- 1Institute of Tropical Bioscience and Biotechnology, Chinese Academy of Tropical Agricultural Sciences, Haikou, China
- 2National Key Laboratory of Plant Molecular Genetics and National Center of Plant Gene Research, Institute of Plant Physiology and Ecology, Shanghai Institutes for Biological Sciences, Chinese Academy of Sciences, Shanghai, China
- 3Haikou Experimental Station, Chinese Academy of Tropical Agricultural Sciences, Haikou, China
Abiotic stress negatively impacts cassava (Manihot esculenta) growth and yield. Several molecular mechanisms of plant response to cold and drought have been identified and described in the literature, however, little is known about the crosstalk of the responses of cassava to these two stresses. To elucidate this question, transcriptome analysis of cassava seedlings under cold or PEG-simulated drought stress treatment was performed. Our results showed that 6103 and 7462 transcripts were significantly regulated by cold and drought stress, respectively. Gene Ontology annotation revealed that the abscisic and jasmonic acid signaling pathways shared between the two stresses responses. We further identified 2434 common differentially expressed genes (DEGs), including 1130 up-regulated and 841 down-regulated DEGs by the two stresses. These co-induced or co-suppressed genes are grouped as stress signal perception and transduction, transcription factors (TFs), metabolism as well as transport facilitation according to the function annotation. Furthermore, a large proportion of well characterized protein kinases, TF families and ubiquitin proteasome system related genes, such as RLKs, MAPKs, AP2/ERFBPs, WRKYs, MYBs, E2 enzymes and E3 ligases, including three complexes of interacting proteins were shown as key points of crosstalk between cold and drought stress signaling transduction pathways in a hierarchical manner. Our research provides valuable information and new insights for genetically improving the tolerance of crops to multiple abiotic stresses.
Introduction
Plant growth and productivity are severely affected by various abiotic stress factors such as cold and drought (Mahajan and Tuteja, 2005). These two stress factors prevent plants from reaching their full genetic potential and limit the crop yields around the world. In general, low temperature mainly results in plant surface lesion, discoloration, tissue break down, accelerated senescence, ethylene production and faster decay due to leakage of plant metabolites (Sharma et al., 2005; Solanke and Sharma, 2008). Cold stress also causes dehydration, mainly due to reduction in water uptake by roots and an impediment to close stomata (Solanke and Sharma, 2008). At freezing temperature, accumulation of ice in the intracellular spaces causes severe cellular dehydration and physical disruption of tissues (Solanke and Sharma, 2008). While drought stress has a profound adverse effect on leaf gas exchange properties, nutrient uptake, cell membrane stability, and proline and ascorbate contents (Lawlor and Cornic, 2002; Quan et al., 2015). Drought stress also exerts its detrimental effects by disrupting the ionic and osmotic equilibrium of the cell (Zhu, 2002). Plants are more vulnerable during germination and during early seedling growth to cold and drought stresses. Therefore, understanding the molecular mechanism of cold and drought stress signal transduction pathway is important for improving stress tolerance of crops.
Plant adaptation to cold and/or drought stress conditions is dependent upon the activation of cascades of molecular networks involved in signal transduction and the expression of specific stress-related genes and metabolites, with interactions and molecular crosstalk happening at several key control points (Chinnusamy et al., 2004). Therefore, understanding the common and specific signaling pathways of plants responding to cold and drought will shed new lights on the mechanisms of plants against various environmental stresses. Recently, some genes of Ca2+-signaling, abscisic acid (ABA)-signaling and nucleic acid pathways have been reported to be up-regulated in response to both cold and drought stresses indicating the presence of crosstalk between these pathways (An et al., 2012; Turyagyenda et al., 2013; Fu et al., 2016). Cytosolic Ca2+ is suggested as an important second messenger and which can be induced by both cold and drought stresses. Increase in cytosolic Ca2+ is sensed by calcium binding proteins (CaM), CaM domain-containing protein kinases (CDPKs), CBL-interacting protein kinases (CIPKs) and mitogen activated protein kinase (MAPKs), which transduce the signals to switch on transcriptional cascades (Solanke and Sharma, 2008; Huang et al., 2012). The overexpression of NPK1 enhances freezing and drought tolerance in transgenic maize (Shou et al., 2004; Solanke and Sharma, 2008). The phytohormone ABA plays central roles in the tolerance of plants to cold and drought stress. Drought stress activates ABA-dependent and ABA-independent gene expression systems involving ABA responsive element binding proteins (AREBs), MYB transcription factors (MYB), Drought responsive element binding 2 (DREB2), and NAC (NAM, ATAF1, 2 and CUC) transcription factors (TFs), while cold stress regulates ABA-independent pathway through DREB1 TFs (Agarwal and Jha, 2010; Roychoudhury et al., 2013; Nakashima et al., 2014). These major TFs facilitate stress signaling and show differential transcript regulation in response to various stresses, and their overexpression resulted in up-regulation of a large number of genes directly or indirectly linked with stress tolerance in plants (Agarwal and Jha, 2010; Nakashima et al., 2014; Joshi et al., 2016).
The ubiquitin 26S proteasome system (UPS) has been shown to act as a critical mediator in plant response and adaptation to various abiotic stresses such as drought, cold and salinity (Stone, 2014). The UPS functions within the nucleus and cytoplasm to affect the accumulation of a wide range of stress-responsive proteins and degrade misfolded proteins that may be induced by abiotic stress treatments (Lyzenga and Stone, 2012). Ubiquitin-dependent proteasomal degradation contains multiple steps requiring the sequential action of three enzymes, namely ubiquitin activating enzyme (E1), ubiquitin conjugating enzyme (E2), and ubiquitin ligase (E3) (Stone, 2014; Sharma et al., 2016). Thousands of ubiquitination-related proteins are predicted in the Arabidopsis genome, which includes four 2 E1s, 37 E2s, and over 1300 E3 ligases (Kraft et al., 2005). E3s can be further divided into three major groups according to the conserved domain contained, including U-box, Really Interesting New Gene (RING) and Homology to E6-Associated Carboxyl-Terminus (HECT) groups (Stone, 2014). In plant, an increasing number of E2 and E3 encoding genes have been shown to be involved in abiotic stress responses. For example, transcript levels of Arabidopsis ICE1 (Inducer of CBF expression 1), a basic helix-loop-helix (bHLH) transcription factor, are up-regulated in response to cold stress. ICE1 activity is inhibited by a RING-type E3 ligase, High Expression of Osmotically Responsive Gene 1 (HOS1), which is capable of catalyzing ICE1 ubiquitination in vitro and in vivo. Overexpression of HOS1 increases sensitivity to freezing conditions of transgenic plants (Dong et al., 2006). In this case, ubiquitination of the regulatory protein would function as a negative regulator. On the contrary, UPS degrading negative players would activate the signaling pathways that are required for tolerance to stress (Stone, 2014). An example is R2R3-type MYB TF, Botrytis Susceptible 1 (BOS1), which plays an important role in plants response to abiotic stresses (Luo et al., 2010). BOS1 can be degraded by RING-type E3 ligase BOI (BOS1 Interactor), which is involved in attaching the ubiquitins to BOS1 (Luo et al., 2010). This research suggests that UPS functions as a positive regulator in response to stress stimulus.
Cassava is an important tropical root crop worldwide and is an essential source of bio-fuel and starch for human consumption and animal feed. Although cassava can effectively utilize light, heat and water resources, cold and drought stresses cause great damage to the production of cassava. Improving the tolerance of cassava to cold and drought is an important strategy to increase root starch yield and supply. Recently, in cassava, many cold or drought stress-inducible genes acting in calcium and reactive oxygen species (ROS) signaling transduction pathways have been identified. In plant, stress-activated Ca2+ signaling transduction pathway is activated by Ca2+ spike from the cytoplasm for overcoming the cold damage and maintaining a balance between the intracellular and extracellular ionic concentrations (An et al., 2012; Zhu et al., 2013). the ROS signaling pathway that plays an important role in ROS detoxification, and hormone signaling pathway that are critical for the regulation of the cold- and/or drought-responsive transcriptome (An et al., 2012; Fu et al., 2016; Liao et al., 2016a). However, most studies have dealt with only one kind of stress, and little is known about how cassava plant integrates multiple signaling pathways, which may crosstalk or diverge at various points and form complex networks in response to both cold and drought stresses. Luckily, transcriptome profiling has been developed as a high-efficiency and extensive used approach to systematically investigate the gene expression in response to a diverse range of stresses globally across distant species (Chen et al., 2010; Zhu et al., 2013; Xie et al., 2015; Zheng et al., 2015; Muthusamy et al., 2016; Zhao et al., 2016). Additionally, transcriptomic data can provide much information on the metabolite, phytohormone, and transcript networks that control plant responses to various environmental stimuli. In this study, we analyzed the transcriptome of cassava seedlings exposed to cold and drought stresses and were able to identify various stress-responsive genes and related crosstalk networks involved in signal perception, transduction, and tolerance.
Results
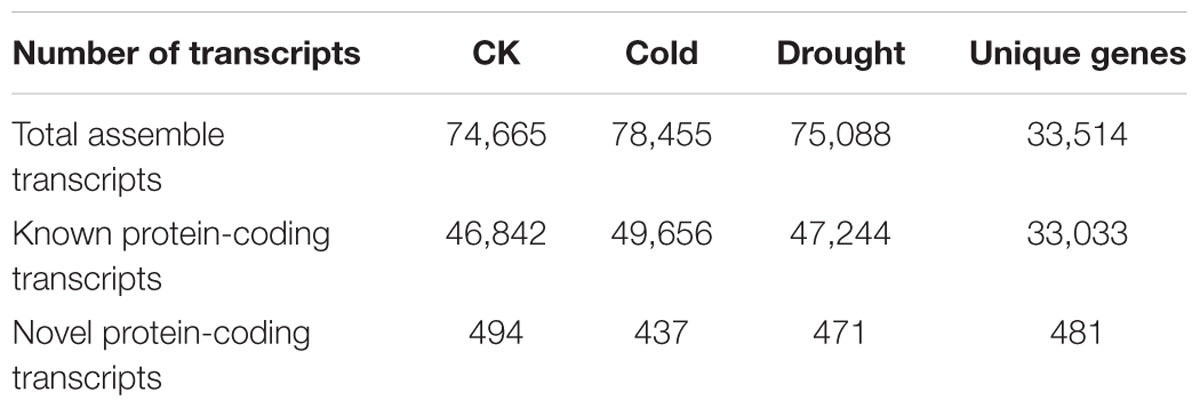
Transcriptome Overview and Novel Transcript Identification
We analyzed our strand-specific RNA sequencing data of cassava shoot apices and leaves exposed to cold, drought stress and control conditions, respectively (Li et al., 2017). A total of ∼2.6 million RNA-Seq clean reads were obtained. After mapping the reads to the cassava genome using TopHat2, 74665, 78455, and 75088 transcript fragments were assembled using Cufflink on the samples under cold, drought stress and control conditions, respectively. By comparing these transcripts with the annotated genes in the cassava genome, we found that approximately 63% of total transcripts matched the predicted cassava genes and 33,033 transcripts were categorized as unique genes (Table 1). Notably, after filtering out low-abundance genes (FPKM < 1) and non-coding RNAs, we found a total of 481 protein-coding transcripts (1.46%) that showed no overlap with any annotated genes, and which were then identified as reference-dependent novel protein-coding genes (Table 1 and Supplementary Table S1).
Identification of Differentially Expressed Genes under Cold and/or Drought Stresses
The expression level of 33033 annotated cassava protein-coding genes for each treatment (control check (CK), cold or drought) was assessed by counting the number of reads from the respective library that matched each contig, and normalizing to FPKM (fragments per kilo base of transcript per million mapped reads) to correct for the differences in sequencing depth for each dataset and gene length differences (Figure 1A). To determine data reproducibility, principal component analysis (PCA) was performed on the correlation of read count of annotated genes among samples. We found that the three replicates with the same treatment were clustered together, and were separated from samples with other treatments, indicating high reproducibility among biological replicates (Supplementary Figure S1). By comparing three RNA-seq libraries, CK, cold and drought, a plenty of differentially expressed genes (DEGs) were identified. To characterize the genes that were associated with cold and/or drought stress, we analyzed the most differentially regulated transcripts with a log2 ratio ≥ 1 or ≤-1 and FDR (false discover rate) ≤ 0.05. Using these standards, 6103 and 7462 genes were identified to be differentially expressed under cold and drought stress, respectively (Figures 1B,C and Supplementary Table S2). Under the cold treatment, 3547 genes were identified to be up-regulated and 2556 were down-regulated (Figure 1B), whereas under the drought treatment 3525 genes were up-regulated and 3937 genes were down-regulated (Figure 1C). Notably, a total of 2434 genes were differentially expressed under both cold and drought stress treatments, of which 1130 were co-induced and 841 were co-repressed by cold and drought (Figures 1D,E).

FIGURE 1. Variation in gene expression under cold or drought stress. (A) The heatmap of the fold change values in the RNA-seq data, and was used to visualize the gene expression pattern under cold and drought stress. (B,C) Volcano plot indicates the significance of DEGs between cold stress and control (B), and between drought stress and control conditions (C), respectively. (D) Venn diagram represents the overlapping DEGs number between cold- and drought-responsive genes. (E) Scatter plot indicates genes that were co-induced, co-repressed and specifically regulated by cold and/or drought treatment.
Hormone Signaling Pathway in Response to Cold and Drought Stresses
According to previous research, both cold and drought stresses caused visible and similar morphological changes, including severe leaf dehydration and apical buds wilting. In addition, a clear impact on metabolites concentration was also observed in leaves of cold- and drought-stressed cassava plants (An et al., 2012). For instance, the concentrations of proline, total soluble sugars and malondialdehyde (MDA) increased after cold or drought treatment (An et al., 2012; Fu et al., 2016). Therefore, the similar physiological and molecular changes of cassava plant response to both stresses suggest the existence of crosstalk between the drought and cold stress signal perception and transduction pathways.
To explore the common and specific responses of cassava leaves to cold and drought stress, Gene Ontology (GO) analyses of all the DEGs were employed, and totally 111 significantly enriched gene categories in response to cold and/or drought stress were identified. The functions of these DEGs were categorized according to biological processes, including response to abiotic stimulus, response to lipid, cell communication, signal transduction and response to ABA stimulus pathways (Supplementary Table S3). Notably, a large number of DEGs were involved in hormone metabolic and signaling transduction pathways. Plant hormones such as auxin, ABA, jasmonic acid (JA), salicylic acid (SA), ethylene (ET), brassinosteroid (BR) and other small molecules play critical roles in regulating plant growth, nutrient allocation and stress tolerance to promote survival and acclimatize to varying environments throughout the life-span of plants (Fahad et al., 2015). In this study, six cold-specific signaling pathways and six drought-specific response pathways were identified. In the cold stress response, DEGs involved in ethylene and SA mediated signaling pathways were significantly enriched. While the “signaling pathway of auxin mediated” were identified in the drought stress response. Notably, two common hormone-related signaling pathways of DEGs that responded to both cold and drought stresses were identified: “response to ABA stimulus” and “response to JA stimulus pathways” (Table 2). Interestingly, we found that ABA-mediated signaling transduction pathway was regulated by cold stress, whereas drought stress mainly affected ABA biosynthetic and metabolic process. In plants, ABA signals are perceived by cellular receptors and a concept of activation of specific cellular ABA responses by perception in the distinct cellular compartments is currently emerging (Golldack et al., 2014). In that study, a total of 34 DEGs were identified as members of the ABA signaling pathway, which were mainly related to ABA transport, binding, signaling perception, transduction and ABA-dependent gene transcription, and included ATP-binding cassette (ABC)-containing transporter proteins, PYRABACTINRESISTANCE-LIKE 2 (PYL2), type 2C protein phosphatases (PP2Cs), SNF1-RELATED PROTEINKINASEs (SnRK2s) (Supplementary Table S4). Furthermore, CORONATINE INSENSITIVE 1 (COI1) and jasmonic acid ZIM-domain protein 2/9 (JAZ2/9), which function as JA receptors, were also identified response to both cold and drought stresses. It was reported that JAZs act as a key negative regulators of JA signaling in response to various abiotic stresses through repressing the activities of a plethora of TFs, such as bHLHs and MYBs (Goossens et al., 2016). The differentially expression of JAZ2 and JAZ9 indicated that JA signaling transduction related proteins were involved in response to cold and drought stresses in cassava.
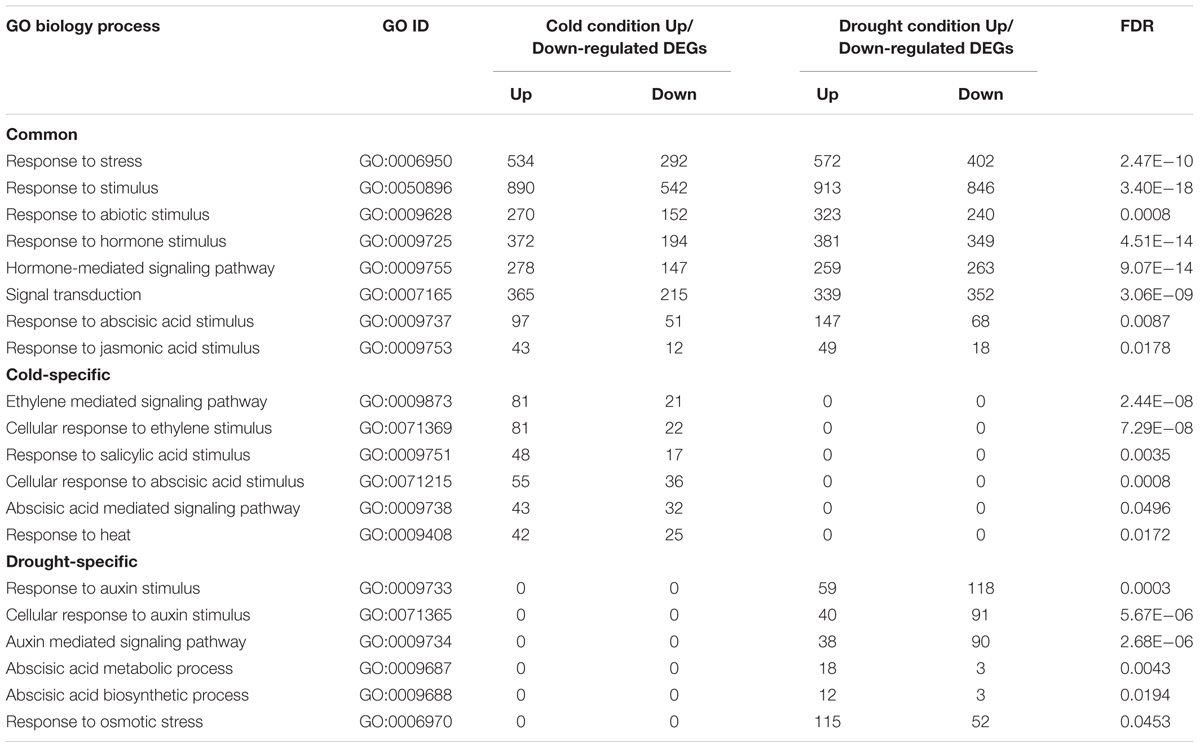
TABLE 2. Gene ontology of biological process classification of DEGs under cold and/or drought stress.
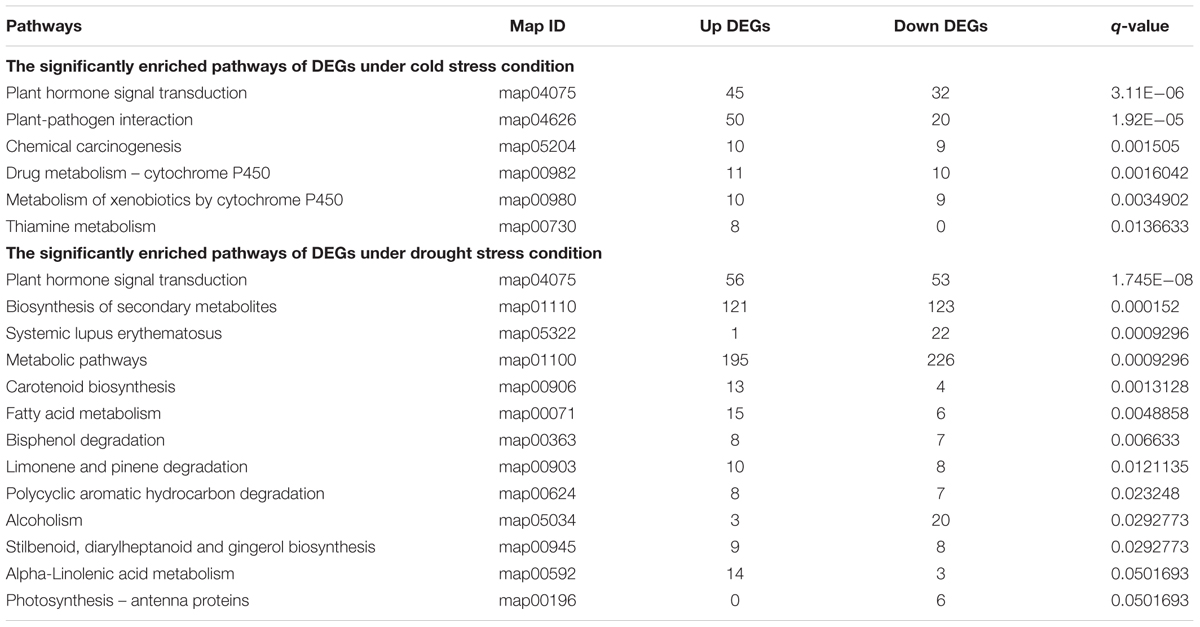
A Kyoto encyclopedia of genes and genomes (KEGG) pathway enrichment analysis was performed to further investigate the changes in the transcriptome profiles between the drought and cold stress responses. Compared to the CK condition, DEGs involved in signaling transduction and metabolic pathways were significantly enriched in both cold and drought stress responses. Table 3 presents the overall response pathways of cassava plants to cold and drought stress, respectively. One common feature was observed by pairwise comparison of both stress responses: plant hormone signal transduction pathways were notably enriched under the two stresses. A few differences between the two stress responses were also observed: (1) the drought-specific pathways were much more enriched than that of cold stress. Meanwhile, the number of DEGs whose expression was repressed by drought was higher than that of cold stress; (2) more metabolism- and photosynthesis-related DEGs were enriched after drought treatment. Taken together, these results pointed, for the first time, to relevant biological processes and pathways enriched as a consequence of the cold and drought stress treatments as compared to the control condition, and also indicated that both ABA and JA were involved in mediating these two stress responses.
Characterization of Cold and Drought Stress Co-responsive DEGs
To gain further insight into whether similar trends existed in the transcriptomic profiles of cassava plants in response to cold and drought stresses, we analyzed the 1971 DEGs up-regulated or down-regulated by the two stress treatments (Supplementary Table S4). To classify these DEGs functionally, the GO database was utilized. In total, 46 GO terms were enriched and distributed in three categories, including biological process, cellular component and molecular function. Under the biological process category, cellular process (58.5% up-regulated, 57.6% down-regulated) was the largest group, followed by metabolic process (56.9% up-regulated, 52.8% down-regulated), response to stimulus (33.8% up-regulated, 23.2% down-regulated) and biological regulation (32.7% up-regulated, 30.5% down-regulated). In relation to the cellular component category, the DEGs associated with cell part (67.5% up-regulated, 70.1% down-regulated) and organelle part (39.2% up-regulated, 37.1% down-regulated), membrane part (25.9% up-regulated, 26.5% down-regulated) represented the most abundant categories. Among the top 3 GO terms enriched in the molecular function category, binding (61.9% up-regulated, 49.2% down-regulated), catalytic activity (48.3% up-regulated, 49.4% down-regulated) and nucleic acid binding transcription factor (15.8% up-regulated, 7.7% down-regulated) were accumulated more than the other terms (Figure 2). In addition, all of the above DEGs were annotated by KEGG pathway analysis, and the most enriched pathway was “biosynthesis of secondary metabolites,” followed by “metabolic pathways” and “microbial metabolism in diverse environments” (Supplementary Table S5). The analysis of the KEGG results suggested that both cold and drought stresses had strong effects on the biosynthesis of secondary metabolites.
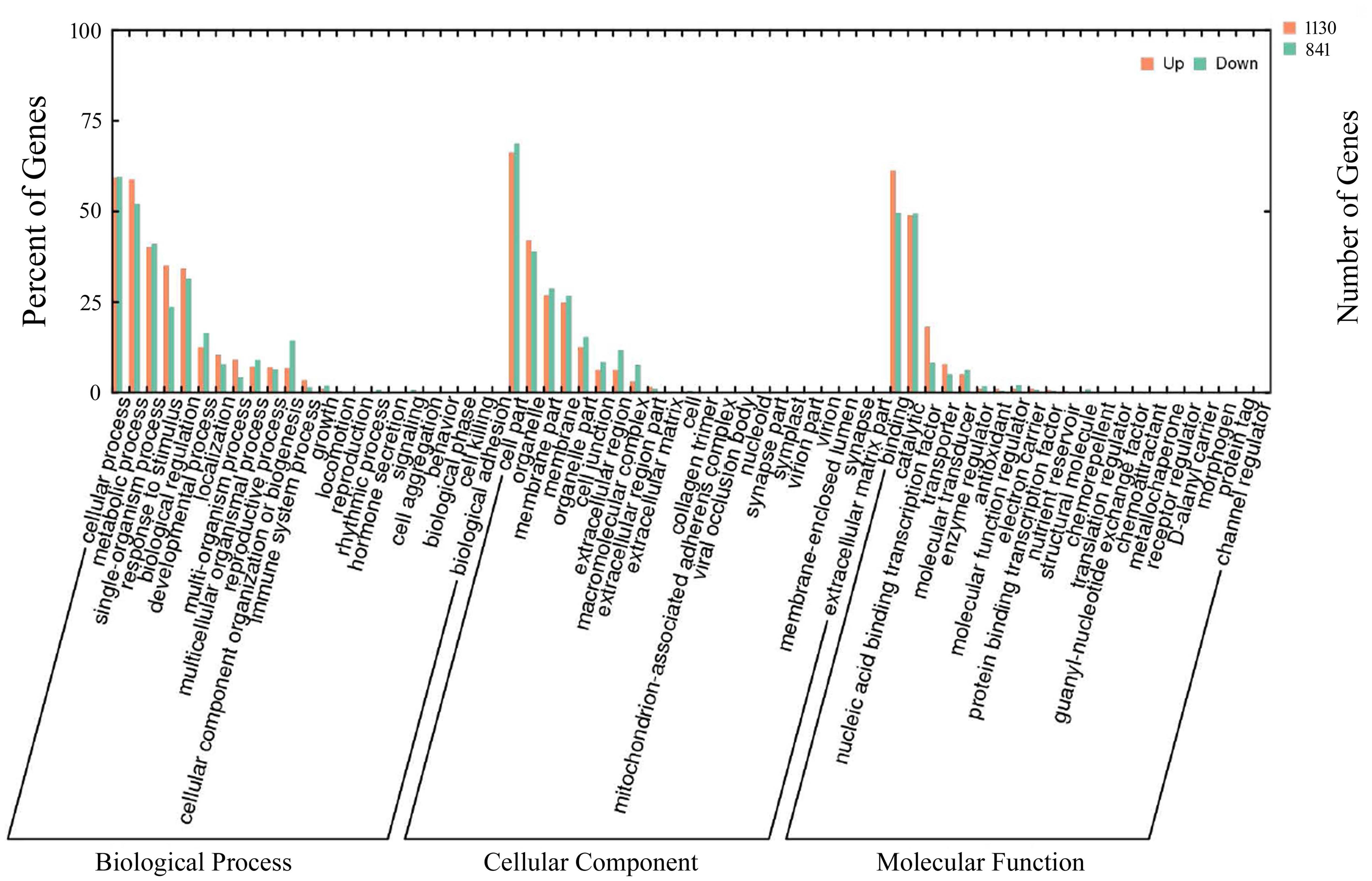
FIGURE 2. Gene ontology enrichment of stress co-responsive DEGs. The results are summarized into three main categories: biological processes, cellular components, and molecular functions. The x-axis corresponds to GO terms, and the y-axis shows the number of up- or down-regulated DEGs. A total of 1971 DEGs co-regulated by cold and drought stress were assigned to GO terms.
Identification of Stress-Related Protein Kinase Genes
According to the “molecular function” GO terms in Figure 2, catalytic activity and transcription factor activity were the two major categories. Therefore, proteins with catalytic function act as master regulators involved in stress signal perception by modulating downstream proteins activities. In the past decades, plant protein kinases have been found to be conserved components of signaling networks such as the perception of plant hormones and various adverse environmental conditions. We identified a wide range of protein kinase family members in the RNA-seq data, mainly composed of 91 pkinase, 26 protein tyrosine kinase and 21 receptor-like protein kinases (RLKs) family members according to the domain classification in the Pfam database (Finn et al., 2014; Figures 3A–C and Supplementary Table S6). Among pkinase and protein tyrosine kinase families, a number of common stress-related DEGs that responded to both cold and drought stresses were identified: 4 MAPK family members and 12 CDPK family members. The majority of these DEGs were up-regulated by both cold and drought stresses, suggesting they may play positive roles in stress tolerance. RLKs are a family of transmembrane receptors with an intracellular serine/threonine kinase domain and perform critical functions in modulating diverse biological processes by perceiving extracellular stimuli and activating downstream signaling responses (Ye et al., 2016). In both the cold and drought stress responses, ten Leucine-Rich Repeat Receptor-Like protein Kinases (LRR-RLKs), seven Malectin or Malectin-like domain-containing Receptor-Like Kinases (MRLKs) and four Legume lectin Receptor-Like Kinases (LecRLKs) were identified. Among 21 RLK family members, a total of 11 RLKs were significantly induced by the two stresses, especially after cold treatment, while the other 10 exhibited decreased expression patterns following all stress conditions (Figure 3C). The diverse expression of RLKs indicated that early cold and drought stress signaling perception and transduction may be enhanced.
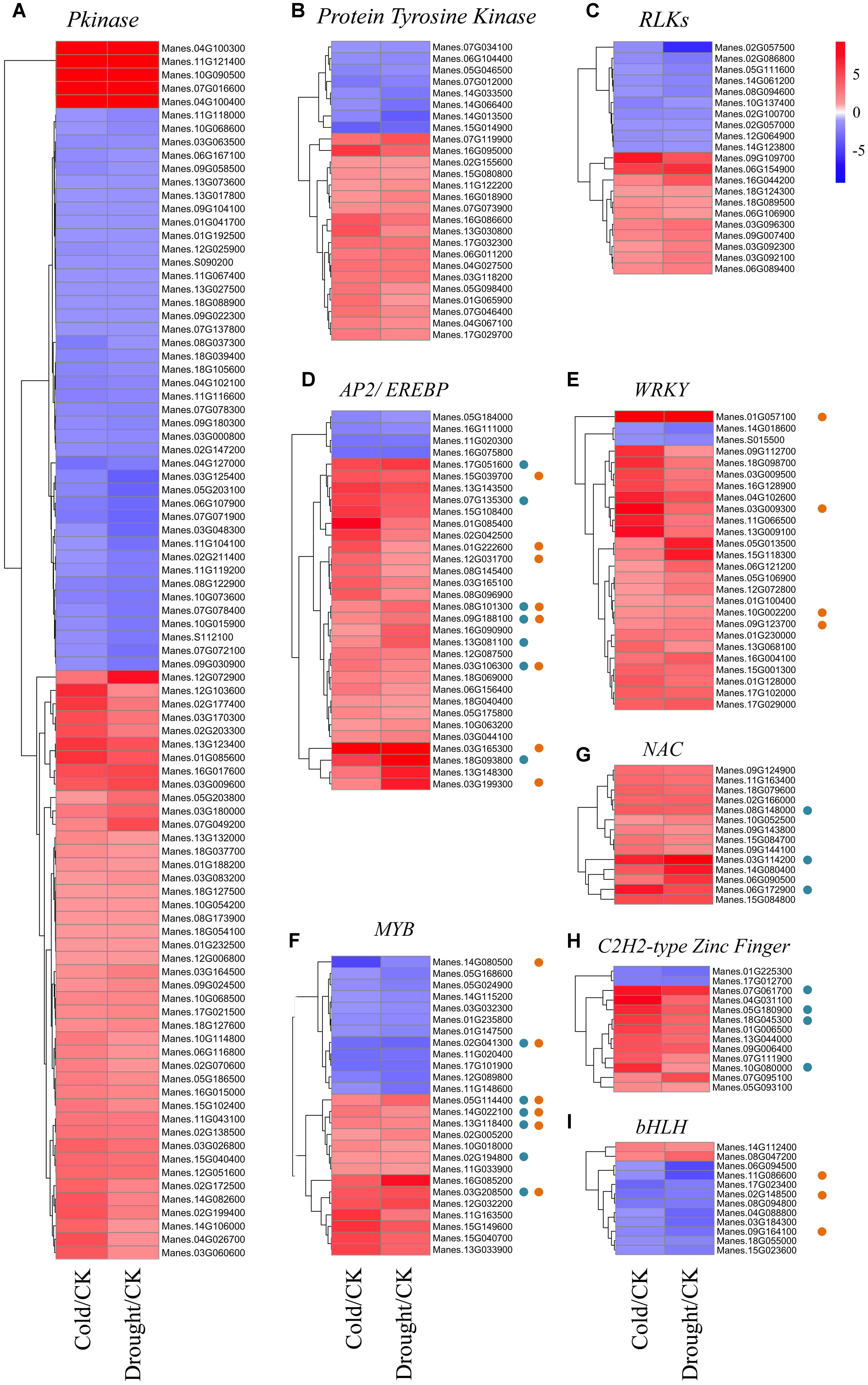
FIGURE 3. Expression pattern of cassava protein kinases and TFs in response to cold and drought stress. (A–I) Heatmaps were generated from the fold change values of Pkinase (A), Protein Tyrosine Kinase (B), RLKs (C), AP2/ERFBP (D), WRKY (E), MYB (F), NAC (G), C2H2-type Zinc Finger (H), and bHLH (I) family numbers under cold and drought stress as compared to the control condition. The blue and orange spots indicated DEGs responding to ABA and JA signaling, respectively.
Identification of Transcription Factors in Responses to Cold and Drought Stress
Transcription factors play an essential role in plant growth and stress responses (Agarwal and Jha, 2010). We investigated the biological functions of TFs that responded both to cold and drought stress in cassava. A total of 211 co-responsive DEGs were identified as TFs, which belonged to 42 gene families based on Pfam database (Finn et al., 2014). The number of stress-induced TFs (125) was much larger than the number of stress-suppressed ones (47), suggesting that transcriptional activation may be dominant over repression (Supplementary Table S6). Among these TFs, the most abundant transcription factor family was APETALA 2/Ethylene-Responsive Element Binding Factor (AP2/EREBP, number of DEGs: 32), followed by WRKY (26), MYB (26), NAC (15), Zinc-Finger C2H2 (13) and bHLH (12) family. The highest number of TFs was induced by the two stresses, except for the bHLH family (Figures 3D–I). Notably, 20 and 21 TFs were identified to be ABA- and JA-responsive according to GO analysis, respectively (Figure 3).
The AP2/EREBP family was originally identified as key regulators in the manifestation of the stress response in plants, as evidenced by many well-characterized DREBs/CBFs, which belongs to DREB subfamily of AP2 TFs (Mizoi et al., 2012). In our study, all members of AP2/EREBP family are classified into four subgroups, including AP2, RAV, ERF and DREB subgroups. For example, Manes.12G031700, Manes.15G039700 and Manes.03G165100 encode members of ERF subgroup (Supplementary Table S6), which is known to be involved in various biotic stress responses (Cheng et al., 2013; Zhu et al., 2014). In many studies TFs defined by the WRKY domain, a highly conserved 60 residues long DNA binding domain (Rushton et al., 2010). Many have been shown WRKYs to be involved in plant responses to abiotic stresses (Rushton et al., 2010; Chen et al., 2012). Our transcriptome analysis showed that most of the differentially expressed WRKYs (92.3%) were induced by cold and drought treatment (Figure 3E). The MYBs are widely distributed in plants, forming a large family characterized by conserved MYB domain, and almost all MYBs are stress- or hormone-responsive (Yanhui et al., 2006; Wang et al., 2016). In our study, 26 MYB or MYB-related family genes were responsive to cold and drought, with 14 stresses-elevated and 12 stresses-repressed (Figure 3F). The number of up- and down-regulated MYBs was nearly equal, suggesting a diverse range of functions for this TF family in response to cold and drought stress. The NAC family has been demonstrated to be associated with diverse biological processes and responding to many biotic and abiotic stresses in plant (Nuruzzaman et al., 2013). We found all of the 15 co-responsive NAC genes identified in this study were up-regulated after stress treatments (Figure 3G), suggesting that these NAC genes may play a positive role in regulating stress-associated genes. The Cys2/His2 (C2H2) zinc finger protein belongs to Zinc-Finger Protein (ZFP) family that play various roles in the plant stress response (Ciftci-Yilmaz and Mittler, 2008). In Arabidopsis, expression of Cys2/His2-type ZFPs AZF1 and STZ are induced by abiotic stresses and ABA treatment. Experiments indicated that they function as positive regulators in stress response (Sakamoto et al., 2004). Members of the bHLH gene family encode TFs and play diverse functions in plant growth and development, such as trichome differentiation, seed development, stamen development, shoot and inflorescence branching (Schmitz and Theres, 2005; Balkunde et al., 2010; Sun et al., 2010). It is worth noting that the majority of bHLH (83.3%) family members were down-regulated based on the RNA-seq data (Figure 3I), suggesting that cold and drought stresses may conserve energy by inhibiting plant growth, and adapt to the environmental stimuli.
In addition to the above-mentioned TFs, we also found four TFs families, including Heat Stress transcription Factors (HSFs, number of DEGs: 10), GRAS (9), Homeobox (8), and Zinc-Finger Dof (7), were cold-and drought-responsive in our study (Supplementary Table S6). Most of these TFs showed the inducible expression pattern after cold and drought treatments. Taken together, the large number and diverse expression patterns of co-responsive TFs indicated their involvement in the complex signaling networks of the two stresses responses.
Identification of DEGs Associated with Protein Degradation
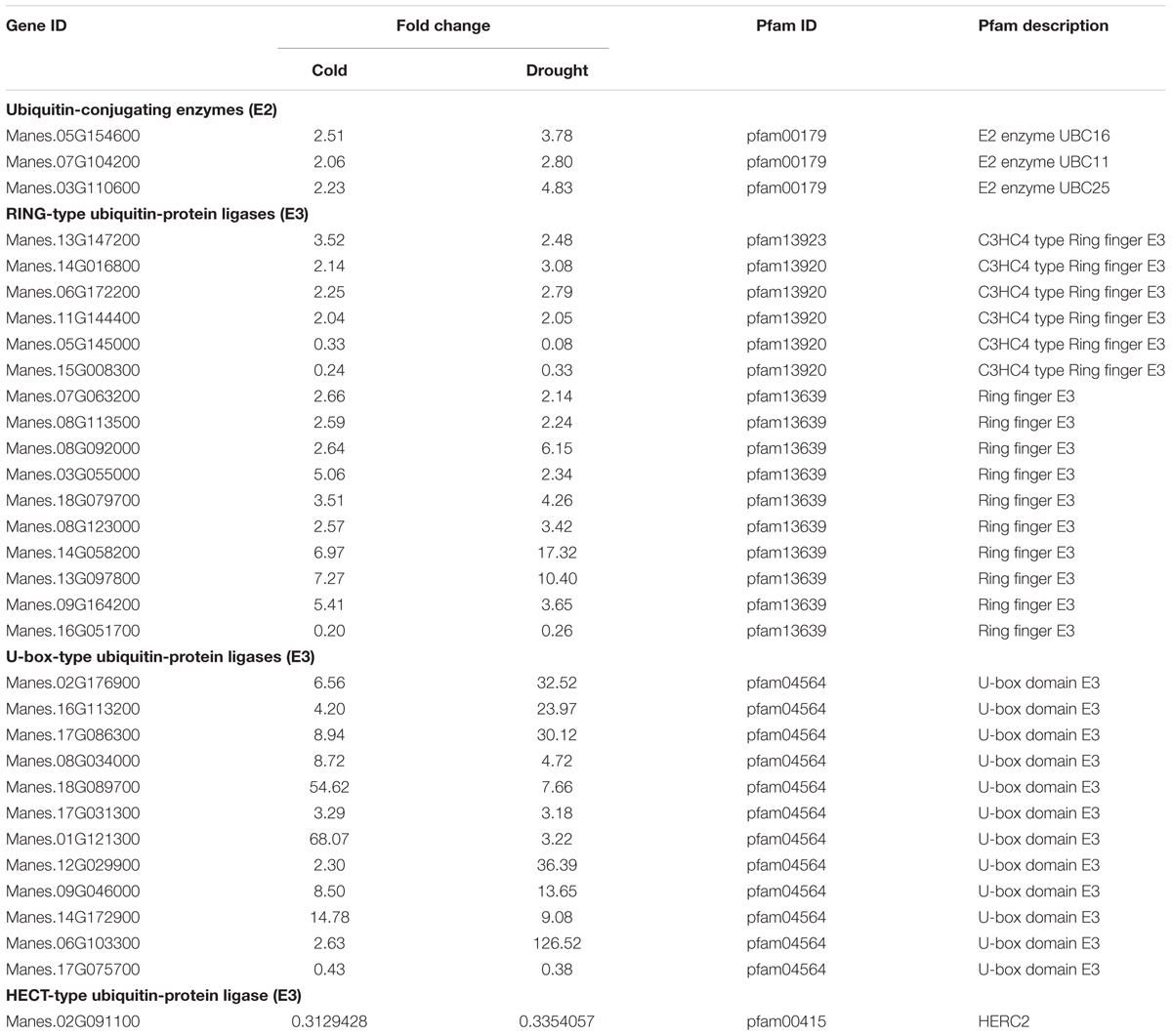
By modulating the activity and accumulation of functional proteins, the UPS acts as key regulator in mediating growth and abiotic stress response of plants. Recent reports in this field have identified numerous ubiquitin-conjugating (E2) enzymes and ubiquitin ligases (E3) exhibiting altered expression levels during abiotic stresses (Lyzenga and Stone, 2012; Stone, 2014). In this study, a great number of degradation-associated proteins were involved in response to both cold and drought stress. Among these DEGs, all of three E2 enzymes were found to be up-regulated under stresses. Meanwhile, a total of 29 E3s composed of 16 RING-type, 12 U-box-type and 1 HECT-type members were also identified and co-regulated by the two stresses (Table 4). As reported in previous studies, E2s and E3s are involved in a range of protective responses that help plants to against various stresses (Stone, 2014). The identification of E3 ubiquitin ligases, in our study, establishes a direct link between the UPS and plant stress tolerance.
Identifying the Interaction Network of Co-Responsive DEGs
In recent years, it has been demonstrated that the computational analysis of protein–protein interaction (PPI) networks is becoming increasingly useful to identify the biological processes and signaling pathways (Natale et al., 2014). To investigate the potential PPI networks of co-responsive DEGs, we identified the homologs of DEGs in Arabidopsis that function in hormone signaling, protein kinase, protein degradation and TFs, and assembled putative networks based on experimentally validated interactions database Arabidopsis interactome map (Arabidopsis Interactome Mapping Consortium, 2011) and BioGRID (Stark et al., 2006). As a result, we found 23 proteins that can self-interact or interact with other members in the network. Among them, there are three interactive protein complex, including LPP2 (LIPID PHOSPHATE PHOSPHATASE 2), BIR1 (BAK1-INTERACTING RECEPTOR-LIKE KINASE 1), and MGT3 (MAGNESIUM TRANSPORTER 3) (Figure 4). AtLPP2 negatively regulates the activity of ABI4, an AP2-type TF, in response to ABA signals (Katagiri et al., 2005). The proteins from these networks were activated, indicating an increase in the ABA response in cassava leaves under stress treatments, which is consistent with our results in Table 2. Previous work showed that BIR1 functions as a negative regulator of plant immunity through regulating multiple plant resistance signaling pathways (Liu et al., 2016). MGT3 was involved in transporting magnesium, which acted as a key osmoticum maintaining growth in low calcium concentrations in plants (Conn et al., 2011). Together, the networks of protein complexes identified in this study were mainly involved in ABA signaling transduction, antimicrobial response and osmotic adjustment, and may represent key crosstalk nodes in the cold and drought stress responses in cassava.
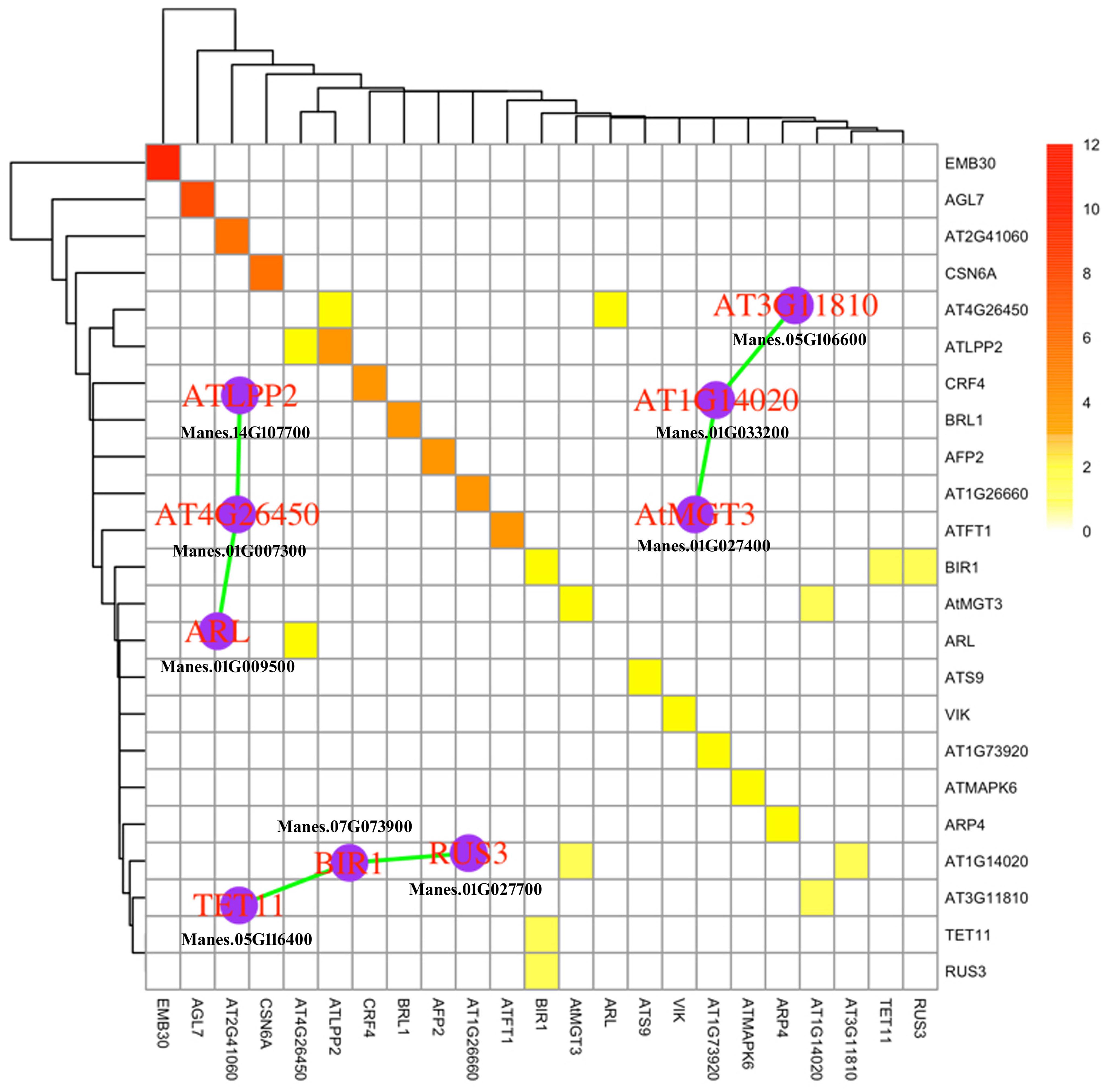
FIGURE 4. Protein–protein Interaction network of cassava co-responsive DEGs homologs in Arabidopsis that function in hormone signal, protein kinase, protein degradation and TFs. PPI database were downloaded from BioGRID (Stark et al., 2006) and Arabidopsis interactome map (Arabidopsis Interactome Mapping Consortium, 2011). The color of heatmap indicates number of evidences for PPI. The signal in diagonal line indicates self-interaction. The graph with purple vertex and green line highlights the PPI network of cassava homologous genes in Arabidopsis.
Validation by Real-Time RT-qPCR of Selected Stress-Responsive Genes as Detected by the RNA-Seq Data
To further study the cold and drought co-responsive DEGs listed in Supplementary Table S4 in cassava, 12 genes were analyzed by real-time quantitative PCR (qRT-PCR). The temporal expression patterns of the DEGs detected by qPCR correlated well with the RNA-seq data. Among the 12 DEGs, 4 protein kinases (Manes.04G026700, Manes.09G024500, Manes.17G021500, and Manes.18G124300) showed differential expression under cold and drought stress (Figures 5A–D). Four TFs MeERF1 (Figure 5E), MeWRKY33 (Figure 5F), MeMYB21 (Figure 5G) and MeAZF3 (Figure 5H) also showed strong induction by cold and drought treatments, except MeWRKY33, which exhibited significant variation between cold and drought-treated plants. Two E2s (MeUBC16 and MeUBC25) and two E3s (Manes.07G063200 and Manes.17G031300) were also up-regulated after both cold and drought treatment (Figures 5I–L). The striking similarity in the response pattern between the two types of stresses suggested that these genes may play important roles in cassava. Thus, the functions of these genes in cassava will be further studied.
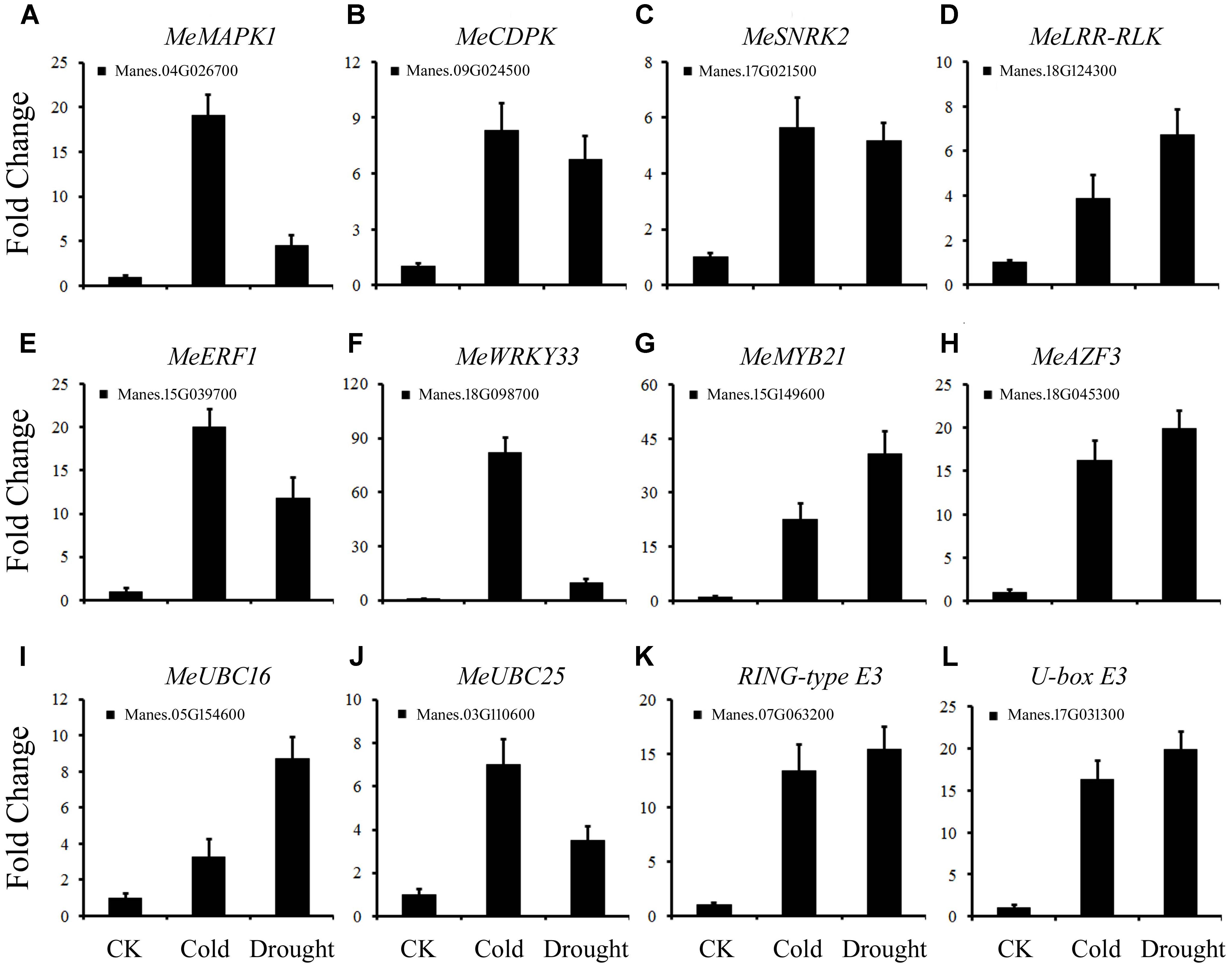
FIGURE 5. Confirmation of the expression patterns of DEGs using quantitative RT-PCR. (A–L) The expression pattern of DEGs under cold or drought stress. Data are presented as mean ± SD of three independent assays. MeACTIN was used as the reference gene.
Discussion
Cassava is an economically important crop grown for its starchy roots and its good adaptation to drought and heat stress conditions. Molecular mechanisms involved in cassava responses to diverse adverse environment stimuli have been studied using microarray chips and high-throughput sequencing (Lokko et al., 2007; An et al., 2012; Utsumi et al., 2012; Turyagyenda et al., 2013; Zhao et al., 2015; Fu et al., 2016). An increasing number of drought- or cold stress-responsive genes have been reported. However, most studies are focused on one kind of stress, no systematic consensus on genes that correspond to crosstalk network for cold and drought stress perception, signal transduction and tolerance of cassava has been reported. Identification of key regulators at the nodes and branches between cold and drought stress signaling pathways will provide insights into the mechanism involved these stress responses in cassava. Here, we focused on the common effects of both cold and drought stresses on the cassava plants response. In this research, we performed a comparative analysis of the transcriptome profiles of cassava leaves under CK, cold, and drought stress conditions, our results would provide fundamental insights into gene expression changes and signaling transduction networks of cassava in response to temperature and water changes.
The Role of Hormones in Plant Response to Cold and Drought Stress
In response to stress, a diverse range of genes involved in various biochemical and physiological processes are involved, which can lead to adjustment of the cellular milieu and plant tolerance. In this study, we obtained the expression information of 33,033 protein-coding genes from cold- and drought-treated cassava plants. Using comparative analysis of stress-responsive genes from different conditions, 1971 genes were identified as commonly induced or repressed by cold and drought treatments. GO analysis revealed that DEGs associated with the hormonal signaling pathways, including ABA and JA signaling pathways, were significantly enriched.
The prominent role of ABA in plant response to abiotic stress conditions has long been studied. Under drought conditions, ABA is known to stimulate closure of stomata, resulting in maintenance of water balance and modulate plant growth through the regulation of stress-responsive genes (Verma et al., 2016). Previous studies have found that levels of endogenous ABA were increased under cold, salt and drought stress conditions in many plant species. Exogenous application of ABA to plants can elevate their tolerances to abiotic stresses (Sah et al., 2016). In our research, a large number of DEGs of “response to ABA stimulus” were significantly up- or down-regulated by cold and drought stress, such as ABC-containing transporter protein (MeABCG25, Manes.18G101300), ABA receptor PYL2 (Manes.16G079500), PP2Cs (Manes.18G136400, Manes.07G131300, Manes.10G010600, and Manes.03G036900), SnRK2 (Manes.17G021500), and TFs (MYBs, NACs and Zinc-finger C2H2s). In Arabidopsis, ABC transporter protein AtABCG25, is required for exporting ABA from vascular tissues (Sah et al., 2016). AtPYL2 acts as an ABA receptor, which can bind ABA directly (Yin et al., 2009). After binding to ABA, this receptor can suppress the phosphatase activity of PP2Cs, the negative regulator of SnRK2 (Park et al., 2009). However, a common feature of PP2Cs is that their expression is induced by high ABA levels and abiotic stresses (Yang et al., 2017). In rice, the overexpression of OsPP108, which encodes a PP2C family protein, confers ABA insensitivity and tolerance to stresses, such as high salt and drought (Singh et al., 2015). The SnRK2 family of protein kinases plays a central role in cellular responses to abiotic stresses and mediates ABA signaling by phosphorylating their substrate proteins such as TFs, metabolic enzymes, and ion channels (Sah et al., 2016). In cassava, the significantly stress-induced expression pattern of MeABCG25, PP2Cs, and SnRK2 suggested that these TFs play regulatory roles in the perception and transduction of ABA signals under stress conditions.
Recent studies have highlighted that JA plays an important role in the regulation of abiotic stress tolerance under osmotic stress conditions (Ahmad et al., 2016). It has been demonstrated that JAs regulate the gene expression involved in stress responses though up-regulating expression of antioxidant enzymes and eliciting plant secondary metabolism (Ahmad et al., 2016). We found that 50 DEGs associated with “response to JA stimulus” signaling pathways were significantly enriched under both cold and drought stress. JA-dependent signaling is initiated by the interaction of JA with the receptor COI1 which causes conformation changes to interact with bHLH TFs. This interaction results in ubiquitination of JAZs by the Skp1/Cullin/F-box (SCF) complex, and releases bHLH TFs suppression, triggering downstream JA responses (Goossens et al., 2016). As previously reported (Valenzuela et al., 2016), COI1, which act as an E3 ubiquitin ligase, was significantly induced under cold and drought treatments. However, two JAZ genes, JAZ2 and JAZ9, the key negative regulator of JA signaling pathway, also significantly increased by stresses, consistent with the results of previously reported studies (Huang et al., 2016; Valenzuela et al., 2016), suggesting possible feedback mechanisms triggered by high levels of JA accumulation. Taken all together, the exploration of ABA and JA signaling molecules can be beneficial to examine the details of underlying molecular mechanisms of these hormone signals transduction, and much work need to be done in near future in cassava.
Signal Transduction in Plant Response to Cold and Drought Stress
In total, we identified 1130 induced and 841 repressed DEGs. Molecular functional classification of the these genes shows a large number of genes that are predicted to be involved in signal transduction, transcription regulation, secondary metabolism, transport facilitation and protein degradation. As expected, protein kinases and TFs corresponded to the most regulated genes, suggesting that cold and drought stress signal transduction pathways overlap at several points. The important Receptor-like Kinases (RLK) group which includes members like LRR-RLK, MRLK, and LecRLK, have been previously shown to be involved in mediating the cellular response to various environmental cues, hormonal signals and stress perception (Ye et al., 2016). Many studies have focused on the characterization and function of RLKs in abiotic stress tolerance, specifically for drought, cold and salinity. In rice, LRR-RLK gene FON1 increased drought tolerance of transgenic rice plants through phosphorylating key components of the ABA signaling pathway and activating the ABA signal (Feng et al., 2014; Ye et al., 2016). Another well studied example is CRLK1, a calcium-regulated RLK, which modulated cold tolerance through phosphorylating MAP kinases in plants, and promoted expression of cold stress response genes, finally regulating plant adaptation to cold stress (Furuya et al., 2013; Ye et al., 2016).
In addition to the stress signal perception cascades, there is a second group of DEGs with protein kinase activity. It was reported that MAPKs and calcium-binding related kinases were central regulators in signal transduction, connecting the perception of external stimuli to cellular responses (Boudsocq and Sheen, 2013; Xie et al., 2015; de Zelicourt et al., 2016). In the study, a total of 4 MAPKs, 12 CDPKs and a large number of serine-threonine protein kinases were identified as commonly stress-responsive members. These kinases may not only function as signals transducers but also work as central regulators through phosphorylating stress-responsive proteins under cold and drought stress. In Physcomitrella patens, ARK encodes a MAP kinase kinase kinase (MAP3K), and acts as a novel regulatory component of ABA signaling transduction pathway via phosphorylating SnRK2s (Saruhashi et al., 2015). Many TFs are also functionally regulated by post-translational phosphorylation mechanisms. For instance, bZIP, NAC and DREB TFs are generally phosphorylated by the serine-threonine kinases, MYB/MYC and WRKY TFs are phosphorylated by MAPKs, and CDPKs are involved in phosphorylation of some bZIP TFs (Gahlaut et al., 2016).
The Role of Transcription Factors in Plant Response to Cold and Drought Stress
It is known that TFs act as regulatory proteins in a synchronized manner by regulating a set of downstream genes under their control and consequently enhance the stress tolerance of the plant. A large number of TFs including WRKYs, ERFs, and MYBs have been shown to exhibit induced expression during exposure to cold or drought stress, suggesting that these genes may be used to improve stress tolerance in cassava (An et al., 2012; Turyagyenda et al., 2013; Fu et al., 2016). In our research, different families of TFs including up-regulated AP2/ERFBP, WRKY, MYB, NAC, GRAS, C2H2 ZFP, and down-regulated bHLH were identified in cassava response to cold and drought stress. Among these TFs, we identified a wide range of ABA-dependent TFs, including 7 AP2/ERFBPs, 6 MYBs, 3 NACs, and 4 zinc-finger TFs. In addition, 7 AP2/ERFBPs, 6 MYBs, 4 WRKYs, and 3 bHLHs TFs were shown to be involved in the regulation of core JA signaling (Figure 3). There are many evidences of implication of AP2/ERFBP genes especially DREBs in drought, salt, heat and cold stress responses in plants (Mizoi et al., 2012). In Arabidopsis, DREB2s have been reported to promote the expression of downstream genes to enhance cold and drought tolerance in transgenic plant (Sakuma et al., 2002). Ectopic overexpression of Arabidopsis DREB1 in cassava improves the plant performance against cold and drought stresses but represses plant growth, and reduces storage root yield (An et al., 2016). In previous studies, a total of 85 WRKY and 166 MYB family numbers in cassava have been identified, respectively, and their expression patterns under drought stress have also been characterized (Liao et al., 2016b; Wei et al., 2016). In our research, abiotic stress related gene like WRKY22 (Manes.01G057100), WRKY33 (Manes.18G098700), MYB78 (Manes.03G208500), and MYB21 (Manes.15G149600) was found to accumulate after cold and drought stress treatment. Based on previous studies, WRKY22 is considered to modulate the interplay between the SA and JA pathways in response to a wide range of biotic and abiotic stimuli (Kloth et al., 2016). WRKY33 is well known as a substrate of MPK3/MPK6 in reprogramming the expression of camalexin biosynthetic genes, which drives the metabolic flow to camalexin production in Arabidopsis challenged by biotic stress (Mao et al., 2011). Furthermore, genetic and physiological experiments demonstrate that the R2R3-MYB transcription factor MYB21 functions as direct targets of JAZs to regulate male fertility specifically, indicating MYB TFs play an essential role in JA signaling transduction pathway (Song et al., 2011). Taken together, according to our results, a large number of TFs were involved in cold and drought stresses responses in a hormone-dependent manner, suggesting that TFs play an important role in modulating the two stresses signal transduction and may crosstalk at many steps.
The Role of Ubiquitin-Mediated Degradation System in Plant Response to Cold and Drought Stresses
Recently, the involvement of the ubiquitin proteasome system (UPS) at the cellular level has received great attention. In plants, ubiquitin-mediated degradation plays an important role in growth, hormonal signaling, abiotic stress, embryogenesis, and senescence (Stone, 2014). The expression of three ubiquitin conjugating enzyme E2 (UBC E2) including UBC11 (Manes.07G104200), UBC16 (Manes.05G154600), and UBC25 (Manes.03G110600) was found to increase after cold and drought stress treatments in cassava. Accumulating evidences show that the expression of UBC E2 genes from a number of plant species is regulated by development and also by environmental conditions (Xu et al., 2009). Overexpression of soybean GmUBC2 resulted in improved drought tolerance in Arabidopsis (Zhou et al., 2010). Therefore cassava UBC11/16/25 might play some role in the response to biotic stresses, given that they were up-regulated after stress treatment.
In addition to E2 enzymes, E3 ligases have also been extensively studied in development and in signaling responses during abiotic stress, and act as either negative or positive regulators in stress signal transduction (Lyzenga and Stone, 2012). We identified 13 RING type and 11 U-box type E3s were strongly induced by cold and drought stress, respectively. It was previously shown that overexpression of Arabidopsis PUB22 and PUB23, which encode U-box E3 ligases, were hypersensitive to drought stress, indicating that PUB22 and PUB23 act as negative regulators in osmotic stress tolerance (Cho et al., 2008). By contrast, RING finger E3 ligases SDIR1 (SALT- AND DROUGHT-INDUCED RING FINGER1) act as positive regulators of ABA signaling. Overexpression of SDIR1 leads to ABA hypersensitivity and ABA-associated phenotypes, such as salt hypersensitivity in germination, enhanced ABA-induced stomatal closing, and enhanced drought tolerance (Zhang et al., 2007). Therefore, positive regulation of RING type and U-box type E3s under cold and drought stress treatment in cassava suggest that E3s may play a role in the response to stress conditions through ubiquitination and the resulting degradation of essential components in these stress signaling pathways.
Hierarchical View of Crosstalk between Cold and Drought Stress
Plant survival under multiple and simultaneous environmental conditions implies complex processes of signal reception, transmission, amplifications and interaction. As shown in Figure 6, signal transduction pathways triggered by cold and drought share a plenty of regulatory proteins that transduce the related signaling into downstream processes that result in resistance to such stresses. In general, the cold and drought stress signals are perceived by several receptors at the cell membrane, such as LRR-RLKs, MRLKs and LecRLKs, followed by calcium molecules transduction to activate downstream stress-responsive genes in response to abiotic stress. The change of second messengers stimulates downstream signals mediated by combinations of protein phosphorylation cascades, such as MAPKs and CDPKs. In addition, ABA and JA usually increase during early stages of stress. In the presence of ABA, PYL receptors bind to ABA and prevent PP2C from phosphorylating SnRK2. The SnRK2s are then phosphorylated by MAP3Ks and activated. The active SnRK2s can phosphorylate downstream TFs and activate the expression of ABA-dependent genes. The cold and drought response has been reported to involve both ABA-dependent and independent pathways (Roychoudhury et al., 2013). In the ABA-independent pathway, the transcription factor of GRAS and bHLH has also been implicated in dehydration stress signaling. On the other hand, in the ABA signaling pathway, TFs such as MYB, AP2 and NAC are believed to activate the transcription of specific target genes. Meanwhile, upon increase of JA/JA-Ile levels by cold and drought stress, SCFCOI1 binds to JA and facilitates the degradation of JAZs protein, repressors of MYCs/MYBs, via the ubiquitin-26S proteasome pathway. Therefore, MYCs/MYBs can switch on transcription of JA -responsive genes. Therefore, ABA- and JA-induced signaling pathways might regulate transcription or directly/indirectly interact with cold and drought stress signaling networks. Moreover, we also obtained a large amount of information on ubiquitin-dependent protein catabolic processes, which was involved in cold and drought stress signaling by either positively or negatively regulating the accumulation of the stress-responsive proteins. Furthermore, we identified PPI network, in which the genes were responsive to both drought and cold, including three complexes of interacting proteins. Taken together, our study indicated that stress signal perception and transduction were critical components of the cold and drought stress responses in cassava plants. Meanwhile, these common signaling-related DEGs could be key nodes of complex networks in response to cold and drought stresses, and are candidates for further studies as potential targets to improve resistance to multiple stresses through genetic engineering.
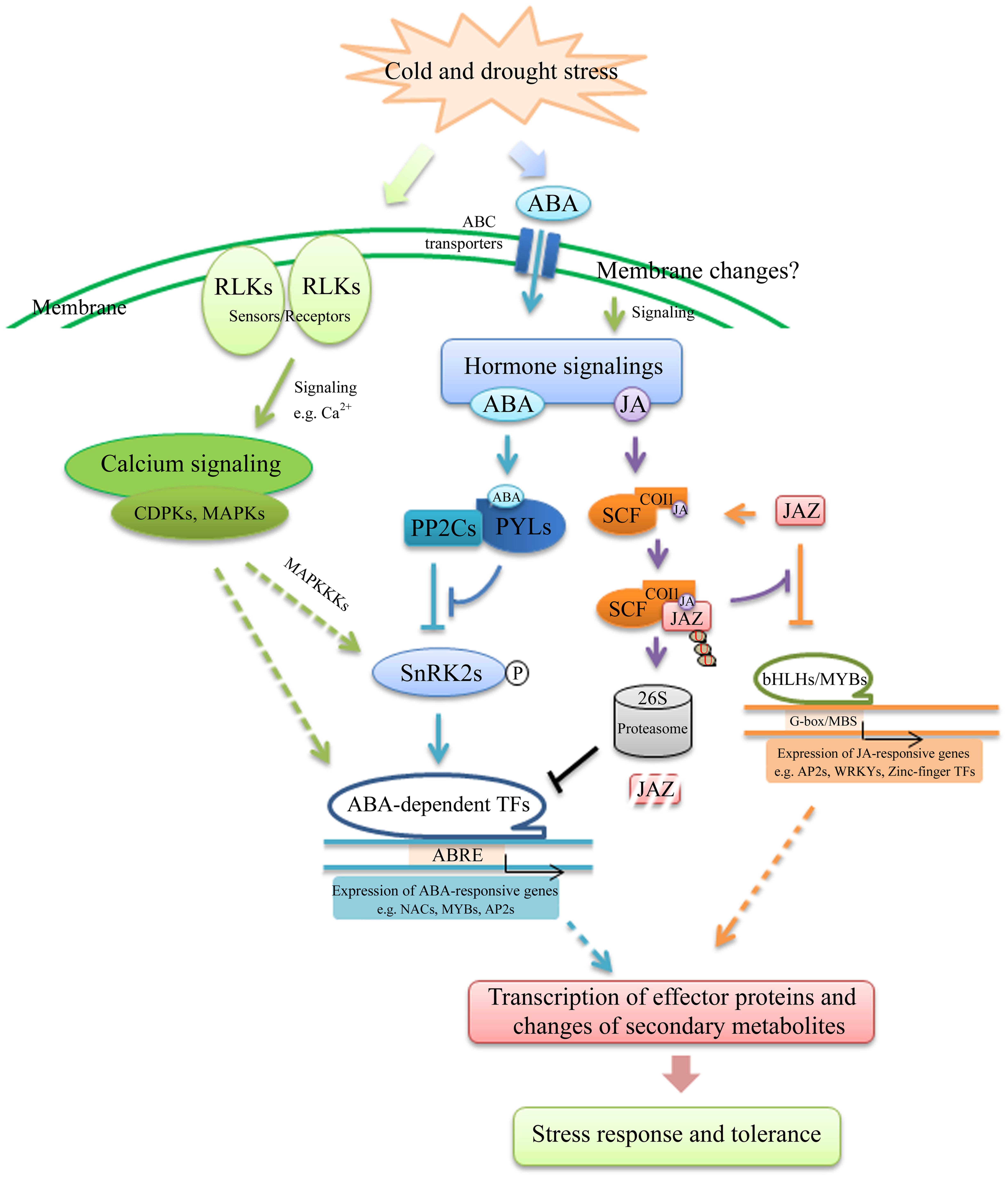
FIGURE 6. Models describing the signaling pathways involved in the acquisition of cold and drought tolerance.
Conclusion
A comprehensive transcriptome-based characterization of cold and drought responsive DEGs was conducted in cassava for the first time. Our results revealed that the expression of 1130 genes was induced, and 841 genes were repressed in cassava following both cold and drought stress treatments. We identified important crosstalk and divergence of the signaling transduction pathways between these two stresses. First, the great majority of the DEGs were related to signal transduction, TFs and the ubiquitin-proteasome system. Second, hormone signaling, transcription activation/repression, and ubiquitin-dependent protein catabolic pathways were important response events in the mechanism underlying the crosstalk of the cold and drought response mechanisms of cassava. Our results revealed functional specialization of gene families when plant cells encountering different stresses. The results of this research provide a theoretical basis for further investigation of the molecular mechanisms involved in the cassava response to cold and drought stress, and also provide valuable information for future work.
Materials and Methods
Plant Material and Stress Treatment
Cassava (Manihot esculenta) cultivar (TMS60444) was used in the study. As previously described, the stems about 1.5 cm in size with one bud were cut and planted in MS plate for 2 weeks in a growth chamber at 26 ± 2°C, with a photoperiod of 16 h light and 8 h dark (Li et al., 2017). For cold treatment, plants with a uniform growth status were treated at 4°C for 24h in a chamber under light (Li et al., 2017). The shoot apex and the youngest leaves were collected and then were frozen in liquid nitrogen for RNA extraction. For drought treatment, seedlings were planted in MS with 20% PEG6000 and harvested at 6 h after treatment (Li et al., 2017). In all cases, parallel and untreated plants at the same stage were used as controls. At least three replicates for each treatment were harvested (Li et al., 2017).
RNA Isolation, DNA Synthesis, and Real-Time Quantitative PCR
Total RNA was extracted using the RNA Plant kit (OMEGA) according to the manufacturer’s instructions. The 1st strand cDNA were synthesized in a 20 μl reaction solution containing 2 μg total RNA samples of CK, as well as abiotic stressed plants using PrimeScriptTM RT reagent Kit (Takara). Real-time qPCR assays were performed using the SYBR Premix Ex TaqTM (Takara) followed the manuals from the manufacturers. A one-step RT-PCR procedure was performed in all experiments. For each sample, qRT-PCR reaction was repeated three times and the relative mRNA expression level was calculated as 2-ΔΔCt. The cassava MeACTIN gene was used as a constitutive reference. All the primers used were as listed in Supplementary Table S7.
RNA-Seq and Data Analysis
For RNA-seq, cassava shoot apices and youngest leaves for each treatment were collected and pooled separately to prepare 3 cDNA libraries (CK- control treatment; cold- cold stressed; drought -drought stressed). As previously described (Li et al., 2017), A total of 140 gigabase in-depth sequencing of library was performed initially on a HiSeq 2500 instrument that generated paired-end reads with 125 nucleotides. After filter out the adapter and low quality sequences, TopHat2 (Trapnell et al., 2012) was used to map the clean reads to cassava genome. Then, reference genome-based transcriptome assembly was performed to generate non-redundant unigenes using Cufflinks v2 method (Trapnell et al., 2010). The expression levels of unigenes were calculated and then normalized to FPKM (fragments per kilo base of transcript per million mapped reads) (Wagner et al., 2012). The DEGs between treatment and control were calculated by cuffdiff program, and the significant DEGs were filtered with ratio more than two fold change and false discovery rate (FDR) less than 0.05. The RNA-seq data was submitted to Sequence Read Archive (SRA) in NCBI and the accession number is SRP101302.
Functional Annotation and Classification
To predict the potential functions and biological pathways of the genes, we annotated the genes using the NR protein database (NCBI), GO (Young et al., 2010) and KEGG databases. As described previously, GO enrichment analysis of genes was implemented by using the GO seq R package. KOBAS software was used for testing the statistical enrichment of DEGs in KEGG pathways1 (Kanehisa et al., 2008).
Protein–Protein Interaction Network
The PPI database were downloaded from Arabidopsis interactome map (Arabidopsis Interactome Mapping Consortium, 2011) and BioGRID (Stark et al., 2006). The Arabidopsis homologs of cassava DEGs were annotated, and number of PPI evidences for pairwise genes from PPI database was extracted. The PPI matrix with evidence number were presented as heatmap using R package “pheatmap,” and the graph network were displayed using R package “igraph.”
Author Contributions
SL and MP participated in the design of the study. SL and XY analyzed the RNA-seq data. ZC, XLY, MBR, and WL carried out the molecular biology experiments. All authors read and approved the final manuscript.
Funding
This work was supported by the Hainan Province Innovative Research Team Foundation (2016CXTD013), the Program of Hainan Association for Science and Technology Plans to Youth R & D Innovation (HAST201627), Central Public-interest Scientific Institution Basal Research Fund for Chinese Academy of Tropical Agricultural Sciences (1630052017022, 1630052016004), and the National Natural Science Foundation of China (31561143012).
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We thank Dr. Rong Di to polish the English for the article.
Supplementary Material
The Supplementary Material for this article can be found online at: http://journal.frontiersin.org/article/10.3389/fpls.2017.01259/full#supplementary-material
FIGURE S1 | Principal component analysis of three samples for each treatment.
Footnotes
References
Agarwal, P. K., and Jha, B. (2010). Transcription factors in plants and ABA dependent and independent abiotic stress signalling. Biol. Plant. 54, 201–212. doi: 10.1007/s10535-010-0038-7
Ahmad, P., Rasool, S., Gul, A., Sheikh, S. A., Akram, N. A., Ashraf, M., et al. (2016). Jasmonates: multifunctional roles in stress tolerance. Front. Plant Sci. 7:813. doi: 10.3389/fpls.2016.00813
An, D., Ma, Q. X., Yan, W., Zhou, W. Z., Liu, G. H., and Zhang, P. (2016). Divergent regulation of CBF regulon on cold tolerance and plant phenotype in cassava overexpressing Arabidopsis CBF3 gene. Front. Plant Sci. 7:1866. doi: 10.3389/fpls.2016.01866
An, D., Yang, J., and Zhang, P. (2012). Transcriptome profiling of low temperature-treated cassava apical shoots showed dynamic responses of tropical plant to cold stress. BMC Genomics 13:64. doi: 10.1186/1471-2164-13-64
Arabidopsis Interactome Mapping Consortium (2011). Evidence for network evolution in an Arabidopsis interactome map. Science 333, 601–607. doi: 10.1126/science.1203877
Balkunde, R., Pesch, M., and Hulskamp, M. (2010). Trichome patterning in Arabidopsis thaliana from genetic to molecular models. Curr. Top. Dev. Biol. 91, 299–321. doi: 10.1016/S0070-2153(10)91010-7
Boudsocq, M., and Sheen, J. (2013). CDPKs in immune and stress signaling. Trends Plant Sci. 18, 30–40. doi: 10.1016/j.tplants.2012.08.008
Chen, L., Ren, F., Zhong, H., Feng, Y., Jiang, W., and Li, X. (2010). Identification and expression analysis of genes in response to high-salinity and drought stresses in Brassica napus. Acta Biochim. Biophys. Sin. 42, 154–164.
Chen, L., Song, Y., Li, S., Zhang, L., Zou, C., and Yu, D. (2012). The role of WRKY transcription factors in plant abiotic stresses. Biochim. Biophys. Acta 1819, 120–128. doi: 10.1016/j.bbagrm.2011.09.002
Cheng, M. C., Liao, P. M., Kuo, W. W., and Lin, T. P. (2013). The Arabidopsis ETHYLENE RESPONSE FACTOR1 regulates abiotic stress-responsive gene expression by binding to different cis-acting elements in response to different stress signals. Plant Physiol. 162, 1566–1582. doi: 10.1104/pp.113.221911
Chinnusamy, V., Schumaker, K., and Zhu, J. K. (2004). Molecular genetic perspectives on cross-talk and specificity in abiotic stress signalling in plants. J. Exp. Bot. 55, 225–236. doi: 10.1093/jxb/erh005
Cho, S. K., Ryu, M. Y., Song, C., Kwak, J. M., and Kim, W. T. (2008). Arabidopsis PUB22 and PUB23 are homologous U-Box E3 ubiquitin ligases that play combinatory roles in response to drought stress. Plant Cell 20, 1899–1914. doi: 10.1105/tpc.108.060699
Ciftci-Yilmaz, S., and Mittler, R. (2008). The zinc finger network of plants. Cell. Mol. Life Sci. 65, 1150–1160. doi: 10.1007/s00018-007-7473-4
Conn, S. J., Conn, V., Tyerman, S. D., Kaiser, B. N., Leigh, R. A., and Gilliham, M. (2011). Magnesium transporters, MGT2/MRS2-1 and MGT3/MRS2-5, are important for magnesium partitioning within Arabidopsis thaliana mesophyll vacuoles. New Phytol. 190, 583–594. doi: 10.1111/j.1469-8137.2010.03619.x
de Zelicourt, A., Colcombet, J., and Hirt, H. (2016). The role of MAPK modules and ABA during abiotic stress signaling. Trends Plant Sci. 21, 677–685. doi: 10.1016/j.tplants.2016.04.004
Dong, C. H., Agarwal, M., Zhang, Y., Xie, Q., and Zhu, J. K. (2006). The negative regulator of plant cold responses, HOS1, is a RING E3 ligase that mediates the ubiquitination and degradation of ICE1. Proc. Natl. Acad. Sci. U.S.A. 103, 8281–8286. doi: 10.1073/pnas.0602874103
Fahad, S., Hussain, S., Bano, A., Saud, S., Hassan, S., Shan, D., et al. (2015). Potential role of phytohormones and plant growth-promoting rhizobacteria in abiotic stresses: consequences for changing environment. Environ. Sci. Pollut. Res. Int. 22, 4907–4921. doi: 10.1007/s11356-014-3754-2
Feng, L., Gao, Z. R., Xiao, G. Q., Huang, R. F., and Zhang, H. W. (2014). Leucine-rich repeat receptor-like kinase FON1 regulates drought stress and seed germination by activating the expression of ABA-responsive genes in rice. Plant Mol. Biol. Rep. 32, 1158–1168.
Finn, R. D., Bateman, A., Clements, J., Coggill, P., Eberhardt, R. Y., Eddy, S. R., et al. (2014). Pfam: the protein families database. Nucleic Acids Res. 42, D222–D230. doi: 10.1093/nar/gkt1223
Fu, L., Ding, Z., Han, B., Hu, W., Li, Y., and Zhang, J. (2016). Physiological investigation and transcriptome analysis of polyethylene glycol (PEG)-induced dehydration stress in cassava. Int. J. Mol. Sci. 17:283. doi: 10.3390/ijms17030283
Furuya, T., Matsuoka, D., and Nanmori, T. (2013). Phosphorylation of Arabidopsis thaliana MEKK1 via Ca2+ signaling as a part of the cold stress response. J. Plant Res. 126, 833–840. doi: 10.1007/s10265-013-0576-0
Gahlaut, V., Jaiswal, V., Kumar, A., and Gupta, P. K. (2016). Transcription factors involved in drought tolerance and their possible role in developing drought tolerant cultivars with emphasis on wheat (Triticum aestivum L.). Theor. Appl. Genet. 129, 2019–2042. doi: 10.1007/s00122-016-2794-z
Golldack, D., Li, C., Mohan, H., and Probst, N. (2014). Tolerance to drought and salt stress in plants: unraveling the signaling networks. Front. Plant Sci. 5:151. doi: 10.3389/fpls.2014.00151
Goossens, J., Fernandez-Calvo, P., Schweizer, F., and Goossens, A. (2016). Jasmonates: signal transduction components and their roles in environmental stress responses. Plant Mol. Biol. 91, 673–689. doi: 10.1007/s11103-016-0480-9
Huang, G. T., Ma, S. L., Bai, L. P., Zhang, L., Ma, H., Jia, P., et al. (2012). Signal transduction during cold, salt, and drought stresses in plants. Mol. Biol. Rep. 39, 969–987. doi: 10.1007/s11033-011-0823-1
Huang, Z., Jin, S. H., Guo, H. D., Zhong, X. J., He, J., Li, X., et al. (2016). Genome-wide identification and characterization of TIFY family genes in Moso Bamboo (Phyllostachys edulis) and expression profiling analysis under dehydration and cold stresses. PeerJ 4:e2620. doi: 10.7717/peerj.2620
Joshi, R., Wani, S. H., Singh, B., Bohra, A., Dar, Z. A., Lone, A. A., et al. (2016). Transcription factors and plants response to drought stress: current understanding and future directions. Front. Plant Sci. 7:1029. doi: 10.3389/fpls.2016.01029
Kanehisa, M., Araki, M., Goto, S., Hattori, M., Hirakawa, M., Itoh, M., et al. (2008). KEGG for linking genomes to life and the environment. Nucleic Acids Res. 36, D480–D484. doi: 10.1093/nar/gkm882
Katagiri, T., Ishiyama, K., Kato, T., Tabata, S., Kobayashi, M., and Shinozaki, K. (2005). An important role of phosphatidic acid in ABA signaling during germination in Arabidopsis thaliana. Plant J. 43, 107–117. doi: 10.1111/j.1365-313X.2005.02431.x
Kloth, K. J., Wiegers, G. L., Busscher-Lange, J., van Haarst, J. C., Kruijer, W., Bouwmeester, H. J., et al. (2016). AtWRKY22 promotes susceptibility to aphids and modulates salicylic acid and jasmonic acid signalling. J. Exp. Bot. 67, 3383–3396. doi: 10.1093/jxb/erw159
Kraft, E., Stone, S. L., Ma, L., Su, N., Gao, Y., Lau, O. S., et al. (2005). Genome analysis and functional characterization of the E2 and RING-type E3 ligase ubiquitination enzymes of Arabidopsis. Plant Physiol. 139, 1597–1611. doi: 10.1104/pp.105.067983
Lawlor, D. W., and Cornic, G. (2002). Photosynthetic carbon assimilation and associated metabolism in relation to water deficits in higher plants. Plant Cell Environ. 25, 275–294.
Li, S., Yu, X., Lei, N., Cheng, Z., Zhao, P., He, Y., et al. (2017). Genome-wide identification and functional prediction of cold and/or drought-responsive lncRNAs in cassava. Sci. Rep. 7:45981. doi: 10.1038/srep45981
Liao, W., Wang, G., Li, Y., Wang, B., Zhang, P., and Peng, M. (2016a). Reactive oxygen species regulate leaf pulvinus abscission zone cell separation in response to water-deficit stress in cassava. Sci. Rep. 6:21542. doi: 10.1038/srep21542
Liao, W., Yang, Y., Li, Y., Wang, G., and Peng, M. (2016b). Genome-wide identification of cassava R2R3 MYB family genes related to abscission zone separation after environmental-stress-induced abscission. Sci. Rep. 6:32006. doi: 10.1038/srep32006
Liu, Y., Huang, X., Li, M., He, P., and Zhang, Y. (2016). Loss-of-function of Arabidopsis receptor-like kinase BIR1 activates cell death and defense responses mediated by BAK1 and SOBIR1. New Phytol. 212, 637–645. doi: 10.1111/nph.14072
Lokko, Y., Anderson, J. V., Rudd, S., Raji, A., Horvath, D., Mikel, M. A., et al. (2007). Characterization of an 18,166 EST dataset for cassava (Manihot esculenta Crantz) enriched for drought-responsive genes. Plant Cell Rep. 26, 1605–1618.
Luo, H., Laluk, K., Lai, Z., Veronese, P., Song, F., and Mengiste, T. (2010). The Arabidopsis Botrytis Susceptible1 Interactor defines a subclass of RING E3 ligases that regulate pathogen and stress responses. Plant Physiol. 154, 1766–1782. doi: 10.1104/pp.110.163915
Lyzenga, W. J., and Stone, S. L. (2012). Abiotic stress tolerance mediated by protein ubiquitination. J. Exp. Bot. 63, 599–616. doi: 10.1093/jxb/err310
Mahajan, S., and Tuteja, N. (2005). Cold, salinity and drought stresses: an overview. Arch. Biochem. Biophys. 444, 139–158. doi: 10.1016/j.abb.2005.10.018
Mao, G. H., Meng, X. Z., Liu, Y. D., Zheng, Z. Y., Chen, Z. X., and Zhang, S. Q. (2011). Phosphorylation of a WRKY transcription factor by two pathogen-responsive MAPKs drives phytoalexin biosynthesis in Arabidopsis. Plant Cell 23, 1639–1653. doi: 10.1105/tpc.111.084996
Mizoi, J., Shinozaki, K., and Yamaguchi-Shinozaki, K. (2012). AP2/ERF family transcription factors in plant abiotic stress responses. Biochim. Biophys. Acta 1819, 86–96. doi: 10.1016/j.bbagrm.2011.08.004
Muthusamy, M., Uma, S., Backiyarani, S., Saraswathi, M. S., and Chandrasekar, A. (2016). Transcriptomic changes of drought-tolerant and sensitive banana cultivars exposed to drought stress. Front. Plant Sci. 7:1609. doi: 10.3389/fpls.2016.01609
Nakashima, K., Yamaguchi-Shinozaki, K., and Shinozaki, K. (2014). The transcriptional regulatory network in the drought response and its crosstalk in abiotic stress responses including drought, cold, and heat. Front. Plant Sci. 5:170. doi: 10.3389/fpls.2014.00170
Natale, M., Benso, A., Di Carlo, S., and Ficarra, E. (2014). FunMod: a Cytoscape plugin for identifying functional modules in undirected protein-protein networks. Genomics Proteomics Bioinformatics 12, 178–186. doi: 10.1016/j.gpb.2014.05.002
Nuruzzaman, M., Sharoni, A. M., and Kikuchi, S. (2013). Roles of NAC transcription factors in the regulation of biotic and abiotic stress responses in plants. Front. Microbiol. 4:248. doi: 10.3389/fmicb.2013.00248
Park, S. Y., Fung, P., Nishimura, N., Jensen, D. R., Fujii, H., Zhao, Y., et al. (2009). Abscisic acid inhibits type 2C protein phosphatases via the PYR/PYL family of START proteins. Science 324, 1068–1071. doi: 10.1126/science.1173041
Quan, W., Liu, X., Wang, H., and Chan, Z. (2015). Comparative physiological and transcriptional analyses of two contrasting drought tolerant alfalfa varieties. Front. Plant Sci. 6:1256. doi: 10.3389/fpls.2015.01256
Roychoudhury, A., Paul, S., and Basu, S. (2013). Cross-talk between abscisic acid-dependent and abscisic acid-independent pathways during abiotic stress. Plant Cell Rep. 32, 985–1006. doi: 10.1007/s00299-013-1414-5
Rushton, P. J., Somssich, I. E., Ringler, P., and Shen, Q. J. (2010). WRKY transcription factors. Trends Plant Sci. 15, 247–258. doi: 10.1016/j.tplants.2010.02.006
Sah, S. K., Reddy, K. R., and Li, J. (2016). Abscisic acid and abiotic stress tolerance in crop plants. Front. Plant Sci. 7:571. doi: 10.3389/fpls.2016.00571
Sakamoto, H., Maruyama, K., Sakuma, Y., Meshi, T., Iwabuchi, M., Shinozaki, K., et al. (2004). Arabidopsis Cys2/His2-type zinc-finger proteins function as transcription repressors under drought, cold, and high-salinity stress conditions. Plant Physiol. 136, 2734–2746. doi: 10.1104/pp.104.046599
Sakuma, Y., Liu, Q., Dubouzet, J. G., Abe, H., Shinozaki, K., and Yamaguchi-Shinozaki, K. (2002). DNA-binding specificity of the ERF/AP2 domain of Arabidopsis DREBs, transcription factors involved in dehydration- and cold-inducible gene expression. Biochem. Biophys. Res. Commun. 290, 998–1009. doi: 10.1006/bbrc.2001.6299
Saruhashi, M., Kumar Ghosh, T., Arai, K., Ishizaki, Y., Hagiwara, K., Komatsu, K., et al. (2015). Plant Raf-like kinase integrates abscisic acid and hyperosmotic stress signaling upstream of SNF1-related protein kinase2. Proc. Natl. Acad. Sci. U.S.A. 112, E6388–E6396. doi: 10.1073/pnas.1511238112
Schmitz, G., and Theres, K. (2005). Shoot and inflorescence branching. Curr. Opin. Plant Biol. 8, 506–511. doi: 10.1016/j.pbi.2005.07.010
Sharma, B., Joshi, D., Yadav, P. K., Gupta, A. K., and Bhatt, T. K. (2016). Role of ubiquitin-mediated degradation system in plant biology. Front. Plant Sci. 7:806. doi: 10.3389/fpls.2016.00806
Sharma, P., Sharma, N., and Deswal, R. (2005). The molecular biology of the low-temperature response in plants. Bioessays 27, 1048–1059. doi: 10.1002/bies.20307
Shou, H., Bordallo, P., and Wang, K. (2004). Expression of the Nicotiana protein kinase (NPK1) enhanced drought tolerance in transgenic maize. J. Exp. Bot. 55, 1013–1019. doi: 10.1093/jxb/erh129
Singh, A., Jha, S. K., Bagri, J., and Pandey, G. K. (2015). ABA inducible rice protein phosphatase 2C confers ABA insensitivity and abiotic stress tolerance in Arabidopsis. PLoS ONE 10:e0125168. doi: 10.1371/journal.pone.0125168
Solanke, A. U., and Sharma, A. K. (2008). Signal transduction during cold stress in plants. Physiol. Mol. Biol. Plants 14, 69–79.
Song, S. S., Qi, T. C., Huang, H., Ren, Q. C., Wu, D. W., Chang, C. Q., et al. (2011). The Jasmonate-ZIM domain proteins interact with the R2R3-MYB transcription factors MYB21 and MYB24 to affect jasmonate-regulated stamen development in Arabidopsis. Plant Cell 23, 1000–1013. doi: 10.1105/tpc.111.083089
Stark, C., Breitkreutz, B. J., Reguly, T., Boucher, L., Breitkreutz, A., and Tyers, M. (2006). BioGRID: a general repository for interaction datasets. Nucleic Acids Res. 34, D535–D539. doi: 10.1093/nar/gkj109
Stone, S. L. (2014). The role of ubiquitin and the 26S proteasome in plant abiotic stress signaling. Front. Plant Sci. 5:135. doi: 10.3389/fpls.2014.00135
Sun, X., Shantharaj, D., Kang, X., and Ni, M. (2010). Transcriptional and hormonal signaling control of Arabidopsis seed development. Curr. Opin. Plant Biol. 13, 611–620. doi: 10.1016/j.pbi.2010.08.009
Trapnell, C., Roberts, A., Goff, L., Pertea, G., Kim, D., Kelley, D. R., et al. (2012). Differential gene and transcript expression analysis of RNA-seq experiments with TopHat and Cufflinks. Nat. Protoc. 7, 562–578. doi: 10.1038/nprot.2012.016
Trapnell, C., Williams, B. A., Pertea, G., Mortazavi, A., Kwan, G., van Baren, M. J., et al. (2010). Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat. Biotechnol. 28, 511–515. doi: 10.1038/nbt.1621
Turyagyenda, L. F., Kizito, E. B., Ferguson, M., Baguma, Y., Agaba, M., Harvey, J. J., et al. (2013). Physiological and molecular characterization of drought responses and identification of candidate tolerance genes in cassava. AoB Plants 5:plt007. doi: 10.1093/aobpla/plt007
Utsumi, Y., Tanaka, M., Morosawa, T., Kurotani, A., Yoshida, T., Mochida, K., et al. (2012). Transcriptome analysis using a high-density oligomicroarray under drought stress in various genotypes of cassava: an important tropical crop. DNA Res. 19, 335–345. doi: 10.1093/dnares/dss016
Valenzuela, C. E., Acevedo-Acevedo, O., Miranda, G. S., Vergara-Barros, P., Holuigue, L., Figueroa, C. R., et al. (2016). Salt stress response triggers activation of the jasmonate signaling pathway leading to inhibition of cell elongation in Arabidopsis primary root. J. Exp. Bot. 67, 4209–4220. doi: 10.1093/jxb/erw202
Verma, V., Ravindran, P., and Kumar, P. P. (2016). Plant hormone-mediated regulation of stress responses. BMC Plant Biol. 16:86. doi: 10.1186/s12870-016-0771-y
Wagner, G. P., Kin, K., and Lynch, V. J. (2012). Measurement of mRNA abundance using RNA-seq data: RPKM measure is inconsistent among samples. Theory Biosci. 131, 281–285. doi: 10.1007/s12064-012-0162-3
Wang, H., Wang, H., Shao, H., and Tang, X. (2016). Recent advances in utilizing transcription factors to improve plant abiotic stress tolerance by transgenic technology. Front. Plant Sci. 7:67. doi: 10.3389/fpls.2016.00067
Wei, Y. X., Shi, H. T., Xia, Z. Q., Tie, W. W., Ding, Z. H., Yan, Y., et al. (2016). Genome-wide identification and expression analysis of the WRKY gene family in cassava. Front. Plant Sci. 7:25.
Xie, Y., Ye, S., Wang, Y., Xu, L., Zhu, X., Yang, J., et al. (2015). Transcriptome-based gene profiling provides novel insights into the characteristics of radish root response to Cr stress with next-generation sequencing. Front. Plant Sci. 6:202. doi: 10.3389/fpls.2015.00202
Xu, L., Menard, R., Berr, A., Fuchs, J., Cognat, V., Meyer, D., et al. (2009). The E2 ubiquitin-conjugating enzymes, AtUBC1 and AtUBC2, play redundant roles and are involved in activation of FLC expression and repression of flowering in Arabidopsis thaliana. Plant J. 57, 279–288. doi: 10.1111/j.1365-313X.2008.03684.x
Yang, W., Zhang, W., and Wang, X. (2017). Post-translational control of ABA signalling: the roles of protein phosphorylation and ubiquitination. Plant Biotechnol. J. 15, 4–14. doi: 10.1111/pbi.12652
Yanhui, C., Xiaoyuan, Y., Kun, H., Meihua, L., Jigang, L., Zhaofeng, G., et al. (2006). The MYB transcription factor superfamily of Arabidopsis: expression analysis and phylogenetic comparison with the rice MYB family. Plant Mol. Biol. 60, 107–124. doi: 10.1007/s11103-005-2910-y
Ye, Y., Ding, Y., Jiang, Q., Wang, F., Sun, J., and Zhu, C. (2016). The role of receptor-like protein kinases (RLKs) in abiotic stress response in plants. Plant Cell Rep. 36, 235–242. doi: 10.1007/s00299-016-2084-x
Yin, P., Fan, H., Hao, Q., Yuan, X., Wu, D., Pang, Y., et al. (2009). Structural insights into the mechanism of abscisic acid signaling by PYL proteins. Nat. Struct. Mol. Biol. 16, 1230–1236. doi: 10.1038/nsmb.1730
Young, M. D., Wakefield, M. J., Smyth, G. K., and Oshlack, A. (2010). Gene ontology analysis for RNA-seq: accounting for selection bias. Genome Biol. 11:R14. doi: 10.1186/gb-2010-11-2-r14
Zhang, Y., Yang, C., Li, Y., Zheng, N., Chen, H., Zhao, Q., et al. (2007). SDIR1 is a RING finger E3 ligase that positively regulates stress-responsive abscisic acid signaling in Arabidopsis. Plant Cell 19, 1912–1929. doi: 10.1105/tpc.106.048488
Zhao, P., Liu, P., Shao, J., Li, C., Wang, B., Guo, X., et al. (2015). Analysis of different strategies adapted by two cassava cultivars in response to drought stress: ensuring survival or continuing growth. J. Exp. Bot. 66, 1477–1488. doi: 10.1093/jxb/eru507
Zhao, P., Liu, P., Yuan, G., Jia, J., Li, X., Qi, D., et al. (2016). New insights on drought stress response by global investigation of gene expression changes in sheepgrass (Leymus chinensis). Front. Plant Sci. 7:954. doi: 10.3389/fpls.2016.00954
Zheng, C., Zhao, L., Wang, Y., Shen, J., Zhang, Y., Jia, S., et al. (2015). Integrated RNA-Seq and sRNA-Seq analysis identifies chilling and freezing responsive key molecular players and pathways in tea plant (Camellia sinensis). PLoS ONE 10:e0125031. doi: 10.1371/journal.pone.0125031
Zhou, G. A., Chang, R. Z., and Qiu, L. J. (2010). Overexpression of soybean ubiquitin-conjugating enzyme gene GmUBC2 confers enhanced drought and salt tolerance through modulating abiotic stress-responsive gene expression in Arabidopsis. Plant Mol. Biol. 72, 357–367. doi: 10.1007/s11103-009-9575-x
Zhu, J. K. (2002). Salt and drought stress signal transduction in plants. Annu. Rev. Plant Biol. 53, 247–273. doi: 10.1146/annurev.arplant.53.091401.143329
Zhu, X., Qi, L., Liu, X., Cai, S., Xu, H., Huang, R., et al. (2014). The wheat ethylene response factor transcription factor pathogen-induced ERF1 mediates host responses to both the necrotrophic pathogen Rhizoctonia cerealis and freezing stresses. Plant Physiol. 164, 1499–1514. doi: 10.1104/pp.113.229575
Keywords: cassava, cold stress, drought stress, RNA sequencing and transcriptome analysis, transcription factors, protein kinases, crosstalk
Citation: Li S, Yu X, Cheng Z, Yu X, Ruan M, Li W and Peng M (2017) Global Gene Expression Analysis Reveals Crosstalk between Response Mechanisms to Cold and Drought Stresses in Cassava Seedlings. Front. Plant Sci. 8:1259. doi: 10.3389/fpls.2017.01259
Received: 14 February 2017; Accepted: 04 July 2017;
Published: 18 July 2017.
Edited by:
Vicent Arbona, Jaume I University, SpainReviewed by:
Jorge E. Mayer, Ag RD&IP Consult P/L, AustraliaHuize Chen, Shaanxi Normal University, China
Copyright © 2017 Li, Yu, Cheng, Yu, Ruan, Li and Peng. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Ming Peng, cGVuZ21pbmdAaXRiYi5vcmcuY24=
†Present address: Xiang Yu, Department of Biology, University of Pennsylvania, Philadelphia, PA, United States
‡These authors have contributed equally to this work.
 Shuxia Li
Shuxia Li Xiang Yu
Xiang Yu Zhihao Cheng3
Zhihao Cheng3 Wenbin Li
Wenbin Li