Abstract
Arctium species are known for a variety of pharmacological effects due to their diverse volatile and non-volatile secondary metabolites. Representatives of Arctium species contain non-volatile compounds including lignans, fatty acids, acetylenic compounds, phytosterols, polysaccharides, caffeoylquinic acid derivatives, flavonoids, terpenes/terpenoids and volatile compounds such as hydrocarbons, aldehydes, methoxypyrazines, carboxylic and fatty acids, monoterpenes and sesquiterpenes. Arctium species also possess bioactive properties such as anti-cancer, anti-diabetic, anti-oxidant, hepatoprotective, gastroprotective, antibacterial, antiviral, antimicrobial, anti-allergic, and anti-inflammatory effects. This review aims to provide a complete overview of the chemistry and biological activities of the secondary metabolites found in therapeutically used Arctium species. Summary of pharmacopeias and monographs contents indicating the relevant phytochemicals and therapeutic effects are also discussed, along with possible safety considerations.
Introduction
Botanical and Ethnobotanical Aspects
The genus Arctium L. (Asteraceae/Compositae, tribe Cardueae, subtribe Carduinae), together with the related genera Cousinia Cass., Hypacanthium Juz. and Schmalhausenia C. Winkl, forms the so-called Arctium–Cousinia group (de Souza et al., 2004). The species of the Arctium genus, also known as ‘burdock,’ comprise biennial herbs occurring in waste places, streams and roadsides, less often in wood and forests, in temperate regions of Europe and Asia and sporadically in subtropical and tropical regions (European Scientific Cooperative on Phytotherapy, 2003). In North and South America, the genus is considered as naturalized, whereas in Africa it is quite rare. The name of the genus comes from the Greek ‘arcteion’ which means ‘bear,’ alluding to the plant habitus characterized by pronounced hairiness.
According to the Plant List1, this genus encompasses 18 recognized species among which five are considered as hybrid species due to the frequent outbreeding occurring between its allogamous representatives (Lopez-Vinyallonga et al., 2010).
Arctium species are represented by hemicryptophyte plants equipped with a stout, erect taproot and entire (sporadically as dentate), rough, unarmed, alternate, tomentose, and cordate leaves. The stem is usually stout, erect, grooved, branched, and reddish. Inflorescences are formed by solitary or corymbose ovoid-conical to spherical capitula equipped with involucres made up of bracts ending with hooked apices. Receptacles are composed of numerous, hard scales. Florets are only tubulose, hermaphrodite, purple or white. Pollination is allowed by insects, mostly belonging to Lepidoptera. Fruits are having oblong, rugose achenes equipped with a golden-yellow pappus (European Scientific Cooperative on Phytotherapy, 2003).
The Arctium genus is highly polymorphic due to variability occurring in hairiness of leaves and capitula, length of floral peduncles, and color of capitula and florets. As a consequence, a sharp distinction between its members cannot occasionally be defined. In the Euro-Mediterranean area, six main species are found: A. atlanticum (Pomel) H. Lindb., A. lappa L., A. minus (Hill) Bernh., A. nemorosum Lej., A. palladini (Marcow) R.E.Fr. & Soderb. and Arctium tomentosum Mill. (European Scientific Cooperative on Phytotherapy, 2003; Figure 1).
FIGURE 1

Plants of Arctium. (A)Arctium lappa; (B)Arctium minus; (C)Arctium tomentosum; (D)Arctium nemorosum.
Arctium lappa enjoys a longstanding use in the traditional medicine (mainly roots, and, to a lesser extent, leaves and seeds) due to the bioactive properties of its metabolites (Zhao et al., 2014). To the best of our knowledge, the most utilized species for therapeutic purposes, are on first place A. lappa, also known as ‘greater burdock,’ and, to a minor extent, A. minus (lesser burdock) and A. tomentosum (wooly burdock).
Arctium lappa is an herbaceous biennial plant up to 150 cm tall, with pubescent to subglabrous epigeal parts. Basal leaves are up to 50 cm in diameter, ovate, cordate and with hollow petioles. Stems are branched and end with corymbose capitula. Florets are as long as the involucral bracts. A. minus differs for the shape of inflorescence (solitary terminal capitula), dimensions of involucral bracts and capitula (smaller and shorter, respectively) and consistency of petiole (hollow). Furthermore, the bracts show a relatively hinted hairiness. A. tomentosum is characterized by petioles and peduncles covered with wooly tufts; petioles are solid. The involucral bracts are similar in dimensions to those of A. minus, but they show a dense covering of hairs. Florets are longer than bracts as in A. minus (European Scientific Cooperative on Phytotherapy, 2003). These three species are quite common in central Europe where often they undergo interspecific hybridization giving rise to questions about their integrity (European Scientific Cooperative on Phytotherapy, 2003).
Greater burdock (A. lappa) has been traditionally used in both Asian and European medicines as depurative, diuretic, carminative, anti-inflammatory, and anti-tubercular agent (Zhao et al., 2014). For therapeutic purposes, its different parts such as roots, fruits, and leaves are used. The latter have been used to treat ulcers and fester wounds (Jaric et al., 2007). They are also applied externally on the forehead to cure headache and fever, on the scalp to treat bruises and hair loss, mixed with oil and honey and applied on the chest to heal cough. In addition, under infusion, they are taken orally to treat enuresis in children (Pieroni et al., 2011). Fruits of burdock (Arctii fructus) are used to purify the blood (Lans and Turner, 2011) and to treat respiratory and infectious diseases (Bai et al., 2016). In addition, A. lappa roots, together with aerial parts of Rumex acetosella L., leaves of Ulmus rubra Muhl. and rhizomes of Rheum officinale Baill., are used to make ‘Essiac,’ a tea used by the Ojibwa tribe of Canada for the treatment of cancer (Leonard et al., 2006). In the veterinary medicine, the root is used to treat mastitis (Lans et al., 2007), whereas the whole plant is applied against endoparasites in poultry (Lans and Turner, 2011). Besides therapeutic uses, A. lappa is also appreciated as an edible plant. For the latter purpose, young leaves, and stalks are eaten raw or cooked (Pieroni et al., 2011).
Lesser burdock (A. minus) leaves are traditionally used externally to treat rheumatic pains, fever, sunstroke, wounds, general infections, skin and body inflammations, alopecia, and bladder diseases (de Souza et al., 2004; Erdemoglu et al., 2009; Neves et al., 2009). They are also disposed above the body of the patient, wetted with vinegar or milk, to stimulate sweating (Sezik et al., 2001). Roots and leaves, under infusion, are also used against snake and scorpion bites and to purify the blood (Mosaddegh et al., 2012). Basal leaves and stems are also eaten raw as a snack or stewed (Tardio et al., 2005). Due to their bitter taste, they are also used to stimulate the appetite and liver functions (Tardio et al., 2005).
Wooly burdock (A. tomentosum) leaves are used as vulnerary, to treat skin rash, ulcers, abscesses, mouth sores and against rheumatic pains, whereas root is applied against alopecia and to wash hairs (Sezik et al., 2004; Saric-Kundalic et al., 2010). Roots are also employed to make a tea used for digestive problems, ulcers, rheumatisms, to purify the blood and increase sweating and as diuretic (Saric-Kundalic et al., 2010).
Medicinal Uses of Arctium Species in Pharmacopeias and Monographs
Burdock species and in particular A. lappa are used in traditional medicine for different purposes. The main traditional use of the roots of A. lappa in Europe comprises treatment of dermatological disorders (Saric-Kundalic et al., 2010; Miglani and Manchanda, 2014) whereas in other Eastern and Asian countries A. lappa fruits and roots are used as an antidiabetic remedy (Tousch et al., 2014; Xu et al., 2014, 2015; Ahangarpour et al., 2017). In Traditional Chinese Medicine (TCM), apart from the antidiabetic activity, the roots of A. lappa are considered as a blood detoxifying agent (Qin et al., 2014). In the Japanese pharmacopeia, the fruit is included as a traditional herbal medicine with recent studies revealing the potential of its extracts in oncology (Ikeda et al., 2016). The leaves of A. lappa have also been reported as an anti-inflammatory agent to relieve gastrointestinal disorders in Brazilian traditional medicine (de Almeida et al., 2013).
According to international institutions that work in the validation of traditional herbal medicines, such as the European Medicines Agency (EMA) and the European Scientific Cooperative on Phytotherapy (ESCOP), A. lappa is recommended and approved for different indications. For example, the EMA monograph approves the use of roots of A. lappa, A. minus, and A. tomentosum as an adjuvant in minor urinary tract complaints, in temporary loss of appetite and for seborrheic skin conditions (European Medicines Agency, 2011). All these indications are based upon long-standing use. In 2016, ESCOP released a monograph where the roots of all the former three species are indicated to be internally and externally used for seborrheic skin, eczema, furuncles, acne, psoriasis and internally for minor urinary tract disorders (European Scientific Cooperative on Phytotherapy-The Scientific Foundation for Herbal Medicinal Products, 2016). For oral and internal administration, the herbal drug can be used as an infusion, extract, tincture or decoction but the fresh pulp of the roots or a decoction can also be directly applied to the skin. The later monograph reveals that A. lappa preparations should not be ingested during pregnancy, lactation, or in case of hypersensitivity to the Compositae and in patients with oedema due to impaired heart or kidney function. Although certain preclinical studies can be found in the literature, clinical trials are not available for these indications approved by ESCOP and EMA.
Phytochemistry
Non-volatile Compounds
Till date, more than two hundred non-volatile compounds have been isolated from Arctium genus. These chemical compounds include lignans, terpenoids, sterols, flavonoids, phenolics, lactones, polyacetylenes, quinic acids, and sugars (polysaccharides). In particular, lignans are the most characteristic components in the Arctium genus. The details of chemical compounds, their occurrence in different plant parts, and the analytical methods used for their quali-quantitative determinations are briefly summarized in Table 1 whereas their description is provided in this section. The chemical structures of some compounds from Arctium species are shown in Figure 2.
Table 1
| No | Compound name | Formula | Species | Plant origin/part | Analytical method | References |
|---|---|---|---|---|---|---|
| Lignans | ||||||
| 1 | Diarctigenin | C42H46O12 | A. lappa | Fruits, roots, seeds | IR/NMR/MS/TLC | Han et al., 1994; Park et al., 2007; Qin et al., 2014 |
| 2 | Arctiin | C27H34O11 | A. lappa, A. tomentosum | Leaves, fruits, roots, seeds | UV/IR/MS/NMR/HPLC/LCMS/ MALDI-QIT-TOF MS | Wang and Yang, 1993; Ting-Guo et al., 2001; Yu et al., 2003; Ming et al., 2004; Liu et al., 2005, 2012, 2015; Wang et al., 2005; Matsumoto et al., 2006; Boldizsar et al., 2010; Ferracane et al., 2010; Zhou et al., 2011; Qin et al., 2014; Su et al., 2015; Lou et al., 2016; Al-Shammaa et al., 2017 |
| 3 | Arctigenin | C12H24O7 | A. lappa, A. tomentosum | Leaves, fruits, seeds, roots | UV/MS/NMR/HPLC/LCMS/ MALDI-QIT-TOF MS/ HRESI-MS | Umehara et al., 1993; Wang and Yang, 1993; Liu et al., 2005, 2012, 2015; Matsumoto et al., 2006; Gao et al., 2008; Boldizsar et al., 2010; Ferracane et al., 2010; Predes et al., 2011; Zhou et al., 2011; Qin et al., 2014; Su et al., 2015; Al-Shammaa et al., 2017 |
| 4 | Arctigenin-4-O-β-D-gentiobioside | C18 H32O16 | A. lappa | Fruits | NMR/UV/IR/ORD/HRESIMS | Yang et al., 2015 |
| 5 | Arctigenin-4-O-α-D-galactopyranosyl-(1→6)-O-β-D-glucopyranoside | C18 H32O16 | A. lappa | Fruits | NMR/UV/IR/ORD/HRESIMS | Yang et al., 2015 |
| 6 | Arctigenin-4-O-β-D-apiofuranosyl-(1→6)-O-β-D-glucopyranoside | C32 H42 O15 | A. lappa | Fruits | NMR/UV/IR/ORD/HRESIMS | Yang et al., 2015 |
| 7 | 3-benzyl-6-(1-hydroxyethyl)-2,5-piperazinedione | C13 H16 N2O3 | A. lappa | Fruits | IR/HR-ESI-MS/NMR/CD | Yang et al., 2012 |
| 8 | 3-benzyl-2,5- piperazinedione | C13H16N2O2 | A. lappa | Fruits | IR/HR-ESI-MS/NMR/CD | Yang et al., 2012 |
| 9 | 5′-propanediolmatairesinoside | C29 H38O13 | A. lappa | Fruits | NMR/UV/IR/ORD/HRESIMS | Yang et al., 2015 |
| 10 | (7′R,8R,8’R)-rafanotrachelogenin-4-O-β-D-glucopyranoside | C27 H34 O12 | A. lappa | Fruits | NMR/UV/IR/ORD/HRESIMS | Yang et al., 2015 |
| 11 | (7′S,8R,8′R)-rafanotrachelogenin-4-O-β-D-glucopyranoside | C27 H34 O12 | A. lappa | Fruits | NMR/UV/IR/ORD/HRESIMS | Yang et al., 2015 |
| 12 | (7S,8S,8’R)-4,7-dihydroxy-3,3’,4-trimethoxyl-9-oxo benzylbutyrolactone lignan-4-O-β-D-glucopyranoside | C27 H34O12 | A. lappa | Fruits | NMR/UV/IR/ORD/HRESIMS | Yang et al., 2015 |
| 13 | (7S,8S,8’R)-4,7- dihydroxy-3,3′,4′-trimethoxyl-9-oxo dibenzylbutyrolactone lignin | C21H24O7 | A. lappa | Fruits | NMR/UV/IR/ORD/HRESIMS | Yang et al., 2015 |
| 14 | (7R,8S,8′R)-4,7,4′-trihydroxy-3,3′- dimethoxyl-9-oxo dibenzylbutyrolactone lignan-4-O-β-D-glucopyranoside | C26 H32O12 | A. lappa | Fruits | NMR/UV/IR/ORD/HRESIMS | Yang et al., 2015 |
| 15 | 7,8-didehydroarctigenin | C21H22O5 | A. lappa | Fruits | HRFAB/EIMS/NMR | Matsumoto et al., 2006 |
| 16 | Arctiidilactone | C20H20O8 | A. lappa | Fruits | NMR/UV/IR/ORD/HRESIMS | Yang et al., 2015 |
| 17 | Arctiiapolignan A | C20H28O10 | A. lappa | Fruits | NMR/UV/IR/ORD/HRESIMS | Yang et al., 2015 |
| 18 | Arctiisesquineolignan A | C42H52O19 | A. lappa | Fruits | NMR/UV/IR/ORD/HRESIMS | Yang et al., 2015 |
| 19 | Arctiisesquineolignan B | C36H46O16 | A. lappa | Fruits | UV/IR/HRESIMS/NMR | He etal., 2016 |
| 20 | Arctiiphenolglycoside A | C19H28O13 | A. lappa | Fruits | UV/IR/HRESIMS/NMR | He etal., 2016 |
| 21 | Arctignan A | C30H34O10 | A. lappa | Seeds | UV/MS/NMR/HPLC | Umehara et al., 1993 |
| 22 | Arctignan B | C30H34O10 | A. lappa | Seeds | UV/MS/NMR/HPLC | Umehara et al., 1993 |
| 23 | Arctignan C | C30H32O10 | A. lappa | Seeds | UV/MS/NMR/HPLC | Umehara et al., 1993 |
| 24 | Arctignan D | C30H34O10 | A. lappa | Seeds | UV/MS/NMR/HPLC/LCMS/ MALDI-QIT-TOF MS | Umehara et al., 1993; Liu et al., 2012 |
| 25 | Arctignan E | C40H44O13 | A. lappa | Seeds | UV/IR/MS/NMR/HPLC | Umehara et al., 1993; Ming et al., 2004; Ferracane et al., 2010; Qin et al., 2014 |
| 26 | Lappaol A | C30H32O9 | A. lappa, A. tomentosum | Seeds/fruits | TLC/UV/IR/MS/NMR/HPLC | Ichihara et al., 1976; Ting-Guo et al., 2001; Ming et al., 2004; Ferracane et al., 2010; Liu et al., 2012; Qin et al., 2014; Su et al., 2015 |
| 27 | Lappaol B | C31H34O9 | A. lappa | Seeds/fruits | NMR/MS/TLC/HPLC | Ichihara et al., 1976; Qin et al., 2014 |
| 28 | Isolappaol C | C30H34O10 | A. lappa, A. tomentosum | Seeds/fruits | NMR/MS/TLC | Park et al., 2007; Qin et al., 2014; Su et al., 2015 |
| 29 | Lappaol C | C30H34O10 | A. lappa | Seeds | TLC/UV/IR/MS/NMR | Ichihara et al., 1977; Ting-Guo et al., 2001; Ming et al., 2004; Park et al., 2007; Ferracane et al., 2010; Liu et al., 2012; Su et al., 2015 |
| 30 | Lappaol D | C31H36O10 | A. lappa | Seeds | NMR/MS/TLC | Ichihara et al., 1977; Park et al., 2007 |
| 31 | Lappaol E | C30H34O10 | A. lappa | Seeds | NMR/MS/TLC | Ichihara et al., 1977; Park et al., 2007 |
| 32 | Lappaol F | C42H46O12 | A. lappa, A. tomentosum | Fruits, seeds | TLC/UV/IR/MS/NMR/HPLC | Ting-Guo et al., 2001; Ming et al., 2004; Park et al., 2007; Ferracane et al., 2010; Qin et al., 2014 |
| 33 | Lappaol H | C40H46O14 | A. lappa | Seeds/fruits | UV/MS/NMR/HPLC/LCMS/ MALDI-QIT-TOF MS | Liu et al., 2012; Qin et al., 2014 |
| 34 | Neoarctin A | C42H46O12 | A. lappa | Seeds | UV, IR, 1H-NMR, 13C-NMR, DEPT, 2D-NMR and MS | Wang and Yang, 1995; Yong et al., 2007 |
| 35 | Neoarctin B | C42H46O12 | A. lappa | Seeds | UV, IR, 1H-NMR, 13C-NMR, DEPT, 2D-NMR and MS | Wang and Yang, 1993 |
| 36 | Matairesinoside | C26H32O11 | A. lappa | Fruits | UV/IR/HPLC | Boldizsar et al., 2010 |
| 37 | Matairesinol | C20H22O6 | A. lappa | Seeds/fruits | UV/MS/NMR/HPLC/LCMS/ MALDI-QIT-TOF MS | Wang and Yang, 1993; Boldizsar et al., 2010; Ferracane et al., 2010; Liu et al., 2012; Qin et al., 2014; Su et al., 2015 |
| 38 | Matairesinol-4,4′-di-O-β-D-glucopyranoside | C27 H34 O12 | A. lappa | Fruits | NMR/UV/IR/ORD/HRESIMS | Yang et al., 2015 |
| 39 | Pinoresinol | C20H22O6 | A. lappa | Fruits | UV/IR/HPLC | Boldizsar et al., 2010 |
| 40 | Phylligenin | C21H24O6 | A. lappa | Fruits | UV/IR/HPLC | Boldizsar et al., 2010 |
| 41 | Styraxlignolide E | C26H32O11 | A. lappa | Fruits | NMR/UV/IR/ORD/HRESIMS | Yang et al., 2015 |
| 42 | Styraxlignolide D | C26H32O11 | A. lappa | Fruits | NMR/UV/IR/ORD/HRESIMS | Yang et al., 2015 |
| 43 | Syringaresinol | C22H26O8 | A. lappa | Roots | UV/IR/ESIMS/NMR | Han et al., 2013 |
| 44 | (7S, 8R)-4,7,9,9′-tetrahydroxy-3,3′-dimethoxyl-7′-oxo-8-4′-oxyneolignan-4-O-β-D-glucopyranoside | C26 H34 O13 | A. lappa | Roots | IR/HR-ESI-MS/NMR/CD | Yang et al., 2012 |
| 45 | (7′S, 8′R, 8S)-4,4′,9′-trihydroxy-3,3′-dimethoxy-7′,9-epoxylignan-7-oxo-4-O-β-D-glucopyranosyl-4′-O-β-D-glucopyranoside | C32 H42 O17 | A. lappa | Roots | IR/HR-ESI-MS/NMR/CD | Yang et al., 2012 |
| 46 | (7S, 8R)-4,7,9,9′-tetrahydroxy-3,3′dimethoxy-8-O-4′-neolignan-9′-O-β-D-apiofuranosyl-(1 → 6)-O-β-D-glucopyranoside | C31H44O16 | A. lappa | Fruits | IR/HR-ESI-MS/NMR/CD | Huang K. et al., 2015; Huang X.Y. et al., 2015 |
| 47 | (8R)-4,9,9′-trihydroxy-3,3′-dimethoxy-7-oxo-8-O-4′-neolignan-4-O-β-D-glucopyranoside | C26 H34 O12 | A. lappa | Fruits | IR/HR-ESI-MS/NMR/CD | Huang K. et al., 2015; Huang X.Y. et al., 2015 |
| 48 | (7R, 8S)-dihydrodehydrodiconiferyl alcohol-7′-oxo-4-O-β-D-glucopyranoside | C26 H32 O12 | A. lappa | Fruits | IR/HR-ESI-MS/NMR/CD | Huang K. et al., 2015; Huang X.Y. et al., 2015 |
| 49 | (7′S, 8′R, 8S)-4,4′,9′-trihydroxy-3,3′-dimethoxy-7′,9-epoxylignan-7-oxo-4-O-β-D-glucopyranoside | C26 H32 O12 | A. lappa | Fruits | IR/HR-ESI-MS/NMR/CD | Huang K. et al., 2015; Huang X.Y. et al., 2015 |
| 50 | Trachelogenin | C21H24O7 | A. lappa | Fruits | – | Ichikawa et al., 1986 |
| Terpenes/Terpenoids | ||||||
| 51 | β-eudesmol | C15H26O | A. lappa | Fruits | – | Yayli et al., 2005 |
| 52 | Ursolic acid | C30H48O3 | A. lappa | Root | UV/IR/ESIMS/NMR | Han et al., 2013 |
| 53 | Oleanolic acid | C30H48O3 | A. lappa | Roots | UV/IR/ESIMS/NMR | Han et al., 2013 |
| 54 | Arctiopicrin | C19H26O6 | A. lappa, A. minus | Leaves | TLC/NMR | Savina et al., 2006 |
| 55 | Onopordopicrin | C19H24O6 | A. lappa, A. nemorosum | Leaves/aerial parts | TLC/HPLC/NMR/HR-ESI-TOF-MS | Barbosa et al., 1993; Savina et al., 2006; Machado et al., 2012; Zimmermann et al., 2012 |
| 56 | Dehydrovomifoliol | C13H18O3 | A. lappa | Leaves | NMR/HR-ESI-TOF-MS | Machado et al., 2012 |
| 57 | Loliolide | C11H16O3 | A. lappa | Leaves | NMR/HR-ESI-TOF-MS | Machado et al., 2012 |
| 58 | Dehydromelitensin-8-(4′-hydroxymethacrylate) | C15 H24 O6 | A. lappa | Leaves | NMR/HR-ESI-TOF-MS | Machado et al., 2012 |
| 59 | Dehydromelitensin | C15H20O4 | A. lappa | Leaves | NMR/HR-ESI-TOF-MS | Machado et al., 2012 |
| 60 | Melitensin | C15H22O4 | A. lappa | Leaves | NMR/HR-ESI-TOF-MS | Machado et al., 2012 |
| 61 | 3α-acetoxyhop-22(29)-ene | C30H49O2 | A. lappa | Leaves | NMR, IR and MS | Jeelani and Khuroo, 2012 |
| 62 | 3α-hydroxylanosta-5,15-diene | C30 H50O | A. lappa | Leaves | NMR, IR and MS | Jeelani and Khuroo, 2012 |
| Flavonoids | ||||||
| 63 | Baicalin | C21H18O11 | A. lappa | Uchiyama et al., 2005 | ||
| 64 | Luteolin | C25H24O12 | A. lappa | Leaves/roots | UPLC/LC/MS/MS | Ferracane et al., 2010; Lou et al., 2010a; Tang et al., 2014 |
| 65 | Rutin | C27H30O16 | A. lappa, A. minus | Leaves | TLC/UPLC/LC/MS/MS | Saleh and Bohm, 1971; Lou et al., 2010a,b |
| 66 | Quercitrin | C21H20O11 | A. lappa | Leaves/roots | UPLC/LC/MS/MS | Lou et al., 2010a |
| 67 | Quercetin | C15H10O7 | A. lappa | Leaves/roots | UPLC/LC/MS/MS/HRESI-MS | Lou et al., 2010a; Predes et al., 2011; Tang et al., 2014 |
| 68 | Quercetin 3-O-glucuronide | C21H18O13 | A. lappa | Roots | HPTLC/LC/ESI–MS/MS | Rajasekharan et al., 2015 |
| 69 | Quercetin 3-vicianoside | C26H28O16 | A. lappa | Roots | HPTLC/LC/ESI–MS/MS | Rajasekharan et al., 2015 |
| 70 | Quercetin rhamnoside | C21H20O11 | A. lappa | roots | HPLC/LC/MS/MS | Ferracane et al., 2010 |
| 71 | Quercimeritrin | C21H20O12 | A. minus | Leaves | TLC | Saleh and Bohm, 1971 |
| 72 | Isoquercetin | C21H20O12 | A. minus | Leaves | TLC | Saleh and Bohm, 1971 |
| 73 | Astragalin | C21H20O11 | A. minus | Leaves | TLC | Saleh and Bohm, 1971 |
| 74 | Kaempferol-3-o-rhamnoglucoside | C27 H30 O15 | A. minus | Leaves | TLC | Saleh and Bohm, 1971 |
| 75 | Biachanin A | C16H12O5 | A. lappa | Roots | – | Tamayo et al., 2000; Eberding et al., 2007 |
| 76 | Genestein | C15H10O5 | A. lappa | Roots | – | Tamayo et al., 2000; Eberding et al., 2007 |
| 77 | Nobiletin | C21H22O8 | A. lappa | Roots | – | Tamayo et al., 2000; Eberding et al., 2007 |
| 78 | Tangeretin | C20H20O7 | A. lappa | Roots | – | Tamayo et al., 2000 |
| Sterols | ||||||
| 79 | β-sitosterol | C29H50O | A. lappa, A. tomentosum | Seeds/roots/fruits | UV/IR/MS/NMR/HPLC | Ting-Guo et al., 2001; Ming et al., 2004; Han et al., 2013 |
| 80 | Sitosterol-beta-D-glucopyranoside | C35 H60 O6 | A. lappa | Roots | IR/NMR/EI-MS | Miyazawa et al., 2005 |
| 81 | Daucosterol | C35H60O6 | A. lappa, A. tomentosum | Seeds/fruits | UV, IR, 1H-NMR, 13C-NMR, DEPT, 2D-NMR and MS/HPLC | Wang and Yang, 1993; Ting-Guo et al., 2001; Han et al., 2013 |
| Fatty acids | ||||||
| 82 | Docosanoic acid | C22H44O2 | A. tomentosum | Seeds | GCMS | Zong et al., 2013 |
| 83 | Eicosanoic acid | C20H40O2 | A. tomentosum | Seeds | GCMS | Zong et al., 2013 |
| 84 | cis-13-eicosenoic acid | C20H38O2 | A. tomentosum | Seeds | GCMS | Zong et al., 2013 |
| 85 | Methyl palmitate | C17H34O2 | A. lappa | – | IR/NMR/EI-MS | Miyazawa et al., 2005 |
| 86 | Methyl linoleate | C19H34O2 | A. lappa | – | IR/NMR/EI-MS | Miyazawa et al., 2005 |
| 87 | Methyl linolenate | C19H32O2 | A. lappa | Roots | IR/NMR/EI-MS/GCMS | Miyazawa et al., 2005; Kuo et al., 2012 |
| 88 | Methyl stearate | C19H38O2 | A. lappa | – | IR/NMR/EI-MS | Miyazawa et al., 2005 |
| 89 | Methyl oleate | C19H36O2 | A. lappa | Roots | IR/NMR/EI-MS/GCMS | Miyazawa et al., 2005; Kuo et al., 2012 |
| 90 | Hexadecanoic acid | C16H32O2 | A. lappa, A. tomentosum | Fruits/seeds | UV/TLC/IR/NMR/EIMS/GCMS | Miyazawa et al., 2005; Boldizsar et al., 2010; Zong et al., 2013 |
| 91 | 9-hexadecenoic acid | C16H30O2 | A. tomentosum | Seeds | GCMS | Zong et al., 2013 |
| 92 | Linoleic acid | C18H32O2 | A. lappa | Roots | IR/NMR/EI-MS/GCMS | Miyazawa et al., 2005; Boldizsar et al., 2010; Kuo et al., 2012 |
| 93 | Linolenic acid | C18H30O2 | A. lappa | Fruits | IR/NMR/EI-MS; GCMS | Miyazawa et al., 2005 |
| 94 | Stearic acid | C17H35CO2H | A. lappa | Fruits | IR/NMR/EI-MS | Miyazawa et al., 2005 |
| 95 | 9,12-octadecadienoic acid | C18H32O2 | A. tomentosum | Seeds | GCMS | Zong et al., 2013 |
| 96 | Oleic acid | C18H34O2 | A. lappa | Fruits | UV/IR/HPLC/NMR/EI-MS | Miyazawa et al., 2005; Boldizsar et al., 2010 |
| 97 | Oxiraneoctanoic acid | C19H36O3 | A. tomentosum | Seeds | GCMS | Zong et al., 2013 |
| 98 | Tetracosanoic acid | C24H48O2 | A. tomentosum | Seeds | GCMS | Zong et al., 2013 |
| Acetylenic compounds | ||||||
| 99 | Arctinone-a | C13H10O2S2 | A. lappa | Roots | UV/TLC/IR/NMR/MS | Washino et al., 1986 |
| 100 | Arctinone-b | C13H10OS2 | A. lappa | Roots | UV/TLC/IR/NMR/MS | Washino et al., 1986 |
| 101 | Arctinol-a | C13H12O2S2 | A. lappa | Roots | UV/TLC/IR/NMR/MS | Washino et al., 1986 |
| 102 | Arctinol-b | C13H12O2S2 | A. lappa | Roots | UV/TLC/IR/NMR/MS | Washino et al., 1986 |
| 103 | Arctinal | C12H8OS2 | A. lappa | Roots | UV/TLC/IR/NMR/MS | Washino et al., 1986 |
| 104 | Arctic acid-b | C13H8O3S2 | A. lappa | Roots | UV/TLC/IR/NMR/MS | Washino et al., 1986 |
| 105 | Arctic acid-c | C13H10O3S2 | A. lappa | Roots | UV/TLC/IR/NMR/MS | Washino et al., 1986 |
| 106 | Methyl arctate-b | C14H10O3S2 | A. lappa | Roots | UV/TLC/IR/NMR/MS | Washino et al., 1986 |
| 107 | Arctinone-a acetate | C15H10O3S2 | A. lappa | Roots | UV/TLC/IR/NMR/MS | Washino et al., 1986 |
| 108 | Dehydrodihydrocostus lactone | C15H21O2 | A. lappa | Roots | UV/TLC/IR/NMR/MS | Washino et al., 1986, 1987 |
| 109 | Dehydrocostus lactone | C15H19O2 | A. lappa | Roots | UV/TLC/IR/NMR/MS | Washino et al., 1986, 1987 |
| 110 | Lappaphen-a | C27H26O4S | A. lappa | Roots | UV/TLC/IR/NMR/MS | Washino et al., 1986, 1987 |
| 111 | Lappaphen-b | C27H26O4S | A. lappa | Roots | UV/TLC/IR/NMR/MS | Washino et al., 1986, 1987 |
| Carboxylic acids/Quinic acids and derivatives | ||||||
| 112 | Caffeic acid | C9H8O4 | A. lappa | Seeds/leaves/roots | TLC/HPLC/UPLC/LC/MS/ HRESI-MS | Chen et al., 2004; Lin and Harnly, 2008; Lou et al., 2010a,b; Ferracane et al., 2010; Predes et al., 2011; Tang et al., 2014; Lou et al., 2016; Al-Shammaa et al., 2017 |
| 113 | Caffeic acid 4-O-glucoside | C15H18O9 | A. lappa | Roots | LC-DAD-ESI/MS | Lin and Harnly, 2008; Lou et al., 2016 |
| 114 | Chlorogenic acid | C16H18O9 | A. lappa | Seeds/leaves/roots | TLC/HPTLC/HPLC/UPLC/LC/MS/MALDI-QIT-TOF MS/ HRESI-MS | Wang et al., 2001; Chen et al., 2004; Lin and Harnly, 2008; Ferracane et al., 2010; Lou et al., 2010a,b; Predes et al., 2011; Liu et al., 2012; Haghi et al., 2013; Qin et al., 2014; Liu et al., 2015; Lou et al., 2016; Al-Shammaa et al., 2017 |
| 115 | p-coumaric acid | C9H8O3 | A. lappa | Seeds/leaves/roots | UPLC/EIMS | Lou et al., 2010a,b; Tang et al., 2014 |
| 116 | Coumaroylquinic acid | C16H18O8 | A. lappa | Roots | HPTLC/LC/ESI–MS/MS | Rajasekharan et al., 2015 |
| 117 | Benzoic Acid | C7H6O2 | A. lappa | Leaves | UPLC/EIMS | Lou et al., 2010a,b |
| 118 | Cynarin | C25H24O12 | A. lappa | Seeds/leaves/roots | UPLC/LC/MS | Ferracane et al., 2010; Lou et al., 2010a, 2016; Tang et al., 2014 |
| 119 | Caffeoyl-hexose-hydroxyphenol | C21 H21O10 | A. lappa | Roots | HPTLC/LC/ESI–MS/MS | Rajasekharan et al., 2015 |
| 120 | 1-O-caffeoylquinic acid | C16H18O9 | A. lappa | Roots | GCMS/LC-DAD-ESI/MS | Lin and Harnly, 2008; Tousch et al., 2014 |
| 121 | 3-O-caffeoylquinic acid | C16H18O9 | A. lappa | Roots | GCMS/LC-DAD-ESI/MS | Lin and Harnly, 2008; Tousch et al., 2014 |
| 122 | 4-O-caffeoylquinic acid | C16H18O9 | A. lappa | Roots | GCMS/LC-DAD-ESI/MS | Lin and Harnly, 2008; Tousch et al., 2014 |
| 123 | 5-O-caffeoylquinic acid | C16H18O9 | A. lappa | Roots | GCMS/LC-DAD-ESI/MS | Lin and Harnly, 2008; Jaiswal and Kuhnert, 2011; Tousch et al., 2014 |
| 124 | 1-O-,5-O-dicaffeoylquinic acid | C25H24O12 | A. lappa | Roots | HPLC/NMR/MS | Maruta et al., 1995; Wang et al., 2001; Han et al., 2013; Rajasekharan et al., 2015 |
| 125 | 1-O-, 5-O-dicaffeoyl-3-O-succinylquinaiccid | C35 H40 015 | A. lappa | Roots | NMR/EI-MS | Maruta et al., 1995 |
| 126 | 1-O,-5-O-dicaffeoyl-4-O-succinylquinic acid | C29H35015 | A. lappa | Roots | NMR/MS | Maruta et al., 1995 |
| 127 | 1-O-,5-O-dicaffeoyl-3-O- | C33H39018 | A. lappa | Roots | NMR/MS | Maruta et al., 1995 |
| 128 | 4-O-disuccinylquaicniidc and 1-O-,3-0-,5-O-tricaffeoyl-4-O-succinylquinic acid | C38H41018 | A. lappa | Roots | NMR/MS | Maruta et al., 1995 |
| 129 | 1,3-di-O-caffeoylquinic acid | C25H24O12 | A. lappa | Seeds/roots | LCMS/ MALDI-QIT-TOF MS | Lin and Harnly, 2008; Liu et al., 2012 |
| 130 | 1,5-di-O-caffeoylquinic acid | C25H24O12 | A. lappa | Leaves/Seeds/roots | UPLC/HPLC/PDA/LCMS/ MALDI-QIT-TOF MS | Maruta et al., 1995; Lin and Harnly, 2008; Liu et al., 2012; Haghi et al., 2013; Tousch et al., 2014 |
| 131 | 1,5-di-O-caffeoyl-4-O-maloylquinic acid | C29H27O16 | A. lappa | Roots | LCMS/ MALDI-QIT-TOF MS | Jaiswal and Kuhnert, 2011; Liu et al., 2012; Tousch et al., 2014 |
| 132 | 1,5-di-O-caffeoyl-3-O-maloylquinic acid | C25H27O16 | A. lappa | Roots | LCMS/ MALDI-QIT-TOF MS | Jaiswal and Kuhnert, 2011; Liu et al., 2012; Tousch et al., 2014 |
| 133 | 1,5-di-O-caffeoyl-3-O-succinoylquinic acid | C29H27O15 | A. lappa | Roots | LCMS/ MALDI-QIT-TOF MS | Maruta et al., 1995; Jaiswal and Kuhnert, 2011; Liu et al., 2012; Tousch et al., 2014 |
| 134 | 1,5-di-O-caffeoyl-3,4-di-O-succinoylquinic acid | C33H31O18 | A. lappa | Roots | LCMS/ MALDI-QIT-TOF MS | Liu et al., 2012; Tousch et al., 2014 |
| 135 | 1,3,5-tri-O-caffeoyl-4-O-succinoylquinic acid | C38H33O18 | A. lappa | Roots | GCMS/LCMS/ MALDI-QIT-TOF MS | Jaiswal and Kuhnert, 2011; Liu et al., 2012; Tousch et al., 2014 |
| 136 | 1,3,5-tri-O-caffeoylquinic acid | C34H29O15 | A. lappa | Roots | GCMS/LCMS/ MALDI-QIT-TOF MS | Liu et al., 2012; Tousch et al., 2014 |
| 137 | 1,5-di-O-caffeoyl-3-O-succinoyl-4-O-maloyquinic acid | – | A. lappa | Roots | LCMS/ MALDI-QIT-TOF MS | Liu et al., 2012 |
| 138 | 5-sinapoylquinic acid | C18H22O10 | A. lappa | Roots | LC-DAD-ESI/MS | Lin and Harnly, 2008 |
| 139 | 3-sinapoyl-5-caffeoylquinic acid | C27H28O13 | A. lappa | Roots | LC-DAD-ESI/MS | Lin and Harnly, 2008 |
| 140 | 3-sinapoyl-5-caffeoyl-1-methoxyoxaloylquinic acid | – | A. lappa | Roots | LC-DAD-ESI/MS | Lin and Harnly, 2008 |
| 141 | 4-sinapoyl-5-caffeoyl-1-methoxyoxaloylquinic acid | – | A. lappa | Roots | LC-DAD-ESI/MS | Lin and Harnly, 2008 |
| 142 | 3,4-dicaffeoylquinic acid | C25H24O12 | A. lappa | Roots/seeds | LC-DAD-ESI/MS | Lin and Harnly, 2008 |
| 143 | 1,4-di-O-caffeoylquinic acid | C25H23O12 | A. lappa | Roots | GCMS/LC-DAD-ESI/MS | Lin and Harnly, 2008; Jaiswal and Kuhnert, 2011; Tousch et al., 2014 |
| 144 | 3,5-di-O-caffeoylquinic acid | C25H24O12 | A. lappa | Roots | GCMS/LC-DAD-ESI/MS | Lin and Harnly, 2008; Jaiswal and Kuhnert, 2011; Tousch et al., 2014 |
| 145 | 4,5-dicaffeoylquinic acid | C25H24O12 | A. lappa | Roots/seeds | LC-DAD-ESI/MS | Lin and Harnly, 2008 |
| 146 | 3,5-dicaffeoyl-1-methoxyoxaloylquinic acid | – | A. lappa | Roots | LC-DAD-ESI/MS | Lin and Harnly, 2008 |
| 147 | 3-feruloyl-5-caffeoylquinic acid | – | A. lappa | Roots | LC-DAD-ESI/MS | Lin and Harnly, 2008 |
| 148 | 4,5-dicaffeoyl-1-methoxyoxaloylquinic acid | – | A. lappa | Roots | LC-DAD-ESI/MS | Lin and Harnly, 2008 |
| 149 | 3-sinapoyl-5-caffeoyl-4-methoxyoxaloylquinic acid | – | A. lappa | Roots | LC-DAD-ESI/MS | Lin and Harnly, 2008 |
| 150 | 1,4,5-tricaffeoylquinic acid | C34H30O15 | A. lappa | Roots | LC-DAD-ESI/MS | Lin and Harnly, 2008 |
| 151 | 3,4,5-tricaffeoylquinic acid | – | A. lappa | Roots | LC-DAD-ESI/MS | Lin and Harnly, 2008 |
| 152 | 1,4,5-tricaffeoyl-3-methoxyoxaloylquinic acid | – | A. lappa | Roots | LC-DAD-ESI/MS | Lin and Harnly, 2008 |
| 153 | 3-succinoyl-4,5-dicaffeoyl | – | A. lappa | Roots | LCMS | Jaiswal and Kuhnert, 2011 |
| 154 | 1,5-dicaffeoyl-3-succinoylquinic acid | – | A. lappa | Roots | HPLC/LCMS | Wang et al., 2001; Jaiswal and Kuhnert, 2011 |
| 155 | 1,5-di-O-caffeoyl-4-O-succinoylquinic acid | C29H27O15 | A. lappa | Roots | GCMS/LCMS | Maruta et al., 1995; Jaiswal and Kuhnert, 2011; Liu et al., 2012; Tousch et al., 2014 |
| 156 | 3,4-dicaffeoyl-5-succinoylquinic acid | C29H28O15 | A. lappa | Roots | LCMS | Jaiswal and Kuhnert, 2011 |
| 157 | 1,3-dicaffeoyl-5-fumaroylquinic acid | – | A. lappa | Roots | LCMS | Jaiswal and Kuhnert, 2011 |
| 158 | 1,5-dicaffeoyl-4-fumaroylquinic acid | – | A. lappa | Roots | LCMS | Jaiswal and Kuhnert, 2011 |
| 159 | 1,5-dicaffeoyl-3-maloylquinic acid | – | A. lappa | Roots | LCMS | Jaiswal and Kuhnert, 2011 |
| 160 | 1,4-di-O-caffeoyl-3-O-maloylquinic Acid | C29H27O16 | A. lappa | Roots | LCMS | Jaiswal and Kuhnert, 2011 |
| 161 | 1,3-di-O-caffeoyl-4,5-di-O-maloylquinic | C33 H31 O20 | A. lappa | Roots | GCMS/LCMS | Jaiswal and Kuhnert, 2011; Tousch et al., 2014 |
| 162 | 1,5-dicaffeoyl-4-maloylquinic acid | – | A. lappa | Roots | LCMS | Jaiswal and Kuhnert, 2011 |
| 163 | 1,4-di-O-maloyl-3,5-di-O-caffeoylquinic acid | C31H33O20 | A. lappa | Roots | GCMS/LCMS | Jaiswal and Kuhnert, 2011; Tousch et al., 2014 |
| 164 | 1,3,5-tricaffeoyl-4-succinoylquinic acid | – | A. lappa | Roots | LCMS | Jaiswal and Kuhnert, 2011 |
| 165 | 1,5-dicaffeoyl-3,4-disuccinoylquinic acid | – | A. lappa | Roots | LCMS | Jaiswal and Kuhnert, 2011 |
| 166 | 1,5-dicaffeoyl-3-fumaroyl-4-succinoylquinic acid | – | A. lappa | Roots | LCMS | Jaiswal and Kuhnert, 2011 |
| 167 | 1-fumaroyl-3,5-dicaffeoyl-4-succinoylquinic acid | – | A. lappa | Roots | LCMS | Jaiswal and Kuhnert, 2011 |
| 168 | 1,5-di-O-caffeoyl-3-O-succinoyl-4-O-maloylquinic acid | C33H31O19 | A. lappa | Roots | GCMS/LCMS | Jaiswal and Kuhnert, 2011; Tousch et al., 2014 |
| 169 | Dimaloyl-dicaffeoylquinic acid isomer 1 | C33H31O20 | A. lappa | Roots | GCMS/LCMS | Tousch et al., 2014 |
| 170 | Succinoyl-tricaffeoylquinic acid isomer | C38H33O18 | A. lappa | Roots | GCMS/LCMS | Tousch et al., 2014 |
| 171 | Maloyl-dicaffeoylquinic acid isomer | C29H27O15 | A. lappa | Roots | GCMS/LCMS | Tousch et al., 2014 |
| 172 | Dicaffeoyl-succinoyl-malonylquinic acid isomer 1 | C33H31O19 | A. lappa | Roots | GCMS/LCMS | Tousch et al., 2014 |
| 173 | Dicaffeoyl-succinoyl-malonylquinic acid isomer 2 | C33H31O20 | A. lappa | Roots | GCMS/LCMS | Tousch et al., 2014 |
| 174 | Dimaloyl-dicaffeoylquinic acid isomer 2 | C33H31O20 | A. lappa | Roots | GCMS/LCMS | Tousch et al., 2014 |
| 175 | Dimaloyl-dicaffeoylquinic acid isomer 3 | C33H31O20 | A. lappa | Roots | GCMS/LCMS | Tousch et al., 2014 |
| 176 | Maloyl-tricaffeoylquinic isomer | C28H32O19 | A. lappa | Roots | GCMS/LCMS | Tousch et al., 2014 |
| 177 | 1,3,5-tri-O-caffeoyl-4-O-maloylquinic Acid | C38H33O19 | A. lappa | Roots | GCMS/LCMS | Tousch et al., 2014 |
| 178 | 5-hydroxymaltol | C6H6O4 | A. lappa | Roots | UV/IR/ESIMS/NMR | Han et al., 2013 |
| 179 | Succinic acid | C4H6O4 | A. lappa | Roots | UV/IR/ESIMS/NMR | Han et al., 2013 |
| Saccharides/Polysaccharides | ||||||
| 180 | Rhamnogalacturonan | C117H178O101 | A. lappa, A. minus | Roots/leaves | Chromatography/NMR/sugar analysis | Kato and Watanabe, 1993; Carlotto et al., 2016 |
| 181 | Xylan | (C5H8O4)n | A. lappa, A. minus | Roots/leaves | Chromatography/NMR/sugar analysis | Kato and Watanabe, 1993 |
| 182 | Arabinan | C9H13N3O5 | A. lappa, A. minus | Roots/leaves | Chromatography/ NMR/sugar analysis | Kato and Watanabe, 1993; Carlotto et al., 2016 |
| 183 | Arabinogalactan | C20H36O14 | A. lappa, A. minus | Roots/leaves | Chromatography/ NMR/sugar analysis | Kato and Watanabe, 1993; Carlotto et al., 2016 |
| 184 | Galactan | C18H32O16 | A. lappa, A. minus | Roots/leaves | Chromatography/ NMR/sugar analysis | Kato and Watanabe, 1993 |
| 185 | Cellulose | C64H124O30 | A. lappa, A. minus | Roots/leaves | Chromatography/ NMR/sugar analysis | Kato and Watanabe, 1993 |
| 186 | Xyloglucan | C51H86O42 | A. lappa, A. minus | Roots/leaves | Chromatography/ NMR/sugar analysis | Kato and Watanabe, 1993 |
| 187 | Galacturonic acid | C6H10O7 | A. lappa | Roots/leaves | Chromatography/ NMR | Carlotto et al., 2016 |
| 188 | Galacturonic acid | C6H10O7 | A. lappa | Roots | Chromatography | Fuchigami et al., 1990 |
| 189 | Galactose | C6H12O6 | A. lappa | Roots/leaves/fruits | Chromatography/ NMR | Fuchigami et al., 1990; Kardosova et al., 2003; Boldizsar et al., 2010; Carlotto et al., 2016 |
| 190 | Glucose | C6H12O6 | A. lappa | Roots/leaves/fruits | UV/NMR/HPLC/GCMS | Kardosova et al., 2003; Boldizsar et al., 2010; Li et al., 2013; Carlotto et al., 2016 |
| 191 | Mannose | C6H12O6 | A. lappa | Roots/leaves | NMR | Carlotto et al., 2016 |
| 192 | Sucrose | C12H22O11 | A. lappa | Roots | UV/NMR/HPLC/GCMS | Boldizsar et al., 2010; Li et al., 2013 |
| 193 | Raffinose | C18H32O16 | A. lappa | Fruits | UV/NMR/HPLC/GCMS | Boldizsar et al., 2010 |
| 194 | Rhamnose | C6H12O5 | A. lappa | Roots/leaves/fruits | UV/NMR/HPLC/GCMS | Boldizsar et al., 2010; Carlotto et al., 2016 |
| 195 | Arabinose | C5H10O5 | A. lappa | Roots/leaves/fruits | UV/NMR/HPLC/GCMS | Fuchigami et al., 1990; Kardosova et al., 2003; Boldizsar et al., 2010; Carlotto et al., 2016 |
| 196 | Inulin (fructan) | (C6H10O5)n | A. lappa, A. tomentosum | Roots | HPTLC/MS/NMR/HPLC-ELSD | Kardosova et al., 2003; Turdumambetov et al., 2004; Milani et al., 2011; Olennikov and Tankhaeva, 2011; Li et al., 2013; Liu et al., 2014 |
| 197 | Fructose | C6H12O6 | A. lappa | Roots | HPLC-ELSD | Li et al., 2013 |
| 198 | Sorbitol | C6H14O6 | A. lappa | Fruits | UV/NMR/HPLC/GCMS | Boldizsar et al., 2010 |
| 199 | Mannitol | C6H14O6 | A. lappa | Fruits | UV/NMR/HPLC/GCMS | Boldizsar et al., 2010 |
| Others | ||||||
| 200 | Crocin | C44H64O24 | A. lappa | Leaves | UPLC | Lou et al., 2016 |
| 201 | b-asparagine | C4H8N2O3 | A. lappa, A. tomentosum | Roots | IR/NMR | Boev, 2005 |
The known non-volatile constituents of Arctium species.
FIGURE 2
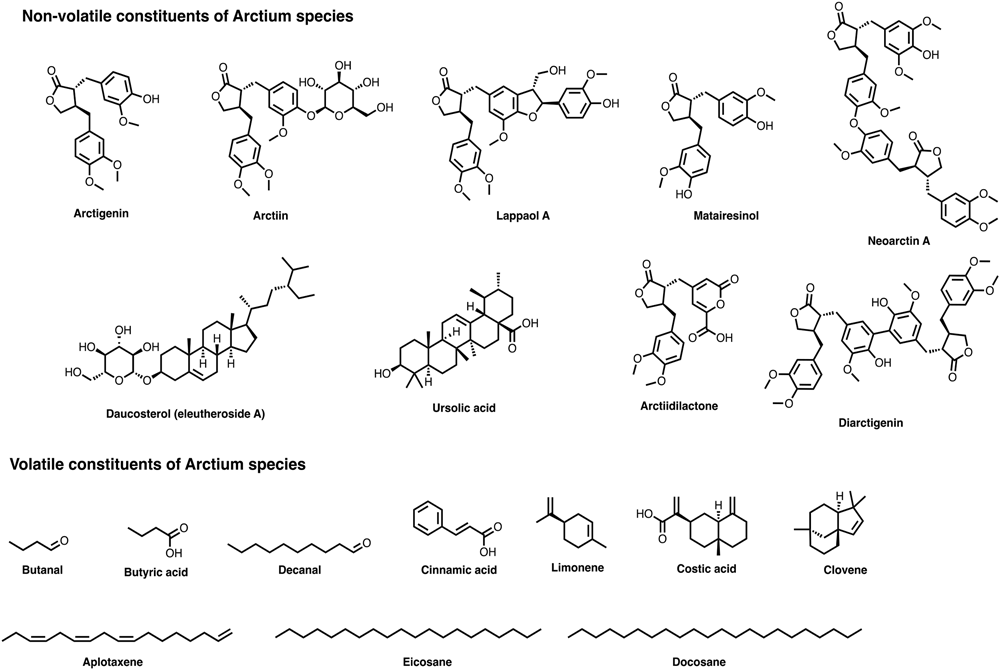
Chemical structures of several relevant components present in Arctium species.
Lignans
Major biologically active lignans include mainly arctigenin (a dietary phytoestrogen) and its glycoside, arctiin (lignanolides) occurring commonly in seeds, roots, fruits, and leaves of A. lappa and A. tomentosum (Yu et al., 2003; Ming et al., 2004; Liu et al., 2005, 2012, 2015; Wang et al., 2005; Matsumoto et al., 2006; Gao et al., 2008; Boldizsar et al., 2010; Ferracane et al., 2010; Zhou et al., 2011; Qin et al., 2014; Su et al., 2015; Lou et al., 2016). In addition, seeds and roots are distributed with low levels of dilignans and sesquilignans. For the first time, two new sesquilignans, namely lappaol A and B were isolated and characterized from A. lappa seeds (Ichihara et al., 1976). Later, 3 more sesquilignans, namely, lappaol C, D, and E, and two dilignans, namely lappaol F and H, were structurally determined from the seeds of A. lappa (Ichihara et al., 1977, 1978; Yong et al., 2007; Su et al., 2015). Lappaol A, C, and F are also found in the fruits of A. tomentosum (Kardosova et al., 2003). Two new lignans, neoarctin A and B, along with other recognized compounds including arctiin, arctigenin, daucosterol, lappaol F, isolappaol C and matairesinol were identified in seeds of A. lappa (Wang and Yang, 1995; Kardosova et al., 2003; Yong et al., 2007; Gao et al., 2008; Qin et al., 2014; Su et al., 2015). A simple RP-HPLC method was developed to identify the presence of arctiin in fruits of A. lappa (Yu et al., 2003; Boldizsar et al., 2010). Using bioactivity-guided fractionation, lappaol A, C and F, arctiin and arctignan E were isolated and characterized from the ethanolic extract (95%) of A. lappa seeds (Ming et al., 2004). Likewise, HPLC/UPLC/LC/MS/MS methods have been developed to identify arctigenin and arctiin in the seeds, leaves and roots of A. lappa (Yu et al., 2003; Liu et al., 2005; Ferracane et al., 2010; Lou et al., 2010a,b; Predes et al., 2011). Further, a supercritical fluid extraction procedure was found to be superior for extracting arctiin from A. lappa fruits (Carlotto et al., 2016). A high-speed counter-current chromatography was employed to obtain the pure compound arctiin from the fruit extracts of A. lappa. Authors obtained 49% of arctiin identified based on LC-MS and NMR techniques (Wang et al., 2005). A novel butyrolactone lignan compound named diarctigenin was found to occur in the methanolic seed extracts of A. lappa (Han et al., 1994). The fruits of A. lappa are reported to contain a total of 13 compounds including 5 new natural products (Umehara et al., 1993). Among them, 6 compounds were identified as arctignan A-E and artctigenin. Later, the occurrence of arctigenin and arctiin was also established from the leaves and seeds of A. lappa (Umehara et al., 1993; Liu et al., 2005; Matsumoto et al., 2006). Besides, the active extract resulted from the bioassay-guided fractionation of seed methanolic extract contained five active compounds including a new sesquilignan named isolappaol C and four known sesquilignan and dilignans namely, diarctigenin, and lappaol C, D, and F (Ferracane et al., 2010). Further, improved methods of extraction and analysis revealed that the seeds and roots of A. lappa contain arctigenin, arctiin, arctignan E, matareisinol, lappaol A, C, and F (Ferracane et al., 2010; Lou et al., 2010a, 2016; Liu et al., 2015; Su et al., 2015). The occurrence of 8 lignans in seeds and 1 lignan in roots of A. lappa was determined. The identified lignans were arctiin, arctigenin arctignan D and E, lappaol A, C, and H, isolappaol C and matairesinol (Liu et al., 2012; Haghi et al., 2013). Likewise, syringaresinol was reported in the chloroform fraction of A. lappa roots (Han et al., 2013). A rare butyrolactone lignan named arctiidilactone, and 11 novel butyrolactone lignans [arctigenin-4-O-β-D-gentiobioside, arctigenin-4-O-α-D-galactopyranosyl-(1→6)-O-β-D-glucopyranoside, arctigenin-4-O-β-D-apiofuranosyl-(1→6)-O-β-D-glucopyranoside, 5′-propanediolmatairesinoside, (7′R,8R,8′R)-rafanotrachelogenin-4-O-β-D-glucopyranoside, (7′S,8R,8′R)-rafanotrachelogenin-4-O-β-D-glucopyranoside, (7S,8S,8′R)-4,7-dihydroxy-3,3′, 4-trimethoxyl-9-oxo benzylbutyrolactone lignan-4-O-β-D-glucopyranoside, (7R,8S,8′R) -4,7,4′-trihydroxy-3,3′-dimethoxyl-9-oxo dibenzylbutyrolactone lignan-4-O-β-D-glucopyranoside, (7S,8S,8′R)-4,7-dihydroxy-3,3′,4′-trimethoxyl-9-oxo dibenzylbutyrolactone lignan, arctiidilactone, arctiiapolignan A and arctiisesquineolignan A] were determined in A. lappa fruits (Yang et al., 2015). Phylligenin, matairesinoside and pinoresinol were reported only in the fruits of A. lappa (Boldizsar et al., 2010). Also, 2 secolignans, styraxlignolide D and styraxlignolide E (Yang et al., 2015) and 4 new neolignan glucosides namely, (8R)-4,9,9′-trihydroxy-3,3′-dimethoxy-7-oxo-8-O-4′-neolignan-4-O-β-D-glucopyranoside, (7S,8R)-4,7,9,9′-tetrahydroxy-3,3′-dimethoxy-8-O-4′-neolignan-9′-O-β-D-apiofuranosyl-(1→6)-O-β-D-glucopyranoside, (7′S,8′R,8S)-4,4′, 9′-trihydroxy-3,3′-dimethoxy -7′,9-epoxylignan-7-oxo-4-O-β-D-glucopyranoside and (7R, 8S)-dihydrodehydrodiconiferyl alcohol-7′-oxo-4-O-β-D-glucopyranoside are reported from A. lappa fruits (Huang X.Y. et al., 2015). Besides, phytochemical analysis of A. lappa fruits revealed the existence of 2 more lignans named arctiisesquineolignan B and arctiiphenolglycoside A (He et al., 2016). Bioassay-guided separation and purification of hydroethanolic extracts of A. lappa fruits allowed to identify a new lignan, (+)-7,8-didehydroarctigenin along with arctigenin and matairesinol identified previously (Matsumoto et al., 2006).
Fatty Acids and Esters
In search of α-glucosidase inhibitory compounds, Miyazawa et al. (2005) found 11 compounds in A. lappa methanol extract. Among them, 10 compounds belonged to fatty acids. The identified compounds were linolenic acid, linoleic acid, methyl linoleate, methyl oleate, methyl linolenate, oleic acid, palmitic acid, methyl palmitate, methyl stearate, and stearic acid. Methanol extract from Arctium lappa L. which was found to contain sitosterol-β-D-glucopyranoside, methyl palmitate, methyl linoleate and methyl linoleneate showed an inhibitory activity against α-glucosidase at 97.3, 73.4, 66.5, and 68.5% respectively at a concentration of 200.0 μM (Miyazawa et al., 2005). Later, Kuo et al. (2012) identified methyl methyl α-linolenate, linolenic acid and methyl oleate as the chief constituents in the n-hexane fraction of A. lappa root (Kuo et al., 2012). The presence of linoleic acid, oleic acid, palmitic acid and stearic acid were also reported from A. lappa fruits (Boldizsar et al., 2010). Fatty acid composition of A. tomentosum seeds showed the occurrence of docosanoic acid, hexadecanoic acid, 9-hexadecenoic acid, 9,12-octadecadienoic acid, oxiraneoctanoic acid, eicosanoic acid, cis-13-eicosenoic acid, and tetracosanoic acid (Zhou et al., 2011).
Acetylenic Compounds
From the roots of A. lappa, Washino et al. (1986) isolated and characterized 9 sulfur-containing acetylenic compounds namely, arctinone-a, arctinone-b, arctinol-a, arctinol-b, arctinal, arctic acid-b, arctic acid-c, methyl arctate-b, and arctinone-a acetate. Based on the chemical and spectral analysis, it was found that all these compounds were derivatives of 5′- (1-propynyl)-2,2′-bithienyl-5-yl. Later, the occurrence of few guaianolides linked with a sulfur-containing acetylenic compounds namely dehydrodihydrocostus lactone, dehydrocostus lactone, lappaphen-a and lappaphen-b were discovered from the acetone extracts of A. lappa roots (Washino et al., 1986, 1987). Several bioactivities of the key A. lappa constituents have been well-described in literature including antibacterial and antifungal activities of acetylenic compounds (Takasugi et al., 1987) and anti-edematogenic activity on carrageenan-induced paw edema (Carlotto et al., 2016).
Phytosterols
Daucosterol, a natural phytosterol-like compound, was obtained from the seeds of A. lappa (Ahangarpour et al., 2017). The fruits of A. tomentosum are reported to contain 2 steroids, such as daucosterol and β-sitosterol. Using bioactivity-guided fractionation, daucosterol and β-sitosterol were recovered from the ethanolic extract (95%) of A. lappa seeds (Ming et al., 2004). Later, sitosterol-beta-D-glucopyranoside was found in the methanolic extracts of A. lappa (Miyazawa et al., 2005). Also, daucosterol and β-sitosterol compounds were detected from the chloroform extracts of A. lappa roots (Han et al., 2013). It was shown that phytosterol daucosterol inhibited cancer cell proliferation by inducing autophagy through reactive oxygen species-dependent manner (Zhao et al., 2015), and exhibited immunoregulatory activity by inducing protective Th1 immune response (Lee et al., 2007).
Polysaccharides
For the first time, Fuchigami et al. (1990; Ferracane et al., 2010) determined the pectic polysaccharides in edible A. lappa roots. Later investigations revealed the occurrence of several kinds of polysaccharides such as pectic substance, rhamnogalacturonan with neutral sugars, hemicellulose (arabinan, arabinogalactan, galactan, xylan, and xyloglucan), galacturonic acid, glucose, galactose, arabinose, rhamnose, mannose, and cellulose in cell walls of A. lappa and A. minus roots and leaves (Kato and Watanabe, 1993; Carlotto et al., 2016). Also, arabinose, glucose, galactose, rhamnose, and raffinose are reported from fruits of A. lappa (Boldizsar et al., 2010). The xyloglucan characterized from A. minus comprised a repeated unit of oligosaccharides of hepta-(Glc–Xyl = 4:3), deca-(Glc–Xyl–Gal–Fuc = 4:3:2:1) and nona-(Glc–Xyl–Gal–Fuc = 4:3:1:1) saccharides in the ratio of 14:5:12 (Kato and Watanabe, 1993). Biologically active inulin-type fructofuranans and other fructooligosaccharides have been identified from the roots of A. lappa (Kardosova et al., 2003). Inulin, a fiber comprising oligomers and polymers of fructose units linked by β(2→1) fructosyl–fructose bonds, has also been reported in the roots of A. lappa (Rajasekharan et al., 2015). A water-soluble polysaccharide fructan with a molecular weight of 4,600 Da, named as ALP1, was purified from A. lappa root and was composed of fructose and glucose in the molar ratio of 13:1. They were linked in →(1)-Fruf-(2)→, Fruf-(2)→ and Glcp-(1)→ (Liu et al., 2014). The structure was similar to the crude fructan obtained previously by Kardosova et al. (2003). In A. tomentosum, the glucofructans content is 24%, constituted by a polymer of 2 inulin type (GF-A and GF-B) and 1 graminan (a mixed type of glucofructans containing 1,2- and 2-6 bonds) type polysaccharides. HPTLC method was developed by Olennikov and Tankhaeva (2011) to quantify fructans in A. tomentosum and A. lappa (Olennikov and Tankhaeva, 2011). Two sugar alcohols, mannitol and sorbitol were reported from the fruits of A. lappa (Boldizsar et al., 2010). The yield of inulin from A. lappa root was successfully increased by adopting an ultrasonic extraction technology (Milani et al., 2011). It was indicated that water-soluble polysaccharide from A. lappa could significantly ameliorate the dysregulation of pro-inflammatory cytokines (IL-1β, IL-6, and TNF-α) and anti-inflammatory cytokine (IL-10) caused by colitis (Wang et al., 2019).
Caffeoylquinic Acid Derivatives (Carboxylic Acids)
Caffeoylquinic acids are the major bioactive phenolic compounds of Arctium species and impart superior antioxidant properties to the plant. The roots of A. lappa were reported to contain caffeoylquinic acid derivatives such as 1-0-,5-O-dicaffeoylquinic acid, 1-0-,5-O-dicaffeoyl-3-O-succinylquinic acid, 1-0,-5-O-dicaffeoyl-4-O-succinylquinic acid, 1-0-,5-O-dicaffeoyl-3-O-,4-O-disuccinylquic acid and 1-0-,3-0-,5-O-tricaffeoyl-4-O- succinylquinic acid (Maruta et al., 1995). Chlorogenic acid content is much higher than the caffeic acid and both occur mainly in the skin of A. lappa roots (Chen et al., 2004). HPTLC analysis was used as a chemical profiling tool to estimate chlorogenic acid in A. lappa roots. The content ranged from 0.107 to 0.140%. Lin and Harnly (2008) and Liu et al. (2012) identified several compounds, including 5-sinapoylquinic acid, 3-sinapoyl-5-caffeoylquinic acid, 3-sinapoyl-5-caffeoyl-1- methoxyoxaloylquinic acid, 4-sinapoyl-5-caffeoyl-1-methoxyoxaloylquinic acid, 1,4-dicaffeoylquinic acid, 3,4-dicaffeoylquinic acid, 4,5-dicaffeoylquinic acid, 3,5-dicaffeoylquinic acid, 3,5-dicaffeoyl-1-methoxyoxaloylquinic acid, 3-feruloyl- 5-caffeoylquinic acid, 4,5-dicaffeoyl-1-methoxyoxaloylquinic acid, 3,5-dicaffeoyl-1- methoxyoxaloylquinic acid, 3-feruloyl-5-caffeoylquinic acid, 4,5-dicaffeoyl-1- methoxyoxaloylquinic acid, 3,4,5-tricaffeoylquinic acid, 1,4,5-tricaffeoylquinic acid and 1,4,5-tricaffeoyl-3-methoxyoxaloylquinic acid from the roots of A. lappa. Jaric et al. (2007) and Jaiswal and Kuhnert (2011) have characterized succinic, fumaric and malic acid-containing chlorogenic acid from the roots of A. lappa. These compounds included 3-succinoyl-4,5-dicaffeoyl, 1,5-dicaffeoyl-4-succinoylquinic acid, 1,5-dicaffeoyl-3-succinoylquinic acid, 3,4-dicaffeoyl-5-succinoylquinic acid, 1,5-dicaffeoyl-4-fumaroylquinic acid, 1,3-dicaffeoyl-5-fumaroylquinic acid, 1,4-dicaffeoyl-3-maloylquinic acid, 1,5-dicaffeoyl-3-maloylquinic acid and 1,5-dicaffeoyl-4-maloylquinic acid, 1,3,5-tricaffeoyl-4-succinoylquinic acid, 1,5-dicaffeoyl-3,4-disuccinoylquinic acid, 1,5-dicaffeoyl-3-fumaroyl-4-succinoylquinic acid, 1-fumaroyl-3,5-dicaffeoyl-4-succinoylquinic acid, 1,5-dicaffeoyl-3-succinoyl- 4-dimaloylquinic acid and dicaffeoyldimaloylquinic acid. Further, Liu et al. (2012) isolated and identified 12 caffeoylquinic acids in both seeds and roots of A. lappa. The identified compounds included chlorogenic acid, 1,5-di-O-caffeoylquinic acid, 1,3-di-O-caffeoylquinic acid, dicaffeoyl-maloylquinic acid, dicaffeoyl-maloylquinic acid, 1,3-di-O-caffeoylquinic acid, 1,5-di-O-caffeoyl-3-O-maloylquinic acid, 1,5-di-O-caffeoyl-3-O-succinoylquinic acid, 1,5-di-O-caffeoyl-4-O-maloylquinic acid, dicaffeoyl-dimaloylquinic acid, 1,5-di-O-caffeoylquinic acid, 1,5-di-O-caffeoyl-3-O-succinoyl-4-Omaloyquinic acid, 1,5-di-O-caffeoyl-3,4-di-O-succinoylquinic acid, and 1,3,5-tri-O-caffeoyl-4-O-succinoylquinic acid. In addition, phytochemical analysis of root extracts of A. lappa showed the occurrence of 8 additional isomers of hydroxycinnamic acids (Liu et al., 2012; Tousch et al., 2014). An average content of chlorogenic acid, 1-O-5-O-dicaffeoylquinic acid and 1,5-dicaffeoyl- 3-succinylquinic acid was observed to be between 1.7 and 7.9 mg/g dry weight of roots (Wang et al., 2001). Two new neolignan glucosides named (70S, 80R, 8S)-4,40,90- trihydroxy-3,30-dimethoxy-70,9-epoxylignan-7-oxo-4-O-b-D-glucopyranosyl-40-O-b-D-glucopyranoside and (7S, 8R)-4,7,9,90-tetrahydroxy-3,30-dimethoxyl- 70-oxo-8-40- oxyneolignan-4-O-b-D-glucopyranoside were determined from the fruit extract of A. lappa (Yang et al., 2012). The occurrence of phenolic acids, caffeic acid, cynarin and chlorogenic acid has been reported for the first time in A. lappa seeds and leaves (Ferracane et al., 2010; Tang et al., 2014; Lou et al., 2016) and later in both seeds and roots (Predes et al., 2011). Chlorogenic acid was determined by Tardio et al. (2005) in the seeds of A. lappa. Further, UPLC analysis revealed the presence of caffeic acid, benzoic acid and p-coumaric acid in the leaves of A. lappa (Tardio et al., 2005; Lou et al., 2010a,b). From the ethyl acetate and n-butanol fractions of A. lappa, 1,5-O-two caffeoylquinic acids, succinic acid and 5-hydroxy maltol were identified for the first time (Han et al., 2013). Likewise, HPLC and UPLC with photodiode array (PDA) detector were used to quantify caffeoyl esters, chlorogenic acid and 1,5-dicaffeoylquinic acid in aerial parts and root samples of A. lappa (Haghi et al., 2013). Two more phenolic compounds, namely, coumaroylquinic acid and caffeoyl-hexose-hydroxyphenol were identified by Rajasekharan et al. (2015) in the root extracts of A. lappa (Rajasekharan et al., 2015). It was reported that caffeoylquinic acids and their derivatives show multiple pharmacological activities including decrease in diet-induced obesity via modulation of PPARα and LXRα transcription (Huang K. et al., 2015) and anti-ulcerogenic effect (Lee et al., 2010).
Flavonoids
The reported flavonoids include flavonols, flavones, and their glycosides. Two major constituents, namely rutin and isoquercetin, along with few other minor flavonoids including kaempferol-3-O-rhamnoglucoside, quercimeritrin and astragalin were identified in the ethanolic extracts of A. minus leaves (Saleh and Bohm, 1971). Likewise, the occurrence of quercetin-3-O-rhamnoside was reported from the leaves of A. lappa. Later, the presence of phenolic compounds such as quercetin, quercitrin, rutin, and luteolin have been reported in seeds, fruits, leaves and roots of A. lappa (Saleh and Bohm, 1971; Tamayo et al., 2000; Yu et al., 2003; Lou et al., 2010a,b, 2016; Predes et al., 2011; Liu et al., 2012; Tang et al., 2014). Also, few isoflavone derivatives including genistein, nobilein, biachanin A and tangeretin have been detected in A. lappa roots (Eberding et al., 2007). A comparative study has shown the existence of chemical differences within the A. lappa organs (Ferracane et al., 2010). According to them, luteolin and quercetin rhamnoside were detected in roots whereas rutin, quercetin, quercitrin and luteolin in leaves. On the other hand, no flavonoids were found in the seeds of A. lappa. Two more flavonols, namely quercetin 3-O-glucuronide and quercetin 3-vicianoside, were identified by Rajasekharan et al. (2015) in the root extracts of A. lappa.
Terpenoids
The fruits of A. lappa were found to contain β-eudesmol, a sesquiterpene alcohol (Rajasekharan et al., 2015; Yang et al., 2015). Pentacyclic triterpenoids, such as ursolic and oleanolic acids were detected by Han et al. in the ethanolic extract of A. lappa roots (Han et al., 2013). Arctiopicrin and onopordopicrin are the sesquiterpene lactones isolated from the leaf extract of A. lappa (Barbosa et al., 1993; Machado et al., 2012). Arctiopicrin occurrence is also evidenced in A. lappa. Later, few more sesquiterpene lactones, namely dehydromelitensin-8- (4′-hydroxymethacrylate), dehydromelitensin, and melitensin and a norisoprenoid along with 2 more terpenes such as dehydrovomifoliol and loliolide were identified in A. lappa leaf (Machado et al., 2012). Onopordopicrin, a germacranolide sesquiterpene lactone was isolated from the aerial parts of A. nemorosum (Zimmermann et al., 2012). Two triterpenoids, namely 3α-acetoxy-hop-22(29)-ene and 3α-hydroxylanosta-5, 15-diene were isolated from the leaves of A. lappa (Jaiswal and Kuhnert, 2011).
Others
From the concentrated sap obtained from A. lappa roots (A. lappa and A. tomentosum), β-asparagine was isolated for the first time (Boldizsar et al., 2010). The carotenoid crocin was reported to occur in the leaves of A. lappa (Lou et al., 2016).
Volatile Compounds
A total of 101 volatile chemical constituents were identified in A. lappa. The details of these compounds are partially summarized in Table 2 and described in this section. Carboxylic acids and fatty acids were more prevalent in A. lappa. On the other hand, there are no available literatures on the identification of volatile components in other Arctium species. The chemical structures of some compounds from Arctium species are shown in Figure 2.
Table 2
| S. No | Compound name | Formula | Species | Plant origin/part | Analytical method | References |
|---|---|---|---|---|---|---|
| Hydrocarbons | ||||||
| 1 | Aplotaxene | C17H28 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 2 | Clovene | C15H24 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 3 | Dihydroaplotaxene | C17H30 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 4 | Docosane | C22H46 | A. lappa | Leaves | GCMS | Aboutabl et al., 2013 |
| 5 | Eicosane | C20H42 | A. lappa | Roots/leaves/seeds | GCMS | Aboutabl et al., 2013 |
| 6 | 1-Heptadecene | C17H34 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 7 | Heptacosane | C27H56 | A. lappa | Roots/leaves | GCMS | Aboutabl et al., 2013 |
| 8 | Hexacosane | C26H54 | A. lappa | Roots/leaves | GCMS | Aboutabl et al., 2013 |
| 9 | Nonadecane | C19H40 | A. lappa | Leaves | GCMS | Aboutabl et al., 2013 |
| 10 | 2-Naphthalenemethanol | C11H10O | A. lappa | Roots | GCMS | Wang et al., 2004 |
| 11 | 1-Pentadecene | C15H30 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 12 | Pentacosane | C25H52 | A. lappa | Roots | GCMS | Aboutabl et al., 2013 |
| 13 | Pentadecane | C15H32 | A. lappa | Roots/leaves | GCMS | Aboutabl et al., 2013 |
| 14 | Tetracosane | C24H50 | A. lappa | Roots/leaves | GCMS | Aboutabl et al., 2013 |
| 15 | Tetradecane | C14H30 | A. lappa | Leaves | GCMS | Aboutabl et al., 2013 |
| Aldehydes | ||||||
| 16 | Benzaldehyde | C7H6O | A. lappa | Roots | GCMS | Washino et al., 1986, 1987; Wang et al., 2004 |
| 17 | Butanal | C4H8O | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 18 | Decanal | C10H20O | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 19 | Dodecanal | C12H24O | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 20 | Heptanal | C7H14O | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 21 | Hexanal | C6H12O | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 22 | (Z)-3-Hexenal | C6H10O | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 23 | (E)-2-Hexenal | C6H10O | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 24 | 2-Methylpropanal | C4H8O | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 25 | 3-Methylbutanal | C5H10O | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 26 | Nonanal | C9H18O | A. lappa | Roots/leaves/seeds | GCMS | Washino et al., 1986, 1987 |
| 27 | Octanal | C8H16O | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 28 | (E)-2-Octanal | C8H14O | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 29 | Phenylacetaldehyde | C8H8O | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 30 | Pentanal | C5H10O | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 31 | Propanal | C3H6O | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 32 | Tridecanal | C13H26O | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 33 | 4-Methoxybenzaldehyde | C8H8O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 34 | Undecanal | C11H22O | A. lappa | Roots | Washino et al., 1986, 1987 | |
| Methoxypyrazines | ||||||
| 35 | 2-Methoxy-3-methylpyrazine | C6H8N2O | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 36 | 2-Isopropyl- 3-methyoxylpyrazine | C8H12N2O | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 37 | 2-Methoxy-3- propylpyrazine | C8H12N2O | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 38 | 2-sec-Butyl-3-methoxypyrazine | C9H14N2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 39 | 2-Isobutyl-3-methoxypyrazine | C9H14N2O | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 40 | 2-Butyl-3- methoxypyrazine | C9H14N2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 41 | 2-Isoamyl-3-methoxypyrazine | C9H14N2O | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| Fatty acids/Carboxylic acids | ||||||
| 42 | Acetic acid | CH3COOH | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 43 | Benzoic acid | C7H6O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 44 | Butyric acid | C4H8O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 45 | Cinnamic acid | C9H8O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 46 | Costic acid | C15H22O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 47 | Decanoic acid | C10H20O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 48 | Dodecanoic acid | C12H24O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 49 | Ethyl oleate | C20H38O2 | A. lappa | Seeds | GCMS | Aboutabl et al., 2013 |
| 50 | Hexanoic acid | C3H6O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 51 | Hexadecanoic acid | C17H34O2 | A. lappa | Roots/seeds | GCMS | Washino et al., 1986, 1987; Aboutabl et al., 2013 |
| 52 | (E)-3-Hexenoic acid | C6H10O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 53 | Heptanoic acid | C7H14O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 54 | (E)-3-Heptenoic acid | C7H12O2 | A. lappa | Roots | GCMS | Washino et al., 1986;, 1987 |
| 55 | Linoleic acid | C18H32O2 | A. lappa | Roots | GCMS | Wang et al., 2004 |
| 56 | 2, 3-Hydroxyoctanoic acid | C8H16O3 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 57 | 2-Methylpropionic acid | C4H8O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 58 | 2-Methylbutyric acid | C5H10O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 59 | 3-Methoxybenzoic acid | C8H8O3 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 60 | Methyl palmitate | C17H34O2 | A. lappa | Roots/seeds | GCMS | Washino et al., 1986, 1987; Aboutabl et al., 2013 |
| 61 | Methyl linolenate | C19H32O2 | A. lappa | Roots | GCMS | Wang et al., 2004 |
| 62 | Methyl oleate | C19H36O2 | A. lappa | Seeds | GCMS | Aboutabl et al., 2013 |
| 63 | Nonanoic acid | C9H18O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 64 | Nonanedioic acid | C9H16O4 | A. lappa | Roots | GCMS | Washino et al., 1986;, 1987 |
| 65 | (E)-3-nonenoic acid | C9H16O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 66 | Octanoic acid | C8H16O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 67 | (E)-3-Octenoic acid | C8H14O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 68 | Octadecanoic acid | C18H36O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 69 | Octadecanoic acid methyl ester | C18H36O2 | A. lappa | Seeds | GCMS | Aboutabl et al., 2013 |
| 70 | Pentanoic acid | C5H10O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 71 | Phenylacetic acid | C8H8O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 72 | Phenylpropionic acid | C9H10O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 73 | Propionic acid | C3H6O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 74 | Pentadecanoic acid | C15H30O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 75 | Salicylic acid | C7H6O3 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 76 | Tridecanoic acid | C13H26O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 77 | Tetradecanoic acid | C14H28O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 78 | Undecanoic acid | C11H22O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| Terpenes/terpenoids | ||||||
| Monoterpenoids | ||||||
| 79 | Carvomenthone | C10H18O | A. lappa | Roots/leaves | GCMS | Aboutabl et al., 2013 |
| 80 | Geraniol | C10H18O | A. lappa | Seeds | GCMS | Aboutabl et al., 2013 |
| 81 | Linalool | C10H18O | A. lappa | Seeds | GCMS | Aboutabl et al., 2013 |
| 82 | Thymol | C10H14O | A. lappa | Seeds | GCMS | Aboutabl et al., 2013 |
| 83 | Z-citral | C10H16O | A. lappa | Seeds | GCMS | Aboutabl et al., 2013 |
| 84 | E-citral | C10H16O | A. lappa | Seeds | GCMS | Aboutabl et al., 2013 |
| Sesquiterpenoids | ||||||
| 85 | Dehydrocostus lactone | C15H18O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 86 | Dehydrodihydrocostus lactone | C15H29O2 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| Oxygenated sesquiterpenes | ||||||
| 87 | Caryophyllene oxide | C15H24O | A. lappa | Roots/leaves | GCMS | Aboutabl et al., 2013 |
| 88 | β-Costol | C15H24O | A. lappa | Roots | GCMS | Aboutabl et al., 2013 |
| Sesquiterpene Hydrocarbons | ||||||
| 89 | Aromadendrene | C15H24 | - | Roots/seeds | GCMS | Aboutabl et al., 2013 |
| 90 | Caryophyllene | C15H24 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 91 | γ-Cadinene | C15H24 | - | Roots/leaves/seeds | GCMS | Aboutabl et al., 2013 |
| 92 | Cyperene | C15H24 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 93 | β-Elemene | C15H24 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987; Aboutabl et al., 2013 |
| 94 | trans-β-Farnesene | C15H24 | A. lappa | Roots/leaves | GCMS | Aboutabl et al., 2013 |
| 95 | α-Guaiene | C15H24 | A. lappa | Roots | GCMS | Washino et al., 1986, 1987 |
| 96 | Isoaromadendrene epoxide | C15H24O | – | Roots/leaves/seeds | GCMS | Aboutabl et al., 2013 |
| 97 | Limonene | C10H16 | A. lappa | Leaves/seeds | GCMS | Washino et al., 1986, 1987 |
| 98 | α-Myrcene | C10H16 | A. lappa | Seeds | GCMS | Aboutabl et al., 2013 |
| 99 | α-Pinene | C10H16 | A. lappa | Roots/leaves | GCMS | Aboutabl et al., 2013 |
| 100 | Squalene | C30H50 | A. lappa | Seeds | GCMS | Aboutabl et al., 2013 |
| Sesquiterpene Alcohol | ||||||
| 101 | β-Copaen-4α-ol | C15H24O | – | Roots/leaves/seeds | GCMS | Aboutabl et al., 2013 |
Volatile constituents of Arctium spp.
Hydrocarbons
Fourteen hydrocarbon compounds, aplotaxene, clovene, dihydroaplotaxene, eicosane, 1-heptadecene, heptacosane, hexacosane, nonadecane, 2-naphthalenemethanol, 1-pentadecene, pentacosane, pentadecane, tetracosane, and tetradecane were detected from the roots, seeds, and leaves of A. lappa (Washino et al., 1986; Wang et al., 2004). In addition, docosane, eicosane, heptacosane, hexacosane, tetracosane, and pentadecane were found only in roots and leaves. Docosane was found only in leaves, while seeds of A. lappa contained only eicosane.
Aldehydes
Nineteen aldehydes, namely, benzaldehyde, butanal, decanal, dodecanal, heptanal, hexanal, (Z)-3-hexenal, (E)-2-hexenal, 2-methylpropanal, 3-methylbutanal, nonanal, octanal, (E)-2-octanal, phenylacetaldehyde, pentanal, propanal, tridecanal, 4-methoxybenzaldehyde, and undecanal were found as root volatile compounds in A. lappa (Washino et al., 1986, 1987; Wang et al., 2004). Interestingly, only the alkyl aldehyde nonanal was present in all plant parts such as roots, leaves, and seeds (Washino et al., 1986, 1987).
Methoxypyrazines
Seven methoxypyrazines, such as 2-methoxy-3-methylpyrazine, 2-methoxy-3- propylpyrazine, 2-isopropyl- 3-methyoxylpyrazine, 2-sec-butyl-3-methoxypyrazine, 2-butyl-3- methoxypyrazine, 2-isobutyl-3-methoxypyrazine, and 2-isoamyl-3-methoxypyrazine were detected in roots of A. lappa (Washino et al., 1986, 1987).
Carboxylic Acids and Fatty Acids
Twenty-two carboxylic acids namely acetic acid, benzoic acid, butyric acid, cinnamic acid, costic acid, dodecanoic acid, hexanoic acid, (E)-3-hexenoic acid, heptanoic acid, (E)-3-heptenoic acid, 2, 3-hydroxyoctanoic acid, 2-methylpropionic acid, 2-methylbutyric acid, 3-methoxybenzoic acid, nonanoic acid, nonanedioic acid, pentanoic acid, phenylacetic acid, phenylpropionic acid, propionic acid, salicylic acid, and undecanoic acid were identified in A. lappa roots (Washino et al., 1986, 1987; Wang et al., 2004). Fatty acids such as decanoic acid, hexadecanoic acid, linoleic acid, octanoic acid, (E)-3-octenoic acid, octadecanoic acid, pentadecanoic acid, tridecanoic acid, and tetradecanoic acid were found in roots while ethyl oleate, methyl oleate, hexadecanoic acid, methyl palmitate, and octadecanoic acid methyl ester were identified in seeds of A. lappa (Washino et al., 1986, 1987; Wang et al., 2004).
Monoterpenes and Sesquiterpenes
Three alcoholic and one phenolic monoterpenoids (carvomenthone, geraniol, linalool, and thymol); 2 sesquiterpene lactones (dehydrocostus lactone and dehydrodihydrocostus lactone, isoaromadendrene epoxide); 2 oxygenated sesquiterpenes (caryophyllene oxide and β-costol) and 12 sesquiterpene hydrocarbons namely, aromadendrene, caryophyllene, γ-cadinene, cyperene, β-elemene, trans-β-farnesene, α-guaiene, limonene, myrcene, α-pinene, and squalene were identified in A. lappa (Washino et al., 1986, 1987; Wang et al., 2004). Geraniol, linalool, thymol, aromadendrene, γ-cadinene, isoaromadendrene epoxide, limonene, α-myrcene, and squalene were identified only in the seeds of A. lappa.α-Pinene, isoaromadendrene epoxide, γ-cadinene, carvomenthone, and caryophyllene oxide were found in the roots and leaves.
Bioactivities of Arctium Species
Arctium lappa is widely used as an ethno-medicinal plant especially in North America, Asia and Europe, and is applied to treat various diseases including diabetes, gout, rheumatism, and skin problems (Chan et al., 2011; Azizov et al., 2012). A. lappa roots have been used as a vegetable in Japanese (referred to as ‘gobo’) and Korean cuisine. Its root has been used to treat constipation, mercury poisoning, upper respiratory infections, inflammation and oxidative stress in patients with knee osteoarthritis (Maghsoumi-Norouzabad et al., 2016), while the leaves were efficacious in healing burns, rashes, and applied in women with labor condition (Force, 2001; Lewis and Elvin-Lewis, 2003; Amish Burn Study et al., 2014). A. lappa has also been found for the treatment of alopecia (loss of hair) among adults (Amish Burn Study et al., 2014). In Western countries, burdock is used as a remedy for several ailments ranging from arthritis, chronic inflammation, and various skin problems (e.g., scaly skin conditions such as psoriasis and eczema) to cancer treatment (Wu et al., 2010; Amish Burn Study et al., 2014).
Studies on the biological activities of extracts of different parts of A. lappa and compounds isolated thereof, were carried out and revealed antipyretic, antimicrobial, diuretic, diaphoretic, hypoglycaemic, antioxidant, anti-inflammatory, anti-hepatotoxicity, antiulcer, antimutagenicity, and antitumour activities.
Anticancer Effects
Arctium lappa fruit has been used in traditional medicine, and it is popular for its various anticancer effects. Arctigenin (ATG), a natural lignan product extracted from the seeds of Arctium lappa, has been shown to have estrogenic properties, that reduced the risk of osteoporosis, heart disease, and menopausal symptoms (Maxwell et al., 2017). It was found to possess antitumor effect by modulating the protein kinase activation pathway and hence rendering the tumor cells susceptible to effects of the nutrient-deprived environment (Awale et al., 2006). Later on, ATG was shown to induce apoptosis (programmed cell death) of estrogen receptor-negative cancer cells (MDA-MB-231) through the ROS/p38 MAPK pathway and epigenetic regulation of Bcl-2 by upregulating trimethylation of histone H3K9 (Hsieh et al., 2014). It was reported that ATG was able to inhibit cell proliferation and may induce apoptosis and cell cycle arrest at the G0/G1 phase in glioma cells (Maimaitili et al., 2017). In more detail, it was found that ATG increased the expression levels of p21, retinoblastoma and p53 proteins, and significantly decreased the expression levels of cyclin D1 and CDK4 proteins (Maimaitili et al., 2017). Furthermore, ATG was able to induce apoptosis in glioma cells, coupled with increased expression levels of cleaved caspase-3 and the pro-apoptotic BCL2-associated X protein (Maimaitili et al., 2017). ATG-induced apoptosis was significantly suppressed by the pretreatment of cells with Z-DEVD-FMK, a caspase-3 inhibitor (Maimaitili et al., 2017). More recently, study by Lou et al. (2017) demonstrated ATG to significantly inhibit in vitro migration and invasion of human breast cancer cells (MDA-MB-231) by downregulation of MMP-2, MMP-9 and heparanase (Lou et al., 2017).
Extracts from A. lappa also showed selective antiproliferative activity against certain human cancer cell lines including K562, MCF-7 and 786-0 (Predes et al., 2011). Lappaol F, a novel natural product isolated from the seeds of A. lappa, was found to suppress cancer cell growth in a dose-dependent manner in various human cancer cell lines through induction of G1 and G2 cell-cycle arrest. This effect was associated with strong induction of p21 and p27 and suppression of cyclin-dependent kinase 1 (CDK1) and cyclin B1 (Sun et al., 2014).
A. lappa is one of the herbs widely used by cancer patients in some Canadian populations to improve quality of life (QOL) and prevent cancer progression. A. lappa is one of the herbs constituting the two proprietary herbal products: Flor-Essence® and Essiac® suggested for prolong survival and the improvement of QOL among cancer patients (Tamayo et al., 2000).
Antidiabetic Effects
Root of A. lappa root has been found to mediate hypoglycemic activities making it a popular choice to be used as a traditional medicine in diabetes. Oral administration of burdock root ethanolic extract in streptozotocin-induced diabetic rats significantly lowered blood glucose and increased insulin level in the diabetic rats compared to the control diabetic group (Cao et al., 2012). Additionally, treatment with A. lappa extract also reduced the levels of serum total cholesterol (TC), triglycerides (TG) and low density lipoprotein (LDL), whereas high density lipoprotein (HDL) level was higher in the control rats. More recently in a similar study, Ahangarpour et al. (2017) investigated the antidiabetic and hypolipidemic properties of the root extract of A. lappa on nicotinamide-streptozotocin (NA-STZ)-induced type 2 diabetes in mice (Ahangarpour et al., 2017). The results show that root extract of A. lappa displays anti-diabetic effect at certain doses. It exerts its effects through hypolipidemic and insulinotropic properties and hence the root extract could serve successfully in treating patients with type 2 diabetes in the future. Moreover, sitosterol-β-D-glucopyranoside from burdock’s root acts as a potent inhibitor of alpha-glucosidases, thereby having the potential to reduce glycogenolysis and help to decrease blood glucose level (Tousch et al., 2014). In addition, Zhao and Zhou (2015) demonstrated that trace elements (e.g., Na, K, Mn, Fe, and Mg) present in the root and fruit extracts of A. lappa exhibit antidiabetic effects. While A. lappa constituents do reduce absorption of glucose, they also elevate inulin content in blood and slow digestion of carbohydrates to confer its anti-diabetic activities. The pharmacological mechanisms of A. lappa roots are slightly different from other classes of oral antihyperglycemic agents such as metformin. Metformin decreases hepatic glucose production, decreases intestinal absorption of glucose, and improves insulin sensitivity by increasing peripheral glucose uptake and utilization (Dumitrescu et al., 2015).
Anti-oxidant, Hepatoprotective and Gastroprotective Activities
It is believed that lignans and caffeoylquinic acids from A. lappa are of value because of their antioxidant capacity (Maruta et al., 1995; Mkrtchian et al., 1998; Jaiswal and Kuhnert, 2011) by which they can scavenge free radicals that are thought to play an important role in many diseases.
The hydroalcoholic extracts of burdock roots possess significant antioxidant potential as seen by the application of various assays. Very recently, Fierascu et al. (2018) quantified antioxidant potential of burdock extracts using DPPH (2,2-diphenyl-1-picrylhydrazyl) and phosphomolybdate assays to demonstrate that burdock extracts have very high antioxidative activities, presumably due to the high content of polyphenols (Fierascu et al., 2018). The potent antioxidative property makes these extracts effective inhibitors of lipid peroxidation in rat liver homogenate in vitro (Duh, 1998) and an excellent hepatoprotective agent in vivo and in vitro (Lin et al., 2000). Due to its radical scavenging ability, A. lappa is also used to treat gastrointestinal ulcers (da Silva et al., 2013).
Antimicrobial Effects
Extracts of different parts of A. lappa have been investigated for their microbial-modulatory properties by many researchers. An organic extract from A. lappa has shown inhibiting properties toward the growth of Pseudomonas aeruginosa, Escherichia coli, Lactobacillus acidophilus, Streptococcus mutans, and Candida albicans residing in the teeth of the oral cavity (Gentil et al., 2006). Pereira et al. (2005) further reported potent growth inhibiting activities of A. lappa extract against a broad spectrum of oral microorganisms, specifically those associated with teeth infections, namely Enterococcus faecalis, Staphylococcus aureus, Pseudomonas aeruginosa, Bacillus subtilis, and Candida albicans (Pereira et al., 2005). Very recently, Fierascu et al. (2018) investigated antifungal potential of hydroalcoholic extract of burdock roots and observed that it is active against the fungal lines Aspergillus niger ATCC 15475 and Penicillium hirsutum ATCC 52323 (Fierascu et al., 2018). Fruit extract of A. lappa was tested (Dias et al., 2017) for in vitro antiviral properties against Herpes simplex virus type-1 (HSV-1) and was found to decrease viral load significantly at all concentrations tested (400, 50, and 3.125 μg/mL). At 400 μg/mL concentration, it showed comparable antiviral activity as acyclovir (50 μg/mL). Arctigenin, one of the key constituents of A. lappa extract, has shown potent activities against human immunodeficiency virus type-1 (HIV-1) both in vivo and in vitro presumably by increasing the expression of Heme oxygenase-2 (HO-2) and blocking HIV-1gag proteins (Schroder et al., 1990).
Anti-inflammatory Effects
Various parts of A. lappa demonstrated anti-inflammatory effects (Liu et al., 2005). Burdock extract is known to alleviate wound irritation and swelling and therefore has been traditionally used for healing burn wounds. This effect might be mediated through the inhibition of the cyclooxygenase-2 (COX-2) enzyme. Cyclooxygenase is a lipid metabolizing enzyme that catalyzes the oxygenation of polyunsaturated fatty acids. This process forms prostanoids, specifically eicosanoids, which are known to be potent cell signaling molecules connected to inflammatory processes (Charlier and Michaux, 2003). Phenolic compounds present in burdock extract (e.g., arctigenin, lappaol F, diarctigenin) are inhibitors of this enzyme (Zhao et al., 2009; Lee and Kim, 2010), thereby suppressing lipopolysaccharide (LPS)-stimulated NO production (Park et al., 2007) and pro-inflammatory cytokines secretion (including TNF-α and IL-6) in a dose-dependent manner (Zhao et al., 2009; Kwon et al., 2016). Arctigenin also strongly inhibited the expression of iNOS (Inducible Nitric Oxide Synthase) and its enzymatic activity (Wang et al., 2007; Zhao et al., 2009). Moreover, it induced endothelial nitric oxide synthase (eNOS) and supressed in a rat model subarachnoid hemorrhage-induced vasospasm by regulation of the PI3K/Akt signaling pathway (Chang et al., 2015). Among the studied phenolic compounds, diarctigenin was found to inhibit the DNA binding ability of NF-κB and to inhibit NF-κB-regulated iNOS expression (Kim et al., 2008), thereby overall targeting NF-κB-activating signaling cascade directly to confer anti-inflammatory response. Luteolin, an important flavonoid from burdock was also reported to possess significant anti-inflammatory properties (Ferracane et al., 2010; Nabavi et al., 2015).
Effects Against Skin Conditions
Leaves of Arctium species have been used in traditional medicinal practices in various skin conditions (e.g., rashes, boils, eczema, ichthyosis, acne, psoriasis, and abscesses) presumably due to the presence of various phenolic compounds. The potent antioxidant and anti-inflammatory properties of these compounds serve to detoxify and mediate healing action (Chan et al., 2011). Several hydroxycinnamic acids which are among the active phytochemicals in the A. lappa extracts (Liu et al., 2012; Tousch et al., 2014) have been found to act as free radical scavengers and possess antioxidant activities, which confer them potential to serve as skin protectors and wound healers (Graf, 1992; Phan et al., 2001; Taofiq et al., 2017). In addition, hydroxycinnamic acid derivatives also display anti-collagenase, anti-inflammatory, antimicrobial and anti-tyrosinase activities, as well as ultraviolet (UV) protective effects, suggesting that they can be exploited as anti-aging and anti-inflammatory agents, preservatives and hyperpigmentation-correcting ingredients (Ahangarpour et al., 2017). These bioactivities are the reason why burdock extracts find their use in various commercial cosmetic products.
Effect on Potency and Fertility
Diabetes mellitus induces many complications among which dysfunctions male reproductive system is worth mentioning. Glucose metabolism plays an important regulatory role on the production or development of mature spermatozoa (spermatogenesis) as well as on maintaining specific functions, such as motility and fertilization ability in mature sperm cells. Therefore, it is not surprising that A. lappa root extract, which has hypoglycemic and antioxidative properties, would have beneficial effects on male potency and fertility. Ahangarpour et al. (2015) investigated the effect of A. lappa root extract on gonadotropin, testosterone, and sperm parameters in nicotinamide/streptozotocin-induced diabetic mice (Ahangarpour et al., 2015). The root extract led to increased level of luteinizing hormone (LH), follicle stimulating hormone (FSH), and testosterone as well as enhancement in sperm viability only in diabetic mice compared with the control group, indicating A. lappa root extract to be a potentially effective treatment for male sterility arising from diabetic conditions.
Effect on NO Production
It was reported that arctigenin inhibited NO release by IFN-γ signal, whereas it significantly enhanced lipopolysaccharide-triggered NO production in RAW264.7 cells, suggesting that arctigenin may regulate immune responses in activated macrophages and lymphocytes including TNF-α and NO production and lymphocyte proliferation (Cho et al., 1999). Another study shows that arctigenin suppressed the overproduction of NO through down-regulation of iNOS expression and iNOS enzymatic activity in LPS-stimulated macrophage (Zhao et al., 2009). Besides, lappaol F and diarctigenin from Arctium lappa were shown to significantly inhibit NO production in the LPS-stimulated RAW264.7 cells with IC50 values of 9.5 and 9.6 μM, respectively (Park et al., 2007).
Safety Considerations on Arctium Species
Several adverse effects have been reported in literature stemming from long-term use of A. lappa. For example, contact dermatitis might develop after several days of applying a burdock root plaster to a wound, or even as fast as within 12 h in some cases (Rodriguez et al., 1995). In one instance, anticholinergic poisoning has been reported upon oral consumption of A. lappa extract (Force, 2001). However, this poisoning later turned out to be caused by products that have been contaminated with root of belladonna (deadly nightshade). The latter herb contains the poisonous chemical atropine. Long-term consumption of burdock also has led to anaphylaxis in one case (Chan et al., 2011). Root oil made from A. lappa was also found to cause unfavorable physiological effects such as redness, and anaphylactic shock (Rodriguez et al., 1995; Lewis and Elvin-Lewis, 2003; Sasaki et al., 2003). Caution is advised for pregnant or nursing women to consume burdock or its extract, as it might have detrimental effects on the fetus (Chan et al., 2011). Burdock can also interfere with blood clotting. People who are already on blood thinning medications are advised not take it without approval from their doctors. Even though burdock is considered a ‘safe’ food, consuming it in large amounts should be avoided due to lack of large amount of safety studies on burdock. More in vivo studies are in particular needed on A. lappa to further evaluate its therapeutic potential and safe application window. Due to the presence of sesquiterpene lactones, the use of Arctium species should be avoided in patients with hypersensitivity to Asteraceae/Compositae (Chan et al., 2011).
Summary
In summary, the volatile and non-volatile secondary metabolites present in different parts of Arctium species showed pharmacological potential in the treatment of various diseases. The literature existing on extracts of different parts of A. lappa and isolated compounds demonstrates antipyretic, antimicrobial, diuretic, diaphoretic, hypoglycaemic, antioxidant, anti-inflammatory, anti-hepatotoxicity, antiulcer, antimutagenicity, and antitumour activities. Hence, Arctium species display a broad therapeutic potential but further studies are needed on potential risks associated with their application.
Statements
Author contributions
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
Funding
This work was supported by the HOMING program of the Foundation for Polish Science co-financed by the European Union under the European Regional Development Fund (Homing/2017-4/41), the Polish KNOW (Leading National Research Centre) Scientific Consortium “Healthy Animal-Safe Food” decision of Ministry of Science and Higher Education No. 05-1/KNOW2/2015, and the Peter und Traudl Engelhorn Foundation for the promotion of Life Sciences.
Acknowledgments
We acknowledge Mr. Paul-Marian Szatmari for providing the pictures of Arctium species in Figure 1.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Footnotes
References
1
AboutablE.El-TantawyM.ShamsM. (2013). Chemical composition and antimicrobial activity of volatile constituents from the roots, leaves, and seeds of Arctium lappa L. (Asteraceae) grown in Egypt.Egypt. Pharm. J.12173–176. 10.4103/1687-4315.124036
2
AhangarpourA.HeidariH.OroojanA. A.MirzavandiF.Nasr EsfehaniK.Dehghan MohammadiZ. (2017). Antidiabetic, hypolipidemic and hepatoprotective effects of Arctium lappa root’s hydro-alcoholic extract on nicotinamide-streptozotocin induced type 2 model of diabetes in male mice.Avicenna J. Phytomed.7169–179.
3
AhangarpourA.OroojanA. A.HeidariH.GhaediE.TaherkhaniR. (2015). Effects of hydro-alcoholic extract from Arctium lappa l.(burdock) root on gonadotropins, testosterone, and sperm count and viability in male mice with nicotinamide/streptozotocin-induced type 2 diabetes.Malays. J. Med. Sci.2225–32.
4
Al-ShammaaD. A.SaourK. Y.Abdul-KhalikZ. M. (2017). Phytochemical investigation for the main active constituents in Arctium lappa L. cultivated in Iraq.Iraqi J. Pharm. Sci.2218–24.
5
Amish Burn StudyG.KolaczN. M.JarochM. T.BearM. L.HessR. F. (2014). The effect of burns & wounds (B&W)/burdock leaf therapy on burn-injured amish patients: a pilot study measuring pain levels, infection rates, and healing times.J. Holist. Nurs.32327–340. 10.1177/0898010114525683
6
AwaleS.LuJ.KalauniS. K.KurashimaY.TezukaY.KadotaS.et al (2006). Identification of arctigenin as an antitumor agent having the ability to eliminate the tolerance of cancer cells to nutrient starvation.Cancer Res.661751–1757. 10.1158/0008-5472.can-05-3143
7
AzizovU. M.KhadzhievaU. A.RakhimovD. A.MezhlumyanL. G.SalikhovS. A. (2012). Chemical composition of dry extract of Arctium lappa roots.Chem. Nat. Compd.471038–1039. 10.1007/s10600-012-0142-3
8
BaiL.ZhangH.LiuQ.ZhaoY.CuiX.GuoS.et al (2016). Chemical characterization of the main bioactive constituents from fruits of Ziziphus jujuba.Food Funct.72870–2877. 10.1039/c6fo00613b
9
BarbosaJ. M.CostaM.GomesC.TrolinG. (1993). Isolation of onopordopicrin, the toxic constituent of Arctium-Lappa L.J. Braz. Chem. Soc.4186–187. 10.5935/0103-5053.19930040
10
BoevR. S. (2005). A substance with cytostatic and apoptosis-inducing activity from burdock roots.Chem. Sustain. Dev.13119–122.
11
BoldizsarI.FuzfaiZ.TothF.SedlakE.BorsodiL.Molnar-PerlI. (2010). Mass fragmentation study of the trimethylsilyl derivatives of arctiin, matairesinoside, arctigenin, phylligenin, matairesinol, pinoresinol and methylarctigenin: their gas and liquid chromatographic analysis in plant extracts.J. Chromatogr. A12171674–1682. 10.1016/j.chroma.2010.01.019
12
CaoJ.LiC.ZhangP.CaoX.HuangT.BaiY.et al (2012). Antidiabetic effect of burdock (Arctium lappa L.) root ethanolic extract on streptozotocin-induced diabetic rats.Afr. J. Biotechnol.119079–9085.
13
CarlottoJ.de SouzaL. M.BaggioC. H.WernerM. F.Maria-FerreiraD.SassakiG. L.et al (2016). Polysaccharides from Arctium lappa L.: chemical structure and biological activity.Int. J. Biol. Macromol.91954–960. 10.1016/j.ijbiomac.2016.06.033
14
ChanY. S.ChengL. N.WuJ. H.ChanE.KwanY. W.LeeS. M.et al (2011). A review of the pharmacological effects of Arctium lappa (burdock).Inflammopharmacology19245–254. 10.1007/s10787-010-0062-4
15
ChangC.-Z.WuS.-C.ChangC.-M.LinC.-L.KwanA.-L. (2015). Arctigenin, a potent ingredient of Arctium lappa L., induces endothelial nitric oxide synthase and attenuates subarachnoid hemorrhage-induced vasospasm through PI3K/Akt pathway in a rat model.Biomed Res. Int.2015:490209. 10.1155/2015/490209
16
CharlierC.MichauxC. (2003). Dual inhibition of cyclooxygenase-2 (COX-2) and 5-lipoxygenase (5-LOX) as a new strategy to provide safer non-steroidal anti-inflammatory drugs.Eur. J. Med. Chem.38645–659. 10.1016/s0223-5234(03)00115-6
17
ChenF. A.WuA. B.ChenC. Y. (2004). The influence of different treatments on the free radical scavenging activity of burdock and variations of its active components.Food Chem.86479–484. 10.1016/j.foodchem.2003.09.020
18
ChoJ. Y.KimA. R.YooE. S.BaikK. U.ParkM. H. (1999). Immunomodulatory effect of arctigenin, a lignan compound, on tumour necrosis factor-alpha and nitric oxide production, and lymphocyte proliferation.J. Pharm. Pharmacol.511267–1273. 10.1211/0022357991777001
19
da SilvaL. M.AllemandA.MendesD. A. G. B.dos SantosA. C.AndreE.de SouzaL. M.et al (2013). Ethanolic extract of roots from Arctium lappa L. accelerates the healing of acetic acid-induced gastric ulcer in rats: involvement of the antioxidant system.Food Chem. Toxicol.51179–187. 10.1016/j.fct.2012.09.026
20
de AlmeidaA. B.Sanchez-HidalgoM.MartinA. R.Luiz-FerreiraA.TrigoJ. R.VilegasW.et al (2013). Anti-inflammatory intestinal activity of Arctium lappa L. (Asteraceae) in TNBS colitis model.J. Ethnopharmacol.146300–310. 10.1016/j.jep.2012.12.048
21
de SouzaG. C.HaasA. P.von PoserG. L.SchapovalE. E.ElisabetskyE. (2004). Ethnopharmacological studies of antimicrobial remedies in the south of Brazil.J. Ethnopharmacol.90135–143. 10.1016/j.jep.2003.09.039
22
DiasM. M.ZuzaO.RianiL. R.de Faria PintoP.PintoP. L. S.SilvaM. P.et al (2017). In vitro schistosomicidal and antiviral activities of Arctium lappa L.(Asteraceae) against Schistosoma mansoni and Herpes simplex virus-1.Biomed. Pharm.94489–498. 10.1016/j.biopha.2017.07.116
23
DuhP. D. (1998). Antioxidant activity of burdock (Arctium lappa Linne): its scavenging effect on free-radical and active oxygen.J. t Am. Oil Chem. Soc.75455–461. 10.1007/s11746-998-0248-8
24
DumitrescuR.MehedintuC.BriceagI.PurcãreaV. L.HuditaD. (2015). Metformin-clinical pharmacology in PCOs.J. Med. Life8187–192.
25
EberdingA.MaderaC.XieS.WoodC. A.BrownP. N.GunsE. S. (2007). Evaluation of the antiproliferative effects of Essiac (TM) on in vitro and in vivo models of prostate cancer compared to paclitaxel.Nutr. Cancer Int. J.58188–196. 10.1080/01635580701328396
26
ErdemogluN.TuranN. N.AkkolE. K.SenerB.AbaciogluN. (2009). Estimation of anti-inflammatory, antinociceptive and antioxidant activities of Arctium minus (Hill) Bernh. ssp. minus.J. Ethnopharmacol.121318–323. 10.1016/j.jep.2008.11.009
27
European Medicines Agency (2011). Community Herbal Monograph on Actium lappa L., Radix. EMA/HMPC/246763/2009. Available: https://www.ema.europa.eu/documents/herbal-monograph/final-community-herbal-monograph-arctium-lappa-l-radix_en.pdf (accessed February 15 2019).
28
European Scientific Cooperative on Phytotherapy (2003). ESCOP Monographs: The Scientific Foundation for Herbal Medicinal Products.Exeter: European Scientific Cooperative on Phytotherapy.
29
European Scientific Cooperative on Phytotherapy-The Scientific Foundation for Herbal Medicinal Products (2016). ESCOP Monograph on Arctii radix (Burdock Root).Available: http://escop.com/downloads/arctii/ (accessed February 15 2019).
30
FerracaneR.GrazianiG.GalloM.FoglianoV.RitieniA. (2010). Metabolic profile of the bioactive compounds of burdock (Arctium lappa) seeds, roots and leaves.J. Pharm. Biomed. Anal.51399–404. 10.1016/j.jpba.2009.03.018
31
FierascuR. C.GeorgievM. I.FierascuI.UngureanuC.AvramescuS. M.OrtanA.et al (2018). Mitodepressive, antioxidant, antifungal and anti-inflammatory effects of wild-growing Romanian native Arctium lappa L.(Asteraceae) and Veronica persica Poiret (Plantaginaceae).Food Chem. Toxicol.11144–52. 10.1016/j.fct.2017.11.008
32
ForceL. H. T. (2001). The Longwood Herbal Task Force.Paris: Task Force.
33
FuchigamiM.KishigamiY.SasakiA. (1990). Pectic polysaccharides in edible burdock root.J. Home Econ. Jpn.41957–963. 10.11428/jhej1987.41.957
34
GaoH.LiG.ZhangJ.ZengJ. (2008). Arctigenin: a lignan from Arctium lappa.Acta Crystallogr. Sec. E Struct. Rep. Online64(Pt 8):o1538. 10.1107/S1600536808021752
35
GentilM.PereiraJ. V.SousaY. T.PietroR.NetoM. D.VansanL. P.et al (2006). In vitro evaluation of the antibacterial activity of Arctium lappa as a phytotherapeutic agent used in intracanal dressings.Phytother. Res.20184–186. 10.1002/ptr.1829
36
GrafE. (1992). Antioxidant potential of ferulic acid.Free Radic. Biol. Med.13435–448. 10.1016/0891-5849(92)90184-i
37
HaghiG.HatamiA.MehranM. (2013). UPLC and HPLC of caffeoyl esters in wild and cultivated Arctium lappa L.Food Chem.138321–326. 10.1016/j.foodchem.2012.10.040
38
HanB. H.KangY. H.YangH. O.ParkM. K. (1994). A butyrolactone lignan dimer from Arctium-Lappa.Phytochemistry371161–1163. 10.1016/s0031-9422(00)89550-3
39
HanZ. W.WangM. Z.WangL. Y.QuH.LiP.WangC. B. (2013). Chemical analysis of burdock root constituents.Asian J. Chem.252573–2576. 10.14233/ajchem.2013.13468
40
HeJ.HuangX. Y.YangY. N.FengZ. M.JiangJ. S.ZhangP. C. (2016). Two new compounds from the fruits of Arctium lappa.J. Asian Nat. Prod. Res.18423–428. 10.1080/10286020.2016.1145671
41
HsiehC.-J.KuoP.-L.HsuY.-C.HuangY.-F.TsaiE.-M.HsuY.-L. (2014). Arctigenin, a dietary phytoestrogen, induces apoptosis of estrogen receptor-negative breast cancer cells through the ROS/p38 MAPK pathway and epigenetic regulation.Free Radic. Biol. Med.67159–170. 10.1016/j.freeradbiomed.2013.10.004
42
HuangK.LiangX. C.ZhongY. L.HeW. Y.WangZ. (2015). 5-Caffeoylquinic acid decreases diet-induced obesity in rats by modulating PPARalpha and LXRalpha transcription.J. Sci. Food Agric.951903–1910. 10.1002/jsfa.6896
43
HuangX. Y.FengZ. M.YangY. N.JiangJ. S.ZhangP. C. (2015). Four new neolignan glucosides from the fruits of Arctium lappa.J. Asian Nat. Prod. Res.17504–511. 10.1080/10286020.2015.1039525
44
IchiharaA.KanaiS.NakamuraY.SakamuraS. (1978). Structures of Lappaol-F and Lappaol-H, dilignans from Arctium-Lappa L.Tetrahedron Lett.193035–3038. 10.1016/s0040-4039(01)94932-9
45
IchiharaA.NumataY.KanaiS.SakamuraS. (1977). New sesquilignans from Arctium-Lappa L - Structure of Lappaol-C, Lappaol-D, and Lappaol-E.Agric. Biol. Chem.411813–1814. 10.1271/bbb1961.41.1813
46
IchiharaA.OdaK.NumataY.SakamuraS. (1976). Lappaol-a and Lappaol-B, novel lignans from Arctium-Lappa L.Tetrahedron Lett.173961–3964. 10.1016/s0040-4039(00)92547-4
47
IchikawaK.KinoshitaT.NishibeS.SankawaU. (1986). The Ca2+ antagonist activity of lignans.Chem. Pharm. Bull.343514–3517. 10.1248/cpb.34.3514
48
IkedaM.SatoA.MochizukiN.ToyosakiK.MiyoshiC.FujiokaR.et al (2016). Phase I trial of GBS-01 for advanced pancreatic cancer refractory to gemcitabine.Cancer Sci.1071818–1824. 10.1111/cas.13086
49
JaiswalR.KuhnertN. (2011). Identification and characterization of five new classes of chlorogenic acids in burdock (Arctium lappa L.) roots by liquid chromatography/tandem mass spectrometry.Food Funct.263–71. 10.1039/c0fo00125b
50
JaricS.PopovicZ.Macukanovic-JocicM.DjurdjevicL.MijatovicM.KaradzicB.et al (2007). An ethnobotanical study on the usage of wild medicinal herbs from Kopaonik Mountain (Central Serbia).J. Ethnopharmacol.111160–175. 10.1016/j.jep.2006.11.007
51
JeelaniS.KhurooM. A. (2012). Triterpenoids from Arctium lappa.Nat. Prod. Res.26654–658. 10.1080/14786419.2010.541886
52
KardosovaA.EbringerovaA.AlfoldiJ.Nosal’ovaG.FranovaS.HribalovaV. (2003). A biologically active fructan from the roots of Arctium lappa L., var. Herkules.Int. J. Biol. Macromol.33135–140. 10.1016/S0141-8130(03)00079-5
53
KatoY.WatanabeT. (1993). Isolation and characterization of a xyloglucan from gobo (Arctium-Lappa L).Biosci. Biotechnol. Biochem.571591–1592. 10.1271/bbb.57.1591
54
KimB. H.HongS. S.KwonS. W.LeeH. Y.SungH.LeeI. J.et al (2008). Diarctigenin, a lignan constituent from Arctium lappa, down-regulated zymosan-induced transcription of inflammatory genes through suppression of DNA binding ability of nuclear factor-kappa B in macrophages.J. Pharmacol. Exp. Ther.327393–401. 10.1124/jpet.108.140145
55
KuoD. H.HungM. C.HungC. M.LiuL. M.ChenF. A.ShiehP. C.et al (2012). Body weight management effect of burdock (Arctium lappa L.) root is associated with the activation of AMP-activated protein kinase in human HepG2 cells.Food Chem.1341320–1326. 10.1016/j.foodchem.2012.03.023
56
KwonK.KoongH. S.KangK. H. (2016). Effect of burdock extracts upon inflammatory mediator production.Technol. Health Care24459–469. 10.3233/thc-151123
57
LansC.TurnerN. (2011). Organic parasite control for poultry and rabbits in British Columbia, Canada.J. Ethnobiol. Ethnomed.7:21. 10.1186/1746-4269-7-21
58
LansC.TurnerN.KhanT.BrauerG.BoeppleW. (2007). Ethnoveterinary medicines used for ruminants in British Columbia, Canada.J. Ethnobiol. Ethnomed.3:11. 10.1186/1746-4269-3-11
59
LeeB. I.NugrohoA.BachriM. S.ChoiJ.LeeK. R.ChoiJ. S.et al (2010). Anti-ulcerogenic effect and HPLC analysis of the caffeoylquinic acid-rich extract from Ligularia stenocephala.Biol. Pharm. Bull.33493–497. 10.1248/bpb.33.493
60
LeeJ. H.LeeJ. Y.ParkJ. H.JungH. S.KimJ. S.KangS. S.et al (2007). Immunoregulatory activity by daucosterol, a beta-sitosterol glycoside, induces protective Th1 immune response against disseminated Candidiasis in mice.Vaccine253834–3840. 10.1016/j.vaccine.2007.01.108
61
LeeJ. Y.KimC. J. (2010). Arctigenin, a phenylpropanoid dibenzylbutyrolactone lignan, inhibits type I-IV allergic inflammation and pro-inflammatory enzymes.Arch. Pharm. Res.33947–957. 10.1007/s12272-010-0619-1
62
LeonardS. S.KeilD.MehlmanT.ProperS.ShiX.HarrisG. K. (2006). Essiac tea: scavenging of reactive oxygen species and effects on DNA damage.J. Ethnopharmacol.103288–296. 10.1016/j.jep.2005.09.013
63
LewisW. H.Elvin-LewisM. P. F. (2003). Medical Botany: Plants Affecting Human Health.Hoboken, NJ: Wiley.
64
LiJ.LiuX.ZhouB.ZhaoJ.LiS. (2013). Determination of fructooligosaccharides in burdock using HPLC and microwave-assisted extraction.J. Agric. Food Chem.615888–5892. 10.1021/jf400534n
65
LinL. Z.HarnlyJ. M. (2008). Identification of hydroxycinnamoylquinic acids of arnica flowers and burdock roots using a standardized LC-DAD-ESI/MS profiling method.J. Agric. Food Chem.5610105–10114. 10.1021/jf802412m
66
LinS. C.ChungT. C.LinC. C.UengT. H.LinY. H.LinS. Y.et al (2000). Hepatoprotective effects of Arctium lappa on carbon tetrachloride- and acetaminophen-induced liver damage.Am. J. Chin. Med.28163–173. 10.1142/S0192415x00000210
67
LiuJ. Y.CaiY. Z.WongR. N. S.LeeC. K. F.TangS. C. W.SzeS. C. W.et al (2012). Comparative analysis of caffeoylquinic acids and lignans in roots and seeds among various burdock (Arctium lappa) genotypes with high antioxidant activity.J. Agric. Food Chem.604067–4075. 10.1021/jf2050697
68
LiuQ. D.QinK. M.ShenB. J.CaiH.CaiB. C. (2015). Optimization of the processing technology of fructus arctii by response surface methodology.Chin. J. Nat. Med.13222–231. 10.1016/S1875-5364(15)30008-X
69
LiuS. M.ChenK. S.SchliemannW.StrackD. (2005). Isolation and identification of arctiin and arctigenin in leaves of burdock (Arctium lappla L.) by polyamide column chromatography in combination with HPLC-ESI/MS.Phytochem. Anal.1686–89. 10.1002/pca.816
70
LiuW.WangJ. J.ZhangZ. Z.XuJ. N.XieZ. H.SlavinM.et al (2014). In vitro and in vivo antioxidant activity of a fructan from the roots of Arctium lappa L.Int. J. Biol. Macromol.65446–453. 10.1016/j.ijbiomac.2014.01.062
71
Lopez-VinyallongaS.ArakakiM.Garcia-JacasN.SusannaA.GitzendannerM. A.SoltisD. E.et al (2010). Isolation and characterization of novel microsatellite markers for Arctium minus (Compositae).Am. J. Bot.97e4–e6. 10.3732/ajb.0900376
72
LouC.ZhuZ.ZhaoY.ZhuR.ZhaoH. (2017). Arctigenin, a lignan from Arctium lappa L., inhibits metastasis of human breast cancer cells through the downregulation of MMP-2/-9 and heparanase in MDA-MB-231 cells.Oncol. Rep.37179–184. 10.3892/or.2016.5269
73
LouZ. X.LiC.KouX. R.YuF. H.WangH. X.SmithG. M.et al (2016). Antibacterial, antibiofilm effect of burdock (Arctium lappa L.) leaf fraction and its efficiency in meat preservation.J. Food Protect.791404–1409. 10.4315/0362-028x.J1t-15-576
74
LouZ. X.WangH. X.LvW. P.MaC. Y.WangZ. P.ChenS. W. (2010a). Assessment of antibacterial activity of fractions from burdock leaf against food-related bacteria.Food Control211272–1278. 10.1016/j.foodcont.2010.02.016
75
LouZ. X.WangH. X.ZhuS.ZhangM.GaoY.MaC. Y.et al (2010b). Improved extraction and identification by ultra performance liquid chromatography tandem mass spectrometry of phenolic compounds in burdock leaves.J. Chromatogr. A12172441–2446. 10.1016/j.chroma.2009.12.022
76
MachadoF. B.YamamotoR. E.ZanoliK.NocchiS. R.NovelloC. R.SchuquelI. T. A.et al (2012). Evaluation of the antiproliferative activity of the leaves from Arctium lappa by a bioassay-guided fractionation.Molecules171852–1859. 10.3390/molecules17021852
77
Maghsoumi-NorouzabadL.AlipoorB.AbedR.Eftekhar SadatB.Mesgari-AbbasiM.Asghari JafarabadiM. (2016). Effects of Arctium lappa L.(Burdock) root tea on inflammatory status and oxidative stress in patients with knee osteoarthritis.Int. J. Rheum. Dis.19255–261. 10.1111/1756-185X.12477
78
MaimaitiliA.ShuZ.ChengX.KaheermanK.SikandeerA.LiW. (2017). Arctigenin, a natural lignan compound, induces G0/G1 cell cycle arrest and apoptosis in human glioma cells.Oncol. Lett.131007–1013. 10.3892/ol.2016.5474
79
MarutaY.KawabataJ.NikiR. (1995). Antioxidative caffeoylquinic acid-derivatives in the roots of burdock (Arctium-Lappa L).J. Agric. Food Chem.432592–2595. 10.1021/jf00058a007
80
MatsumotoT.Hosono-NishiyamaK.YamadaH. (2006). Antiproliferative and apoptotic effects of butyrolactone lignans from Arctium lappa on leukemic cells.Planta Med.72276–278. 10.1055/s-2005-916174
81
MaxwellT.ChunS. Y.LeeK. S.KimS.NamK. S. (2017). The anti-metastatic effects of the phytoestrogen arctigenin on human breast cancer cell lines regardless of the status of ER expression.Int. J. Oncol.50727–735. 10.3892/ijo.2016.3825
82
MiglaniA.ManchandaR. K. (2014). Observational study of Arctium lappa in the treatment of acne vulgaris.Homeopathy103203–207. 10.1016/j.homp.2013.12.002
83
MilaniE.KoochekiA.GolimovahhedQ. A. (2011). Extraction of inulin from Burdock root (Arctium lappa) using high intensity ultrasound.Int. J. Food Sci. Technol.461699–1704. 10.1111/j.1365-2621.2011.02673.x
84
MingD. S.GunsE. S.EberdingA.TowersG. H. N. (2004). Isolation and characterization of compounds with anti-prostate cancer activity from Arictium lappa L. using bioactivity-guided fractionation.Pharm. Biol.4244–48. 10.1080/13880200490505474
85
MiyazawaM.YagiN.TaguchiK. (2005). Inhibitory compounds of α-glucosidase activity from Arctium lappa L.J. Oleo Sci.54589–594. 10.5650/jos.54.589
86
MkrtchianT. A.SnapianG. G.NikogosianG. A. (1998). Pectin production from burdock (Arctium sp.).Ukr Biokhim Zh7098–105.
87
MosaddeghM.NaghibiF.MoazzeniH.PiraniA.EsmaeiliS. (2012). Ethnobotanical survey of herbal remedies traditionally used in Kohghiluyeh va Boyer Ahmad province of Iran.J. Ethnopharmacol.14180–95. 10.1016/j.jep.2012.02.004
88
NabaviS. F.BraidyN.GortziO.Sobarzo-SanchezE.DagliaM.Skalicka-WoźniakK.et al (2015). Luteolin as an anti-inflammatory and neuroprotective agent: a brief review.Brain Res. Bull.1191–11. 10.1016/j.brainresbull.2015.09.002
89
NevesJ. M.MatosC.MoutinhoC.QueirozG.GomesL. R. (2009). Ethnopharmacological notes about ancient uses of medicinal plants in Tras-os-Montes (northern of Portugal).J. Ethnopharmacol.124270–283. 10.1016/j.jep.2009.04.041
90
OlennikovD. N.TankhaevaL. M. (2011). A quantitative assay for total fructans in burdock (Arctium spp.) roots.Russ. J. Bioorg. Chem.37893–898. 10.1134/S1068162011070181
91
ParkS. Y.HongS. S.HanX. H.HwangJ. S.LeeD.RoJ. S.et al (2007). Lignans from Arctium lappa and their inhibition of LPS-induced nitric oxide production.Chem. Pharm. Bull.55150–152. 10.1248/cpb.55.150
92
PereiraJ. V.BergamoD. C. B.PereiraJ. O.FrançaS. D. C.PietroR. C. L. R.Silva-SousaY. T. C. (2005). Antimicrobial activity of Arctium lappa constituents against microorganisms commonly found in endodontic infections.Braz. Dent. J.16192–196. 10.1590/s0103-64402005000300004
93
PhanT.-T.WangL.SeeP.GrayerR. J.ChanS.-Y.LeeS. T. (2001). Phenolic compounds of Chromolaena odorata protect cultured skin cells from oxidative damage: implication for cutaneous wound healing.Biol. Pharm. Bull.241373–1379. 10.1248/bpb.24.1373
94
PieroniA.GiustiM. E.QuaveC. L. (2011). Cross-cultural ethnobiology in the western balkans: medical ethnobotany and ethnozoology among albanians and serbs in the Pešter Plateau, Sandžak, South-Western Serbia.Hum. Ecol.39333–349. 10.1007/s10745-011-9401-3
95
PredesF. S.RuizA. L. T. G.CarvalhoJ. E.FoglioM. A.DolderH. (2011). Antioxidative and in vitro antiproliferative activity of Arctium lappa root extracts.BMC Compl. Altern. Med.11:25. 10.1186/1472-6882-11-25
96
QinK.LiuQ.CaiH.CaoG.LuT.ShenB.et al (2014). Chemical analysis of raw and processed fructus arctii by high-performance liquid chromatography/diode array detection-electrospray ionization-mass spectrometry.Pharmacogn. Mag.10541–546. 10.4103/0973-1296.141806
97
RajasekharanS. K.RameshS.BakkiyarajD.ElangomathavanR.KamalanathanC. (2015). Burdock root extracts limit quorum-sensing-controlled phenotypes and biofilm architecture in major urinary tract pathogens.Urolithiasis4329–40. 10.1007/s00240-014-0720-x
98
RodriguezP.BlancoJ.JusteS.GarcesM.PerezR.AlonsoL.et al (1995). Allergic contact-dermatitis due to burdock (Arctium-Lappa).Contact Dermat.33134–135. 10.1111/j.1600-0536.1995.tb00524.x
99
SalehN. A. M.BohmB. A. (1971). Flavonoids of arctium-minus (Compositae).Experientia27:1494. 10.1007/Bf02154314
100
Saric-KundalicB.DobesC.Klatte-AsselmeyerV.SaukelJ. (2010). Ethnobotanical study on medicinal use of wild and cultivated plants in middle, south and west Bosnia and Herzegovina.J. Ethnopharmacol.13133–55. 10.1016/j.jep.2010.05.061
101
SasakiY.KimuraY.TsunodaT.TagamiH. (2003). Anaphylaxis due to burdock.Int. J. Dermatol.42472–473. 10.1046/j.1365-4362.2003.01716_2.x
102
SavinaA. A.SheichenkoV. I.StikhinY. V.StikhinV. A.Sokol’skayaT. A.AnisimovaO. S.et al (2006). Sesquiterpene lactones in juice of great burdock leaves.Pharm. Chem. J.40624–626. 10.1007/s11094-006-0207-3
103
SchroderH. C.MerzH.SteffenR.MullerW. E.SarinP. S.TrummS.et al (1990). Differential in vitro anti-HIV activity of natural lignans.Z Naturforsch C451215–1221. 10.1515/znc-1990-11-1222
104
SezikE.YesiladaE.HondaG.TakaishiY.TakedaY.TanakaT. (2001). Traditional medicine in Turkey X. folk medicine in central anatolia.J. Ethnopharmacol.7595–115. 10.1016/s0378-8741(00)00399-8
105
SezikE.YesiladaE.ShadidoyatovH.KuliveyZ.NigmatullaevA. M.AripovH. N.et al (2004). Folk medicine in Uzbekistan. I. Toshkent, Djizzax, and Samarqand provinces.J. Ethnopharmacol.92197–207. 10.1016/j.jep.2004.02.016
106
SuS.ChengX. L.WinkM. (2015). Natural lignans from Arctium lappa modulate P-glycoprotein efflux function in multidrug resistant cancer cells.Phytomedicine22301–307. 10.1016/j.phymed.2014.12.009
107
SunQ.LiuK.ShenX.JinW.JiangL.SheikhM. S.et al (2014). Lappaol F, a novel anticancer agent isolated from plant Arctium Lappa L.Mol. Cancer Ther.1349–59. 10.1158/1535-7163.MCT-13-0552
108
TakasugiM.KawashimaS.KatsuiN.ShirataA. (1987). Studies on stress metabolites.5. 2 polyacetylenic phytoalexins from Arctium-Lappa.Phytochemistry262957–2958. 10.1016/S0031-9422(00)84570-7
109
TamayoC.RichardsonM. A.DiamondS.SkodaI. (2000). The chemistry and biological activity of herbs used in Flor-Essence (TM) herbal tonic and Essiac (TM).Phytother. Res.141–14. 10.1002/(sici)1099-1573(200002)14:1<1::aid-ptr580>3.3.co;2-f
110
TangY. X.LouZ. X.RahmanM. R. T.Al-HajjN. Q.WangH. X. (2014). Chemical composition and anti-biofilm activity of burdock (Arctium lappa L Asteraceae) leaf fractions against Staphylococcus aureus.Trop. J. Pharm. Res.131933–1939. 10.4314/tjpr.v13i11.23
111
TaofiqO.González-ParamásA. M.BarreiroM. F.FerreiraI. C. (2017). Hydroxycinnamic acids and their derivatives: cosmeceutical significance, challenges and future perspectives, a review.Molecules22281–304. 10.3390/molecules22020281
112
TardioJ.PascualH.MoralesR. (2005). Wild food plants traditionally used in the province of Madrid, central Spain.Econ. Bot.59122–136. 10.1663/0013-0001(2005)059%5B0122:wfptui%5D2.0.co;2
113
Ting-GuoK.Wen-JunZ.HitoshiT.TomokoK.Zhao-HuiX. U.Song-SongY.et al (2001). Pharmacognostical evaluation of arctii fructus (4) : chemical constituents from fruits ofarctium tomentosum(Natural Medicine Note).
 .Nat. Med.55:153.
.Nat. Med.55:153.114
TouschD.BidelL. P. R.CazalsG.FerrareK.LeroyJ.FaucanieM.et al (2014). Chemical analysis and antihyperglycemic activity of an original extract from burdock root (Arctium lappa).J. Agric. Food Chem.627738–7745. 10.1021/jf500926v
115
TurdumambetovK.BakirovG. A.RakhimovD. A. (2004). Glucofructans from arctium tomentosum roots.Chem. Nat. Compd.40211–214. 10.1023/B:CONC.0000039125.29910.a1
116
UchiyamaY.TagamiJ.KamisukiS.KasaiN.OshigeM.ChikuH.et al (2005). Selective inhibitors of terminal deoxyribonucleotidyltransferase (TdT): baicalin and genistin.Biochim. Biophys. Acta1725298–304. 10.1016/j.bbagen.2005.06.017
117
UmeharaK.SugawaA.KuroyanagiM.UenoA.TakiT. (1993). Studies on differentiation-inducers from arctium-fructus.Chem. Pharm. Bull.411774–1779. 10.1248/cpb.41.1774
118
WangB. S.YenG. C.ChangL. W.YenW. J.DuhP. D. (2007). Protective effects of burdock (Arctium lappa Linne) on oxidation of low-density lipoprotein and oxidative stress in RAW 264.7 macrophages.Food Chem.101729–738. 10.1016/j.foodchem.2006.01.051
119
WangH. Y.YangJ. S. (1993). Studies on the chemical constituents of Arctium lappa L.Yao Xue Xue Bao28911–917.
120
WangH. Y.YangJ. S. (1995). Neoarctin A from Arctium lappa L.Chin. Chem. Lett.6217–220.
121
WangR.AyanoH.FurumotoT.KondoA.FukuiH. (2001). Variation of the content of chlorogenic acid derivatives among cultivars and market items of burdock (Arctium lappa L.).Nippon Shokuhin Kagaku Kogaku Kaishi48857–862. 10.3136/nskkk.48.857
122
WangX.ChengC.YangY.ZhengC. (2004). Analysis of the essential oil from Arctium lappa L.Nat. Prod. Res. Dev.1633–35.
123
WangX.LiF. W.SunQ. L.YuanJ. P.JiangT.ZhengC. C. (2005). Application of preparative high-speed counter-current chromatography for separation and purification of arctiin from Fructus Arctii.J. Chromatogr. A1063247–251. 10.1016/j.chroma.2004.11.077
124
WangY.ZhangN.KanJ.ZhangX.WuX.SunR.et al (2019). Structural characterization of water-soluble polysaccharide from Arctium lappa and its effects on colitis mice.Carbohydr. Polym.21389–99. 10.1016/j.carbpol.2019.02.090
125
WashinoT.KobayashiH.IkawaY. (1987). Structures of Lappaphen-a and Lappaphen-B, new guaianolides linked with a sulfur-containing acetylenic-compound, from Arctium-Lappa-L.Agric. Biol. Chem.511475–1480. 10.1271/bbb1961.51.1475
126
WashinoT.YoshikuraM.ObataS. (1986). New sulfur-containing acetylenic-compounds from Arctium-Lappa.Agric. a Biol. Chem.50263–269. 10.1271/bbb1961.50.263
127
WuY.-C.LinL.-F.YehC.-S.LinY.-L.ChangH.-J.LinS.-R.et al (2010). Burdock essence promotes gastrointestinal mucosal repair in ulcer patients.Fooyin J. Health Sci.226–31. 10.1016/S1877-8607(10)60010-0
128
XuZ. H.GuC. C.WangK.JuJ. X.WangH. Y.RuanK. F.et al (2015). Arctigenic acid, the key substance responsible for the hypoglycemic activity of Fructus Arctii.Phytomedicine22128–137. 10.1016/j.phymed.2014.11.006
129
XuZ. H.JuJ. X.WangK.GuC. C.FengY. (2014). Evaluation of hypoglycemic activity of total lignans from Fructus Arctii in the spontaneously diabetic Goto-Kakizaki rats.J. Ethnopharmacol.151548–555. 10.1016/j.jep.2013.11.021
130
YangY. N.HuangX. Y.FengZ. M.JiangJ. S.ZhangP. C. (2015). New butyrolactone type lignans from arctii fructus and their antiinflammatory activities.J. Agric. Food Chem.637958–7966. 10.1021/acs.jafc.5b02838
131
YangY. N.ZhangF.FengZ. M.JiangJ. S.ZhangP. C. (2012). Two new neolignan glucosides from Arctii Fructus.J. Asian Nat. Prod. Res.14981–985. 10.1080/10286020.2012.729050
132
YayliN.YasarA.GulecC.UstaA.KolayliS.CoskuncelebiK.et al (2005). Composition and antimicrobial activity of essential oils from Centaurea sessilis and Centaurea armena.Phytochemistry661741–1745. 10.1016/j.phytochem.2005.04.006
133
YongM.KunG.QiuM. H. (2007). A new lignan from the seeds of Arctium lappa.J. Asian Nat. Prod. Res.9541–544. 10.1080/10286020600882577
134
YuB. S.YanX. P.XiongJ. Y.XinQ. (2003). Simultaneous determination of chlorogenic acid, forsythin and arctiin in Chinese traditional medicines preparation by reversed phase-HPLC.Chem. Pharm. Bull.51421–424. 10.1248/cpb.51.421
135
ZhaoC.SheT.WangL.SuY.QuL.GaoY.et al (2015). Daucosterol inhibits cancer cell proliferation by inducing autophagy through reactive oxygen species-dependent manner.Life Sci.13737–43. 10.1016/j.lfs.2015.07.019
136
ZhaoF.WangL.LiuK. (2009). In vitro anti-inflammatory effects of arctigenin, a lignan from Arctium lappa L., through inhibition on iNOS pathway.J. Ethnopharmacol.122457–462. 10.1016/j.jep.2009.01.038
137
ZhaoJ. L.EvangelopoulosD.BhaktaS.GrayA. I.SeidelV. (2014). Antitubercular activity of Arctium lappa and Tussilago farfara extracts and constituents.J. Ethnopharmacol.155796–800. 10.1016/j.jep.2014.06.034
138
ZhaoY.ZhouX. (2015). “Arctium lappa L.
 (Niubangzi, Great Burdock),” inDietary Chinese Herbs: Chemistry, Pharmacology and Clinical EvidenceedsLiuY.WangZ.ZhangJ. (Vienna: Springer Vienna) 301–305. 10.1007/978-3-211-99448-1_33
(Niubangzi, Great Burdock),” inDietary Chinese Herbs: Chemistry, Pharmacology and Clinical EvidenceedsLiuY.WangZ.ZhangJ. (Vienna: Springer Vienna) 301–305. 10.1007/978-3-211-99448-1_33139
ZhouX.ZhangH.GeL.GongH.TianS. (2011). Determination of arctiin and arctigenin contents in arctium tomentosum mill. by HPLC method.E J. Chem.8S372–S376. 10.1155/2011/517681
140
ZimmermannS.ThomiS.KaiserM.HamburgerM.AdamsM. (2012). Screening and HPLC-based activity profiling for new antiprotozoal leads from european plants.Sci. Pharm.80205–213. 10.3797/scipharm.1111-13
141
ZongG.ZhuJ.SunL.YeX.LuL.JinQ.et al (2013). Associations of erythrocyte fatty acids in the de novo lipogenesis pathway with risk of metabolic syndrome in a cohort study of middle-aged and older Chinese.Am. J. Clin. Nutr.98319–326. 10.3945/ajcn.113.061218
Summary
Keywords
Arctium species, secondary metabolites, volatile compounds, non-volatile compounds, chemodiversity, bioactivity
Citation
Wang D, Bădărau AS, Swamy MK, Shaw S, Maggi F, da Silva LE, López V, Yeung AWK, Mocan A and Atanasov AG (2019) Arctium Species Secondary Metabolites Chemodiversity and Bioactivities. Front. Plant Sci. 10:834. doi: 10.3389/fpls.2019.00834
Received
27 November 2018
Accepted
11 June 2019
Published
09 July 2019
Volume
10 - 2019
Edited by
Helen Skaltsa, National and Kapodistrian University of Athens, Greece
Reviewed by
Hiroshi Noguchi, Nihon Pharmaceutical University, Japan; Nishikant Wase, University of Nebraska–Lincoln, United States
Updates
Copyright
© 2019 Wang, Bădărau, Swamy, Shaw, Maggi, da Silva, López, Yeung, Mocan and Atanasov.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Dongdong Wang, dongdong.wang@univie.ac.atAndrei Mocan, mocan.andrei@umfcluj.roAtanas G. Atanasov, atanas.atanasov@univie.ac.at
†These authors have contributed equally to this work as co-first authors
This article was submitted to Plant Metabolism and Chemodiversity, a section of the journal Frontiers in Plant Science
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.