- 1World Vegetable Center, West and Central Africa, Coastal and Humid Regions, Cotonou, Benin
- 2Council for Scientific and Industrial Research - Crops Research Institute, Kumasi, Ghana
- 3Genetic Resources Unit, National Horticultural Research Institute, Ibadan, Nigeria
- 4World Vegetable Center Mexico Office hosted by International Maize and Wheat Improvement Center (CIMMYT), Carretera México-Veracruz, Texcoco, Mexico
Okra is a nutritious vegetable of global significance. This crop serves various purposes and presents numerous untapped opportunities. However, several challenges hinder Africa from fully harnessing okra’s economic and nutritional benefits, including pest and disease pressures, salinity and cold stress, weak seed systems, insufficient market knowledge, and a lack of value addition. A wealth of okra genetic resources is conserved in gene banks worldwide; however, only a handful have been characterized for agronomic and value-added traits, limiting their use in breeding programs. Although traditional breeding has focused on enhancing yield, disease resistance, and pod quality traits, which remain important, the growing demand for new okra-based products such as pectin, oil, snacks, and coffee substitutes offers new opportunities for farmers, processors, and breeders. Meeting these demands will require incorporating value-added traits into breeding programs, as identified in this review. Understanding the diversity of okra germplasm for value-added traits, their genetic architecture, and developing efficient screening methods is crucial for creating improved varieties that meet the needs of farmers, processors, and consumers. Enhancing the value-added traits of okra will increase production to meet the rising demand. We identified key traits of interest for improvement across different okra uses. Improving okra for biotic and abiotic challenges, as well as integrating horticultural and value-added traits, requires an in-depth understanding of the okra market to define and prioritize market segments along with target product profiles that fulfill their requirements, increased investment in okra breeding, collaboration between public and private seed and processing firms, resource sharing, and strengthening seed systems. Building capacity in okra breeding and seed technologies is critical to catalyzing okra improvement in Africa.
1 Introduction
Okra (Abelmoschus spp.) is a highly valued crop cultivated extensively in tropical and subtropical regions, recognized for its nutritional and culinary significance (Elkhalifa et al., 2021). The Asian cultivated species (A. esculentus (L.) Moench), also referred to as common okra (or Bhendi in India), is an allotetraploid (2n = 130-140), while the West African cultivated okra, A. caillei (A. Chev.) Stevels, is an amphipolyploid (allotetraploid, 2n = 196-200) between A. esculentus (2n = 130-140) and A. manihot (L.) Medik. (2n = 60-68) (Siemonsma, 1982; Ahiakpa et al., 2017). Okra is predominantly cultivated in Asia and Africa, with production also occurring in Southern Europe and the Americas, underscoring its global significance (Mangan et al., 2008; Sabitha et al., 2011). Okra is mainly grown for its immature pods, which can be consumed fresh or dried and incorporated into various dishes. Although less commonly utilized, okra seeds and leaves are also consumed (Adetuyi and Adelabu, 2011). Immature pods are rich in essential nutrients, including fiber, vitamins A and C, potassium, and iron, while the seeds contain 20% oil and high-quality protein, making them a valuable addition to the human diet (Gemede et al., 2015). Okra is considered a functional food due to its high content of mucilage and bioactive compounds, such as beta-carotene and ascorbic acid, which offer various health benefits (Adetuyi and Adelabu, 2011; Arlai et al., 2012; Ghori et al., 2014; Petropoulos et al., 2018; Dantas et al., 2021). Okra is a nutrient-dense vegetable; for instance, one cup (100 grams) of raw okra contains approximately 36 kilocalories, 8.20 grams of carbohydrates, and 2.10 grams of protein, while being low (0.2 grams) in fat (Adetuyi and Dada, 2014). Okra is exceptionally high in dietary fiber, which aids digestion and helps regulate blood sugar levels. Polyphenols, a type of antioxidant, may enhance okra’s health benefits by reducing oxidative stress and decreasing the risk of chronic diseases (Kendall and Jenkins, 2004; Tseng et al., 2004; Amin and Mahmood, 2011). In addition to its culinary uses, okra holds significant cultural relevance (Woldetsadik et al., 2022). In Nigeria, for instance, the consumption of okra leaves is linked to cultural beliefs about fertility and multiple births (Omonkhua et al., 2020).
Okra is well-suited for warm regions, with an ideal growth temperature of around 30°C, thriving throughout the year in various agro-ecological zones. The significance of okra goes beyond mere subsistence farming; it is vital for crop diversification and income generation for those involved in its value chains (Amin et al., 2023). The okra seed market is a growing sector, with a global value estimated at USD 215.8 million in 2024 and projected to reach USD 380 million by 2030 (Okra Seeds Market Report, 2025). Nonetheless, in Africa, cultivation is limited by minimal investment in breeding improved varieties, the prevalence of farmer seed systems providing seeds of variable quality, and pests and diseases that affect okra globally, resulting in low yields. Additionally, global okra breeding has largely overlooked value addition traits. To address these challenges, it is essential to develop breeding programs that align with consumer preferences, cropping systems, and environmental issues. Current knowledge regarding okra production, its economic significance, and breeding goals in Africa is limited and not well-documented. This lack of comprehensive information hinders understanding of the crop’s importance and the associated challenges and prospects for its improvement. Consolidating and discussing existing information on okra will support investment in improving the crop in Africa. Extensive reviews are focusing on okra breeding (Mishra et al., 2017; Dhankhar and Koundinya, 2020; Mishra et al., 2021; Singh et al., 2023), as well as its uses, properties, and health benefits (Gemede et al., 2015; Dantas et al., 2021; Elkhalifa et al., 2021). This review contributes to the current body of research on okra by summarizing its production and economic value in Africa and examining the breeding needs related to various uses of the crop. Additionally, we mapped existing okra breeding programs and suggested strategies for okra improvement in Africa.
2 Cultivated species in Africa
The Asian cultivated species (A. esculentus), commonly known as lady’s finger or okra, and referred to as Bhendi in India, has been reported as an amphidiploid with varying chromosome number (2n = 130-140), 2n = 130 being the most frequently reported chromosome number (Singh et al., 2023). Recent genomic information confirmed the allotetraploid nature of A. esculentus, with two sub-genomes: one containing 30 chromosomes and another comprising 35 chromosomes (Wang et al., 2023; Nieuwenhuis et al., 2024). For A. moschata, a full-length transcriptome has been produced (Guangying et al., 2023). The West African cultivated okra (A. caillei) is an amphipolyploid (allotetraploid) with 2n = 194 chromosomes (Singh and Bhatnagar, 1975). This species arose from the cross between A. esculentus (2n = 130-140) and A. manihot (2n = 60-68) (Siemonsma, 1982). A. caillei, or West African okra, is reportedly well-suited to the growing conditions in West and Central Africa, where it is cultivated as a subsistence crop. It is a short-day crop that is sensitive to photoperiodism (Kumar et al., 2010), which limits its adaptability to intensive cultivation conditions. Earliness has become vital for farmers to adapt to their cropping systems and climate change and respond to market demand. The introduction of higher-yielding A. esculentus varieties has reduced the land area allocated to A. caillei, which is now primarily found in home gardens, accounting for less than 5% of the total okra cultivation area. A. caillei has a longer harvest duration and drought tolerance, which can be transferred to A. esculentus, an essential trait for okra growers. However, considering its adaptation to growing conditions, beyond using A. caillei as a source of traits for A. esculentus improvement (Kumar et al., 2010), this species can also benefit from efforts to improve its adaptation to commercial production systems. Gathering market information on consumer acceptability of A. caillei will guide decision-making on investing in its improvement. Key traits requiring improvement in A. caillei to fit into commercial production systems include photo-insensitivity, earliness, and plant architecture. Additionally, due to its large leaves and extended life cycle, A. caillei can be utilized as a dual-purpose crop in communities where both leaves and pods are widely consumed.
3 Okra uses and breeding implications
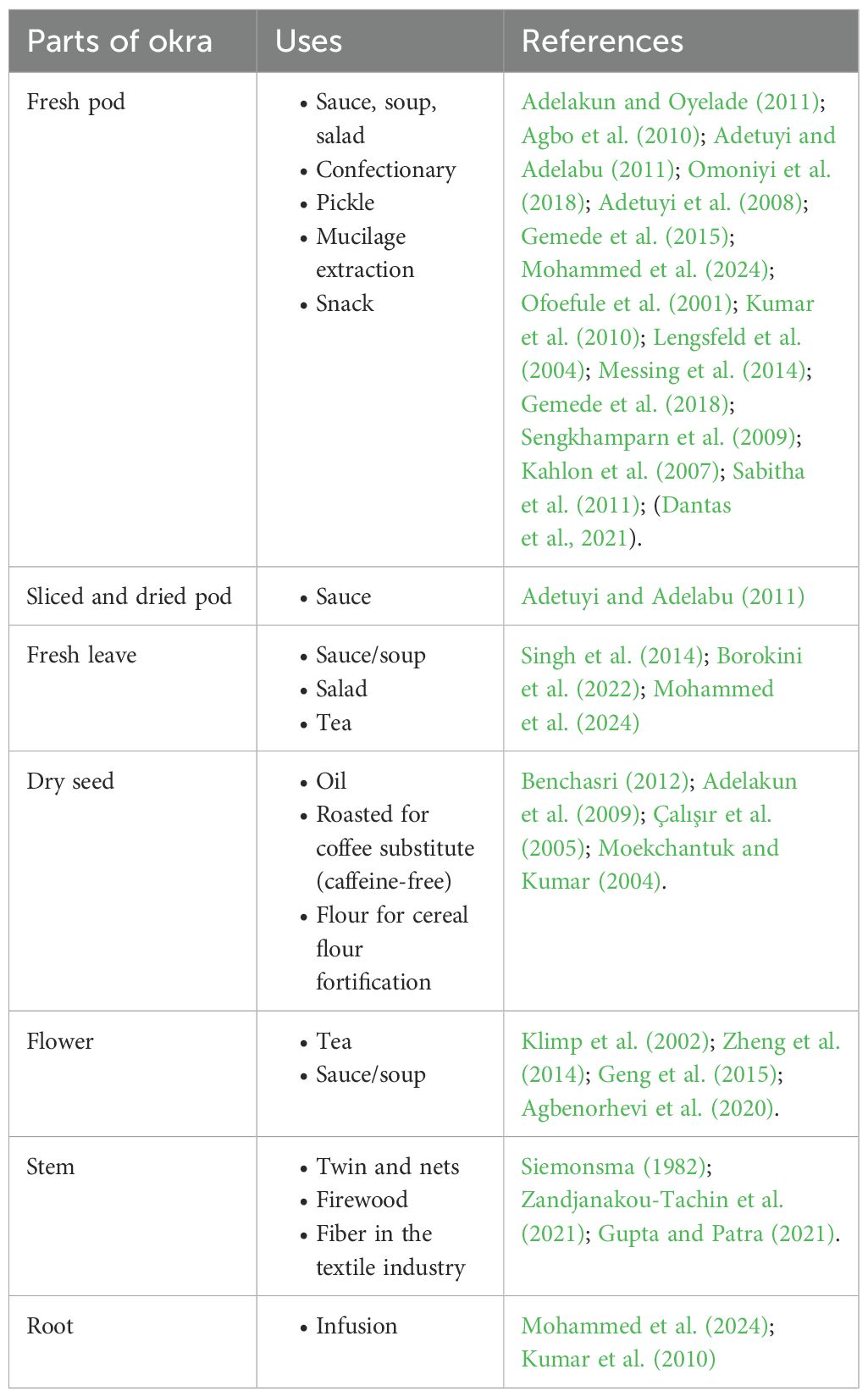
Okra is a multipurpose crop. All the plant parts are used for human consumption or in the non-food industry (Table 1). Fresh pods are widely consumed in various forms, such as salads, soups, and stews (Salameh, 2014). The fresh pods are also used for confectionery (Gemede et al., 2015), pickles, and snacks (Gemede et al., 2015). Mucilage is extracted from fresh pods for various uses, including edible films, additives in food formulations, and excipients in the pharmaceutical industry due to its stabilizing, emulsifying, and thickening properties (de Alvarenga Pinto Cotrim et al., 2016; Dantas et al., 2021; Kalkan and Maskan, 2023). Okra pods are amenable to drying to increase storability and reduce shipping costs. Dried okra sauce is widely consumed in West Africa (Kumar et al., 2010). Okra leaves are used fresh or dried for sauces in many communities in West Africa Omonkhua et al., 2020; Borokini et al., 2022). Okra is used in folk medicine. A decoction of the immature capsules is demulcent, diuretic and emollient (Kumar et al., 2013). Okra mucilage has medicinal applications and can be used as a plasma replacement or blood expander due to its ability to increase blood volume (Akinyele and Temikotan, 2007). In Nepal, the juice of the root is used externally to treat cuts, wounds, and boils. It is also considered a suitable coagulant for industrial wastewater treatment (Freitas et al., 2015).
Mature okra fruits and stems are utilized in the paper industry, while the roots are used to prepare jaggery (Gupta et al., 2021). Seeds are roasted and ground and used as a substitute for coffee (Moekchantuk and Kumar, 2004; Gupta and Patra, 2021).
While immature okra pods are primarily cooked, there are diverse uses in the food, pharmaceutical, and non-food industries. Okra breeding programs have traditionally focused on improving yield, resistance to pests and disease, and pod quality for cooking with less emphasis on value added traits (Mishra et al., 2021; Singh et al., 2023). Expanding the okra market and tapping into its diverse uses require the integration of value-added traits into breeding objectives.
3.1 Fresh pods for food and increased nutrients
Breeding okra for enhanced nutritional quality and yield is essential for addressing food and nutrition security, particularly in sub-Saharan Africa, where mineral deficiencies are prevalent (Alake, 2020). Okra is recognized for its high dietary fiber content and rich profile of essential nutrients, including vitamins and minerals such as calcium, potassium, and magnesium (Gemede et al., 2016; Karmakar et al., 2022). However, anti-nutritional factors like phytic acid can hinder nutrient absorption, necessitating breeding efforts aimed at reducing antinutrient compounds while nutrient levels (Adetuyi and Adelabu, 2011). Genetic improvement of okra can yield varieties with potential to significantly improve the nutritional quality of diets and promote public health (Alake, 2020). Previous research has primarily focused on the genetic diversity underlying the nutritional content of okra in various regions worldwide. For instance, Gerrano (2018) conducted a genetic diversity study based on nutritional traits on 50 accessions obtained from the World Vegetable Center, identifying genotypes VI056457, VI033796, VI060824, VI060802, VI055423, and VI049632 as the most genetically distinct. Similarly, Srivastava et al. (2023) reported that genotypes IC169472 and IC169453 from India exhibited high levels of iodine, crude protein, crude fiber, sodium, and total soluble solids. Kumar et al. (2016) identified genotypes USDO-2546, Punjab Padmini, and Arka Abhay as promising founder lines for breeding high nutritional-quality cultivars. Moreover, Kolawole et al. (2022) identified accessions NHOK-0165, NHOK-0171, NHOK-0188, NHOK-0418, NHOK-0462, NHOK-0544, NHOK-0601, NHOK-0602, NHOK-0622, NHOK-0623, and NHOK-0635 as having desirable agronomic traits along with favorable mineral and proximate composition based on a variability study conducted on 29 okra accessions from the National Horticultural Research Institute genebank in Nigeria. These findings suggest that the genetic diversity within okra germplasm provides a valuable resource for developing cultivars that meet market demands while contributing to improved public health outcomes (Fujimoto et al., 2018). Understanding the genetic architecture of quality traits is crucial for advancing breeding programs (Fayad, 2021). However, there remains a significant gap in research focusing on the genetic control of key biochemical compounds related to okra’s nutritional quality. Furthermore, the influence of environmental factors and agronomic practices on the nutritional content of okra remains to be elucidated.
Varietal differences in protein content have been reported for okra, and this trait can be targeted to enhance the nutritional value of okra (Oyelade et al., 2003). Okra is rich in methionine and tryptophan, making it a good complement to improve the quality of legume- and cereal-based diets. The highest proportion of lipids and proteins in okra is found in the seeds, whether fresh or dry. Understanding whether there is an association between seed content and nutritional content in okra can provide a quick and easy way to phenotype for high-nutrient content. If such an association is established, increasing seed content in okra could increase the crop’s nutritional value. While increased seed content could be desirable for dried and ground okra, consumer acceptability for high seed content, especially where fresh okra pods are used, would need to be understood and factored into the breeding process.
3.2 Fresh pods for mucilage extraction
High mucilage content is a must-have trait for okra varieties in West Africa, unlike in many other okra production regions (Kumar et al., 2010; Ahiakpa et al., 2014; Agbenorhevi et al., 2020). The main components of okra mucilage polysaccharides are mannose, rhamnose, glucuronic acid, glucose, arabinose, galacturonic acid, galactose, and xylose (Gao et al., 2018; Wang et al., 2018; Elkhalifa et al., 2021). Breeding programs do not traditionally target high mucilage content (Singh et al., 2023). Besides being a preferred trait in West Africa, powdered high-mucilage okra can be utilized in industrial wastewater treatment as a natural coagulant, removing turbidity, color, and chemical oxygen demand, reducing the environmental impact of industrial processes (Gemede et al., 2018). To address this, breeders should prioritize traits like pod size, mucilage content, and ease of processing, selecting accessions with longer, thicker pods that are rich in mucilage. Few studies have examined variability in okra germplasm collections for mucilage content. Ahiakpa et al. (2014) identified accessions DKA, Amanfrom, Asontem NV, Yeji-Local, and Kortebortor-BAR as having higher mucilage content out of 21 West African okra accessions. (Gemede et al., 2018) reported that accession OPA#7 exhibited the highest mucilage contents in Ethiopia. However, our current knowledge of the genetic architecture of mucilage content in okra is limited. In-depth investigations combining biochemical and genomic approaches are necessary to identify the genetic determinants underlying variations in mucilage rate (Fayad, 2021).
Additionally, it is crucial to assess the environmental influence on mucilage production. The current method for quantifying mucilage content using a viscometer is destructive, time-consuming, and variable, depending on the extraction method and conditions (Ahiakpa et al., 2014; Alba et al., 2015; Agbenorhevi et al., 2020). Extensive screening of germplasm collections and routine phenotyping of breeding materials for this trait will require the development of user-friendly, fast, reliable, and cost-effective methods. Okra mucilage is made up of a complex mixture of polysaccharides, including pectin, cellulose, and hemicelluloses such as xylan and xyloglucan (Sengkhamparn et al., 2009). Pectin is the main polysaccharide responsible for the viscous texture of okra mucilage (Kpodo et al., 2017). The highest mucilage yield (17.65%) and intrinsic viscosity value (11.9 dL/g) were recorded in intermediate-matured okra pods (14–15 days after flowering) and immature pods, respectively (Agbenorhevi et al., 2020). Exploring the possibility of developing a near-infrared reflectance spectroscopy (NIRS) calibration model to screen for mucilage content accurately will improve breeding efficiency for this trait in okra. Near-infrared hyperspectral imaging was used to quantify pectin content in orange peel non-destructively (Badaró et al., 2020), in mulberry fruit (Yang et al., 2021), and to quantify flavonoid content in okra (Cui et al., 2023). The application of near-infrared spectroscopy to quantify pectin content in other crops and compounds in okra suggests that it can be deployed to assess pectin content in okra.
3.3 Fresh pods for snack production
Developing okra varieties with market-preferred characteristics is crucial to meet this demand. Mid-sized pods (7 to 10 cm) are particularly well-suited for snack production, as they are smooth and bright green (Abba et al., 2020). Fruit length and pod texture are important traits in okra varieties for adaptation to snack production (Singh et al., 2023). Previous research has identified several promising okra accessions with traits needed for snack production. For instance, Temam et al. (2021) reported accession 29622 as having smooth, green pods. Similarly, Reddy et al. (2016) identified multiple accessions with smooth pod textures, including RNO-201 to RNO-205, RNO-207, RNO-208, RNO-210, RNO-211, RNO-213, and RNO-217 to RNO-220. Regarding fruit length, Binalfew and Alemu (2016) identified accessions 240201-C, 240203-A, 240203-B, 240203-D, 240203-F, 240586-A, 240587-B, and 240592-A as having small fruits. Kumari et al. (2017) further identified the following accessions as possessing small fruit sizes: Pusa Makhamali, VRO-6, Kashi Mohini, Pusa Sawani, Punjab-8, SB-2, 307-10-1, Kashi Satadhari, CO-3, VROB-178, Arka Amanika, IBS-02, Azad Blindi-1, and VRO-106. To date, an okra variety possessing all the desired traits for snack production remains elusive. To address this gap, we propose utilizing the accessions identified in previous studies as parents for hybridization (Yadav et al., 2024). This approach will facilitate the development of hybrids that align with market demands. Additionally, conducting a genome-wide association study (GWAS) for key traits of okra in snack production could help unravel the genetic basis of these traits (Fujimoto et al., 2018).
3.4 Seed for oil production
The global demand for vegetable oils is steadily increasing, driven by population growth and the expansion of industrial applications (Hossain et al., 2001; Abba et al., 2020). The cultivation of okra holds potential to significantly contribute to the vegetable oil market, particularly in Asia and Africa. Okra seeds contain comparable edible oil (13.0% to 38.1%) to soybean (17.0%–21.0%), cotton (15.0%–24.0%) and safflower (27.0%–41.0%) (Ergönül and Özbek, 2020; Kammili and Yadav, 2022). Okra oil shares similarities with cottonseed oil, particularly as a good source of unsaturated linoleic acid, which ranges from 32.22% to 43.07% (Liu et al., 2021). The gossypol content in a set of 26 okra accessions was lower (< LOQ-62.459 mg/kg) than the international limit (Kantar et al., 2024), suggesting okra oil is edible. Okra seed is rich in palmitic acid, with the highest value recorded in A. esculentus (30.42%), oleic acid (31.885% in A. manihot), and linoleic acid (49.638% in A. tuberculatus) (Jarret et al., 2011).
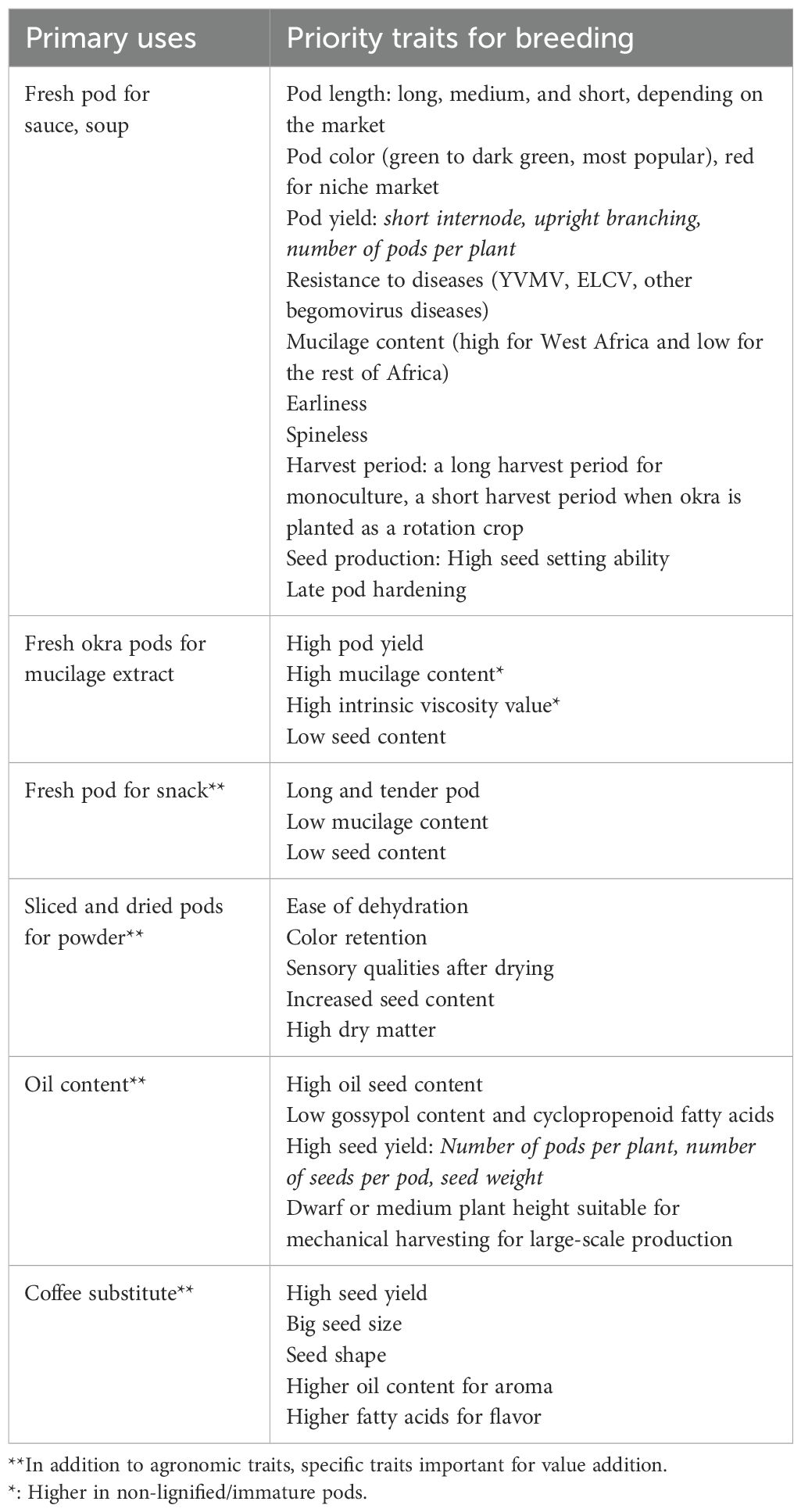
The growing interest in sustainable and nutritious oils highlights the need for breeding programs to improve the oil content and quality of okra seeds. Specific traits of interest in breeding for oil content in okra include seed number per pod, seed yield, and oil content per seed (Zibelo et al., 2016; Kumar et al., 2017; Mohammed et al., 2022) (Table 2).
Additionally, Kantar et al. (2024) reported several high-oil-content landraces from Turkey, and Jarret et al. (2011) evaluated about 1,100 okra accessions of seven species and found that oil content ranged from 2.51 to 23.2%, with the highest oil content recorded in A. tuberculatus accession PI639680 (23%). High oil content was also recorded in accessions PI274350 (21.5%), PI538082 (20.9%), and PI538097 (20.9%), suggesting the potential of the cultivated okra as an oil source (Jarret et al., 2011).
The differences in oil content and profile observed in previous studies (Jarret et al., 2011; Sami et al., 2013; Kantar et al., 2024) provide pieces of evidence to support the development of new cultivars with higher oil content, which could improve the potential of okra as an oil crop. Because okra has not been traditionally grown for its grains, yield evaluation data are limited. However, there are a few studies where grain yield was quantified. There was a wide variation in grain yield, ranging from 3.39 to 5.87 t/ha (Makinde, 2014), 0.368 to 0.684 t/ha (Chattopadhyay et al., 2011), and 0.303 t/ha to 2.3 t/ha (Mohammadi et al., 2016). The reported differences likely depend on growing conditions, genotypes, and agronomic practices.
Overall, a concerted effort to select for traits such as seed quantity, oil percentage, oil functional and nutritional properties is essential in developing new okra varieties that meet consumer preferences and enhance agricultural sustainability and food security. To further advance okra breeding for oil production, a comprehensive characterization of okra germplasm collections for oil content and seed yield is essential to identify accessions with significant oil content potential. Seed oil yield is a complex trait, and a better understanding of its component traits and their genetic architecture is crucial for designing breeding strategies to improve it.
3.5 Seed as a coffee substitute
The potential of okra as a coffee substitute, due to its caffeine-free nature, has garnered increasing attention, with several studies highlighting the need for targeted breeding programs to enhance specific traits critical for optimal roasting quality and flavor. Gulsen et al. (2007); Kumar et al. (2023), and Kumar et al. (2010) emphasized the importance of traits such as seed yield, size, shape, and oil content for improving roasting efficiency and flavor profile (Table 2). However, current breeding programs have not sufficiently focused on developing varieties tailored to the emerging coffee substitute market (Kumar et al., 2023), indicating a significant research gap that needs to be addressed to meet the growing consumer demand for innovative beverage options.
The success of enhancing okra production through value addition largely relies on processing companies. Value addition in okra will require developing varieties that meet the industry’s specific needs, whether for mucilage extraction, processing, or seed for oil extraction. Table 2 summarizes key essential traits for value addition in okra. The priority traits identified here should serve as a starting point for a more refined prioritization, depending on the context. The decision to integrate valued-added traits into a breeding pipeline necessitates further prioritization exercises, including market surveys and consultations with okra value chains stakeholders.
4 Okra improvement for biotic stresses
Worldwide, one of the most devastating diseases is Yellow Vein Mosaic Disease (YVMD), transmitted by the whitefly (Bemisia tabaci), which can cause yield losses ranging from 50% to 100%, depending on the timing of infection during the plant’s growth stages (Jose and Usha, 2003; Fauquet and Stanley, 2005). However, in Africa, Enation Leaf Curl Virus (ELCV) is the most serious viral threat to okra production, with yield penalties of up to 100% (Yadav et al., 2018). In Burkina Faso, a yield loss of up to 55% due to okra leaf curl disease was reported in a local variety, resulting in a financial loss of $11,100 per hectare (Tiendrébéogo et al., 2010b). Insect pests, particularly whiteflies, jassids, and aphids, exacerbate these issues by facilitating viral transmission and directly damaging the plants (Rashida et al., 2005; Kumari et al., 2020). Moreover, root-knot nematodes (Meloidogyne spp.), cercosporiose caused by Cercospora malayensis and Pseudocercospora abelmoschii (Armand et al., 2013), and powdery mildew caused by the air-borne fungus Erysiphe cichoracearum (Moharam and Ali, 2012) are known to cause significant damage to okra roots, leading to stunted growth and reduced yields (Suma et al., 2023).
Abang et al. (2014); Abang et al. (2018), and Abang et al. (2024) screened okra accessions for resistance to aphids under various conditions and identified VI033805, VI036213, and VI051114 (Abang et al., 2014), and VI033824 (Abang et al., 2021) as resistant to aphids. A. esculentus was overall less attacked than A. caillei accessions. The yield of resistant accessions remained very low (less than 2 t/ha) (Abang et al., 2021). Accession VI036213, with a low yield, demonstrated stable resistance to aphids across agroecologies in Central Africa (Abang et al., 2024) and could be a potential donor of aphid resistance.
Jassid infestation on okra was reported in West Africa in 2021, leading to significant yield loss and increased pesticide use by farmers. Putative tolerant okra accessions were identified (Kouadio et al., 2024). However, using these accessions in breeding programs will require further screening and trait development.
Breeding for tolerance to these stresses requires prior knowledge of the spread and diversity of their causal agents across the target regions. However, except for studies on begomovirus (Tiendrébéogo et al., 2010a; Leke et al., 2013) focusing on specific countries, we currently lack information on the diversity of causal agents of the major okra diseases, which is a limitation for accurate screening and breeding. A better understanding of the diversity of causal agents, identification of pest and disease hotspots, and seasonal dynamics of okra pests and diseases will provide valuable information for germplasm screening and selection. Breeding for resistance to biotic stress in okra increasingly focuses on utilizing related A. esculentus species that exhibit resistance traits. For instance, A. caillei and A. manihot are sources of resistance against YVMD (Suma et al., 2023) and ELCV. Integrating advanced breeding techniques, including wide hybridization and molecular approaches (marker-assisted selection, genomic selection), is essential for enhancing genetic diversity and developing resilient okra varieties capable of withstanding these biotic challenges (Singh et al., 2014; Zhan et al., 2019). Wide crosses with A. esculentus have been challenging due to fertility issues in F1 due to differences in chromosome number. However, by using A. esculentus as a female parent and employing chromosome doubling with colchicine to restore F1 fertility, Suma et al. (2023) have successfully developed interspecific progenies with desirable, essential agronomic traits that have been transferred from related species, thereby broadening the genetic base of cultivated okra.
5 Okra improvement for abiotic stress tolerance
Abelmoschus esculentus is a warm-season crop adapted to relatively high temperatures. The crop thrives well at temperatures between 25 and 32°C. However, various abiotic stresses, particularly drought, heat, salinity, and cold, negatively affect okra, which significantly impacts its growth and yield. Drought duration and intensity are expected to increase across Africa (Yahaya et al., 2024). Drought stress is a significant concern, as it negatively impacts photosynthesis, resulting in reduced productivity (Asante et al., 2024). The co-occurrence of drought and lower or higher temperature stress further exacerbates these effects, highlighting the need for improved varieties that can withstand these stresses (Asante et al., 2024).
Salinity is a significant abiotic stress that disrupts osmotic balance and ion homeostasis in plants, resulting in stunted growth and yield loss (Ashraf and Harris, 2004). Okra exhibits varying degrees of tolerance to salinity, with specific accessions showing better performance under saline conditions (Haq et al., 2012).
Okra is sensitive to low temperatures, which can cause chilling injury and hinder growth. Exposure to temperatures below 20°C results in physiological stress in okra, characterized by yellowing leaves and reduced flowering (Omran and Powell, 1971; Huang et al., 2012). Cold stress is significant in the northern regions of coastal and Sahelian countries in West Africa, which account for more than 90% of Africa’s total okra production and harvested area (FAOStat, 2024). Average minimum temperatures (19°C) from November to February are lower than the optimal temperatures for okra growth (Figure 1). This period also coincides with a higher incidence of viral diseases. Considering the temperature patterns, February to September is the optimal growing season for okra in West Africa, provided irrigation facilities are available to supply water from February to March-April.
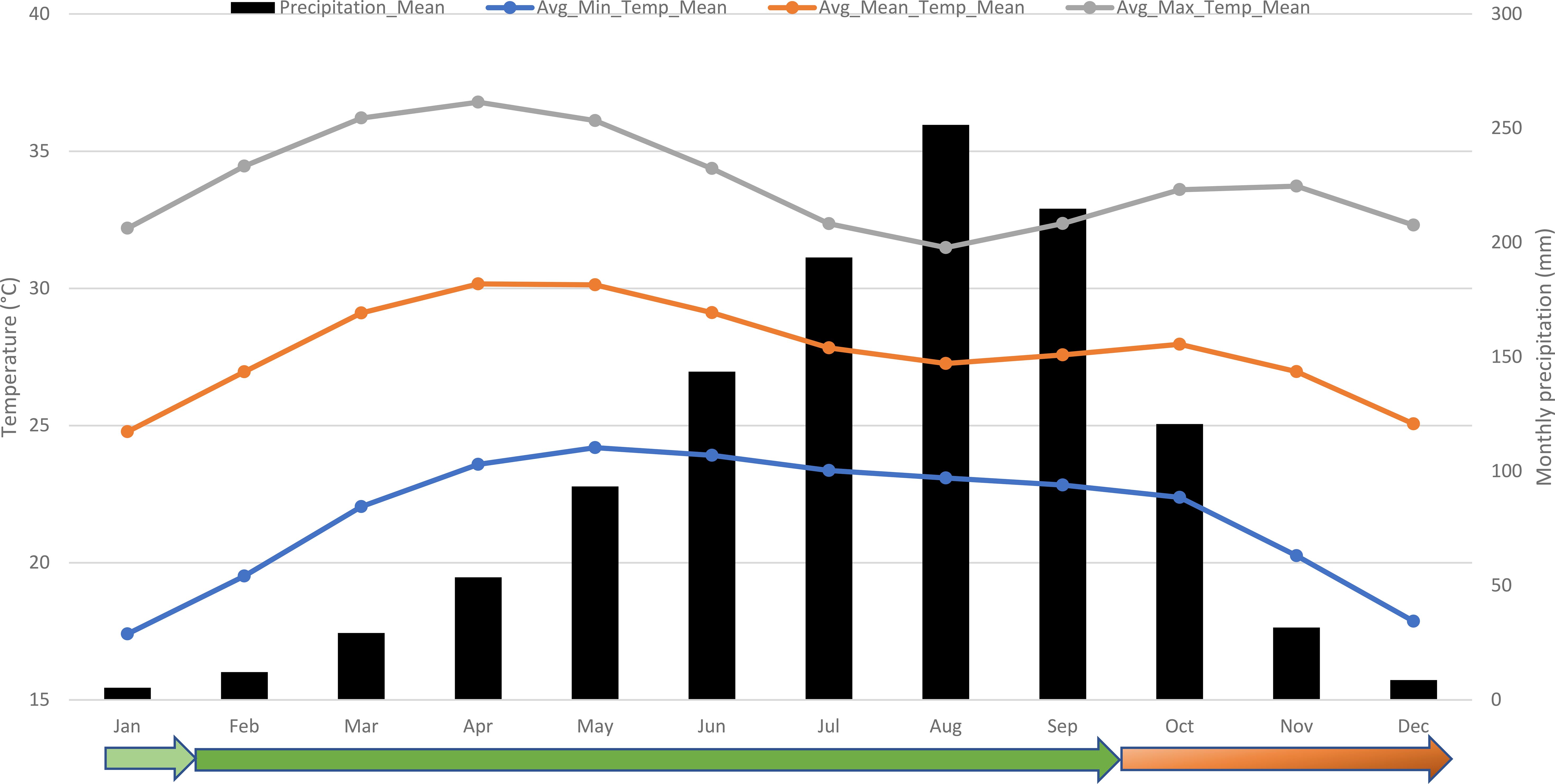
Figure 1. Temperatures and precipitations across West Africa and the okra production seasons in the region. Source: Climate data retrieved from the World Bank Climate Knowledge Portal (https://climateknowledgeportal.worldbank.org/). The color gradient, from light green (moderate suitability) to red (poor suitability), indicates the suitability of the season for okra production.
To adapt to abiotic stresses, increased attention and investment should be devoted to developing resilient okra varieties that can thrive under adverse environmental conditions. Given the complex nature of abiotic stress tolerance, investing in trait discovery to identify surrogate traits with less complex genetic architecture for improvement will be critical.
6 Seed systems in Africa
The okra seed market in Africa remains underdeveloped, presenting a significant opportunity for growth if the seed sector receives substantial investment and enabling policy and regulatory measures (Kuhlmann et al., 2023). In sub-Saharan Africa (SSA), the okra seed system is primarily informal, with most farmers cultivating local landraces due to cultural preferences and perceived risks associated with new technologies (Nkongho et al., 2022). For instance, in Mali, 14% of okra seeds are locally produced and certified, while the remainder comes from imported seeds and the farmer seed system (Dembélé et al., 2021). Various factors, including limited availability and access to certified seeds—partly due to insufficient investment in local seed production, promotion, and distribution—along with inadequate storage, logistical infrastructure, and policies, impede the availability and use of quality okra seeds, especially in remote areas (Mihretu, 2019; Nkongho et al., 2022). The reliance on seed saving and exchange without quality assurance and control can lead to low seed quality, poor crop establishment, reduced seedling vigor, and lower yields (Hirpa et al., 2010; Nkongho et al., 2022). Additionally, weak extension services and insufficient farmer education exacerbate these challenges, as farmers often lack proper knowledge regarding storage and planting techniques, leading to suboptimal variety performance (Kassie et al., 2015; Nkongho et al., 2022). Okra seed systems in SSA are comparable to some global vegetables, such as tomatoes and peppers, characterized by low local seed production, seed import, and overall high saved seed, exchange, and sourcing from open markets (Ayenan et al., 2021). Okra seed systems hold high potential for development in terms of private sector investment and increased use of improved varieties and high-quality seeds. This is exemplified by the current status of the okra seed sector in Asia especially in India, which is dominated by hybrid varieties (over 80% of the market) mostly developed and marketed by private seed companies (Tikoo, 2025). Addressing okra seed sector’ challenges necessitates a multi-faceted approach that includes investments in seed system development, strengthening extension services, improving infrastructure, and promoting farmer education.
7 Diversity of okra germplasm collection
7.1 Okra phenotypic diversity assessment in Africa
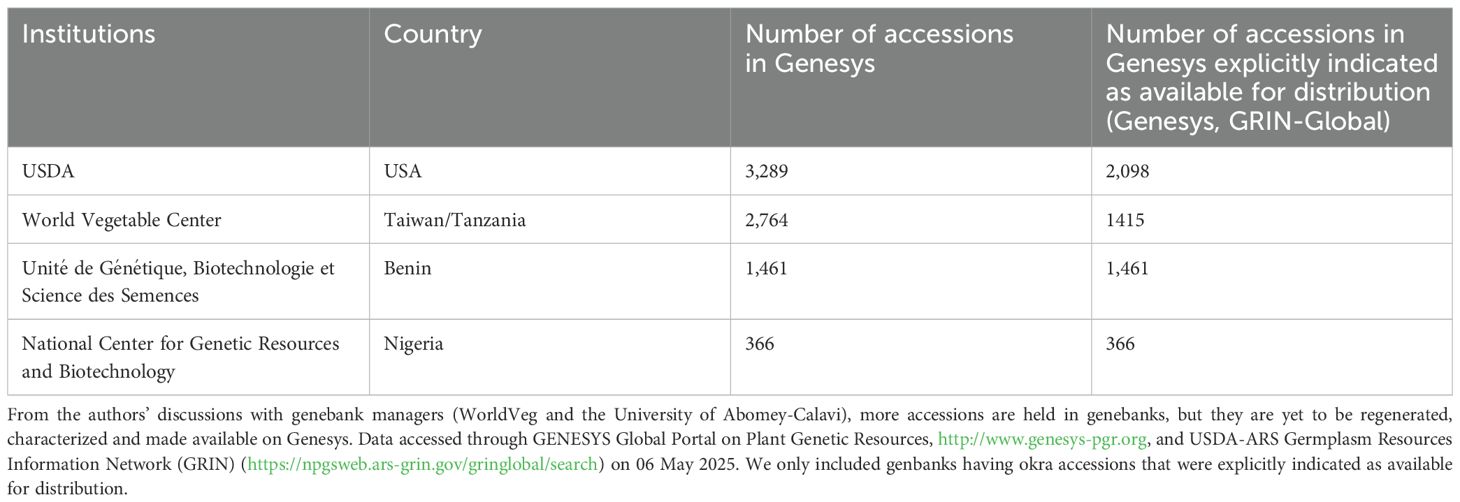
As of 06 May 2025, approximately 8,000, of which about 5,340 were available for distribution okra accessions, held in various genebanks worldwide, are recorded in the Genesys database (Table 3). Knowledge of the existing diversity in these okra collections is crucial for guiding the selection and use of germplasm in breeding programs. We identified 46 studies from Web of Science and Scopus on the characterization and evaluation of okra germplasm, of which 16 were conducted in Africa, reporting the characterization and evaluation of okra accessions under various conditions (Supplementary Table S1). In our search, we only included papers published in English, and papers published in languages other than English might have been overlooked. The studies include 160 accessions from Ghana and Nigeria (Annor et al., 2023), 50 accessions (Ibitoye and Kolawole, 2022) and 22 accessions (Kolawole et al., 2022) from Nigeria, 21 accessions (Kenaw et al., 2023) and 33 accessions (Temam et al., 2021) from Ethiopia. All these studies rely solely on phenotypic traits, known to be influenced by environmental factors. Evaluation of okra germplasm for important agronomic and quality traits is even more scarce. Recent studies included germplasm evaluation for mucilage content (Ahiakpa et al., 2014), resistance to okra mosaic virus (OkMV) and Okra yellow vein mosaic virus (OYVMV) (Appiah et al., 2020), drought tolerance (Mkhabela et al., 2022), and resistance to aphids (Aphis gossypii Glover) (Abang et al., 2014, 2018, 2021). The characterized accessions represent only a handful of the collected and conserved okra accessions. Characterization of okra germplasm for bioactive compounds is currently scarce.
This lack of comprehensive knowledge about the diversity of okra germplasm collections significantly hampers efforts to improve the crop’s productivity and adaptability to various growing conditions (Gulsen et al., 2007; Reddy et al., 2016; Singh et al., 2018; Komolafe et al., 2021; Kumar et al., 2023). Okra faces numerous production constraints, including pest and disease pressures, poor soil fertility, and abiotic stresses (Zibelo et al., 2016; Cobbinah and Kwoseh, 2023). Without a thorough understanding of the genetic diversity within existing germplasm, researchers struggle to identify and utilize sources of important agronomic, adaptation, and quality traits to overcome the numerous biotic and abiotic stresses affecting okra (Kumar et al., 2010). Moreover, limited access to diverse germplasm collections restricts the ability to breed new cultivars with traits such as disease resistance, improved nutritional content, and better tolerance to environmental stresses (Kiiza et al., 2012; Reddy et al., 2016), as well as to identify processing and value-added traits. Addressing these knowledge gaps and expanding the characterization of the okra germplasm collection will significantly enhance the crop’s productivity and utilization.
7.2 Molecular diversity of okra germplasm collections
Genetic variation within okra germplasm is crucial for identifying genetically distinct genotypes for hybrid breeding and establishing diversity panels to screen for desirable horticultural and nutritional traits, as well as adaptive mechanisms to breed for tolerance to abiotic and biotic stresses. The genetic diversity of okra germplasm has been extensively studied using various molecular markers (Supplementary Table S2), including single nucleotide polymorphism (Sun et al., 2023), random amplified polymorphic DNA (RAPD) (Aladele et al., 2008; Haq et al., 2012; Kaur et al., 2013; Goswami et al., 2016; Hamdan et al., 2024), inter-simple sequence repeats (ISSR) (Yuan et al., 2014; El-Sherbeny et al., 2018), amplified fragment length polymorphism (AFLP) (Akash et al., 2013; Kyriakopoulou et al., 2014; Salameh, 2014; Massucato et al., 2019), sequence-related amplified polymorphism (SRAP) (Gulsen et al., 2007; Yıldız et al., 2016), and simple sequence repeats (SSR) (Sawadogo et al., 2009; Fougat et al., 2015; Yıldız et al., 2015; Kumar et al., 2017; Mohammed et al., 2020; Das et al., 2022) and Isozymes (Torkpo et al., 2006). Most of these studies have focused on a small number of accessions. Additionally, in Africa, very few studies have examined the molecular aspects of diversity. A better understanding of the genetic diversity of the African collection is needed to support the selection of parental lines for breeding programs.
8 Prospects for okra breeding in Africa
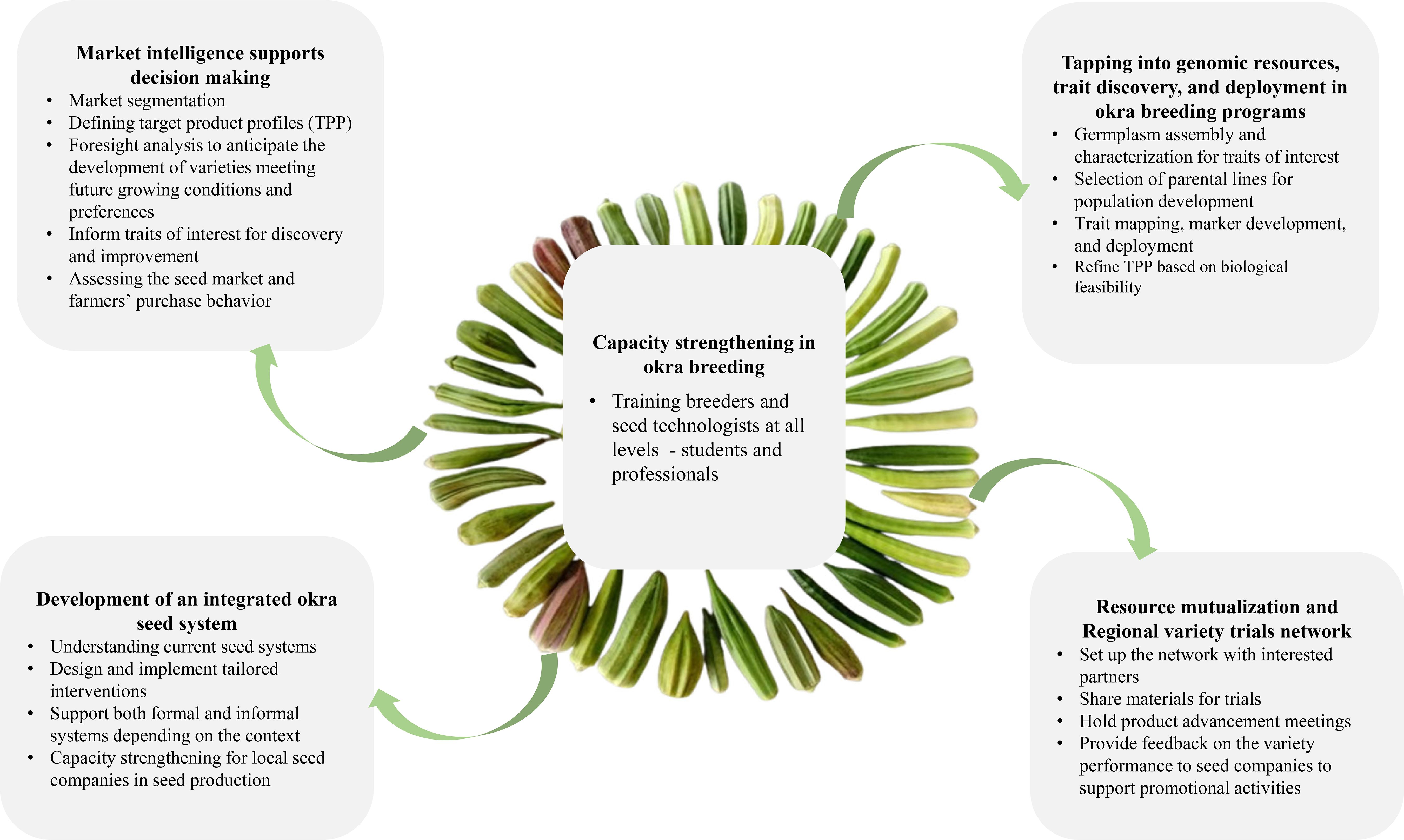
Enhancing okra breeding programs in Africa necessitates building capacity in okra improvement technologies and methods, collaborating with the private sector to boost investment and adoption of developed varieties, increasing funding for breeding activities, and creating a regional trial network for testing okra varieties. These proposed actions are interconnected, with capacity building at the core of the strategy (Figure 2).
8.1 Market intelligence for better decision making
Integrating market-driven needs into breeding programs is crucial for effectively deploying improved okra varieties throughout Africa (Tiwari et al., 2022; Singh et al., 2023). Gaining a deeper insight into market requirements is vital for creating competitive and adoptable okra varieties. The market segments for okra in Africa can generally be categorized into long and ridge pod hybrids/OPVs, medium and ridge pods, and short or baby pods. Nonetheless, this broad categorization often fails to acknowledge the significant differences in consumers’ and traders’ preferences, which can vary not only between countries but also within particular regions. Therefore, it is essential to refine the market segmentation. By identifying subtle variations in product preferences and incorporating these preferences as targets in breeding strategies through well-defined, region-specific target product profiles (TPPs), we can considerably enhance the likelihood of variety adoption (Donovan et al., 2022). Varieties that are more closely aligned with consumer expectations, trader needs, and production challenges will experience greater acceptance across the value chain from farmers to markets. This alignment will promote uptake and help farmers and seed companies capitalize on higher-value segments, thereby improving both the competitiveness and profitability of okra production.
8.2 Tapping into genomic resources, trait discovery, and deployment in okra
The application of genomics, transcriptomics, metabolomics, and phenomics tools can facilitate our understanding of the genetic foundation of key traits and/or proxy traits such as disease resistance, seed oil quality, and mucilage content, as well as assist in the development and application of markers. Genome-wide association studies (GWAS) are widely used to dissect the genetic basis of complex traits in crops. However, in okra, such studies remain limited due to its complex polyploid genome. Despite this, recent GWAS efforts on okra have identified SNPs associated with important agronomic traits, including seedling tolerance to salinity (Sun et al., 2023), and seed germination under salinity stress (Xu et al., 2024). Schafleitner et al. (2013) generated gene sequence data and marker resources from transcriptomic analyses of leaves and pods in okra. Nevertheless, these genomic resources have not been consistently utilized in okra breeding programs, indicating the need for the creation of customized genomic resources, particularly markers that function effectively without reference sequences and consider not just sequence variation but also copy number polymorphisms (Schafleitner and Lin, 2025). Such genomic resources for marker-assisted selection in okra could be developed through K-mer mapping (Voichek and Weigel, 2020; Schafleitner and Lin, 2025). Overall, there is limited availability of molecular markers for okra breeding due to the complexity of its genome and the absence of a genetic linkage map (Mishra et al., 2017). Despite these challenges, marker-assisted selection (MAS) holds significant promise for accelerating the development of improved okra varieties. Although MAS in okra is still in its early stages, Shwetha et al. (2024) identified molecular markers associated with YVMV resistance in okra, paving the way for early detection, early-stage screening, and the selection of resistant plants.
Whole genome sequences facilitate the development and use of molecular markers such as SNP-markers for genetic analyses. Several whole genome sequences have been produced for okra. A chromosome-scale genome sequence of okra spanning 1.19 Gb over 63 chromosomes was made available (Wang et al., 2023). Another whole genome reference sequence comprises 65 chromosomes and a genome size of 1.45 Gb (Nieuwenhuis et al., 2024). For A. moschata, a full-length transcriptome has been produced (Guangying et al., 2023).
The increased availability of genomic information on A. esculentus and insight into its genome structure pave the way for genomic-assisted breeding in okra. Marker panels can be developed for quality control and assurance, including verifying varietal identity, testing for genetic purity, and detecting seed lot contamination, thus addressing the gap in this aspect within the public sector. This approach can also benefit small-scale private seed companies. More efforts are needed to sequence and make available the genomes of related species such as A. caillei, A. manihot, and A. moschatus, which serve as donor sources for many traits.
Innovative phenotyping approaches are essential to understand okra response to abiotic stresses and develop resilient okra varieties. High-throughput phenotyping using unmanned aerial vehicles (UAVs) equipped with multispectral cameras offers a significant opportunity to identify novel traits associated with abiotic stress tolerance (Feng et al., 2020; Zhang et al., 2020). While underutilized in okra research, this approach holds considerable promise for trait discovery. Furthermore, understanding the behavior of belowground plant traits is critical for developing drought-tolerant varieties. Root phenotyping can identify promising accessions that exhibit tolerance to abiotic stresses (Wasaya et al., 2018; Canales et al., 2019; Dutta and Sarma, 2022). Integrating aboveground (aerial) and belowground (root) phenotyping provides a comprehensive understanding of plant responses to environmental challenges through trait discovery, ultimately facilitating the breeding of resilient crop varieties (Shi et al., 2023).
In contrast to crops such as tomatoes, where a variety of populations are publicly accessible, this is currently not the case for okra. The development of different population types, including biparental, interspecific, and multi-parent generation inter-cross (MAGIC) populations for specific traits of interest, will facilitate the speed of trait discovery through QTL mapping, marker development, and deployment, as well as product development. Indeed, numerous research teams can utilize these populations for in-depth investigations into the genetic and physiological architecture of important agronomic traits. QTL mapping is a valuable approach for identifying genes associated with agronomic, nutritional, and stress-resistance traits, thereby accelerating crop improvement. However, in polyploid crops, such as okra, QTL mapping is more complex. Recent advances in statistical tools, such as the PolymapR package in R, have enabled effective QTL mapping in allopolyploids. These tools have been successfully applied in other polyploid species, for example, in mapping black spot resistance in climbing rose (Zurn et al., 2018), anthocyanin content in tetraploid roses (Cheng et al., 2021), and canker resistance in Actinidia chinensis kiwifruit, an allohexaploid (Tahir et al., 2020). Their application in okra could facilitate the identification of key genomic regions and support the development of market-preferred, and resilient varieties. Most value-added traits in okra are likely quantitative, hence amenable to genomic selection. To date, there have been no public reports on the application of genomic selection in okra; however, approaches developed in other allopolyploid crops, such as white clover, an allotetraploid (Ehoche et al., 2025), can provide a basis for such an application.
8.3 Capacity strengthening in okra breeding
Capacity strengthening is crucial for improving okra breeding in Africa. Analysis of current breeding programs shows a lack of qualified personnel. Enhancing skills for more okra breeders is essential for fostering local efforts. Until enough breeders, seed technologists, and support staff are trained, substantial changes are unlikely. Offering short courses on market segmentation, target product profiles, using okra-related species to enhance traits, marker-assisted selection, experimental design, managing segregating populations, genomic selection, maintaining parental lines, and seed quality control will equip breeders with essential knowledge to enhance their breeding efficiency programs. Internship and research in full-fledged okra breeding programs are good avenues to enhance skills. Current breeding courses across the continent, like the Africa Breeding Academy and other breeding initiatives, offer promising opportunities for capacity building. International agricultural research institutions with breeding programs on okra can also host professionals and students to provide hands-on okra breeding.
8.4 Resource mutualization and Regional variety trials network
Resource mutualization is key for success, considering the limited resources available for okra breeding programs. When available, sharing expertise, germplasm, laboratory, and field facilities can enhance okra breeding capabilities on the continent. Access and use of a breeding management system will enhance collaboration and data sharing. However, resource mutualization needs collaboration among institutions. Additionally, better coordination of okra breeding activities could help limit the duplication of resources. Such a coordinate needs a network that offers breeders an exchange platform. A starting point for this network can be a regional variety trial. Regional trials are essential for collecting valuable data to improve breeding efficiency and gathering information on advanced materials for release. Such trials can enable the identification of “regional champion” varieties, which have wide adaptation and could interest seed companies in increasing their market share. The okra breeding programs in Africa are currently too small to bear the cost of regional trials. However, creating a network will minimize trials and variety release costs, since the network of seed companies can take advantage of regionally harmonized seed regulations to release “regional champion” in one country (West Africa) and two countries (Common Market for Eastern and Southern Africa - COMESA, Southern African Development Community - SADC) for seed production and marketing. Such a network will also provide a platform for exchanging materials, knowledge, and valuable market information on okra improvement.
8.5 Support the development of an integrated seed system
Seed systems play a crucial role as the primary channels through which farmers obtain high-quality seeds of the crop varieties essential for their production needs (Nkongho et al., 2022). A well-functioning seed system can significantly enhance agricultural productivity by facilitating rapid and cost-effective increases in crop yields. Improving access to high-quality seeds in Africa is paramount for boosting agricultural productivity (Biemond, 2013; Mihretu, 2019). Breeding programs cannot be sustainable unless a vibrant seed system drives the chain. Regulatory and seed policies are in place to support the okra seed system. Increased investment in capacity strengthening for local okra seed production, along with a more conducive environment, for instance, by easing requirements for mandatory variety testing and releasing to locally produce and certify seeds, will favor the development of local and regional seed companies. These companies and seed cooperatives will take up locally bred varieties for seed production and marketing. In the long run, local seed companies can invest in pre-breeding and breeding activities to access new varieties and diversify their portfolio. Seed cooperatives that produce and serve their communities will also be included in seed capacity strengthening activities to ensure that no one is left behind. A more flexible quality assurance scheme can be implemented for these cooperatives to alleviate the burden of the formal seed certification process.
The execution of these strategic initiatives will result in:
● Full-fledged okra breeding programs that use innovative technologies and methods to develop and deliver improved varieties consistently.
● Providing farmers with market-preferred varieties and products, thus improving livelihoods and nutrition.
● Boosting private sector investment in okra breeding and seed systems.
Author contributions
MATA: Conceptualization, Writing – original draft, Writing – review & editing, Funding acquisition, Project administration. FV: Writing – original draft, Writing – review & editing. MA: Writing – review & editing. JAO: Writing – review & editing. DOI: Funding acquisition, Writing – review & editing. RS: Funding acquisition, Writing – review & editing.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. This work was supported by the Vision for Adapted Crops and Soils and the UK International Development from the UK government (FCDO Project Number: 205271). We also acknowledge strategic donors to the World Vegetable Center: Taiwan, the United Kingdom, the United States, Australia, Germany, Thailand, the Philippines, South Korea, and Japan.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fpls.2025.1631221/full#supplementary-material
References
Abang, A., Kekeunou, S., Bilong Bilong, C., Srinivasan, R.-A., Chang, J.-A., Hanna, R., et al. (2014). Identification of okra (Abelmoschus spp.) accessions resistant to aphid (Aphis gossypii Glover) in Cameroon. Afr. Entomology 22, 273–284. doi: 10.4001/003.022.0201
Abang, A. F., Ramasamy, S., Hanna, R., Kekeunou, S., Fotso Kuate, A., Tenkouano, A., et al. (2024). New okra genotypes for the management of aphids (Hemiptera: Aphididae) in the diverse ecological landscapes of Central Africa. J. Economic Entomology 117, 2123–2134. doi: 10.1093/jee/toae165
Abang, A. F., Srinivasan, R., Hanna, R., Fotso, A. K., Kekeunou, S., Tenkouano, A., et al. (2021). Productivity and resistance of okra (Abelmoschus spp.) to the cotton aphid Aphis gossypii Glover (Hemiptera: Aphididae) under tropical conditions. Int. J. Trop. Insect Sci. 41, 197–208. doi: 10.1007/s42690-020-00193-w
Abang, A. F., Srinivasan, R., Kekeunou, S., Hanna, R., Kamga, R., and Bilong, C.-F. B. (2018). Influence of okra (Abelmoschus spp.) accessions on colonization by Aphis gossypii (Hemiptera: Aphididae) and their effects on aphid biological parameters. Florida Entomologist 101, 549–558. doi: 10.1653/024.101.0415
Abba, A., Umar, K. J., Hassan, L. G., Yakubu, M. K., and Rabiu, A. K. (2020). Evaluation of some varieties of Okra (Abelmoschusesculentus) seed oil for consumption and industrial uses. Equity J. Sci. Technol. 7, 148–151.
Adelakun, O. E. and Oyelade, O. J. (2011). “Chemical and antioxidant properties of okra (Abelmoschus esculentus moench) seed,” in Nuts and seeds in health and disease prevention (Elsevier), 841–846.
Adelakun, O., Oyelade, O., Ade-Omowaye, B., Adeyemi, I., Van de Venter, M., and Koekemoer, T. (2009). Influence of pre-treatment on yield chemical and antioxidant properties of a Nigerian okra seed (Abelmoschus esculentus moench) flour. Food Chem. Toxicol. 47, 657–661. doi: 10.1016/j.fct.2008.12.023
Adetuyi, F. and Adelabu, H. (2011). Impact of okra (Abelmoschus esculentus) seed flour on nutrients, functional properties and zinc bioavailability of plantain flour. Malaysian J. Nutr. 17, 359–366.
Adetuyi, F. and Dada, I. (2014). Nutritional, phytoconstituent and antioxidant potential of mucilage extract of Okra (Abelmoschus esculentus), water leaf (Talinum triangulare) and Jews mallow (Corchorus olitorius). Int. Food Res. J. 21, 2345.
Adetuyi, F., Osagie, A., and Adekunle, A. (2008). Comparative study of quality changes in okra Abelmoschus esculentus (L) Moench stored at different relative humidities. Biol. Sciences-PJSIR 51, 304–308.
Agbenorhevi, J. K., Kpodo, F. M., Banful, B. K., Oduro, I. N., Abe-Inge, V., Datsomor, D. N., et al. (2020). Survey and evaluation of okra pectin extracted at different maturity stages. Cogent Food Agric. 6, 1760476. doi: 10.1080/23311932.2020.1760476
Agbo, E. A., Nemlin, J. G., Anvoh, B. K., and Gnakri, D. (2010). Characterisation of lipids in okra mature seeds. Int. J. Biol. Chem. Sci. 4, 184–192. doi: 10.4314/ijbcs.v4i1.54243
Ahiakpa, J., Amoatey, H., Amenorpe, G., Apatey, J., Ayeh, E., and Agbemavor, W. (2014). Mucilage Content of 21 accessions of Okra (Abelmoschus spp L.). Scientia Agriculturae 2, 96–101. doi: 10.15192/PSCP.SA.2014.2.2.96101
Ahiakpa, J., Magdy, M., Werner, O., Amoatey, H., Yeboah, M., Appiah, A., et al. (2017). Intra-specific variation in West African and Asian germplasm of okra (Abelmoschus spp L.). Ann. Agric. Sci. 62, 131–138. doi: 10.1016/j.aoas.2017.08.002
Akash, M. W., Shiyab, S. M., and Saleh, M. I. (2013). Yield and AFLP analyses of inter-landrace variability in okra (Abelmoschus esculentus L.). Life Sci. J. 10, 2771–2779.
Akinyele, B. and Temikotan, T. (2007). Effect of variation in soil texture on the vegetative and pod characteristics of okra (Abelmoschus esculentus (L.) Moench). Int. J. Agric. Res. 2, 165–169. doi: 10.3923/ijar.2007.165.169
Aladele, S. E., Ariyo, O., and De Lapena, R. (2008). Genetic relationships among West African okra (Abelmoschus caillei) and Asian genotypes (Abelmoschus esculentus) using RAPD. Afr. J. Biotechnol. 7, 1426–1431.
Alake, C. O. (2020). Traits associated with pod yield, seed Fe, and Zn contents in Okra landraces: a path analysis. Int. J. Vegetable Sci. 26, 573–590. doi: 10.1080/19315260.2019.1685619
Alba, K., Laws, A. P., and Kontogiorgos, V. (2015). Isolation and characterization of acetylated LM-pectins extracted from okra pods. Food Hydrocolloids 43, 726–735. doi: 10.1016/j.foodhyd.2014.08.003
Amin, M. H. A., Akter, M. M., Jutidamrongphan, W., and Techato, K.-a. (2023). Okra tree crop agroforestry model: economic and environmental impact. Environment Dev. Sustainability 25, 12527–12542. doi: 10.1007/s10668-022-02577-z
Amin, B. and Mahmood, H. (2011). Effect of drought stress and its interaction with ascorbate and salicylic acid on okra (Hibiscus esculents L.) germination and seedling growth. J. Stress Physiol. Biochem. 7, 55–65.
Annor, B., Kena, A.W., and Bediako, K.A. (2023). Variability among West African okra (Abelmoschus spp. L.) accessions based on quantitative traits. Ecological Genetics and Genomics 26, 100156. doi: 10.1016/j.egg.2022.100156
Appiah, A., Amiteye, S., Boateng, F., and Amoatey, H. (2020). Evaluation of okra (Abelmoschus esculentus L. Moench) cultivars for resistance to okra mosaic virus and okra yellow vein mosaic virus. Australas. Plant Pathol. 49, 541–550. doi: 10.1007/s13313-020-00727-3
Arlai, A., Nakkong, R., Samjamin, N., and Sitthipaisarnkun, B. (2012). The effects of heating on physical and chemical constitutes of organic and conventional okra. Proc. Eng. 32, 38–44. doi: 10.1016/j.proeng.2012.01.1234
Armand, N. M., Martin, B. J., Célestin, D., Elmith, M. J., Bille, N. H., Ntsefong, G. N., et al. (2013). Assessment of some agro-morphological parameters of some local and exotic varieties of okra [Abelmoschus Esculentus (Moench). Int. J. Biotechnol. Food Sci. 1, 6–12.
Asante, J., Opoku, V. A., Hygienus, G., Andersen, M. N., Asare, P. A., and Adu, M. O. (2024). Photosynthetic efficiency and water retention in okra (Abelmoschus esculentus) contribute to tolerance to single and combined effects of drought and heat stress. Sci. Rep. 14, 28090. doi: 10.1038/s41598-024-79178-5
Ashraf, M. and Harris, P. J. (2004). Potential biochemical indicators of salinity tolerance in plants. Plant Sci. 166, 3–16. doi: 10.1016/j.plantsci.2003.10.024
Ayenan, M. A. T., Aglinglo, L. A., Zohoungbogbo, H. P. F., N’Danikou, S., Honfoga, J., Dinssa, F. F., et al. (2021). Seed systems of traditional African vegetables in eastern Africa: A systematic review. Front. Sustain. Food Syst. 5. doi: 10.3389/fsufs.2021.689909
Badaró, A. T., Garcia-Martin, J. F., del Carmen López-Barrera, M., Barbin, D. F., and Alvarez-Mateos, P. (2020). Determination of pectin content in orange peels by near infrared hyperspectral imaging. Food Chem. 323, 126861. doi: 10.1016/j.foodchem.2020.126861
Benchasri, S. (2012). Okra (Abelmoschus esculentus (L.) Moench) as a valuable vegetable of the world. Field Vegetable Crops Research/Ratarstvo i povrtarstvo 49, 105–112. doi: 10.5937/ratpov49-1172
Biemond, C. (2013). Seed quality in informal seed systems (Netherlands: Wageningen University and Research).
Binalfew, T. and Alemu, Y. (2016). Characterization of okra (Abelmoschus esculentus (L.) Moench) germplasms collected from Western Ethiopia. Int. J. Res. Agric. For 3, 11–17.
Borokini, F. B., Oladipo, G. O., Komolafe, O. Y., and Ajongbolo, K. F. (2022). Phytochemical, nutritional and antioxidant properties of Abelmoschus esculentus Moench L leaf: A pointer to its fertility potentials. Measurement: Food 6, 100034. doi: 10.1016/j.meafoo.2022.100034
Çalışır, S., Özcan, M., Hacıseferoğulları, H., and Yıldız, M. U. (2005). A study on some physico-chemical properties of Turkey okra (Hibiscus esculenta L.) seeds. J. Food Eng. 68, 73–78. doi: 10.1016/j.jfoodeng.2004.05.023
Canales, F. J., Nagel, K. A., Müller, C., Rispail, N., and Prats, E. (2019). Deciphering root architectural traits involved to cope with water deficit in oat. Front. Plant Sci. 10. doi: 10.3389/fpls.2019.01558
Chattopadhyay, A., Dutta, S., and Chatterjee, S. (2011). Seed yield and quality of okra as influenced by sowing dates. Afr. J. Biotechnol. 10, 5461–5467.
Cheng, B., Wan, H., Han, Y., Yu, C., Luo, L., Pan, H., et al. (2021). Identification and QTL analysis of flavonoids and carotenoids in tetraploid roses based on an ultra-high-density genetic map. Front. Plant Sci. 12, 682305. doi: 10.3389/fpls.2021.682305
Cobbinah, P. and Kwoseh, C. (2023). Evaluation and improvement of the health status of farmer-saved okra (Abelmoschus esculentus (L.) Moench) seeds in the Ashanti Region of Ghana. Ghana J. Agric. Sci. 58, 85–99-85–99. doi: 10.4314/gjas.v58i1.8
Cui, Y., Wu, J., Chen, Y., Ji, F., Li, X., Yang, J., et al. (2023). Optimization of near-infrared reflectance models in determining flavonoid composition of okra (Abelmoschus esculentus L.) pods. Food Chem. 418, 135953. doi: 10.1016/j.foodchem.2023.135953
Dantas, T. L., Alonso Buriti, F. C., and Florentino, E. R. (2021). Okra (Abelmoschus esculentus L.) as a potential functional food source of mucilage and bioactive compounds with technological applications and health benefits. Plants 10, 1683. doi: 10.3390/plants10081683
Das, A., Yadav, R. K., Choudhary, H., Lata, S., Singh, S., Kumar, C., et al. (2022). Population structure, gene flow and genetic diversity analyses based on agro-morphological traits and microsatellite markers within cultivated and wild germplasms of okra [Abelmoschus esculentus (L.) Moench. Genet. Resour. Crop Evol. 69, 771–791. doi: 10.1007/s10722-021-01263-9
de Alvarenga Pinto Cotrim, M., Mottin, A. C., and Ayres, E. (2016) “Preparation and characterization of okra mucilage (Abelmoschus esculentus) edible films,” in Macromolecular Symposia, 90–100. doi: 10.1002/masy.201600019
Dembélé, S., Tignegre, J. B., and Diarra, B. G. (2021). Development of the vegetable seed sector in Mali and opportunities for irrigated seed production. World Vegetable Center. 21-1035.
Dhankhar, S. and Koundinya, A. (2020). “Accelerated breeding in Okra,” in Accelerated Plant Breeding, Volume 2: Vegetable Crops. Switzerland AG: Springer Nature, 337–353. doi: 10.1007/978-3-030-47298-6_12
Donovan, J., Coaldrake, P., Rutsaert, P., Bänzinger, M., Gitonga, A., Naziri, D., et al. (2022). “Market intelligence for informing crop-breeding decisions by CGIAR and NARES,” in Market Intelligence Brief Series 1, Montpellier: CGIAR. (Montpellier, France: CGIAR) Available online at: https://hdl.handle.net/10883/22248. (Accessed October 8, 2024).
Dutta, C. and Sarma, R. N. (2022). Role of root traits and root phenotyping in drought tolerance. Int. J. Environ. Climate Change 12, 2300–2309. doi: 10.9734/IJECC/2022/v12i1131224
Ehoche, O. G., Arojju, S. K., Jahufer, M. Z. Z., Jauregui, R., Larking, A. C., Cousins, G., et al. (2025). Genomic selection shows improved expected genetic gain over phenotypic selection of agronomic traits in allotetraploid white clover. Theor. Appl. Genet. 138, 34. doi: 10.1007/s00122-025-04819-w
Elkhalifa, A. E. O., Al-Shammari, E., Adnan, M., Alcantara, J. C., Mehmood, K., Eltoum, N. E., et al. (2021). Development and characterization of novel biopolymer derived from Abelmoschus esculentus L. extract and its antidiabetic potential. Molecules 26, 3609. doi: 10.3390/molecules26123609
El-Sherbeny, G., Khaled, A., Obiadalla-Ali, H., and Ahmed, A. (2018). ISSR markers linked to agronomic traits in okra. Int. J. Modern Agric. 7, 1–7.
Ergönül, P. G. and Özbek, Z. A. (2020). “Cold pressed safflower (Carthamus tinctorius L.) seed oil,” in Cold Pressed Oils, Cold Pressed Oils: Green Technology, Bioactive Compounds, Functionality, and Applications. Ed. Ramadan, M. F. (Academic Press, Cambridge, Massachusetts, USA), 323–333. doi: 10.1016/B978-0-12-818188-1.00029-3
FAOStat (2024). FAO online statistical database (Rome, Italy: Food and Agriculture Organization of the United Nations (FAO)). Available online at: https://www.fao.org/faostat/en/.
Fauquet, C. and Stanley, J. (2005). Revising the way we conceive and name viruses below the species level: a review of geminivirus taxonomy calls for new standardized isolate descriptors. Arch. Virol. 150, 2151–2179. doi: 10.1007/s00705-005-0583-0
Fayad, A. (2021). Combining Ability and Heritability in Round Eggplant (Solanum melongena L.) for Vegetative Traits, Yield and its Component. J. Plant Production 12, 1301–1305. doi: 10.21608/jpp.2021.219995
Feng, X., Zhan, Y., Wang, Q., Yang, X., Yu, C., Wang, H., et al. (2020). Hyperspectral imaging combined with machine learning as a tool to obtain high-throughput plant salt-stress phenotyping. Plant J. 101, 1448–1461. doi: 10.1111/tpj.14597
Fougat, R. S., Purohit, A. R., Kumar, S., Parekh, M. J., and Kumar, M. (2015). SSR based genetic diversity in Abelmoschus species. Indian J. Agric. Sci. 85, 1223–1228. doi: 10.56093/ijas.v85i9.51636
Freitas, T., Oliveira, V., De Souza, M., Geraldino, H., Almeida, V., Fávaro, S., et al. (2015). Optimization of coagulation-flocculation process for treatment of industrial textile wastewater using okra (A. esculentus) mucilage as natural coagulant. Ind. Crops Products 76, 538–544. doi: 10.1016/j.indcrop.2015.06.027
Fujimoto, R., Uezono, K., Ishikura, S., Osabe, K., Peacock, W. J., and Dennis, E. S. (2018). Recent research on the mechanism of heterosis is important for crop and vegetable breeding systems. Breed. Sci. 68, 145–158. doi: 10.1270/jsbbs.17155
Gao, H., Zhang, W., Wang, B., Hui, A., Du, B., Wang, T., et al. (2018). Purification, characterization and anti-fatigue activity of polysaccharide fractions from okra (Abelmoschus esculentus (L.) Moench). Food Funct. 9, 1088–1101. doi: 10.1039/C7FO01821E
Gemede, H. F., Haki, G. D., Beyene, F., Rakshit, S. K., and Woldegiorgis, A. Z. (2018). Indigenous Ethiopian okra (Abelmoschus esculentus) mucilage: A novel ingredient with functional and antioxidant properties. Food Sci. Nutr. 6, 563–571. doi: 10.1002/fsn3.596
Gemede, H. F., Haki, G. D., Beyene, F., Woldegiorgis, A. Z., and Rakshit, S. K. (2016). Proximate, mineral, and antinutrient compositions of indigenous Okra (Abelmoschus esculentus) pod accessions: implications for mineral bioavailability. Food Sci. Nutr. 4, 223–233. doi: 10.1002/fsn3.282
Gemede, H. F., Ratta, N., Haki, G. D., Woldegiorgis, A. Z., and Beyene, F. (2015). Nutritional quality and health benefits of okra (Abelmoschus esculentus): A review. J. Food Process Technol. 6, 2. doi: 10.4172/2157-7110.1000458
Geng, S., Liu, Y., Ma, H., and Chen, C. (2015). Extraction and antioxidant activity of phenolic compounds from okra flowers. Trop. J. Pharm. Res. 14, 807–814. doi: 10.4314/tjpr.v14i5.10
Gerrano, A. S. (2018). Agronomic performance, nutritional phenotyping and trait associations of Okra (Abelmoschus esculentus) Genotypes in South Africa. Rediscovery Landraces as Resource Future 69. doi: 10.5772/intechopen.70813
Ghori, M. U., Alba, K., Smith, A. M., Conway, B. R., and Kontogiorgos, V. (2014). Okra extracts in pharmaceutical and food applications. Food Hydrocolloids 42, 342–347. doi: 10.1016/j.foodhyd.2014.04.024
Goswami, A., Singh, B., and Sharma, A. (2016). Analysis of molecular diversity in okra (Abelmoschus esculentus) genotypes using RAPD markers. Indian J. Agric. Sci. 86, 1310–1315. doi: 10.56093/ijas.v86i10.62112
Guangying, Y., Zaihua, W., Hailin, L., and Jie, L. (2023). Medicinal and edible Abelmoschus Manihot: Sequencing and characteristic analysis of Full-Length transcriptome. Chin. Agric. Sci. Bull. 39, 29–35. doi: 10.11924/j.issn.1000-6850.casb2022-0219
Gulsen, O., Karagul, S., and Abak, K. (2007). Diversity and relationships among Turkish okra germplasm by SRAP and phenotypic marker polymorphism. Biologia 62, 41–45. doi: 10.2478/s11756-007-0010-y
Gupta, P. and Patra, S. (2021). Okra plant: A multi-purpose underutilized vegetable crop: A review. Bhartiya Krishi Anusandhan Patrika 36, 208–211. doi: 10.18805/BKAP261
Gupta, P. K., Patra, S., and Samanta, K. K. (2021). Potential of okra for application in textiles: A review. J. Natural Fibers 18, 1788–1800. doi: 10.1080/15440478.2019.1697997
Hamdan, Y. A., Hawamda, A. I., Basheer-Salimia, R., and Salman, M. (2024). Genetic diversity assessment of Palestinian okra landraces (Abelmoschus esculentus L.) through RAPD marker. Genet. Resour. Crop Evol. 71, 3555–3562. doi: 10.1007/s10722-024-01859-x
Haq, I.-u., Khan, A. A., Khan, I. A., and Azmat, M. A. (2012). Comprehensive screening and selection of okra (Abelmoschus esculentus) germplasm for salinity tolerance at the seedling stage and during plant ontogeny. J. Zhejiang Univ. Sci. B 13, 533–544. doi: 10.1631/jzus.B1200027
Hirpa, A., Meuwissen, M. P., Tesfaye, A., Lommen, W. J., Oude Lansink, A., Tsegaye, A., et al. (2010). Analysis of seed potato systems in Ethiopia. Am. J. potato Res. 87, 537–552. doi: 10.1007/s12230-010-9164-1
Hossain, M., Focken, U., and Becker, K. (2001). Evaluation of an unconventional legume seed, Sesbania aculeata, as a dietary protein source for common carp, Cyprinus carpio L. Aquaculture 198, 129–140. doi: 10.1016/S0044-8486(00)00574-3
Huang, S., Li, T., Jiang, G., Xie, W., Chang, S., Jiang, Y., et al. (2012). 1-Methylcyclopropene reduces chilling injury of harvested okra (Hibiscus esculentus L.) pods. Scientia Hortic. 141, 42–46. doi: 10.1016/j.scienta.2012.04.016
Ibitoye, D. O. and Kolawole, A. O. (2022). Farmers’ Appraisal on Okra [Abelmoschus esculentus (L.)] production and phenotypic characterization: a synergistic Approach for Improvement. Front. Plant Sci. 13. doi: 10.3389/fpls.2022.787577
Jarret, R. L., Wang, M. L., and Levy, I. J. (2011). Seed oil and fatty acid content in okra (Abelmoschus esculentus) and related species. J. Agric. Food Chem. 59, 4019–4024. doi: 10.1021/jf104590u
Jose, J. and Usha, R. (2003). Bhendi yellow vein mosaic disease in India is caused by association of a DNA β satellite with a begomovirus. Virology 305, 310–317. doi: 10.1006/viro.2002.1768
Kahlon, T., Chapman, M., and Smith, G. (2007). In vitro binding of bile acids by okra, beets, asparagus, eggplant, turnips, green beans, carrots, and cauliflower. Food Chem. 103, 676–680. doi: 10.1016/j.foodchem.2006.07.056
Kalkan, E. and Maskan, M. (2023). Mucilage in okra: extraction, modelling, optimization and application. J. Food Measurement Characterization 17, 4812–4822. doi: 10.1007/s11694-023-01979-5
Kammili, A. and Yadav, P. (2022). Enhancing oleic acid and oil content in low oil and oleic type Indian safflower (Carthamus tinctorius L.). Ind. Crops Products 175. doi: 10.1016/j.indcrop.2021.114254
Kantar, F., Cengiz, M. F., Erbaş, S., and Babacan, Ü. (2024). Seed oil contents, fatty acid compositions, and gossypol concentrations of some okra landraces. Food Sci. Nutr. 12, 10087–10098. doi: 10.1002/fsn3.4535
Karmakar, P., Sagar, V., and Singh, P. (2022). Dynamics of anthocyanin and chlorophyll content in red fruited okra var. Kashi Lalima. Vegetable Sci. 49, 197–203. doi: 10.61180/vegsci.2022.v49.i2.10
Kassie, M., Teklewold, H., Jaleta, M., Marenya, P., and Erenstein, O. (2015). Understanding the adoption of a portfolio of sustainable intensification practices in eastern and southern Africa. Land Use Policy 42, 400–411. doi: 10.1016/j.landusepol.2014.08.016
Kaur, K., Pathak, M., Kaur, S., Pathak, D., and Chawla, N. (2013). Assessment of morphological and molecular diversity among okra [Abelmoschus esculentus (L.) Moench.] germplasm. Afr. J. Biotechnol. 12, 3160–3170.
Kenaw, W., Mohammed, W., and Woldetsadik, K. (2023). Morpho-agronomic variability of okra [Abelmoschus esculentus (L.) Moench] genotypes in Dire Dawa, eastern Ethiopia. PloS One 18, e0288534. doi: 10.1371/journal.pone.0288534
Kendall, C. W. and Jenkins, D. J. (2004). A dietary portfolio: maximal reduction of low-density lipoprotein cholesterol with diet. Curr. Atheroscl. Rep. 6, 492–498. doi: 10.1007/s11883-004-0091-9
Kiiza, B., Kisembo, L. G., and Mwanga, R. O. M. (2012). Participatory plant breeding and selection impact on adoption of improved sweetpotato varieties in Uganda. J. Agric. Sci. Technol. A 2, 673.
Klimp, A., De Vries, E., Scherphof, G., and Daemen, T. (2002). A potential role of macrophage activation in the treatment of cancer. Crit. Rev. oncology/hematology 44, 143–161. doi: 10.1016/S1040-8428(01)00203-7
Kolawole, A. O., Ibitoye, D. O., and Aderibigbe, O. R. (2022). Phenotypic diversity and performance of agro-nutritional characters in okra germplasm collection from Nigeria. J. Trop. Agric. 60, 151–164.
Komolafe, R. J., Ariyo, O. J., and Alake, C. O. (2021). Diversity in phenotypic traits and mineral elements of Okra (Abelmoschus esculentus L. Moench) genotypes. Int. J. Agron. 2021, 5528703. doi: 10.1155/2021/5528703
Kouadio, H., Kouakou, M., Bini, K. K. N., Koffi, K. J. I., Ossey, C. L., Kone, P. W. E., et al. (2024). Annual and geographical variations in the specific composition of jassids and their damage on cotton in Ivory Coast. Sci. Rep. 14, 2094. doi: 10.1038/s41598-024-52127-y
Kpodo, F. M., Agbenorhevi, J. K., Alba, K., Bingham, R. J., Oduro, I. N., Morris, G., et al. (2017). Pectin isolation and characterization from six okra genotypes. Food Hydrocolloids 72, 323–330. doi: 10.1016/j.foodhyd.2017.06.014
Kuhlmann, K. A., Francis, T., Thomas, I., and Schreinemachers, P. (2023). Laws and regulations enabling and restricting Africa’s vegetable seed sector. Int. J. Agric. Sustainability 21, 2210005. doi: 10.1080/14735903.2023.2210005
Kumar, S., Dagnoko, S., Haougui, A., Ratnadass, A., Pasternak, N., and Kouame, C. (2010). Okra (Abelmoschus spp.) in West and Central Africa: Potential and progress on its improvement. African Journal of Agricultural Research. 5(25), 3590–3598.
Kumar, S., Parekh, M. J., Fougat, R. S., Patel, S. K., Patel, C. B., Kumar, M., et al. (2017). Assessment of genetic diversity among okra genotypes using SSR markers. J. Plant Biochem. Biotechnol. 26, 172–178. doi: 10.1007/s13562-016-0378-2
Kumar, D., Singh, J., Pathania, R., Dogra, B. S., and Chandel, V. (2023). Revealing genetic diversity for the improvement of pod yield in okra (Abelmoschus esculentus L. Moench). Electronic J. Plant Breed. 14, 1497–1504. doi: 10.37992/2023.1404.178
Kumar, D. S., Tony, D. E., Kumar, A. P., Kumar, K. A., Rao, D. B. S., and Nadendla, R. (2013). A review on: Abelmoschus esculentus (Okra). Int. Res. J. Pharm. Appl. Sci. 3, 129–132.
Kumar, R., Yadav, R., Bhardwaj, R., Baranwal, V., Chaudhary, H., and VINOD, V. (2016). Assessment of genetic diversity among okra (Abelmoschus esculentus) genotypes for quality traits. Indian J. Agric. Sci. 86, 785–791. doi: 10.56093/ijas.v86i6.58981
Kumari, M., Solankey, S., Akhtar, S., and Neha, P. (2017). Assessment of genetic variability and character association in okra genotypes for yield and contributing characters. J. Appl. Natural Sci. 9, 1825. doi: 10.31018/jans.v9i3.1446
Kumari, D. A., Suresh, V., Nayak, M. H., and Lavanya, A. (2020). Efficacy of insecticides against leaf hoppers and whitefly in okra. Agric. Res. J. 57, 943–946. doi: 10.5958/2395-146X.2020.00139.8
Kyriakopoulou, O. G., Arens, P., Pelgrom, K. T., Karapanos, I., Bebeli, P., and Passam, H. C. (2014). Genetic and morphological diversity of okra (Abelmoschus esculentus [L.] Moench.) genotypes and their possible relationships, with particular reference to Greek landraces. Scientia Hortic. 171, 58–70. doi: 10.1016/j.scienta.2014.03.029
Leke, W. N., Sattar, M. N., Ngane, E. B., Ngeve, J. M., Kvarnheden, A., and Brown, J. K. (2013). Molecular characterization of begomoviruses and DNA satellites associated with okra leaf curl disease in Cameroon. Virus Res. 174, 116–125. doi: 10.1016/j.virusres.2013.03.010
Lengsfeld, C., Titgemeyer, F., Faller, G., and Hensel, A. (2004). Glycosylated compounds from okra inhibit adhesion of Helicobacter pylori to human gastric mucosa. J. Agric. Food Chem. 52, 1495–1503. doi: 10.1021/jf030666n
Liu, Y., Qi, J., Luo, J., Qin, W., Luo, Q., Zhang, Q., et al. (2021). Okra in food field: Nutritional value, health benefits and effects of processing methods on quality. Food Rev. Int. 37, 67–90. doi: 10.1080/87559129.2019.1695833
Makinde, E. A. (2014). Plant population on growth, fruit size, seed yield, and cost of production of okra. Int. J. Vegetable Sci. 20, 254–263. doi: 10.1080/19315260.2013.801384
Mangan, F. X., de Mendonça, R. U., Moreira, M., Nunes, S., Finger, F. L., Barros, Z., et al. (2008). Production and marketing of vegetables for the ethnic markets in the United States. Horticultura Bras. 26, 6–14. doi: 10.1590/S0102-05362008000100002
Massucato, L. R., Nakamura, K. K., Ruas, P. M., Zeffa, D. M., Silva, D., and Gonçalves, L. S. A. (2019). Genetic diversity among Brazilian okra landraces detected by morphoagronomic and molecular descriptors. Acta Scientiarum. Agron. 42, e43426. doi: 10.4025/actasciagron.v42i1.43426
Messing, J., Thöle, C., Niehues, M., Shevtsova, A., Glocker, E., Boren, T., et al. (2014). Antiadhesive properties of Abelmoschus esculentus (Okra) immature fruit extract against Helicobacter pylori adhesion. PloS One 9, e84836. doi: 10.1371/journal.pone.0084836
Mihretu, F. B. (2019). Challenges and opportunities of vegetable quality seed production and seed system in Ethiopia. Int. J. Res. Stud. Agric. Sci. 5, 15–25. doi: 10.20431/2454-6224.0508003
Mishra, G. P., Seth, T., Karmakar, P., Sanwal, S. K., Sagar, V., et al. (2021). “Breeding strategies for yield gains in Okra (Abelmoschus esculentus L.),” in Advances in Plant Breeding Strategies: Vegetable Crops: Volume 9: Fruits and Young Shoots (Switzerland AG: Springer), 205–233.
Mishra, G. P., Seth, T., Karmakar, P., Sanwal, S. K., and Sagar, V. (2017). Biotechnological advancements and begomovirus management in okra (Abelmoschus esculentus L.): status and perspectives. Front. Plant Sci. 8. doi: 10.3389/fpls.2017.00360
Mkhabela, S. S., Shimelis, H., Gerrano, A. S., and Mashilo, J. (2022). Phenotypic response of okra (Abelmoschus esculentus [L.] Moench) genotypes under drought-stressed and non-stressed conditions. South Afr. J. Bot. 145, 293–302. doi: 10.1016/j.sajb.2021.11.008
Moekchantuk, T. and Kumar, P. (2004). Export okra production in Thailand Vol. 56 (Bangkok, Thailand: Inter-country programme for vegetable IPM in South & SE Asia phase II Food & Agriculture Organization of the United Nations).
Mohammadi, G., Khah, E. M., Petropoulos, S. A., and Chachalis, D. (2016). Effect of nitrogen application on seed yield, pod and seed characteristics of okra. J. Plant Nutr. 39, 1899–1905. doi: 10.1080/01904167.2016.1187750
Mohammed, W., Amelework, B., and Shimelis, H. (2020). Simple sequence repeat markers revealed genetic divergence and population structure of okra [‘Abelmoschus esculentus’] collections of diverse geographic origin. Aust. J. Crop Sci. 14, 1032–1041. doi: 10.21475/ajcs
Mohammed, J., Mohammed, W., and Shiferaw, E. (2022). Performance and genetic variability of okra (abelmoschus esculentus (L.) Moench) genotypes in Ethiopia for agromorphology and biochemical traits. Adv. Agric. 2022, 5521151. doi: 10.1155/2022/5521151
Mohammed, H. Y., Teferra, T. F., and Sime, G. (2024). Indigenous knowledge and nutritional and morphological characterization of okra (Abelmoschus esculentus (L.) Moecnh) varieties in Western Ethiopia. Food Sci. Nutr. 12, 2537–2550. doi: 10.1002/fsn3.3936
Moharam, M. H. A. and Ali, H.A.E.-R.O. (2012). Preventative and curative effects of several plant derived agents against powdery mildew disease of okra. Notulae Scientia Biologicae 4, 76–82. doi: 10.15835/nsb437765
Nieuwenhuis, R., Hesselink, T., van den Broeck, H. C., Cordewener, J., Schijlen, E., Bakker, L., et al. (2024). Genome architecture and genetic diversity of allopolyploid okra (Abelmoschus esculentus). Plant J. 118, 225–241. doi: 10.1111/tpj.16602
Nkongho, R. N., Efouba-Mbong, J. T., Ndam, L. M., Ketchem, G. A., Etchu-Takang, E. G., and Agbor, D. T. (2022). Seed production system and adaptability of okra (Abelmoschus esculentus L.) cultivars in Buea, Cameroon. PloS One 17, e0278771. doi: 10.1371/journal.pone.0278771
Ofoefule, S., Chukwu, A., Anyakoha, N., and Ebebe, I. (2001). Application of Abelmoschus esculentus in solid dosage formulation: Use as a binder for a poorly water soluble drug. Indian J. Pharm. Sci. 63, 234–238.
Okra Seeds Market Report. (2025). Cognitive Market Research, Maharashtra, India. Available at https://www.cognitivemarketresearch.com/okra-seeds-market-report (Accessed April 15, 2025).
Omoniyi, S. A., Idowu, M. A., and Adeola, A. A. (2018). Potential domestic and industrial utilizations of okra seed: a review. Annals: Food Sci. Technol. 19, 722–730.
Omonkhua, A. A., Okonofua, F. E., Ntoimo, L. F., Aruomaren, A. I., Adebayo, A. M., and Nwuba, R. (2020). Community perceptions on causes of high dizygotic twinning rate in Igbo-Ora, South-west Nigeria: A qualitative study. PloS One 15, e0243169. doi: 10.1371/journal.pone.0243169
Omran, R. G. and Powell, R. D. (1971). Chilling injury in okra (Hibiscus esculentus L.) in relation to plant development and nitrogen metabolism. Plant Soil 35, 357–369. doi: 10.1007/BF01372667
Oyelade, O., Ade-Omowaye, B., and Adeomi, V. (2003). Influence of variety on protein, fat contents and some physical characteristics of okra seeds. J. Food Eng. 57, 111–114. doi: 10.1016/S0260-8774(02)00279-0
Petropoulos, S., Fernandes, Â., Barros, L., and Ferreira, I. C. (2018). Chemical composition, nutritional value and antioxidant properties of Mediterranean okra genotypes in relation to harvest stage. Food Chem. 242, 466–474. doi: 10.1016/j.foodchem.2017.09.082
Rashida, P., Sultan, M., Khan, M., and Noor-ul-Islam, N.-u. (2005). Screening of cotton germplasm against cotton leaf curl begomovirus (CLCuV). Journal of Agriculture and Social Sciences. 1(3), 235–238
Reddy, M. T., Pandravada, S., Sivaraj, N., and Sunil, N. (2016). Characterization of Indian landrace germplasm and morphological traits desirable for designing a customer-driven variety in okra (Abelmoschus esculentus L. Moench). J. Global Agric. Ecol. 6, 7–34.
Sabitha, V., Ramachandran, S., Naveen, K., and Panneerselvam, K. (2011). Antidiabetic and antihyperlipidemic potential of Abelmoschus esculentus (L.) Moench. in streptozotocin-induced diabetic rats. J. Pharm. bioallied Sci. 3, 397–402. doi: 10.4103/0975-7406.84447
Salameh, N. M. (2014). Genetic diversity of okra (Abelmoschus esculentus L.) landraces from different agro-ecological regions revealed by AFLP analysis. American-Eurasian J. Agric. Environ. Sci. 14, 155–160. doi: 10.5829/idosi.aejaes.2014.14.02.12289
Sami, R., Lianzhou, J., Yang, L., Ma, Y., and Jing, J. (2013). Evaluation of fatty acid and amino acid compositions in okra (Abelmoschus esculentus) grown in different geographical locations. BioMed. Res. Int. 2013, 574283. doi: 10.1155/2013/574283
Sawadogo, M., Ouedraogo, J. T., Balma, D., Ouedraogo, M., Gowda, B. S., Botanga, C., et al. (2009). The use of cross species SSR primers to study genetic diversity of okra from Burkina Faso. Afr. J. Biotechnol. 8, 2476–2482.
Schafleitner, R., Kumar, S., Lin, C.-y., Hegde, S. G., and Ebert, A. (2013). The okra (Abelmoschus esculentus) transcriptome as a source for gene sequence information and molecular markers for diversity analysis. Gene 517, 27–36. doi: 10.1016/j.gene.2012.12.098
Schafleitner, R. and Lin, Y.-P. (2025). “Genomic resources for okra breeding,” in Okra: Status, Challenges and Opportunities (Singapore: Springer), 37–40.
Sengkhamparn, N., Verhoef, R., Schols, H. A., Sajjaanantakul, T., and Voragen, A. G. (2009). Characterisation of cell wall polysaccharides from okra (Abelmoschus esculentus (L.) Moench). Carbohydr. Res. 344, 1824–1832. doi: 10.1016/j.carres.2008.10.012
Shi, R., Seiler, C., Knoch, D., Junker, A., and Altmann, T. (2023). Integrated phenotyping of root and shoot growth dynamics in maize reveals specific interaction patterns in inbreds and hybrids and in response to drought. Front. Plant Sci. 14. doi: 10.3389/fpls.2023.1233553
Shwetha, A., Gunnaiah, R., Basavaraja, N., Krishna, K., UdayKumar, H., and Sameer, S. (2024). Discovery of an SSR marker linked to Yellow Vein Mosaic Virus resistance in Okra (Abelmoschus esculentus L. Moench). Genetic Resources and Crop Evolution 71 (6), 2573-2584 . doi: 10.1007/s10722-023-01799-y
Siemonsma, J. (1982). West african okra—Morphological and cytogenetical indications for the existence of a natural amphidiploid of Abelmoschus esculentus (L.) Moench and A. manihot (L.) Medikus. Euphytica 31, 241–252. doi: 10.1007/BF00028327
Singh, H. B. and Bhatnagar, A. (1975). Chromosome number in okra from Ghana. Indian J. Genet. Plant Breed. 36, 24–27.
Singh, P., Chauhan, V., Tiwari, B. K., Chauhan, S. S., Simon, S., Bilal, S., et al. (2014). An overview on okra (Abelmoschus esculentus) and its importance as a nutritive vegetable in the world. Int. J. Pharm. Biol. Sci. 4, 227–233.
Singh, B., Karmakar, P., Singh, P., Maurya, B., Singh, H., Sagar, V., et al. (2023). Okra: breeding and genomics. Vegetable Sci. 50, 261–273. doi: 10.61180/vegsci.2023.v50.i2.01
Singh, H. K., Singh, K., and Meraj, M. (2018). Growth and yield performance of okra [Abelmoschus esculentus (L.) Moench] varieties on farmer’s field. Int. J. Curr. Microbiol. Appl. Sci. 7, 1411–1417.
Srivastava, M., Kathayat, K., and Mashkey, V. (2023). Performance of okra genotypes for different quality parameters. J. Food Chem. Nanotechnol 9, S285–S290. doi: 10.17756/jfcn.2023-s1-037
Suma, A., Joseph John, K., Bhat, K. V., Latha, M., Lakshmi, C. J., Pitchaimuthu, M., et al. (2023). Genetic enhancement of okra [Abelmoschus esculentus (L.) Moench] germplasm through wide hybridization. Front. Plant Sci. 14. doi: 10.3389/fpls.2023.1284070
Sun, J., Xu, G., Hu, Y., Cheng, Y., Wang, X., Yang, J., et al. (2023). Genome-wide assessment of genetic diversity and association mapping for salt tolerance traits in okra (Abelmoschus esculentus L. Moench) using genotyping-by-sequencing. Scientia Hortic. 313, 111922. doi: 10.1016/j.scienta.2023.111922
Tahir, J., Brendolise, C., Hoyte, S., Lucas, M., Thomson, S., Hoeata, K., et al. (2020). QTL mapping for resistance to cankers induced by Pseudomonas syringae pv. actinidiae (Psa) in a tetraploid Actinidia chinensis kiwifruit population. Pathogens 9, 967. doi: 10.3390/pathogens9110967
Temam, N., Mohammed, W., and Aklilu, S. (2021). Variability assessment of okra (Abelmoschus esculentus (L.) Moench) genotypes based on their qualitative traits. Int. J. Agron. 2021, 6678561. doi: 10.1155/2021/6678561
Tiendrébéogo, F., Lefeuvre, P., Hoareau, M., Villemot, J., Konaté, G., Traoré, A. S., et al. (2010a). Molecular diversity of Cotton leaf curl Gezira virus isolates and their satellite DNAs associated with okra leaf curl disease in Burkina Faso. Virol. J. 7, 1–10. doi: 10.1186/1743-422X-7-48
Tiendrébéogo, F., Traoré, V. E., Lett, J.-M., Barro, N., Konaté, G., Traoré, A. S., et al. (2010b). Impact of okra leaf curl disease on morphology and yield of okra. Crop Prot. 29, 712–716. doi: 10.1016/j.cropro.2010.02.007
Tikoo, S. K. (2025). “Introduction,” in Okra: Status, Challenges and Opportunities. Eds. Tikoo, S. K., Angadi, S., Tiwari, A., Yadav, R. K., Tomar, B. S., and Adeniyi, A. H. (Springer, Singapore). doi: 10.1007/978-981-97-9963-3_1
Tiwari, A., Tikoo, S. K., Angadi, S. P., Kadaru, S. B., Ajanahalli, S. R., and Rao, M. V. (2022). Market-driven plant breeding for practicing breeders (Singapore: Springer).
Torkpo, S., Danquah, E., Offei, S., and Blay, E. (2006). Esterase, total protein and seed storage protein diversity in Okra (Abelmoschus esculentus L. Moench). West Afr. J. Appl. Ecol. 9, 1–7. doi: 10.4314/wajae.v9i1.45677
Tseng, M., Breslow, R. A., DeVellis, R. F., and Ziegler, R. G. (2004). Dietary patterns and prostate cancer risk in the National Health and Nutrition Examination Survey Epidemiological Follow-up Study cohort. Cancer Epidemiol. Biomarkers Prev. 13, 71–77. doi: 10.1158/1055-9965.EPI-03-0076
Voichek, Y. and Weigel, D. (2020). Identifying genetic variants underlying phenotypic variation in plants without complete genomes. Nat. Genet. 52, 534–540. doi: 10.1038/s41588-020-0612-7
Wang, R., Li, W., He, Q., Zhang, H., Wang, M., Zheng, X., et al. (2023). The genome of okra (Abelmoschus esculentus) provides insights into its genome evolution and high nutrient content. Horticulture Res. 10, uhad120. doi: 10.1093/hr/uhad120
Wang, K., Li, M., Wen, X., Chen, X., He, Z., and Ni, Y. (2018). Optimization of ultrasound-assisted extraction of okra (Abelmoschus esculentus (L.) Moench) polysaccharides based on response surface methodology and antioxidant activity. Int. J. Biol. macromolecules 114, 1056–1063. doi: 10.1016/j.ijbiomac.2018.03.145
Wasaya, A., Zhang, X., Fang, Q., and Yan, Z. (2018). Root phenotyping for drought tolerance: a review. Agronomy 8, 241. doi: 10.3390/agronomy8110241
Woldetsadik, D., Llorent-Martínez, E. J., Gebrezgabher, S., Njenga, M., Mendum, R., Castillo-López, R., et al. (2022). Okra (Abelmoschus esculentus) in a refugee context in East Africa: Kitchen gardening helps with mineral provision. SN Appl. Sci. 4, 32. doi: 10.1007/s42452-021-04898-6
Xu, G., Cheng, Y., Wang, X., Dai, Z., Kang, Z., Ye, Z., et al. (2024). Identification of single nucleotide polymorphic loci and candidate genes for seed germination percentage in okra under salt and no-salt stresses by genome-wide association study. Plants 13, 588. doi: 10.3390/plants13050588
Yadav, Y., Maurya, P. K., Devi, A. P., Jamir, I., Bhattacharjee, T., Banerjee, S., et al. (2018). Enation leaf curl virus (ELCV): A real threat in major okra production belts of India: A review. J. Pharmacogn. Phytochem. 7, 3795–3802.
Yadav, B., Mehata, D. K., Bhandari, S., Shrestha, S., and Sangroula, G. (2024). Evaluation of different okra (Abelmoschus esculentus L.) cultivars for yield and biomass production. Asian J. Res. Agric. Forestry 10, 99–105. doi: 10.9734/AJRAF/2024/v10i1273
Yahaya, I., Zhou, J., Jiang, S., Xu, R. H., Huang, J., Su, B., et al. (2024). Depiction of Drought Over Africa in the Light of Changing Climate from CMIP6 Models. Earth Syst. Environ. 8, 1011–1035. doi: 10.1007/s41748-024-00502-0
Yang, L., Gao, H., Meng, L., Fu, X., Du, X., Wu, D., et al. (2021). Nondestructive measurement of pectin polysaccharides using hyperspectral imaging in mulberry fruit. Food Chem. 334, 127614. doi: 10.1016/j.foodchem.2020.127614
Yıldız, M., Ekbiç, E., Düzyaman, E., Serçe, S., and Abak, K. (2016). Genetic and phenotypic variation of Turkish Okra (Abelmoschus esculentus L. Moench) accessions and their possible relationship with American, Indian and African germplasms. J. Plant Biochem. Biotechnol. 25, 234–244. doi: 10.1007/s13562-015-0330-x
Yıldız, M., Koçak, M., and Baloch, F. S. (2015). Genetic bottlenecks in Turkish okra germplasm and utility of iPBS retrotransposon markers for genetic diversity assessment. Genet. Mol. Res. 14 (3), 10588-602. doi: 10.4238/2015.September.8.20
Yuan, C., Zhang, C., Wang, P., Hu, S., Chang, H., Xiao, W., et al. (2014). Genetic diversity analysis of okra (Abelmoschus esculentus L.) by inter-simple sequence repeat (ISSR) markers. Genet. Mol. Res. 13, 3165–3175. doi: 10.4238/2014.April.25.1
Zandjanakou-Tachin, M., Zinsou, V. A., Dannon, E., Towanou, M. A., Kone, D., Sorho, F., et al. (2021). First report of Okra leaf virus (OLCV) in Benin its distribution and farmers’ cultural practices for the disease management. J. Appl. Biosci. 162, 16726–16743. doi: 10.35759/JABs.162.4
Zhan, Y., Wu, Q., Chen, Y., Tang, M., Sun, C., Sun, J., et al. (2019). Comparative proteomic analysis of okra (Abelmoschus esculentus L.) seedlings under salt stress. BMC Genomics 20, 1–12. doi: 10.1186/s12864-019-5737-7
Zhang, C., Marzougui, A., and Sankaran, S. (2020). High-resolution satellite imagery applications in crop phenotyping: An overview. Comput. Electron. Agric. 175, 105584. doi: 10.1016/j.compag.2020.105584
Zheng, W., Zhao, T., Feng, W., Wang, W., Zou, Y., Zheng, D., et al. (2014). Purification, characterization and immunomodulating activity of a polysaccharide from flowers of Abelmoschus esculentus. Carbohydr. polymers 106, 335–342. doi: 10.1016/j.carbpol.2014.02.079
Zibelo, H., Wtsadik, K., and Sharma, J. (2016). Effect of inter-and intra-row spacing on growth and yield of okra (Abelmoschus esculentus L. Moench) at Humera, Northern Ethiopia. J. Biology Agric. Healthcare 6, 92–108.
Keywords: Abelmoschus esculentus, opportunity crops, value-added traits, trait improvement, pectins, mucilage
Citation: Ayenan MAT, Vihou F, Ambali M, Opoku JA, Ibiotye DO and Schafleitner R (2025) Tapping into the potential of okra (Abelmoschus spp.) in Africa: integrating value-added traits into breeding. Front. Plant Sci. 16:1631221. doi: 10.3389/fpls.2025.1631221
Received: 19 May 2025; Accepted: 18 July 2025;
Published: 14 August 2025.
Edited by:
Zeynep Aksoylu Özbek, Manisa Celal Bayar University, TürkiyeReviewed by:
Abe Shegro Gerrano, Agricultural Research Council of South Africa (ARC-SA), South AfricaSandeep N, ICAR-Indian Institute of Millets Research (IIMR), India
Thomas Wilbur Davis, University of Nebraska-Lincoln Libraries, United States
Kubra Unal, Selçuk University, Türkiye
Copyright © 2025 Ayenan, Vihou, Ambali, Opoku, Ibiotye and Schafleitner. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Mathieu A. T. Ayenan, bWF0aGlldS5heWVuYW5Ad29ybGR2ZWcub3Jn
 Mathieu A. T. Ayenan
Mathieu A. T. Ayenan Fabrice Vihou
Fabrice Vihou Mwasilwa Ambali1
Mwasilwa Ambali1 Jacinta Adoma Opoku
Jacinta Adoma Opoku Dorcas Olubunmi Ibiotye
Dorcas Olubunmi Ibiotye Roland Schafleitner
Roland Schafleitner