- 1Department of Surgery, Etablissement Hospitalier du Nord Vaudois eHnv, Yverdon-les-Bains, Switzerland
- 2Department of Pediatric Surgery, Centre Hospitalier Universitaire Vaudois CHUV, Lausanne, Switzerland
- 3Department of Gastro-enterology, Centre Hospitalier Universitaire Vaudois CHUV, Lausanne, Switzerland
Introduction: Duodenal duplication cysts (DDC) are rare congenital malformations which are generally diagnosed in the first decade of life. The clinical presentation of DDC is highly variable and may be complicated by pancreatitis. When pancreatic pseudocysts (PPC) develop, definitive DDC treatment is delayed and exposes the patient to recurrent episodes of pancreatitis which further lengthen the process. We present a novel approach to the management of such cases by using echo-endoscopic cystogastric drainage of a large retrogastric PPC as a bridge to surgery. To our knowledge, this is the youngest reported case.
Case: A 21-month-old girl presented with abdominal pain, bloating, vomiting and failure to thrive lasting for 3 months. Her prior medical history was normal.
Diagnosis, therapeutic intervention and outcomes: Blood work showed pancreatitis. Ultrasound (US) showed multiple cysts inside the abdomen. A thoraco-abdominal magnetic resonance imaging (MRI) scan allowed differentiation between multiple PPC and a DDC, which had caused a complicated obstructive pancreatitis. The DDC was confirmed by biopsies. Further imaging identified a large persistent retrogastric pseudocyst. Due to poor feeding and stable but compromised general condition, a two-step procedure was scheduled with echo-endoscopic cystogastric drainage of the large retrogastric PPC to reduce the convalescence time after the last episode of pancreatitis, followed by surgical resection of the DDC. The patient was released from the hospital the day after this procedure as oral intake had normalized. Unfortunately, 3 weeks after this procedure, the patient developed a septic shock due to infection of the remaining cysts. As surgery was required to treat the sepsis, the DDC was resected at the same time.
Conclusion: Echo-endoscopic cystogastric drainage is feasible and effective in children as young as 21 months. Pediatric guidelines have yet to be determined for this procedure.
1 Introduction
Digestive tract duplications are congenital malformations situated on the mesenteric side of the digestive tract, which share a common blood supply with the native bowel. They can occur anywhere from the mouth to the anus. 85% of them are diagnosed before the age of 2, with 60% before 6 months (1). Duodenal duplication cysts (DDC) represent 2%–12% of digestive tract duplications and have an estimated prevalence of 1/100,000 live births (2). Most DDCs occur in continuity with the second and third parts of the duodenum, sharing a common muscularis propria (3). Some occur as separate entities within or around the pancreas, between the inferior vena cava and portal vein, or adherent to the stomach (4, 5). Pancreatitis is a well-documented complication of DDCs. When pancreatic pseudocysts (PPC) complicate an episode of pancreatitis caused by a DDC, a vicious cycle can ensue where the surgical treatment of the DDC is delayed because of recurring episodes of pancreatitis. Challenges in these cases range from diagnosis to treatment.
This case is presented to share our experience with echo-endoscopic drainage of retrogastric PPC as a bridge to surgery in complicated cases of pancreatitis resulting from DDC. To our knowledge, this is the youngest case where this procedure has been used. This approach is uncommon in children, due to equipment size limitations, but is slowly gaining in popularity due to industry improvements (6).
2 Case report
2.1 Timeline of the main events
Day 1: We present the case of a 21-month-old girl who was brought to the emergency department of a regional hospital in Switzerland for acute abdominal pain in October of 2022 (Figure 1). Her previous medical history was normal, including during pregnancy. For four months, the patient had been suffering from isolated episodes of abdominal pain, distension and low-grade fever which were attributed to viral infections. The reason for the consultation in the emergency department was the refusal of solid food intake for 4 days which resulted in the loss of 400 g. The recent medical history showed no gastro-intestinal, respiratory, ENT or other systemic symptoms which could orient the diagnosis. There was also no history of abdominal or other trauma. The vital signs were as follows: HR 156/min, RR 32/min, Temperature 36.2°C, O2 saturation 97%, a weight of 10.2 kgs and normal hydration and perfusion status. The abdomen was distended, the auscultation was normal, and an uncomplicated umbilical hernia was present. Complete physical examination was otherwise normal. The initial blood work showed a normal white blood cell count (WBC), normal hemoglobin, and a C-Reactive Protein level (CRP) of 30.5 mg/L.
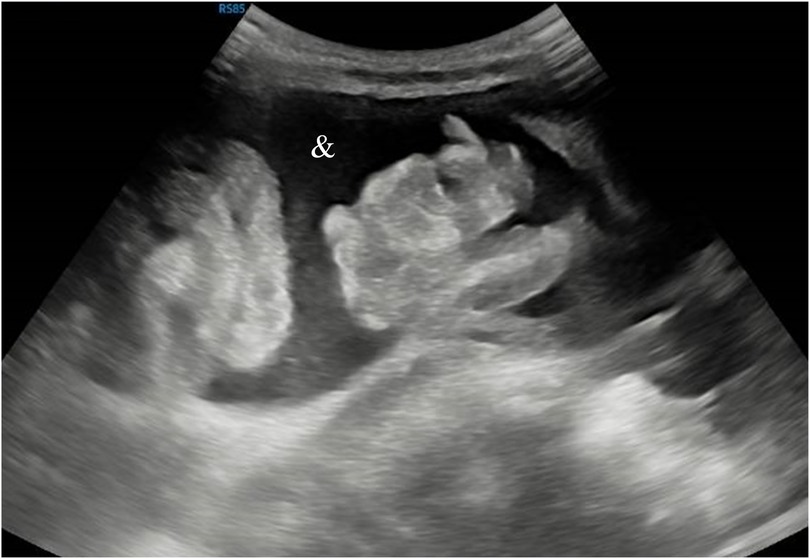
An abdominal ultrasound (US) (Figure 2) was performed which showed multiples cysts and ascites.
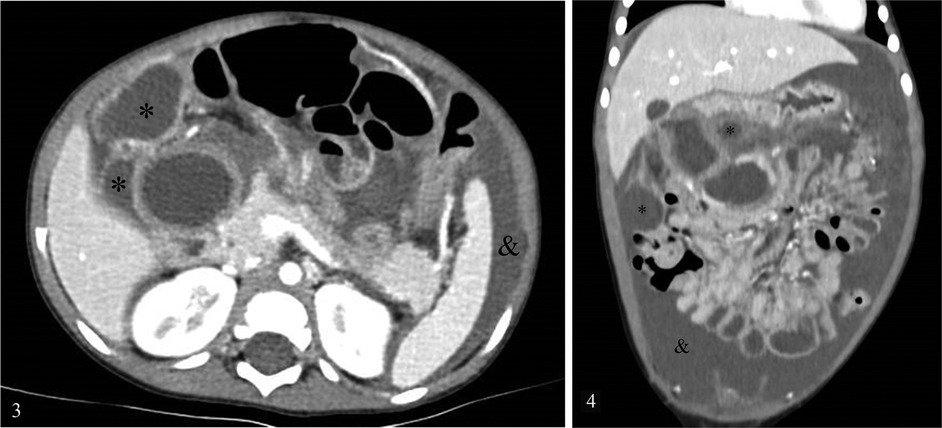
This was followed by a thoraco-abdominal computed tomography (CT) scan (Figures 3, 4) which confirmed the presence of multiple, large, abdominal cysts and free abdominal fluid. As cystic lymphangioma was suspected, the patient was transferred to a tertiary hospital center for etiologic diagnosis and treatment. Extensive blood work showed Lipase 667 UI/L, CRP 30 mg/L, normal liver function tests, and normal alpha-feto-protein and Beta-hCG levels. The criteria for pancreatitis were met and the patient was treated accordingly.
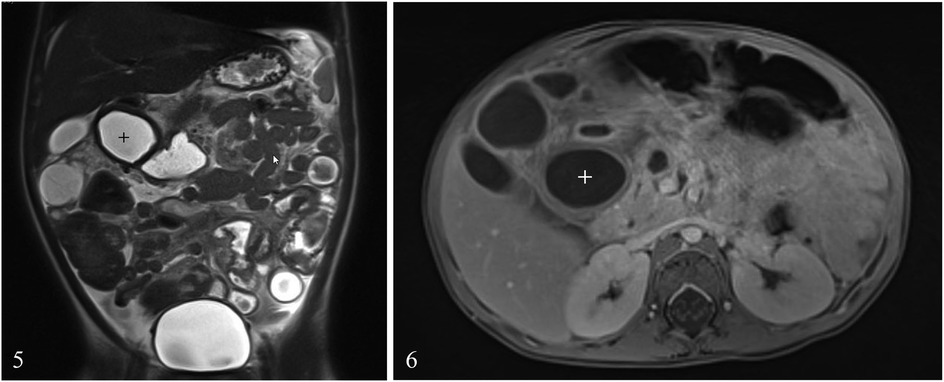
Day 4: A thoraco-abdominal magnetic resonance imaging (MRI) scan (Figures 5, 6) was performed which showed multiple thin-walled cysts and a single thick-walled paraduodenal cyst compatible with a DDC. A naso-jejunal feeding tube (NJFT) was placed and standard enteral nutrition (Nutrini®) was administered.
Day 5: As the initial differential diagnosis included cystic lymphangioma, an US-guided ascites and cyst aspiration was also performed. The culture was sterile and no malignant cells were present. The analysis of the fluid showed a normal triglyceride and WBC count, but lipase and pancreatic amylase were 1,159 UI/L and 863 UI/L respectively, suggesting that the thin-walled cysts were PPC. The double-walled cyst was then highly suspected to be a DDC. The patient then presented intercurrent rotavirus gastroenteritis delaying further investigations.
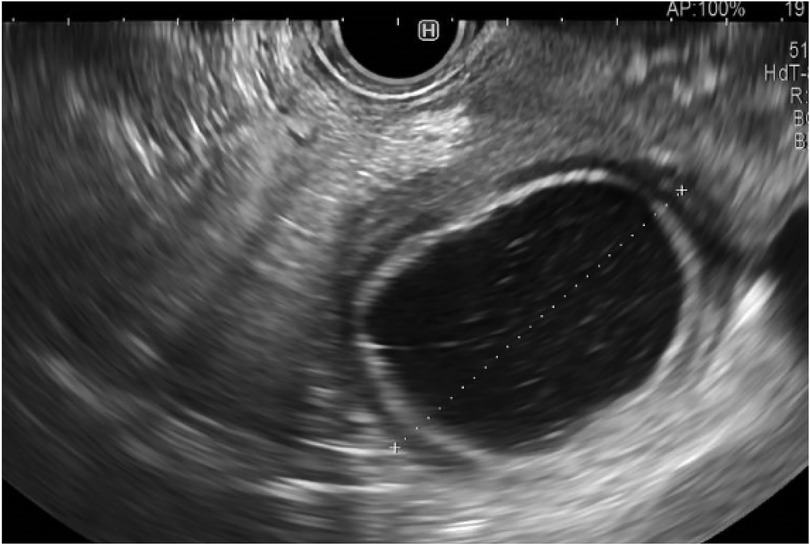
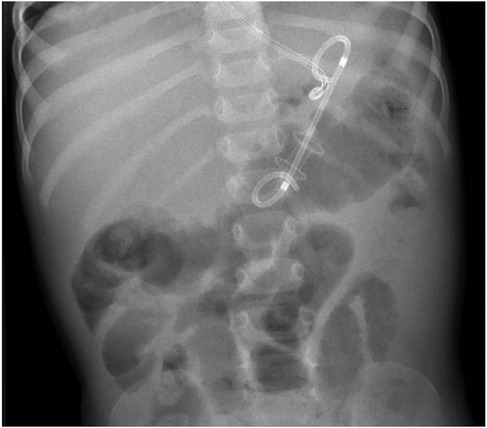
Day 22: Two weeks after admissions, as investigations into the etiologic diagnosis started again, the patient showed a stagnant weight curve, poor oral intake due to gastroparesis and low jejunal feeding tolerance. Along with biopsies of the suspected DDC, a drainage of the retrogastric PPC was decided to alleviate gastroparesis symptoms and speed-up recovery. The echo-endoscopy confirmed the presence of a single thick-walled DDC, signs of inflammation involving the pancreatic head and several thin-walled PPC, the largest being retrogastric. A Cystogastric drainage was performed using the smallest stent available, a Hot Axios 6 mm stent. A Pigtail 7Fr was placed through it (Figures 7, 8). The drainage revealed a white fluid which was positive for anaerobes. Biopsy of the suspected duplication cyst showed digestive epithelial lining confirming that the cyst was a DDC.
Day 23: Normal oral intake and enteral nutrition were well tolerated after this procedure. The patient was released from the hospital the day after the procedure with painkillers and ambulatory nutritional support via our dietician team. The removal of the stent was scheduled 6 weeks after its placement.
Day 30: At the 1-week follow-up consultation, the patient kept a stable weight but suffered from intermittent abdominal pain only during feeding. The dietary prescription was changed, and the patient went back home.
Day 48: The patient was hospitalized for abdominal pain and fever. The WBC was 9 G/L and CRP was 280 mg/L. A Norovirus was found in the stool test. The MRI scan showed no residual retrogastric PPC, but multiple infected PPC. A large spectrum antibiotic therapy was initiated.
Day 53: The patient developed a septic shock and presented increased signs of infection on the control MRI scan. A laparotomy was performed which found multiple infected pseudo-cysts and a 6 cm long, perforated, tubular, non-communicating, duodenal duplication, on the mesenteric side of D1. The dome of the cyst and the mucosa found on the common wall were excised and sent for analysis. The gastro-cystic stent, which had spontaneously migrated in the omental bursa, was removed and the wall of the stomach was closed. Lavage and drainage of the infected collections was performed before closure. The pathology report showed a DDC with typical duodenal wall characteristics. The antibiotics were stopped 10 days after surgery and the patient was released from the hospital on day 78.
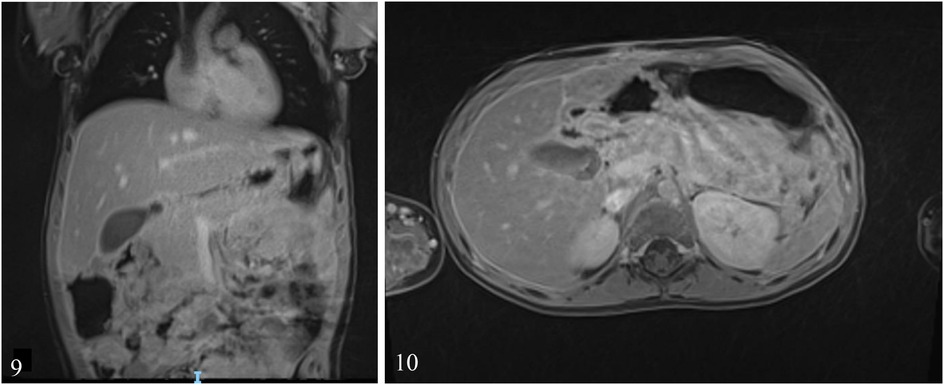
Day 151: Three months after surgery, a control echo-endoscopy was performed which showed no residual DDC. An MRI scan was also performed at the same time which also showed no residual DDC and no residual collections or pseudocysts (Figures 9, 10). At the 1-year follow-up consultation, the patient had caught-up on her growth curves and was thriving. The 1-year control US was normal.
Unfortunately, 15 months after surgery, the patient presented with adhesion-related small bowel obstruction which did not respond to conservative treatment. She had an exploratory laparotomy with extensive adhesiolysis and a short segmental small bowel resection. Clinical course was thereafter uneventful.
3 Discussion
Pancreatitis has an incidence of 1/10,000 children per year, and this rate is increasing (7). Anatomical variations should be suspected and investigated in children with recurrent acute pancreatitis. DDC are such anatomical variations and cause nonspecific symptoms such as abdominal pain, nausea and vomiting. Common complications include volvulus, intussusception, recurrent hemorrhage and perforation (8). Pancreatitis is the most reported complication of DDCs and is caused by 3 main mechanisms: (1) transient obstruction of the major papilla outflow by the nearby cyst due to peristalsis; (2) compression of the main pancreatic duct by a large cyst; and (3) obstruction of the pancreatic duct by biliary sludge, viscous mucous secretions or blood clots from the cyst (9). We suspect that the second mechanism caused the symptoms of our patient.
In our case, establishing the diagnosis was challenging. The CT scan showed only undifferentiable cysts. Although the MRI scan suggested a DDC, the definitive diagnosis was confirmed after the pathology results were obtained. Only then was surgical resection of the DDC planned. Regarding the imagery modalities used, we argue that for complex cases such as this, MRI scans, rather that CT scans, should be favored for the following reasons: (1) 5%–20% of children with pancreatitis have pancreato-biliary anomalies, for which MRI scans are the imaging gold standard (10); (2) MRI scans are equally effective as CT scans for severity scoring and follow-up of pancreatitis in children (11); (3) MRI scans provide detailed anatomical images of the DDC and its blood supply, facilitating surgical planning.
Surgical, endoscopic, and percutaneous methods have been described for the drainage of PPCs in pediatric patients. No specific guidelines compare these approaches in this population. Generally, adult guidelines are adapted for pediatric use. The 2019 European Society of Gastrointestinal Endoscopy (ESGE) guidelines recommend echo-endoscopic drainage of accessible PPCs in chronic pancreatitis, over percutaneous or surgical treatment (12). The American Gastroenterological Association (AGA) has not published specific guidelines for PPCs. However, their 2020 Clinical Practice Update on the management of pancreatic necrosis (13) aligns with the 2019 ESGE guidelines. It recommends echo-endoscopic drainage as the preferred initial method for PPCs, percutaneous drainage when endoscopic drainage is unavailable, unsuccessful or not feasible due to the risk of pancreaticocutaneous fistulae, and surgery as last resort for infected pancreatic necrosis or sterile pancreatic necrosis causing persistent organ dysfunction or failure to thrive.
Although echo-endoscopic drainage of retrogastric PPCs is commonly performed in adults, these procedures have recently been conducted in pediatric patients with good outcomes. A PubMed literature review of PPC managed with endoscopic drainage, using search terms including “PPC”, “walled-off necrosis”, “endoscopic ultrasound”, “cystogastric stent” and “pediatric”, yielded 211 results. Of these, 41 studies were relevant, primarily case reports and retrospective case series. The only meta-analysis, by Nabi et al., summarized the best case series (14). The youngest patient in these studies was 2 years old (15). Table 1 gives a brief summary of some of these studies.
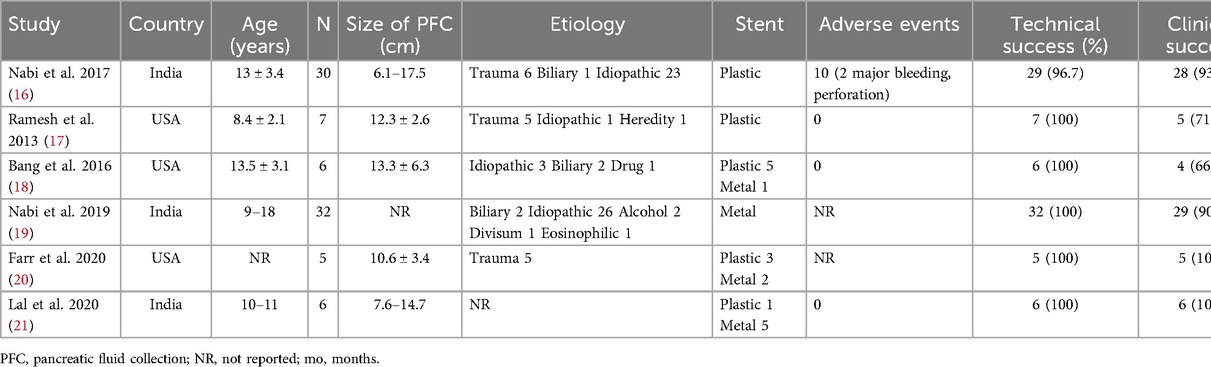
Table 1. Characteristics and outcomes of studies on Echo-endoscopic drainage of pancreatic fluid collections in children.
Complications of echo-endoscopic drainage of PPCs include bleeding, hematemesis, intestinal perforation, infection, and stent migration (16). External stent migration is generally benign, whereas internal migration, as in our case, requires endoscopic or surgical intervention. Double-pigtail stents are recommended to reduce migration. Metal stents are associated with higher bleeding risk. To reduce perforation risk, echo-endoscopy and fluoroscopy can be used (14).
In our case, a conservative approach to the retrogastric PPC was not feasible because the patient suffered from severe gastroparesis. Oral intake and NJFT were significantly reduced, slowing recovery and exposing the patient to recurrent episodes of pancreatitis. Surgery was not considered as a viable option because intra-abdominal inflammation due to pancreatitis increased the risk of complications, such as bleeding and bowel perforation. Percutaneous drainage was deemed unsuitable for our case as echo-endoscopy was readily available. Thus, echo-endoscopic drainage of the retrogastric PPC was planned as a bridge-to-surgery. The procedure was successful, markedly improving tolerance of oral intake and NJFT and significantly reducing abdominal discomfort. The patient was discharged the day after the procedure, while waiting for surgery.
We do not believe that the biopsy of the DDC or the ascites aspiration caused the septic shock, as they were performed 3 and 4 weeks prior, respectively. Additionally, the DDC perforation is unlikely to have caused the septic shock because the cyst was non-communicating. Instead, we hypothesize that the cystogastric stenting infected all the PPCs, as they were initially connected. Consequently, the septic shock may have resulted from one of two mechanisms. First, the migration of the stent impaired drainage. Alternately, as the PPC decreased in size over time, some became isolated and were no longer drained.
The preferred treatment for DDC is complete surgical resection when feasible to prevent malignant transformation (22). Partial resection or internal derivation is recommended if bilio-pancreatic structures are connected to the DDC (23). Endoscopic marsupialization, an emerging technique, has been described for partial cyst resection (24). In our case, due to the septic shock, surgery was performed.
Written informed consent was obtained from the patient's legal guardians for the publication of this case. When asked about their perspective, the guardians reported feeling relieved after months of uncertainty once the diagnosis and treatment plan were established. They also reported feeling heard by the medical and care teams. Their questions were answered in clear, jargon-free language, and they actively participated in the decision-making process. They also expressed frustration with initial pain management due to their daughter's high pain tolerance. They believed treatment delays could have been avoided with direct access to senior surgeons, the primary decision-makers, rather than consulting interns first, particularly on weekends.
4 Conclusion
The diagnosis and management of acute recurrent pancreatitis complicated by multiple PPCs caused by a DDC can be challenging. Cystogastric drainage of retrogastric PPCs is an effective and feasible bridge-to-surgery in children as young as 21 months old. It can speed up recovery and enable earlier surgical intervention for the DDC.
Due to the rarity of echo-endoscopic drainage of retrogastric PPCs in children, specific guidelines for this procedure are lacking. Rather, adult guidelines are adapted for pediatric cases. The increasing incidence of pediatric pancreatitis and the availability of pediatric echo-endoscopes and stents highlight the need for prospective studies. These should determine optimal timing, stent types, and PPC sizes requiring drainage.
Data availability statement
The original contributions presented in the study are included in the article, further inquiries can be directed to the corresponding author.
Ethics statement
Written informed consent was obtained from the minor(s)’ legal guardian/next of kin for the publication of any potentially identifiable images or data included in this article.
Author contributions
JP: Conceptualization, Data curation, Investigation, Methodology, Supervision, Writing – original draft, Writing – review & editing, Formal analysis, Project administration, Validation. NJ: Formal analysis, Methodology, Project administration, Supervision, Validation, Writing – review & editing. ND: Methodology, Supervision, Validation, Writing – review & editing. SG: Supervision, Writing – review & editing. SV: Methodology, Supervision, Validation, Writing – review & editing.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. All the funding was provided by the first author and the Department of Pediatric Surgery, Centre Hospitalier Universitaire Vaudois CHUV, Lausanne, Switzerland.
Acknowledgments
We would like to thank Dr. Sebastien GODAT for his expertise and experience on the case and case report.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Stern LE, Warner BW. Gastrointestinal duplications. Semin Pediatr Surg. (2000) 9(3):135–40. doi: 10.1053/spsu.2000.7565
2. Chen JJ, Lee HC, Yeung CY, Chan WT, Jiang CB, Sheu JC. Meta-analysis: the clinical features of the duodenal duplication cyst. J Pediatr Surg. (2010) 45(8):1598–606. doi: 10.1016/j.jpedsurg.2010.01.010
3. Liu R, Adler D. Duplication cysts: diagnosis, management, and the role of endoscopic ultrasound. Endosc Ultrasound. (2014) 3(3):152. doi: 10.4103/2303-9027.138783
4. Kawahara H, Takahashi T, Okada A. Characteristics of duodenal duplications causing pancreatitis in children and adolescents: a case report and review of the literature. J Pediatr Gastroenterol Nutr. (2002) 35(3):372–6. doi: 10.1002/j.1536-4801.2002.tb07840.x
5. Tantemsapya N, Chin A, Melin-Aldana H, Superina RA. Intrapancreatic duodenal duplication cyst as a cause of chronic pancreatitis in a child. Eur J Pediatr Surg. (2010) 20(02):125–8. doi: 10.1055/s-0029-1224192
6. Grover AS, Gugig R, Barakat MT. Endoscopy and pediatric pancreatitis. Gastrointest Endosc Clin N Am. (2023) 33(2):363–78. doi: 10.1016/j.giec.2022.11.002
7. Pohl JF, Uc A. Paediatric pancreatitis. Curr Opin Gastroenterol. (2015) 31(5):380–6. doi: 10.1097/MOG.0000000000000197
8. Anand S, Aleem A. Duplication Cyst. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing (2022). Available online at: https://www.ncbi.nlm.nih.gov/books/NBK564347/
9. Dipasquale V, Barraco P, Faraci S, Balassone V, De Angelis P, Di Matteo FM, et al. Duodenal duplication cysts in children: clinical features and current treatment choices. Biomed Hub. (2020) 5(2):1–13. doi: 10.1159/000508489
10. Uc A, Husain SZ. Pancreatitis in children. Gastroenterology. (2019) 156(7):1969–78. doi: 10.1053/j.gastro.2018.12.043
11. Trout AT, Anupindi SA, Freeman AJ, Macias-Flores JA, Martinez JA, Parashette KR, et al. North American society for pediatric gastroenterology, hepatology and nutrition and the society for pediatric radiology joint position paper on noninvasive imaging of pediatric pancreatitis: literature summary and recommendations. J Pediatr Gastroenterol Nutr. (2021) 72(1):151–67. doi: 10.1097/MPG.0000000000002964
12. Dumonceau JM, Delhaye M, Tringali A, Arvanitakis M, Sanchez-Yague A, Vaysse T, et al. Endoscopic treatment of chronic pancreatitis: European society of gastrointestinal endoscopy (ESGE) guideline—updated August 2018. Endoscopy. (2019) 51(02):179–93. doi: 10.1055/a-0822-0832
13. Baron TH, DiMaio CJ, Wang AY, Morgan KA. American gastroenterological association clinical practice update: management of pancreatic necrosis. Gastroenterology. (2020) 158(1):67–75.e1. doi: 10.1053/j.gastro.2019.07.064
14. Nabi Z, Talukdar R, Lakhtakia S, Reddy DN. Outcomes of endoscopic drainage in children with pancreatic fluid collections: a systematic review and meta-analysis. Pediatr Gastroenterol Hepatol Nutr. (2022) 25(3):251. doi: 10.5223/pghn.2022.25.3.251
15. Walsh LT, Groff A, Mathew A, Moyer MT. Endoscopic management of large peripancreatic fluid collections in two pediatric patients by endoscopic ultrasound-guided transmural drainage. Pediatr Gastroenterol Hepatol Nutr. (2020) 23(1):105–9. doi: 10.5223/pghn.2020.23.1.105
16. Nabi Z, Lakhtakia S, Basha J, Chavan R, Gupta R, Ramchandani M, et al. Endoscopic drainage of pancreatic fluid collections: long-term outcomes in children. Dig Endosc. (2017) 29(7):790–7. doi: 10.1111/den.12884
17. Ramesh J, Bang JY, Trevino J, Varadarajulu S. Endoscopic ultrasound–guided drainage of pancreatic fluid collections in children. J Pediatr Gastroenterol Nutr. (2013) 56(1):30–5. doi: 10.1097/MPG.0b013e318267c113
18. Bang JY, Varadarajulu S. Endoscopic treatment of walled-off necrosis in children: clinical experience and treatment outcomes. J Pediatr Gastroenterol Nutr. (2016) 63(3):e31–5. doi: 10.1097/MPG.0000000000001269
19. Nabi Z, Basha J, Lakhtakia S, Shava U, Pal P, Ramchandani M, et al. Disconnected pancreatic duct in children with walled OFF necrosis: impact on outcomes of endoscopic drainage. J Pediatr Gastroenterol Nutr. (2019) 69(1):116–9. doi: 10.1097/MPG.0000000000002367
20. Farr BJ, Fox VL, Mooney DP. Endoscopic cyst gastrostomy for traumatic pancreatic pseudocysts in children: a case series. Trauma Surg Acute Care Open. (2020) 5(1):e000456. doi: 10.1136/tsaco-2020-000456
21. Lal SB, Venkatesh V, Rana SS, Anushree N, Bhatia A, Saxena A. Paediatric acute pancreatitis: clinical profile and natural history of collections. Pancreatology. (2020) 20(4):659–64. doi: 10.1016/j.pan.2020.03.007
22. Ma H, Xiao W, Li J, Li Y. Clinical and pathological analysis of malignancies arising from alimentary tract duplications. Surg Oncol. (2012) 21(4):324–30. doi: 10.1016/j.suronc.2012.09.001
23. Merrot T, Anastasescu R, Pankevych T, Tercier S, Garcia S, Alessandrini P, et al. Duodenal duplications. Clinical characteristics, embryological hypotheses, histological findings, treatment. Eur J Pediatr Surg. (2006) 16(1):18–23. doi: 10.1055/s-2006-923798
Keywords: duodenal duplication cyst, echo-endoscopy, pancreatic pseudocyst, cystogastric stent, bridge to surgery, case report
Citation: Pacifique JX, Jauquier N, Divjak N, Godat S and Vasseur Maurer S (2025) Echo-endoscopic drainage of retrogastric pancreatic pseudocysts as a bridge-to-surgery for complicated cases of duodenal duplication cyst: case report. Front. Pediatr. 13:1588823. doi: 10.3389/fped.2025.1588823
Received: 6 March 2025; Accepted: 29 May 2025;
Published: 26 June 2025.
Edited by:
Di Li, First Hospital of Jilin University, ChinaCopyright: © 2025 Pacifique, Jauquier, Divjak, Godat and Vasseur Maurer. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Nicolas Jauquier, bmljb2xhcy5qYXVxdWllckBjaHV2LmNo
 Joseph Xavier Pacifique
Joseph Xavier Pacifique Nicolas Jauquier
Nicolas Jauquier Natalie Divjak
Natalie Divjak Sebastien Godat3
Sebastien Godat3 Sabine Vasseur Maurer
Sabine Vasseur Maurer