in raising pH at the product rates applied, with the pre-plant sample pH elevated by only 0.1 unit and the post-harvest sample by 0.5 unit above that of the untreated bulk soil used in the trial (Tables 2, 5).
Calmasil Si 2 significantly reduced acid saturation percent below that of all other treatments in the pre-plant sample, including lime when compared at the same product rates (Table 8). The reduction of acid saturation (elimination of reactive Al) by Calmasil at the lower application rate (Si 1) was not significantly different from that produced by lime at the higher application rate (Si 2) (Table 8). Calsimag-P Si 1 and Turbo-Grow Si 1 had similar (and the least) effects on acid saturation, although Calsimag-P Si 2 reduced it to a level not significantly different from that of lime at the lower application rate (dolomitic lime 1) (Table 8). The higher application rate of Turbo-Grow (Si 2) had little effect in reducing acid saturation below that of the lower rate (Si 1) of this product (Table 8). All products however, lowered acid saturation substantially compared with that of the untreated bulk soil (38.8%, Table 2).
Similar differences and similarities between treatments were evident in the post-harvest soil sample; however, it was apparent that acid saturation decreased from pre-plant levels in the lime, Calmasil and Calsimag-P treatments, while that in the Turbo-Grow treatments increased above those of the pre-plant values (values between pre-plant and post-harvest samples were not compared statistically) (Table 8). In the post-harvest sample, acid saturation in the Calsimag-P Si 2 treatment did not differ significantly from that in any of the lime or Calmasil treatments (Table 8).
Effects of Si Sources on Leaf Si, Ca, Mg, and P Concentrations—Trial 2
The soil treatments significantly affected leaf Si concentration (P < 0.001; ANOVA); however, Calmasil Si 2 was the only treatment that significantly increased leaf Si above the dolomitic lime control (Figure 2B). Calmasil Si 2 increased leaf Si 2-fold over that of lime applied at the same product rate (lime 2). Calsimag-P - notwithstanding its substantially greater release of soluble Si into the soil, especially at the higher rate (Figure 2A)-had a small and non-significant effect in raising leaf Si above the lime controls (Figure 2B). Turbo-Grow had no discernible effect on leaf Si content (Figure 2B). As for Trial 1, it is evident for Trial 2 (Figure 2) that leaf Si in the Calmasil and Calsimag-P treatments did not increase in direct relation to the concentration of extractable soil Si.
Leaf concentrations of Ca and Mg were significantly affected by the soil treatments (Table 9). Calmasil Si 1 and Si 2 produced significantly higher leaf Ca than Turbo-Grow Si 2, and Calmasil Si 2 produced significantly higher leaf Ca than lime 2 and Calsimag-P Si 1 and Si 2 (Table 9). For Mg, the only significant difference occurred between Calmasil Si 2 and Turbo-Grow Si 2 (Table 9). The treatments had no significant effect on leaf P concentration (Table 9).
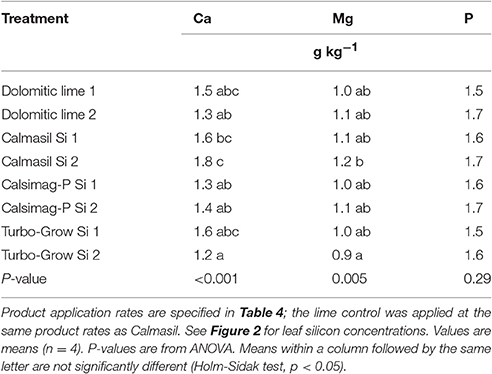
Table 9. Leaf concentrations of elements (calcium, magnesium, phosphorus) provided by the silicon sources applied at lower and higher rates (Si 1 and Si 2) in Trial 2.
Discussion
A striking outcome of this study was that although Calmasil had the lowest total Si content (10.3% Si; Table 1), a larger proportion of the Si it provided was taken up by the plant, as it consistently produced the highest leaf Si concentrations in sugarcane. By contrast, sources with high total Si content (K silicate, Prosil Plus, Turbo-Grow; 16.3–30.8% Si; Table 1) produced leaf Si concentrations that were substantially (between 2.2- and 15.4-fold) lower than Calmasil and not significantly different from the lime or untreated controls, other than K silicate type MC (Figures 1B, 2B). The above high-Si-content sources also provided no detectable increases in CaCl2-extractable soil Si compared with the controls in both pre-plant and post-harvest samples (Figures 1A, 2A). The low Si provision (even soon after their application) and plant uptake from these sources indicates that they provided little in the way of plant-available Si, despite their high total Si content. Also striking, was the much lower than expected uptake of Si from the Calsimag-P treatments, especially in Trial 1 (Figures 1B, 2B), in direct contrast with this product's substantial and extended provision of extractable soil Si in pre-plant and post-harvest soil samples (Figures 1A, 2A). Although all Si sources were applied at product rates intended to achieve equivalent Si rates of 300 or 750 kg ha−1 (Tables 3, 4), the results of the soil analyses clearly show that the total Si content of sources, as stipulated by the manufacturers, cannot be used as a basis for predicting a product's performance in terms of the release of extractable soil Si following its application. These conclusions are borne out by the results of other studies. For example, Haynes et al. (2013) found that a negligible quantity of the very high total Si content (29.1%) of fly ash was in extractable form (using several extractants) compared with steel slag and processing mud, which had the lowest total Si contents but relatively high extractable Si. Korndörfer and Gascho (1999) reported high Si content in steel slag (29%) and Mg silicate (39%), but low availability and uptake (by rice) from these sources compared with wollastonite and thermophosphate. Notably, Elephant et al. (2016) showed from soil incubation studies that Calmasil produced higher concentrations of CaCl2-extractable soil Si than Langfos® (crushed sedimentary phosphate rock) and quarry dust (crushed dwyka tillite), with 14.2 and 24.4% total Si, respectively.
A further conundrum with respect to the Si supplying capacity of the sources studied here is the role of particle size. Studies have generally shown that Si availability increases as particle size decreases and surface area of dissolution increases (Medina-Gonzales et al., 1988; Datnoff et al., 1992; Gascho, 2001; Ma and Takahashi, 2002; Haynes et al., 2013). However, in the present study, the materials with the finest particle size (Turbo-Grow and Prosil Plus; Table 5) did not have high Si supplying capacity, while Calsimag-P, a granular product (Table 5), released substantial quantities of Si. The form of Si in the product and its solubility are clearly critical, as emphasized by Kingston (2011), Haynes et al. (2013), Babu et al. (2016b) and Tubana et al. (2016). For example, Babu et al. (2016b) noted that slag is a recently formed polycrystalline material and supplies Si at a relatively fast rate and high concentration, while wollastonite, a geologically formed pure crystalline mineral, releases Si at slower rates and lower concentrations.
The potential of thermophosphates, such as Calsimag-P, to be highly efficacious sources of plant-available Si has been demonstrated in other studies (Gascho and Korndörfer, 1999; Pereira et al., 2004; Kingston, 2011). In line with this, Gascho (2001) pointed out that although the total Si content of thermophosphate may be low compared to certain silicate slags (e.g., electric furnace slag, 18.2% Si), the proportion of soluble Si is high. While Calsimag-P did not significantly raise leaf Si content above controls in the present study, this source has previously produced leaf Si values comparable with those of Calmasil using the same soil and sugarcane variety (Keeping et al., 2017); this indicates that uptake from this source can occur, but may be strongly dependent on specific environmental or soil conditions. Such results support Snyder's (2001) point that laboratory analyses of Si-containing materials can only be used as initial screening procedures to identify promising Si sources, and that glasshouse and field studies of plant uptake are ultimately required to provide certainty about the Si supplying capacity of sources.
In the present study, Calmasil significantly increased soil pH in both trials and above that of the equivalent lime treatments in Trial 2 (Table 8); it also reduced acid saturation in pre-plant and post-harvest soil samples, with significantly greater efficiency than dolomitic lime in Trial 2 (Table 8). Notably, Calmasil Si 2 in Trial 2 was the only Si treatment that raised soil pH(CaCl2) well above 5.0 in the pre-plant sample (Table 8), at which point Al would precipitate out (Fageria et al., 1988) and its reaction with Si would be reduced. This emphasizes the value of Ca silicate slags in ameliorating soil acidity and Al toxicity, while also supplying Si, Ca, and Mg (Korndörfer and Gascho, 1999; Meyer and Keeping, 2001; Haynes et al., 2013; Castro et al., 2016; Ning et al., 2016). In Trial 1, pH was not increased above 5.0 in the pre-plant sample or above 5.3 in the post-harvest sample (Table 6). Here, Calmasil was applied at only one rate (2,913 kg ha−1, equal to the lower Calmasil Si 1 rate in Trial 2), yet the leaf Si concentration of the Calmasil treatment was substantially higher than that in Trial 2 (10-fold more than Si 1 and 7-fold more than Si 2; Figures 1B, 2B). This was not likely due to differences in available soil Si concentration, which was in fact higher in the Calmasil Si 2 treatment in Trial 2 than in the Calmasil treatment in Trial 1 (Figures 1A, 2A). Possibly the already low acid saturation and H++Al3+ levels in the soil used in Trial 1 (Table 2) contributed to greater Si uptake and higher leaf Si levels in this trial, given that the soil had probably been limed by the grower many months or even years prior to its use. In contrast to Calmasil, Calsimag-P did not raise soil pH or reduce acid saturation in Trial 2 (Table 8); this may have lowered its effectiveness in elevating leaf Si content as a result of rapid complexation of released Si with soluble, reactive Al3+ (Farmer et al., 1979; Doucet et al., 2001; Schneider et al., 2004; Exley, 2012).
Where Si sources are high in total Si but nonetheless provide little or no plant-available Si to the soil, as in the case of the Mg silicate (Prosil Plus), K silicate (Figure 1A), crushed volcanic rock (Turbo-Grow, Figure 2A), diatomaceous earth or fly ash (Kingston, 2011; Haynes et al., 2013; Keeping et al., 2017) and quarry dust (Elephant et al., 2016), the effects of Al3+ in reducing their provision of Si to plants would be largely immaterial. However, the situation for slags may be different. Babu et al. (2016a) pointed out that trace amounts of Al3+ reduce the equilibrium solubility of Si due to the co-deposition of these elements as hyrdroxyaluminosilicate (HAS) within the soil environment (see Cocker et al., 1998, for a review of this process). They also argued that the presence of Al3+ ions on slag particles can reduce the rate of dissolution of silica and act as catalysts in accelerating the process of polymerization of monomeric H4SiO4 to colloidal silica, which cannot be taken up by plants. Adsorption of the silicate anion (H3) to hydroxides of Al and Fe (sesquioxides) increases at higher pH (especially above pH 9) and is of critical importance in constraining the concentration of Si in soil solution (Beckwith and Reeve, 1963; Jones and Handreck, 1963; McKeague and Cline, 1963b; Kato and Owa, 1996; Tavakkoli et al., 2011; Haynes, 2014). This is especially so when higher rates of calcium silicate slag are applied, wherein the higher pH and concentration of solubilizing Si produced by the slag promotes increasing adsorption of H3 to sesquioxide surfaces (Haynes et al., 2013; Babu et al., 2016a). While Calmasil contains on average 1.07% Al, the greatest source of this element in the present study, by a very large margin, would be the acid soil used (Table 2). Notwithstanding the substantial reductions in acid saturation in the Calmasil treatments in both trials (Tables 6, 8), small quantities of native soluble Al3+ ions may have reduced equilibrium Si solubility or increased its polymerization in the manner described by Babu et al. (2016a). Under field conditions, where wetting and drying cycles would serve to concentrate Si solubilized from slag, polymerization may be especially important in this regard (Keeping et al., 2013).
The mechanisms discussed above do not, however, satisfactorily account for the low concentrations of Si in leaf tissue from the Calsimag-P treatments, where abundant levels of soluble Si were present in the treated soil (Figures 1A, 2A). This suggests that the Si was either not taken up by the plants or, if taken up, not translocated to the shoot. The substantial reductions in soil Si from pre-plant to post-harvest samples in both trials indicate that appreciable plant uptake occurred and/or that some of the available Si was converted during the course of the trials to forms not readily extractable with 0.01 M CaCl2. Under conditions of low pH, high acid saturation (and therefore presence of soluble Al3+), and high Si concentration in the rhizosphere, it is likely that the plant will simultaneously take up Al3+ and H4SiO4 into the root cells, where they may react and co-precipitate (Hodson and Evans, 1995; Cocker et al., 1998; Hodson and Sangster, 1999). As stated by Hodson (2011), co-deposition in planta of Al with Si in solid phytoliths is a relatively widespread phenomenon in higher plants, and in roots Al is often co-deposited with Si in epidermal and cortical cells. An in planta mechanism that immobilizes Si and inhibits its translocation from the roots to the shoot, may explain the abundant soil Si levels but low (or relatively low) leaf Si concentrations in the Calsimag-P treatments in this study. On the other hand, the robust liming effect of Calmasil may have been sufficient to reduce solubility and plant uptake of Al3+ to the extent that its co-deposition with Si within the plant had a much-reduced effect on translocation of Si to the shoot. As mentioned, the already low acid saturation of the soil used in Trial 1 (Table 6) may have accentuated such an effect and promoted the high leaf Si accumulation in the Calmasil treatment (Figure 1B). Detailed studies of Al and Si co-deposition in roots of sugarcane, such as those performed by Cocker et al. (1997) in wheat and Prabagar et al. (2011) in Norway spruce, may reveal in planta interactions between these elements, their possible effects on Si translocation, and the extent to which low shoot Si accumulation is a reflection of reactions within the soil (which affect uptake) or reactions within the plant (which affect translocation).
Attention by sugarcane growers to addressing problems of Si and other nutrient deficiencies is an important step in avoiding plant stress and reducing infestation by sugarcane borer (White and White, 2013; Keeping et al., 2014; Nikpay et al., 2015). The present study emphasizes a further essential step, which is to improve soil health and root growth by reducing soil acidity and Al toxicity through liming and/or calcium silicate provision. With increasing acidification of soils due to long periods of monocropping and intensive use of nitrogenous fertilizers, not only in South Africa but across many tropical and sub-tropical crop-growing regions (Meyer et al., 1998; Barnard and du Preez, 2004; Ma, 2004; Fageria and Baligar, 2008; Van der Laan and Miles, 2010; Marafon and Endres, 2013; Meena et al., 2014), the use of calcium silicate slags presents a valuable substitute for conventional dolomitic limes. As demonstrated in the present study and previous studies, slags also provide Ca and Mg (Table 8); both of these nutrients, along with P, were nutritionally adequate in Trials 1 and 2 (Tables 7, 9) (Miles and Rhodes, 2013). Moreover, slags have the additional advantages of supplying Si, being more reactive than lime (and in the current study more effective on a mass for mass basis), and correcting acidity and eliminating Al3+ to a greater soil depth (Korndörfer and Gascho, 1999; Pereira and Cabral, 2005; Bokhtiar et al., 2012; Marafon and Endres, 2013; Haynes, 2014; Castro and Crusciol, 2015; Castro et al., 2016). The results presented here indicate that Si sources (such as thermophosphate) that provide ample soluble Si but have limited liming capacity, may not be effective sources in acid soils due to reaction of their solubilized Si with Al3+ within the soil and perhaps to a larger extent within the plant. This underscores the advantages of alkaline Si sources that can simultaneously eliminate Al3+ in the rhizosphere and reduce its uptake. Future field studies should focus on means to further eliminate Al3+ or prevent its reaction with Si, by combining treatments of slag with gypsum and sources of organic matter, such as crop residues, manure or bagasse (the sugarcane stalk residue remaining after juice extraction). Reduction of sub-soil acidity, and the retention of soil moisture and improved rainwater infiltration associated with higher soil organic matter, are crucial practices in ensuring root health (Thorburn et al., 1999; Bell et al., 2001; Pankhurst et al., 2005; Sumner, 2011, 2012), and may be equally important in augmenting Si uptake from both native and applied sources.
Finally, there has been increasing focus on the importance of recycling of crop residues, which may contain large quantities of amorphous Si in the form of phytoliths (i.e., phytogenic Si), back into soils in an effort to compensate for the large-scale and ongoing removal of Si from agricultural landscapes when crops are harvested (Struyf et al., 2010; Clymans et al., 2011; Guntzer et al., 2012; Keller et al., 2012; Vandevenne et al., 2012; Cornelis and Delvaux, 2016). In sugarcane, the dry leaf matter is an important potential source of Si, as the concentration of Si in dry leaf may reach 3% DM (Van Dillewijn, 1952), following the deposition of amorphous Si to form phytoliths in green leaves (Kaufman et al., 1979; Tripathi et al., 2011). A rainfed crop that yields a total biomass of 80 t ha−1 at harvest could produce 16 t ha−1 dry leaf matter, as dry leaf matter may account for 20% DM of total biomass (Purchase et al., 2008), potentially providing 480 kg Si ha−1, assuming the above 3% Si composition. As in the case for rice (Ma and Takahashi, 1991; Savant et al., 1999; Haynes, 2014; Klotzbücher et al., 2016), this alone could provide much of the Si that would otherwise need to be provided in the form of silicate amendments, and highlights the substantial yearly removal of Si from sugarcane fields effected in the process of crop residue removal and burning. Silicon provision through retention of crop residues should therefore be viewed as a prominent but generally overlooked benefit of crop residue retention along with the many other benefits of this practice (Thorburn et al., 1999; Bell et al., 2001; van Antwerpen et al., 2001; Pankhurst et al., 2005) in sugarcane production.
Conclusions
Vendors of new Si-bearing materials frequently lay considerable emphasis on the high Si content of their products, without clear evidence as to how much of the total Si is plant-available. Yet this and previous studies have shown that the total Si content of sources is not a reliable indicator of how effective they may be in, firstly, releasing sufficient quantities of plant-available Si into soil solution and, secondly, in facilitating its uptake by the plant. In weathered acid soils dominated by Al and Fe sesquioxides, and which occur across much of the tropical and sub-tropical regions where Si-accumulating crops such as rice and sugarcane are grown, calcium silicate appears to be the most effective source in both respects, as it dissolves readily in an acid soil environment to release silicic acid, and is also an efficient liming agent capable of significantly reducing acid saturation and Al toxicity. The latter factor may be critical in constraining reaction of available Si with Al3+, either in the soil or in the plant roots, and thereby maximizing Si uptake and translocation to the shoot. Other sources with high Si content either provide very small quantities of soluble Si or, if they do provide adequate Si, may have limited or no liming capacity; consequently they are unable to counteract the direct toxic effects on Al3+ on roots or its reaction with silicic acid. As a result of these properties and their provision of ample Ca and Mg, calcium silicate slag appears still to offer the most effective and affordable Si source for sugarcane growers, at least in the acid, sandy soils of the dryland production regions of the South African sugar industry. However, attention should also be directed toward practices that promote recycling of phytogenic Si back into soils, principally through retention of crop residues and preservation of soil organic matter, which in themselves may also promote uptake of Si from silicate slags.
Author Contributions
MK conceived and conducted the research, performed the statistical analyses, and wrote the manuscript.
Funding
Funding and infrastructural support for the research was provided by the South African Sugarcane Research Institute (SASRI), and additional financing of research expenses by the Incentive Funding Programme for Rated Researchers of the National Research Foundation (NRF), South Africa (Grant No. IFR2011040700004).
Conflict of Interest Statement
The author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The following representatives and their companies (company and their respective product/s given in parentheses) are gratefully acknowledged for provision, free of charge, of the silicon sources used in this study: Andrew Perkins (Turbo-Grow (Pty) Ltd., volcanic rock dust), Christian Menay (AgroMatChem Ltd., Prosil Plus WP), Flippie van Dyk (Farmsecure Agriscience, Calsimag-P®), Mike Thom (Tangmere Resources (Pty) Ltd., potassium silicate types M and MC), Siegfried Schubert (PDB Lime (Pty) Ltd., Calmasil®). I thank Denis Sibisi and the SASRI Technical Team for invaluable technical assistance. Neil Miles is thanked for critical comments on an earlier version of the manuscript and, together with Stuart Rutherford, for fruitful discussions and ideas related to this study.
References
Adrees, M., Ali, S., Rizwan, M., Zia-ur-Rehman, M., Ibrahim, M., Abbas, F., et al. (2015). Mechanisms of silicon-mediated alleviation of heavy metal toxicity in plants: a review. Ecotox. Environ. Safe 119, 186–197. doi: 10.1016/j.ecoenv.2015.05.011
Alvarez, J., and Datnoff, L. E. (2001). “The economics of silicon for integrated management and sustainable production of rice and sugarcane,” in Silicon in Agriculture, eds L. E. Datnoff, G. H. Snyder, and G. H. Korndörfer (Amsterdam: Elsevier), 221–239.
Anon (2006). Information Sheet 13.3 Variety N12. Mount Edgecombe: South African Sugarcane Research Institute.
Babu, T., Tubana, B., Datnoff, L., Yzenas, J., and Maiti, K. (2016a). Release and sorption pattern of monosilicic acid from silicon fertilizers in different soils of Louisiana: a laboratory incubation study. Commun. Soil Sci. Plan. 47, 1559–1577. doi: 10.1080/00103624.2016.1194995
Babu, T., Tubana, B., Paye, W., Kanke, Y., and Datnoff, L. (2016b). Establishing soil silicon test procedure and critical silicon level for rice in Louisiana soils. Commun. Soil Sci. Plan. 47, 1578–1597. doi: 10.1080/00103624.2016.1194996
Barnard, R. O., and du Preez, C. C. (2004). Soil fertility in South Africa: the last twenty five years. S. Afr. J. Plant Soil 21, 301–315. doi: 10.1080/02571862.2004.10635066
Beckwith, R. S., and Reeve, R. (1963). Studies on soluble silica in soils. I. The sorption of silicic acid by soils and minerals. Aust. J. Soil Res. 1, 157–168. doi: 10.1071/SR9630157
Bell, M. J., Halpin, N. V., Orange, D. N., and Haines, M. (2001). Effect of compaction and trash blanketing on rainfall infiltration in sugarcane soils. Proc. Aust. Soc. Sugar Cane Technol. 23, 161–167.
Berthelsen, S., Hurney, A., Kingston, G., Rudd, A., Garside, A. L., and Noble, A. D. (2001a). Plant cane responses to silicated products in the Mossman, Innisfail and Bundaberg districts. Proc. Aust. Soc. Sugar Cane Technol. 23, 297–303.
Berthelsen, S., and Korndörfer, G. H. (2005). “Methods for Si analysis in plant, soil and fertilizers,” in Proceedings of the Third Silicon in Agriculture Conference (Uberlândia: Federal University of Uberlândia), 86–93.
Berthelsen, S., Noble, A. D., and Garside, A. L. (2001b). “Silicon research down under: past, present, and future,” in Silicon in Agriculture, eds L. E. Datnoff, G. H. Snyder, and G. H. Korndörfer (Amsterdam: Elsevier), 241–255.
Bokhtiar, S. M., Huang, H., and Li, Y. (2012). Response of sugarcane to calcium silicate on yield, gas exchange characteristics, leaf nutrient concentrations, and soil properties in two different soils. Commun. Soil Sci. Plan. 43, 1363–1381. doi: 10.1080/00103624.2012.670516
Castro, G. S. A., and Crusciol, C. A. C. (2015). Effects of surface application of dolomitic limestone and calcium-magnesium silicate on soybean and maize in rotation with green manure in a tropical region. Bragantia 74, 311–321. doi: 10.1590/1678-4499.0346
Castro, G. S. A., Crusciol, C. A. C., Costa, C. H. M., Ferrari Neto, J., and Mancuso, M. A. C. (2016). Surface application of limestone and calcium-magnesium silicate in a tropical no-tillage system. J. Soil Sci. Plant Nutr. 16, 362–379. doi: 10.4067/S0718-95162016005000034
Cheong, Y. W. Y., and Halais, P. (1969). “Nutrition and soils: 1. The effect of calcium silicate slag and coral sand on sugar cane yields in field trials” in Mauritius Sugar Industry Research Institute Annual Report for 1968, (Port Louis: Claude Marrier d'Unienville, The Mauritius Printing Cy. Ltd), 71–73.
Clymans, W., Struyf, E., Govers, G., Vandevenne, F., and Conley, D. J. (2011). Anthropogenic impact on amorphous silica pools in temperate soils. Biogeosciences 8, 2281–2293. doi: 10.5194/bg-8-2281-2011
Cocker, K. M., Evans, D. E., and Hodson, M. J. (1998). The amelioration of aluminium toxicity by silicon in higher plants: solution chemistry or an in planta mechanism? Physiol. Plantarum 104, 608–614. doi: 10.1034/j.1399-3054.1998.1040413.x
Cocker, K. M., Hodson, M. J., Evans, D. E., and Sangster, A. G. (1997). Interaction between silicon and aluminum in Triticum aestivum L. (cv. Celtic). Israel J. Plant Sci. 45, 285–292. doi: 10.1080/07929978.1997.10676691
Cooke, J., and Leishman, M. R. (2011). Is plant ecology more siliceous than we realise? Trends Plant Sci. 16, 61–68. doi: 10.1016/j.tplants.2010.10.003
Cooke, J., and Leishman, M. R. (2012). Tradeoffs between foliar silicon and carbon-based defences: evidence from vegetation communities of contrasting soil types. Oikos 121, 2052–2060. doi: 10.1111/j.1600-0706.2012.20057.x
Cornelis, J. T., and Delvaux, B. (2016). Soil processes drive the biological silicon feedback loop. Funct. Ecol. 30, 1298–1310. doi: 10.1111/1365-2435.12704
Crusciol, C. A. C., Foltran, R., Rossato, O. B., McCray, J. M., and Rossetto, R. (2014). Effects of surface application of calcium-magnesium silicate and gypsum on soil fertility and sugarcane yield. Rev. Bras. Ciênc. Solo 38, 1843–1854. doi: 10.1590/S0100-06832014000600019
Datnoff, L. E., Rodrigues, F. A., Seebold, K. W., Elmer, W. H., and Huber, D. M. (2007). “Silicon and plant diseas,” in Mineral Nutrition and Plant Disease, ed L. E. Datnoff (St. Paul, MN: The American Phytopathological Society), 233–246.
Datnoff, L. E., Snyder, G. H., and Deren, C. W. (1992). Influence of silicon fertilizer grades on blast and brown spot development and on rice yields. Plant Dis. 76, 1011–1013. doi: 10.1094/PD-76-1011
D'Hotman De Villiers, O. (1961). Soil rejuvenation with crushed basalt in Mauritius. Part I - Consistent results of worldwide interest. Int. Sugar J. December, 363–364.
D'Hotman De Villiers, O. (1962). Soil rejuvenation with crushed basalt in Mauritius Part I - The fertility of basalt and its nutritional effects. Int. Sugar J. January, 3–5.
Doucet, F. J., Schneider, C., Bones, S. J., Kretchmer, A., Moss, I., Tekely, P., et al. (2001). The formation of hydroxyaluminosilicates of geochemical and biological significance. Geochim. Cosmochim. Ac. 65, 2461–2467. doi: 10.1016/S0016-7037(01)00571-3
Elawad, S. H., Gascho, G. J., and Street, J. J. (1982). Response of sugarcane to silicate source and rate. I. Growth and yield. Agron. J. 74, 481–484.
Elephant, D., Miles, N., and Dlungele, P. (2016). Evaluation of the impact of a range of soil ameliorants on soil chemical and biological properties. Proc. S. Afr. Sug. Technol. Ass. 89, 170–180.
Epstein, E. (1994). The anomaly of silicon in plant biology. Proc. Nat. Acad. Sci. U.S.A. 91, 11–17. doi: 10.1073/pnas.91.1.11
Epstein, E. (2001). “Silicon in plants: facts vs. concepts,” in Silicon in Agriculture, eds L. E. Datnoff, G. H. Snyder, and G. H. Korndörfer (Amsterdam: Elsevier), 1–15.
Epstein, E. (2009). Silicon: its manifold roles in plants. Ann. Appl. Biol. 155, 155–160. doi: 10.1111/j.1744-7348.2009.00343.x
Exley, C. (2012). Reflections upon and recent insight into the mechanism of formation of hydroxyaluminosilicates and the therapeutic potential of silicic acid. Coordin. Chem. Rev. 256, 88. doi: 10.1016/j.ccr.2011.06.022
Fageria, N. K., and Baligar, V. C. (2008). Ameliorating soil acidity of tropical oxisols by liming for sustainable crop production. Adv. Agron. 99, 345–399. doi: 10.1016/S0065-2113(08)00407-0
Fageria, N. K., Baligar, V. C., and Wright, R. J. (1988). Aluminium toxicity in crop plants. J. Plant Nutr. 11, 303–319. doi: 10.1080/01904168809363804
Farmer, V. C., Fraser, A. R., and Tait, J. M. (1979). Characterization of the chemical structures of natural and synthetic aluminosilicate gels and sols by infrared spectroscopy. Geochim. Cosmochim. Ac. 43, 1417–1420. doi: 10.1016/0016-7037(79)90135-2
Gascho, G. J. (2001). “Silicon sources for agriculture,” in Silicon in Agriculture, eds L. E. Datnoff, G. H. Snyder, and G. H. Korndörfer (Amsterdam: Elsevier), 197–207.
Gascho, G. J., and Korndörfer, G. H. (1999). Availability of silicon from several sources as determined by chemical and biological methods. Soil Crop Sci. Soc. Fl 58, 109–113.
Guntzer, F., Keller, C., Poulton, P. R., McGrath, S. P., and Meunier, J. D. (2012). Long-term removal of wheat straw decreases soil amorphous silica at Broadbalk, Rothamsted. Plant Soil 352, 173–184. doi: 10.1007/s11104-011-0987-4
Halais, P., and Parish, D. H. (1964). “Silica and manganese contents of cane leaf sheaths in relation to soil and nutrition,” in Reports of the Mauritius Sugar Industry Research Institute for 1963 (Réduit), 74–76.
Haynes, R. J. (2014). A contemporary overview of silicon availability in agricultural soils. J. Plant Nutr. Soil Sci. 177, 831–844. doi: 10.1002/jpln.201400202
Haynes, R. J., Belyaeva, O. N., and Kingston, G. (2013). Evaluation of industrial wastes as sources of fertilizer silicon using chemical extractions and plant uptake. J. Plant Nutr. Soil Sci. 176, 238–248. doi: 10.1002/jpln.201200372
Hodson, M. J. (2011). “Recent progress on aluminium/silicon interactions in higher plants,” in Proceedings of the Fifth International Silicon in Agriculture Conference (Beijing: Chinese Academy of Agricultural Sciences), 65–67
Hodson, M. J., and Evans, D. E. (1995). Aluminium/silicon interactions in higher plants. J. Exp. Bot. 46, 161–171. doi: 10.1093/jxb/46.2.161
Hodson, M. J., and Sangster, A. G. (1999). Aluminium/silicon interactions in conifers. J. Inorg. Biochem. 76, 89–98. doi: 10.1016/S0162-0134(99)00119-1
Jones, L. H. P., and Handreck, K. A. (1963). Effects of iron and aluminium oxides on silica in solution in soils. Nature 198, 852–853. doi: 10.1038/198852a0
Kato, N., and Owa, N. (1996). The factors affecting Si concentration in the soil solution: effects of soil solution pH, Ca concentration, CO2 gas and slag application. Jap. J. Soil Sci. Plant Nutr. 67, 655–661.
Kaufman, P. B., Takeoka, Y., Carlson, T. J., Bigelow, W. C., Jones, J. D., Moore, P. H., et al. (1979). Studies on silica deposition in sugarcane (Saccharum spp.) using scanning electron microscopy, energy-dispersive X-ray analysis, neutron activiation analysis, and light microscopy. Phytomorphology 29, 185–193.
Keeping, M. G., and Meyer, J. H. (2006). Silicon-mediated resistance of sugarcane to Eldana saccharina Walker (Lepidoptera: Pyralidae): effects of silicon source and cultivar. J. Appl. Entomol. 130, 410–420. doi: 10.1111/j.1439-0418.2006.01081.x
Keeping, M. G., Meyer, J. H., and Sewpersad, C. (2013). Soil silicon amendments increase resistance of sugarcane to stalk borer Eldana saccharina Walker (Lepidoptera: Pyralidae) under field conditions. Plant Soil 363, 297–318. doi: 10.1007/s11104-012-1325-1
Keeping, M. G., Miles, N., and Rutherford, R. S. (2017). Liming an acid soil treated with diverse silicon sources: effects on silicon uptake by sugarcane (Saccharum spp. hybrids). J. Plant Nutr. doi: 10.1080/01904167.2016.1267751. [Epub ahead of print].
Keeping, M. G., Miles, N., and Sewpersad, C. (2014). Silicon reduces impact of plant nitrogen in promoting stalk borer (Eldana saccharina) but not sugarcane thrips (Fulmekiola serrata) infestations in sugarcane. Front. Plant Sci. 5:289. doi: 10.3389/fpls.2014.00289
Keller, C., Guntzer, F., Barboni, D., Labreuche, J., and Meunier, J. D. (2012). Impact of agriculture on the Si biogeochemical cycle: input from phytolith studies. Comptes Rendus Geoscience 344, 739–746. doi: 10.1016/j.crte.2012.10.004
Kingston, G. (2011). “Cationic resin and plant bioassays to assess suitability of silicated fertilisers,” in Proceedings of the Fifth International Silicon in Agriculture Conference (Beijing: Chinese Academy of Agricultural Sciences), 82–88.
Klotzbücher, T., Marxen, A., Jahn, R., and Vetterlein, D. (2016). Silicon cycle in rice paddy fields: insights provided by relations between silicon forms in topsoils and plant silicon uptake. Nut. Cycl. Agroecosys. 105, 157–168. doi: 10.1007/s10705-016-9782-1
Korndörfer, G. H., and Gascho, G. J. (1999). “Avaliação de fontes de silício para o arroz,” in Congresso Nacional de Arroz Irrigado (Pelotas: Embrapa), 313–316.
Kvedaras, O. L., and Keeping, M. G. (2007). Silicon impedes stalk penetration by the borer Eldana saccharina in sugarcane. Entomol. Exp. Appl. 125, 103–110. doi: 10.1111/j.1570-7458.2007.00604.x
Kvedaras, O. L., Keeping, M. G., Goebel, F. R., and Byrne, M. J. (2007). Water stress augments silicon-mediated resistance of susceptible sugarcane cultivars to the stalk borer Eldana saccharina (Lepidoptera: Pyralidae). Bull. Ent. Res. 97, 175–183. doi: 10.1017/S0007485307004853
Liang, Y. C., Sun, W. C., Zhu, Y. G., and Christie, P. (2007). Mechanisms of silicon-mediated alleviation of abiotic stresses in higher plants: a review. Environ. Pollut. 147, 422–428. doi: 10.1016/j.envpol.2006.06.008
Liang, Y., Nikolic, M., Bélanger, R., Gong, H., and Song, A. (2015). Silicon in Agriculture: From Theory to Practice. Dordrecht: Springer.
Ma, J. F. (2004). Role of silicon in enhancing the resistance of plants to biotic and abiotic stresses. Soil Sci. Plant Nutr. 50, 11–18. doi: 10.1080/00380768.2004.10408447
Ma, J. F., and Takahashi, E. (1991). Availability of rice straw [as a soil amendment] Si to rice plants. Soil Sci. Plant Nutr. 37, 111–116. doi: 10.1080/00380768.1991.10415016
Ma, J. F., and Takahashi, E. (2002). Soil, Fertilizer and Plant Silicon Research in Japan. Amsterdam: Elsevier Science B.V.
Marafon, A. C., and Endres, L. (2013). Silicon: fertilization and nutrition in higher plants. Amaz. J. Agric. Environ. Sci. 56, 380–388. doi: 10.4322/rca.2013.057
McCray, J. M., and Ji, S. N. (2013). Comparison of silicon sources for sugarcane on mineral and organic soils in Florida. J. Amer. Soc. Sugar Cane Technol. 33, 1–19.
McKeague, J. A., and Cline, M. G. (1963a). Silica in soil solutions I. The form and concentration of dissolved silica in aqueous extract of some soils. Can. J. Soil Sci. 43, 70–82.
McKeague, J. A., and Cline, M. G. (1963b). Silica in soil solutions II. The adsorption of monosilicic acid by soil and by other substances. Can. J. Soil Sci. 43, 83–96.
Medina-Gonzales, O. A., Fox, R. L., and Bosshart, R. P. (1988). Solubility and availability to sugarcane (Saccharum spp.) of two silicate materials. Fert. Res. 16, 3–13. doi: 10.1007/BF01053310
Meena, V. D., Dotaniya, M. L., Vassanda, C., Rajendiran, S., Kundu, A. S., and Rao, A. S. (2014). A case for silicon fertilization to improve crop yields in tropical soils. Proc. Natl. Acad. Sci. India B 84, 505–518. doi: 10.1007/s40011-013-0270-y
Meyer, J. H., Harding, R., Rampersad, A. L., and Wood, R. A. (1998). Monitoring long term soil fertility trends in the South African sugar industry using the FAS analytical database. Proc. S. Afr. Sug. Technol. Ass. 72, 61–68.
Meyer, J. H., and Keeping, M. G. (2001). “Past, present and future research of the role of silicon for sugarcane in southern Africa,” in Silicon in Agriculture, eds L. E. Datnoff, G. H. Snyder, and G. H. Korndörfer (Amsterdam: Elsevier), 257–275.
Meyer, J. H., Wood, R. A., and du Preez, P. (1971). A nutrient survey of sugarcane in the South African industry with special reference to trace elements. Proc. S. Afr. Sug. Technol. Ass. 45, 196–203.
Miles, N., Elephant, D., and Mathadeen, P. (2014a). Prediction of phosphorus availability and fixation in soils of the southern African sugar industry. Proc. S. Afr. Sug. Technol. Ass. 86, 145–148.
Miles, N., Manson, A. D., Rhodes, R., van Antwerpen, R., and Weigel, A. (2014b). Extractable silicon in soils of the South African Sugar industry and relationships with crop uptake. Commun. Soil Sci. Plan. 45, 2949–2958. doi: 10.1080/00103624.2014.956881
Miles, N., and Rhodes, R. (2013). Information Sheet 7.17 Guidelines for the Interpretation of Leaf Analyses for Sugarcane. Mount Edgecombe: South African Sugarcane Research Institute.
Miles, N., van Antwerpen, R., van Heerden, P. D. R., Rhodes, R., Weigel, A., and McFarlane, S. A. (2011). Extractable silicon in soils of the sugar industry and relationships with crop uptake. Proc. S. Afr. Sug. Technol. Ass. 84, 189–192.
Moberly, P. K., and Meyer, J. H. (1975). The amelioration of acid soils in the South African Sugar Industry. Fert. Soc. S. Afr. J. 2, 57–66.
Nikpay, A., Soleyman-Nejadian, E., Goldasteh, S., and Farazmand, H. (2015). Response of sugarcane and sugarcane stalk borers Sesamia spp. (Lepidoptera: Noctuidae) to calcium silicate fertilization. Neotrop. Entomol. 44, 498–503. doi: 10.1007/s13744-015-0298-1
Ning, D., Liang, Y., Liu, Z., Xiao, J., and Duan, A. (2016). Impacts of steel-slag-based silicate fertilizer on soil acidity and silicon availability and metals-immobilization in a paddy soil. PLoS ONE 11:e0168163. doi: 10.1371/journal.pone.0168163
Pankhurst, C. E., Blair, B. L., Magarey, R. C., Stirling, G. R., Bell, M. J., Garside, A. L., et al. (2005). Effect of rotation breaks and organic matter amendments on the capacity of soils to develop biological suppression towards soil organisms associated with yield decline of sugarcane. Appl. Soil Ecol. 28, 271–282. doi: 10.1016/j.apsoil.2004.07.010
Pereira, H. S., and Cabral, N. B. (2005). “Slag as silicon source: application criterion,” in Proceedings of the Third Silicon in Agriculture Conference (Uberlândia: Federal University of Uberlândia), 94–105.
Pereira, H. S., Korndörfer, G. H., Vidal, A. D., and de Camargo, M. S. (2004). Silicon sources for rice crop. Sci. Agric. 61, 522–528. doi: 10.1590/S0103-90162004000500010
Prabagar, S., Hodson, M. J., and Evans, D. E. (2011). Silicon amelioration of aluminium toxicity and cell death in suspension cultures of Norway spruce (Picea abies (L.) Karst.). Environ. Exp. Bot. 70, 266–276. doi: 10.1016/j.envexpbot.2010.10.001
Purchase, B. S., Wynne, A. T., Meyer, E., and van Antwerpen, R. (2008). Is there profit in cane trash? Another dimension to the assessment of trashing versus burning. Proc. S. Afr. Sug. Technol. Ass. 81, 86–99.
Ramouthar, P. V., Caldwell, P. M., and McFarlane, S. A. (2016). Effect of silicon on the severity of brown rust of sugarcane in South Africa. Eur. J. Plant Pathol. 145, 53–60. doi: 10.1007/s10658-015-0812-7
Reynolds, O. L., Keeping, M. G., and Meyer, J. H. (2009). Silicon-augmented resistance of plants to herbivorous insects: a review. Ann. Appl. Biol. 155, 171–186. doi: 10.1111/j.1744-7348.2009.00348.x
Rhodes, R., Miles, N., and Keeping, M. G. (2013). Crop nutrition and soil textural effects on eldana damage in sugarcane. Proc. S. Afr. Sug. Technol. Ass. 86, 212–136.
Ross, L., Nababsing, P., and Cheong, Y. W. Y. (1974). Residual effect of calcium silicate applied to sugarcane soils. Proc. Intl. Soc. Sug. Technol. 15, 539–542.
Sanchez, P. A. (1976). Properties and Management of Soils in the Tropics. New York, NY: John Wiley and Sons.
Sauer, D., Saccone, L., Conley, D. J., Herrmann, L., and Sommer, M. (2006). Review of methodologies for extracting plant-available and amorphous Si from soils and aquatic sediments. Biogeochemistry 80, 89–108. doi: 10.1007/s10533-005-5879-3
Savant, N. K., Datnoff, L. E., and Snyder, G. H. (1997a). Depletion of plant-available silicon in soils: a possible cause of declining rice yields. Commun. Soil Sci. Plan. 28, 1245–1252.
Savant, N. K., Korndörfer, G. H., Datnoff, L. E., and Snyder, G. H. (1999). Silicon nutrition and sugarcane production: a review. J. Plant Nutr. 22, 1853–1903. doi: 10.1080/01904169909365761
Savant, N. K., Snyder, G. H., and Datnoff, L. E. (1997b). Silicon management and sustainable rice production. Adv. Agron. 58, 151–199.
Schneider, C., Doucet, F., Strekopytov, S., and Exley, C. (2004). The solubility of an hydroxyaluminosilicate. Polyhedron 23, 3185–3191. doi: 10.1016/j.poly.2004.10.003
Schroeder, B. L., Robinson, J. B., Wallace, M., and Turner, P. (1994). Soil acidification: occurrence and effects in the South African Sugar Industry. Proc. S. Afr. Sug. Technol. Ass. 68, 70–74.
Snyder, G. H. (2001). “Methods of silicon analysis in plants, soils, and fertilizers,” in Silicon in Agriculture, eds L. E. Datnoff, G. H. Snyder, and G. H. Korndörfer (Amsterdam: Elsevier), 185–196.
Soil Survey Staff (2006). Keys to Soil Taxonomy. Washington, DC: United States Department of Agriculture, Natural Resources Conservation Service.
Sommer, M., Kaczorek, D., Kuzyakov, Y., and Breuer, J. (2006). Silicon pools and fluxes in soils and landscapes - a review. J. Plant Nutr. Soil Sci. 169, 310–329. doi: 10.1002/jpln.200521981
Struyf, E., Smis, A., Van Damme, S., Garnier, J., Govers, G., Van Wesemael, B., et al. (2010). Historical land use change has lowered terrestrial silica mobilization. Nat. Commun. 1:129. doi: 10.1038/ncomms1128
Sumner, M. E. (1970). Aluminium toxicity - a growth limiting factor in some Natal soils. Proc. S. Afr. Sug. Technol. Ass. 44, 197–203.
Sumner, M. E. (2011). Out of sight out of mind: subsurface factors in sugarcane yield decline. Proc. S. Afr. Sug. Technol. Ass. 84, 1–27.
Sumner, M. E. (2012). Review of Brazilian research on subsoil acidity. Proc. S. Afr. Sug. Technol. Ass. 85, 67–79.
Tavakkoli, E., Lyons, G., English, P., and Guppy, C. N. (2011). Silicon nutrition of rice is affected by soil pH, weathering and silicon fertilisation. J. Plant Nutr. Soil Sci. 174, 437–446. doi: 10.1002/jpln.201000023
Thorburn, P. J., Probert, M. E., Lisson, S., Wood, A. W., and Keating, B. A. (1999). Impacts of trash retention on soil nitrogen and water: an example from the Australian sugarcane industry. Proc. S. Afr. Sug. Technol. Ass. 73, 75–79.
Tripathi, D. K., Kumar, R., Chauhan, D. K., Rai, A. K., and Bicanic, D. (2011). Laser-induced breakdown spectroscopy for the study of the pattern of silicon deposition in leaves of Saccharum species. Instrum. Sci. Technol. 39, 510–521. doi: 10.1080/10739149.2011.623206
Tsujimoto, Y., Muranaka, S., Saito, K., and Asai, H. (2014). Limited Si-nutrient status of rice plants in relation to plant-available Si of soils, nitrogen fertilizer application, and rice-growing environments across Sub-Saharan Africa. Field Crop. Res. 155, 1–9. doi: 10.1016/j.fcr.2013.10.003
Tubana, B. S., Babu, T., and Datnoff, L. E. (2016). A review of silicon in soils and plants and its role in US agriculture: history and future perspectives. Soil Sci. 181, 393–411. doi: 10.1097/SS.0000000000000179
van Antwerpen, R., Meyer, J. H., and Turner, P. E. T. (2001). The effects of cane trash on yield and nutrition from the long-term field trial at Mount Edgecombe. Proc. S. Afr. Sug. Technol. Ass. 75, 235–241.
Van der Laan, M., and Miles, N. (2010). Nutrition of the South African sugar crop: current status and long-term trends. Proc. S. Afr. Sug. Technol. Ass. 83, 195–204.
Vandevenne, F., Struyf, E., Clymans, W., and Meire, P. (2012). Agricultural silica harvest: have humans created a new loop in the global silica cycle? Front. Ecol. Environ. 10, 243–248. doi: 10.1890/110046
Van Dillewijn, C. (1952). Botany of Sugarcane. Waltham, MA: The Chronica Botanica Co., Book Department.
Wedepohl, K. H. (1995). The composition of the continental crust. Geochim. Cosmochim. Ac. 59, 1217–1232. doi: 10.1016/0016-7037(95)00038-2
White, W. H., and White, P. M. (2013). Sugarcane borer resistance in sugarcane as affected by silicon applications in potting medium. J. Am. Soc. Sugar Cane Technol. 33, 38–54.
Yamauchi, M., and Winslow, M. D. (1989). Effect of silica and magnesium on yield of upland rice in the humid tropics. Plant Soil 113, 265–269. doi: 10.1007/BF02280190
Keywords: acid soils, aluminum, calcium silicate, liming, plant stress, silicon uptake, soil pH, thermophosphate
Citation: Keeping MG (2017) Uptake of Silicon by Sugarcane from Applied Sources May Not Reflect Plant-Available Soil Silicon and Total Silicon Content of Sources. Front. Plant Sci. 8:760. doi: 10.3389/fpls.2017.00760
Received: 30 November 2016; Accepted: 24 April 2017;
Published: 15 May 2017.
Edited by:
Rupesh Kailasrao Deshmukh, Laval University, CanadaReviewed by:
Soichi Kojima, Tohoku University, JapanFangsen Xu, Huazhong Agricultural University, China
Durgesh Kumar Tripathi, Banaras Hindu University, India
Copyright © 2017 Keeping. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Malcolm G. Keeping, bWFsY29sbS5rZWVwaW5nQHN1Z2FyLm9yZy56YQ==
 Malcolm G. Keeping
Malcolm G. Keeping