- 1Institute of Plant Protection, Chinese Academy of Agricultural Sciences, Beijing, China
- 2College of Plant Protection, Southwest University, Chongqing, China
Ralstonia solanacearum phylotype I mulberry strains are causative agent of bacterial wilt of mulberry. Current diagnostic methods are not adopted to the mulberry wilt disease. In this study, we developed a rapid method, loop-mediated isothermal amplification (LAMP), to detect R. solanacearum phylotype I mulberry strains. A set of six primers was designed to target the clone MG67 sequence in this LAMP detection which can be completed in 20 min at 64°C. The results of the LAMP reaction could be observed with the naked eye due to magnesium pyrophosphate precipitate produced during the reaction or the color change after adding SYBR Green I. The specificity of the LAMP was confirmed using DNA from 46 representative strains of R. solanacearum and 7 other soil-borne bacteria strains. This method was also of high sensitivity and could be used to detect the presence of less than 160 fg genomic DNA or 2.2 × 102 CFU/ml of bacterial cells per 25 μl reaction volume, moreover, the presence of plant tissue fluid did not affect the sensitivity. Since it does not require expensive equipment or specialized techniques, this LAMP-based diagnostic method has the potential to be used under field conditions to make disease forecasting more accurate and efficient.
Introduction
Bacterial wilt caused by Ralstonia solanacearum has an extremely wide geographic distribution in tropical, subtropical and some warm temperate regions of the world, and poses a huge threat to the production of many economically important food and cash crops (Hayward, 1991). R. solanacearum is metabolically versatile and can survive not only in soil but also in latently infected plants and water (Xue et al., 2011). The species complex has been subdivided into four phylotypes (Phylotypes I, II, III, and IV) corresponding to the four genetic groups identified via sequence analysis (Fegan and Prior, 2005).
The causative agents of BWM was designated as phylotype I mulberry strains. BWM was initially found in Guangdong province of Southern China (He et al., 1983), and has been discovered as a restricted distribution in Guangxi, Yunnan, Shandong, Jiangxi, and Zhejiang provinces (Pan et al., 2013). Since 2002, this vascular disease has increased severely in Zhejiang province and has been regarded as an important bottleneck in the local sericulture industry. BWM has been dramatically reduced to a low damage level attributed to the implementation of integrated management strategies, i.e., phytosanitation, rotation with corn and use of tolerant cultivars. However, the propagating materials, contaminated by R. solanacearum phylotype I mulberry strains, have important impact on re-outbreaks of BWM in some orchards. Therefore, rapid detection and accurate diagnosis of R. solanacearum phylotype I mulberry strains in the field is essential to prevent the bacterial dissemination with asymptomatic and symptomless germplasms from poorly controlled areas to well managed ones.
Several diagnostic methods have been widely used to detect R. solanacearum. For instance, serological techniques relying on the composition of bacteria (Griep et al., 1998) and molecular techniques that are based on the DNA or RNA of viable bacterial cells (Seal et al., 1993; Weller et al., 2000; Bentsink et al., 2002) have been reported. Furthermore, some PCR-based methods have been developed for subspecies-specific detection (Horita et al., 2004; Prior and Fegan, 2005; Thammakijjawat et al., 2006; Smith and De Boer, 2009; Paret et al., 2010; Ha et al., 2012). However, since the conserved genomic sequence of R. solanacearum phylotype I mulberry strains have not been identified, less work has been done on the detection of them. A multiplex PCR method based on the genetic diversity within BWM strains in China was developed in 2013 to specifically detect the mulberry strains (Pan et al., 2013). Although this was a simple and efficient method using the primer set MG67-F/MG67-R in combination with the species-specific primer pair AU759f/AU760r, there were still shortcomings to overcome. It required a high precision thermal cycler for the amplification as well as electrophoresis gel imaging equipment for the determination of the results, which made this method only available in the laboratory and unsuitable in the field. In addition, it could not be used for rapid detection, since it took more than 2 h.
Loop-mediated isothermal amplification (LAMP), first described by Notomi et al. (2000), has the potential to be more suitable for field use than PCR-based methods since it allows the synthesis of large amounts of DNA in a short time and under isothermal conditions ranging from 60 to 65 °C (Tomotada et al., 2003; Tomlinson et al., 2010). As a novel method to amplify nucleic acid, LAMP employing a set of six primers and a Bst DNA polymerase with strand displacement activity is characterized by its high specificity, efficiency, and rapidity. (Nagamine et al., 2002).
Loop-mediated isothermal amplification requires a set of four specifically designed primers that recognize six distinct sequences on the target DNA; its specificity is extremely high. The primers essential for the LAMP reaction include a FIP, a BIP, a forward outer primer (F3) and a backward outer primer (B3). FIPs and BIPs, of which both the 3′ region and 5′ region match the target, interact with upstream external primers resulting in the displacement of strands containing self-complementary regions that form stem-loop structures (Tomlinson et al., 2010). Nagamine et al. (2002) developed a method that accelerates the LAMP reaction by using additional primers, termed loop primers. Those primers can also improve the selectivity since they need appropriate starting material for LAMP cycling (Nagamine et al., 2002). The results of LAMP can be observed by the naked eye since the reaction by-product pyrophosphate ions bind to magnesium ions and form a white precipitate of magnesium pyrophosphate (Mori et al., 2001). In addition, the LAMP products can also be directly detected by adding SYBR Green I to the tube and observing the color-change of the solution, which turns green if the LAMP amplification occurs, while it remains orange if there is no amplification (Tomotada et al., 2003). Mori et al. (2004) designed an assay to monitor LAMP products in real-time by measurement of turbidity or fluorescence with a real-time thermal cycler or a real-time turbidimeter.
Given the great importance of the detection, identification and surveillance of R. solanacearum phylotype I mulberry strains in BWM integrated management, and that there have been no simple, rapid and sensitive LAMP-based methods available, we designed a subspecies-specific LAMP method targeting the MG67 DNA fragment which is generated from suppressive subtractive hybridization and proved to be well conserved in phylotype I mulberry strains (Pan et al., 2013; Cao et al., 2015). And we further evaluated the method on naturally infected mulberry samples in this study.
Materials and Methods
Strains Used in This Study
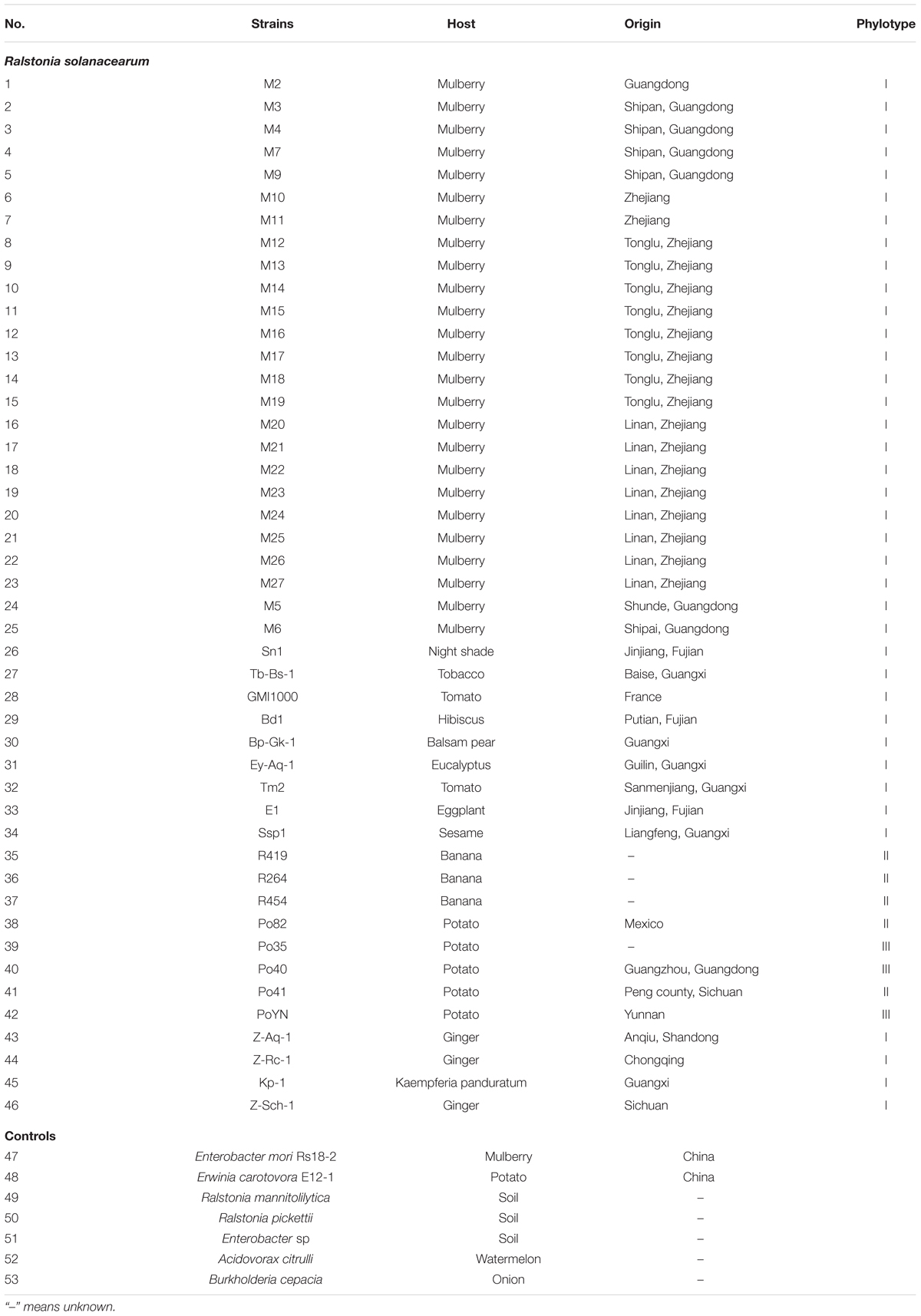
A total of 53 bacterial strains including 46 R. solanacearum strains and 7 other control bacteria strains were used in this study, and the characteristics of all strains are briefly presented in Table 1.
DNA Preparation
The LAMP assays were evaluated on 53 bacterial strains. All strains were maintained in sterile distilled water at room temperature, streaked onto TZC agar (nutrient agar supplemented with 0.05% tetrazolium chloride) (Kelman, 1954), and incubated at 28°C for 48 h. Single slimy, milky colonies with a pink center were transferred to nutrient agar (0.3% beef extract, 1.0% glucose, 0.5% peptone, 0.05% yeast extract, and 1.8% agar) at 28°C for 48 h. Purified genomic DNAs were isolated with a commercial kit (Genomic DNA Purification kit; BioTeKe, Beijing, China) according to the manufacturer’s instructions. DNA concentrations were quantified photometrically (absorbance measurements at 260 and 280 nm, with a NanoVue Plus spectrophotometer; NanoDrop Technologies, USA).
Primer Design
The LAMP primers (Figure 1) of the R. solanacearum phylotype I mulberry strains were designed targeting the gene MG67 obtained from GenBank (accession number GS923075) and using the Primer Explorer V.4 software tool available on the Eiken Genome site1 provided by Eiken Chemical Co., Ltd. (Tokyo, Japan). The detailed sequences of the primers used for amplification of R. solanacearum phylotype I mulberry strains are shown in Table 2.
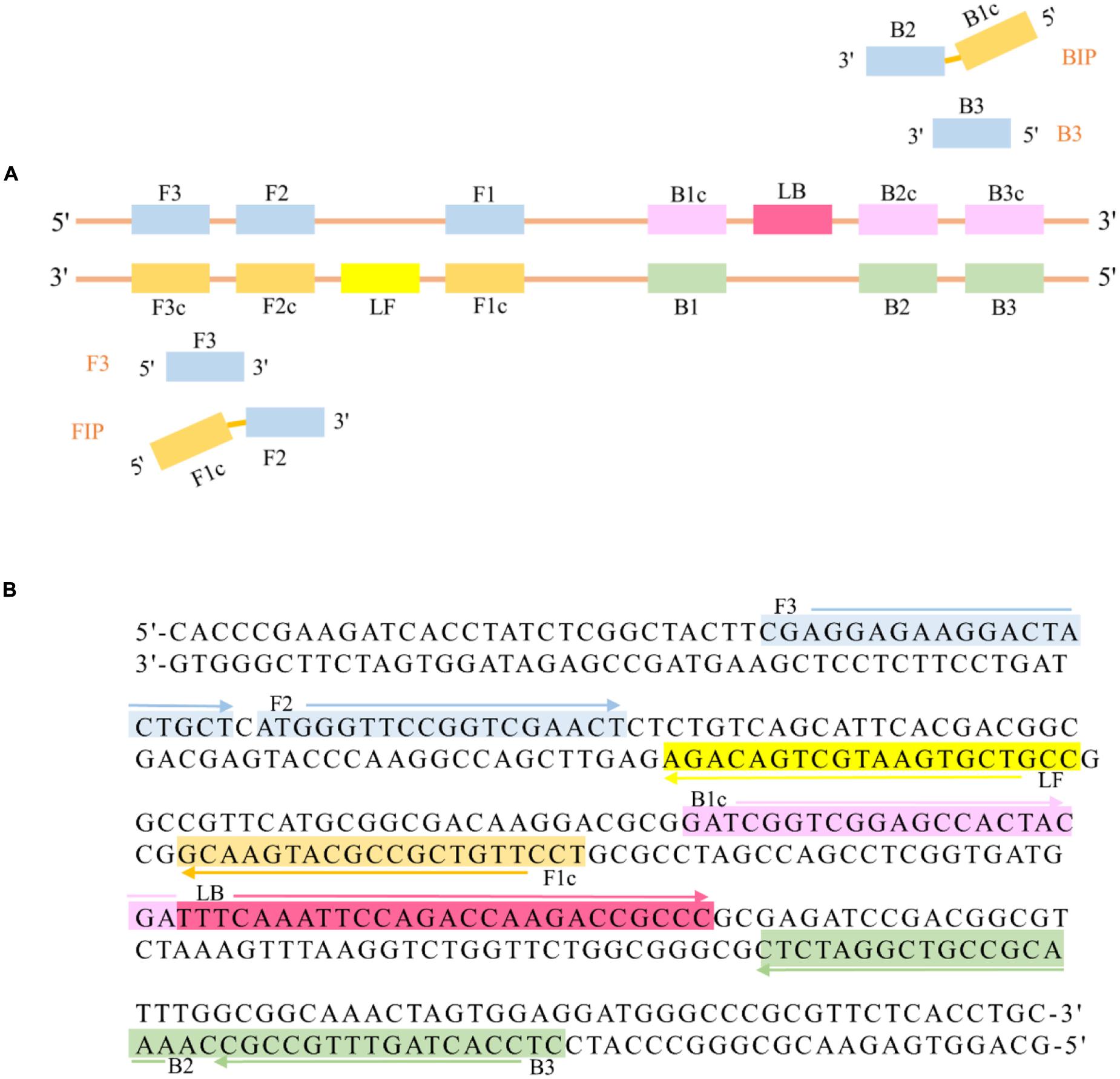
FIGURE 1. (A) Schematic diagram of two inner primers (FIP, BIP), two outer primers (F3, B3), and two loop primers (LF, LB) for LAMP. (B) MG67 target DNA fragment. This sequence was used to design three pairs of primers, which are shown in different colors, with the arrows showing the orientation of the primers.
LAMP Reaction
Twenty five microliter of the LAMP reactions mixture contained 1.6 μM FIP and BIP, 0.2 μM F3 and B3, 0.4 μM LF and LB, 1.4 mM dNTPs, 1.0 M betaine (Sigma–Aldrich Corp, St. Louis, MO, USA), 20 mM Tris-HCl (pH 8.8), 50 mM KCl, 10 mM (NH4)2SO4, 8 mM MgSO4, 0.1% Tween-20, 8 U of the Bst DNA polymerase large fragment (New England Biolabs, USA), and 1 μl of the template DNA. Sterile deionized water was used as the template for the negative control.
These reactions were carried out in 0.2 ml microtubes, 1 μl of 1/10 diluted original SYBR Green I (Molecular Probes Inc.) was added in the inner lid of every microtube before incubating the reactions in a Veriti 96 well thermal cycler (Applied Biosystems, USA).
Positivity Criteria and Running-Time Determination
Time of positivity (Tp) and melting temperature (Tm) are two basic parameters for positive signals. Tp is the time when the second derivative of fluorescent amplification reaches its peak above the baseline value, and this is expressed in minutes. The Tm is the temperature expressed in °C at which the amplification products melt into two single-stranded DNA molecules. In order to determine the optimal reaction temperature and time, we measured the turbidity of seven samples at different temperatures ranging from 60 to 65°C in real time.
ROX reference dye II (TaKaRa) (0.15 μl) and Eva Green (Biotium) (1.2 μl) were added to the LAMP reaction for the real-time LAMP. A real-time thermal cycler ABI 7500 (Applied Biosystems) with version 2.3 of the software was used to amplify and measure fluorescence at different reaction temperature and cycle. The program was: 1 s to react and 59 s to collect data for 50 cycles or more (1 cycle is 1 min) followed by the melting curve.
Conventional Phylotype I Mulberry Strains-Specific Multiplex PCR
Phylotype I mulberry strains-specific primer set MG67-F/MG67-R, in combination with the species-specific primer pair AU759f/AU760r, was used to identify R. solanacearum phylotype I mulberry strains strains in a single PCR assay characterized by providing the expected amplicons of 156 and 282 bp (Pan et al., 2013). Each PCR mix had a total volume of 25 μl and contained 1× Taq MasterMix (PCR buffer, 1.5 mM MgCl2, 250 μM of each dNTP, 50 mM KCl, 10 mM Tris-HCl, and 1.25 U of Taq DNA polymerase; Tiangen Biotech), 6 pmoles of the primers MG67-F and MG67-R, and 4 pmoles of the primers AU759f and AU760r).
Initial denaturation was conducted at 94°C for 5 min, followed by 30 cycles of denaturation (30 s at 94°C), annealing (30 s at 60°C) and extension (30 s at 72°C), with a final extension step for 10 min. Subsequently, the PCR products were observed by being subjected to agarose gel electrophoresis.
Verification of LAMP Detection Specificity
Fifty three representative strains that included 23 typical R. solanacearum phylotype I mulberry strains, 2 atypical R. solanacearum phylotype I mulberry strains, 21 R. solanacearum strains isolated from other plant species and 7 other soil-borne bacteria strains (Enterobacter mori Rs18-2, Erwinia carotovora E12-1, R. mannitolilytica, R. pickettii, Enterobacter sp., Acidovorax citrulli, and Burkholderia cepacia) were used to determine the specificity of the primers.
Sensitivity of LAMP
In order to assess the sensitivity of the LAMP, serial 10-fold dilutions of the reference strain M7 pure culture and genomic DNA (at an initial concentration of 160 ng/μl) were prepared in sterile deionized water. A pure culture of M7 was plated on nutrient agar at 28°C for 48 h. The cell suspension from 2-day-old culture of M7 strain were adjusted to 108 CFU/ml, and a series of decimal dilutions ranging from 108 to 102 CFU/ml were prepared. Genomic DNA was extracted as mentioned above and followed by 10-times gradient dilution.
Bacterial suspension or genomic DNA (1 μl) from each dilution series was added to the LAMP and conventional PCR reaction mixture, respectively, as the amplification template.
In addition, healthy mulberry leaves collected from Beijing were crushed in 10 ml of sterile deionized water and then mixed with newly prepared bacterial suspension of strain M7 to result in the following concentrations: 107, 106, 105, 104, 103 CFU/ml, 102, 101 CFU/ml. A 1 μl sample of the resulting supernatant was then assayed using the LAMP method.
Application of LAMP for Plant Samples
To demonstrate the field application of LAMP as a diagnostic tool for R. solanacearum phylotype I mulberry strains, 20 samples were collected from Tonglu district, Hangzhou city, in east China’s Zhejiang province, where is the epidemic areas. And 2 mulberry samples were collected from Beijing, where is the BWM-free areas.
The surfaces of stems or roots were flushed with clean water and disinfected with 70% ethyl alcohol. 1-cm-long plant tissues taken from these samples were cut into pieces with disinfected garden shears in sterile Petri dishes containing 10 ml of sterile deionized water, then suspended for 30 min. A total of 1 μl of each sample suspension was used as the amplification template for both the LAMP method and the conventional PCR.
Results
Visual Detection of the LAMP Reaction
One of the characteristics of LAMP is its ability to give results that can be identified with the naked eye due to the magnesium pyrophosphate precipitate produced during the reaction. We used 1 μl of genomic DNA of strain M7 as a template for the positive control and 1 μl of water as a template for the negtive control tn the LAMP assay. White turbidity could be seen in the positive reaction (+), whereas the negative control (-) remained clear (Figure 2A). A 1 μl aliquot of the 1/10 diluted original SYBR Green I was added to the solution of the positive and negative controls. After brief centrifugation, the positive control (+) turned green, while the negative control (-) remained orange (Figure 2B).
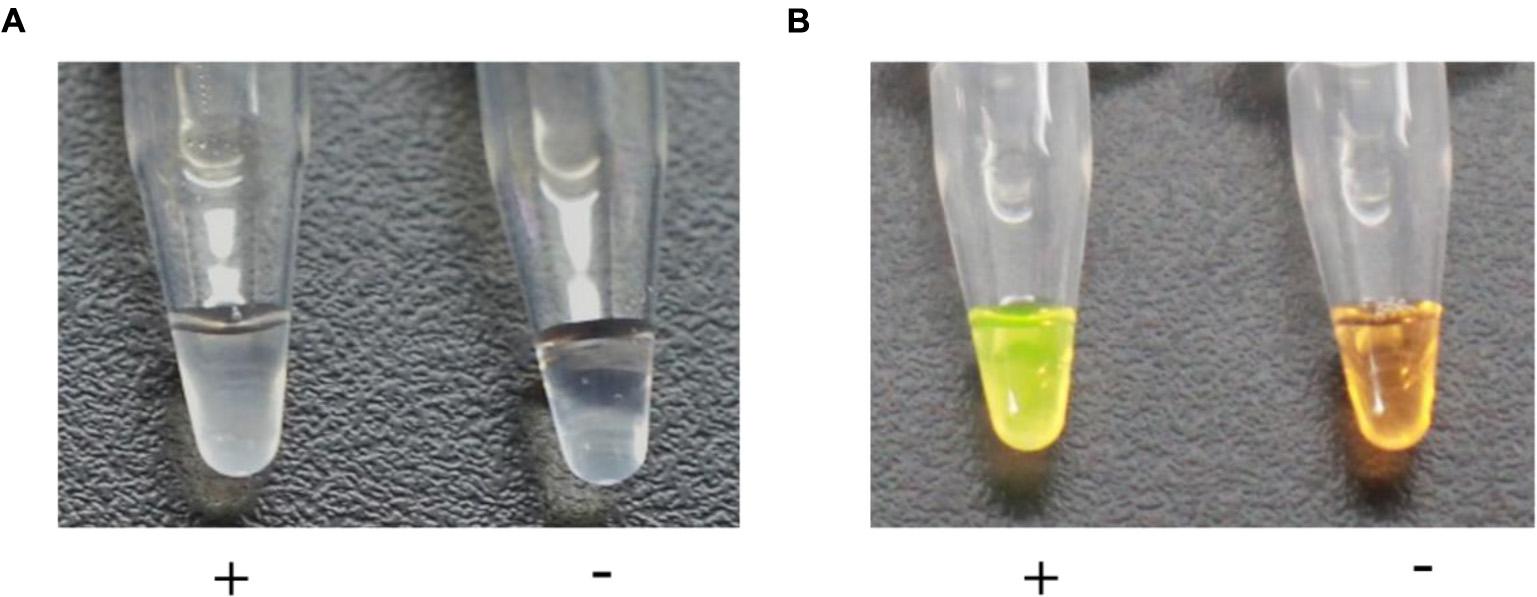
FIGURE 2. Visual assessment of DNA amplification by LAMP. (A) The turbidity indicating a positive reaction (+) was caused by the magnesium pyrophosphate precipitate produced during DNA synthesis, whereas the negative control (-) remained clear. (B) The solution turned green (+) in the presence of a LAMP amplification, while it remained orange (-) with no amplification.
Melting Curve Analysis
In order to analyze the LAMP reactions in real-time, we used the genomic DNA of strain M7 as a template to perform real-time LAMP at given temperatures with a real-time thermal cycler ABI 7500. The real-time kinetics of the LAMP reaction were studied by monitoring fluorescence as described in Materials and Methods. The LAMP reactions were performed at different temperatures from 60 to 65°C; as shown in Figure 3, reaction at 64°C was the most productive as well as the most time-saving, and this amplification reached its peak in the 17th minute. Therefore, the LAMP reaction was finally determined to be best carried out at 64°C for 20 min.
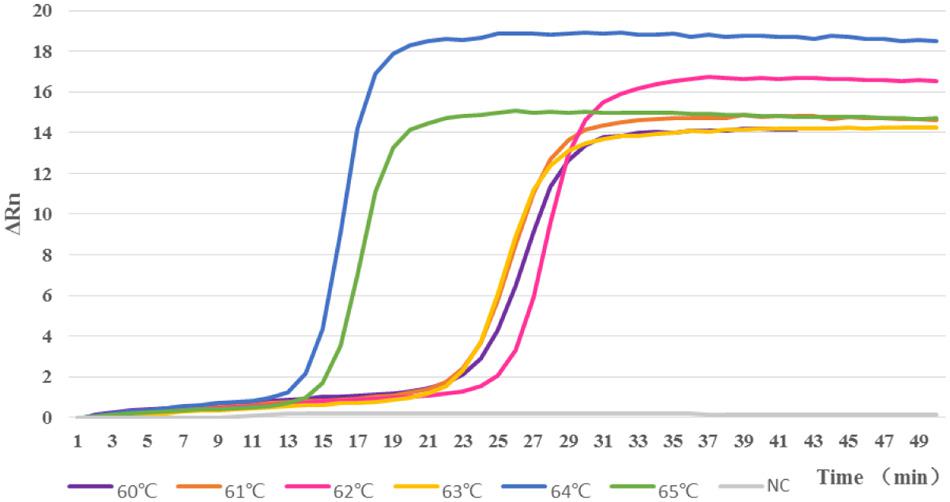
FIGURE 3. Reactions were carried out at different temperatures from 60 to 65°C. When tested at 64°C, the LAMP assay had the highest amplification efficiency, compared with the reactions at other temperatures.
Specificity of LAMP
The specificity of the LAMP for detecting R. solanacearum phylotype I mulberry strains was analyzed using genomic DNA isolated from 46 representative R. solanacearum strains that belonged to Phylotypes I (sample 1–34; 43–46), II (sample 35–38; 41), and III (sample 39–40; 42), and 7 other pathogens, of which Enterobacter mori Rs18-2 (sample 47) and Erwinia carotovora E12-1 (sample 48) cause bacterial diseases in mulberry in China, R. mannitolilytica (sample 49) and R. pickettii (sample 50) are closely related to R. solanacearum, and B. cepacia (sample 51), Enterobacter sp. (sample 52) and A. citrulli (sample 53) are common soil-borne bacteria in nature.
Positive reactions showed bright green fluorescence, while negative ones remained light orange after brief centrifugation. Amplifications were detected in all R. solanacearum phylotype I mulberry populations; in contrast, no amplifications were observed from other strains (Figure 4). This result is indicative that the primer set could be used to specifically detect R. solanacearum phylotype I mulberry strains. The conventional PCR with multiplex primers (MG67-F/MG67-R and AU759f/AU760r) also showed good specificity.

FIGURE 4. Specificity of LAMP detection. Tubes 1–53 correspond to strains 1–53 listed in Table 1. Tube N, negative control.
Sensitivity of LAMP
The sensitivity was first evaluated with a 10-fold dilution series of genomic DNA of strain M7. The results (Figure 5) showed that LAMP could detect original, 10-1, 10-2, 10-3, 10-4, 10-5, and 10-6 diluent genomic DNA of strain M7, and the detection limit was 160 fg; while the conventional PCR could only detect original, 10-1, 10-2, 10-3, 10-4, and 10-5, diluent genome DNA of strain M7, which showed the sensitivity of the former was 10 times higher than that of the latter.
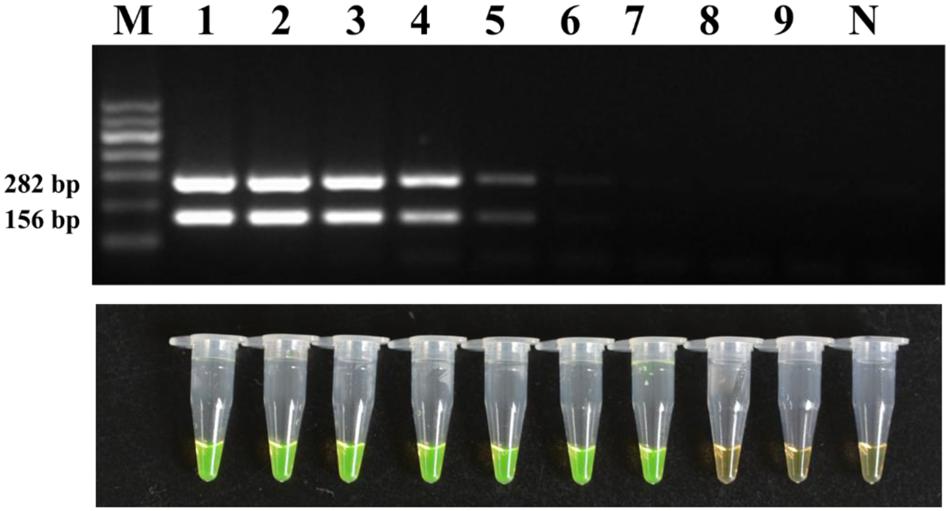
FIGURE 5. Detection limit of the conventional PCR and LAMP method for DNA. Tubes/lanes 1–9, nine different DNA concentrations made from a series of 10-fold dilutions: 160, 160 × 10-1, 160 × 10-2, 160 × 10-3, 160 × 10-4, 160 × 10-5, 160 × 10-6, 160 × 10-7, 160 × 10-8 ng/μl, respectively. Tube/lane N, negative control. Lane M, Marker I (700, 600, 500, 400, 300, 200, 100 kb) (BingDa Biotech).
The sensitivity was then evaluated with a 10-fold dilution series of pure cultures of strain M7. The detection limit reached as low as 2.2 × 102 CFU/ml of bacterial cells which is 100 times higher than the conventional PCR (Figure 6).
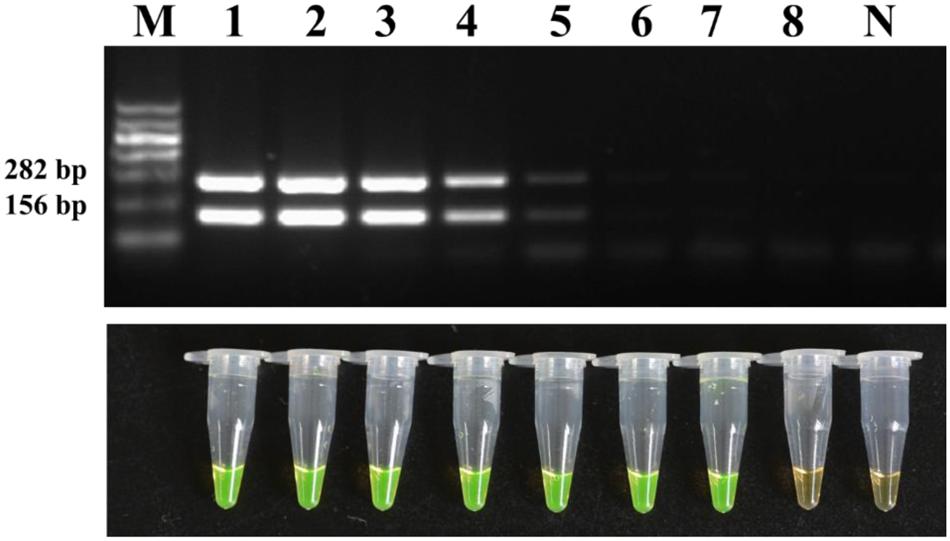
FIGURE 6. Detection limit of the conventional PCR and LAMP method for the bacterial suspensions. Tubes/lanes 1–8, eight different cell concentrations made from a series of 10-fold dilutions: 2.2 × 108, 2.2 × 107, 2.2 × 106, 2.2 × 105, 2.2 × 104, 2.2 × 103, 2.2 × 102, 2.2 × 101 CFU/ml, respectively. Tube/lane N, negative control. Lane M, Marker I (700, 600, 500, 400, 300, 200, 100 kb) (BingDa Biotech).
The LAMP method could also detect 2.2 × 102 CFU/ml of bacterial suspension of strain M7 mixed with the mulberry extract (Figure 7), which was indicative that the LAMP reaction was not affected by the presence of plant tissue fluid.
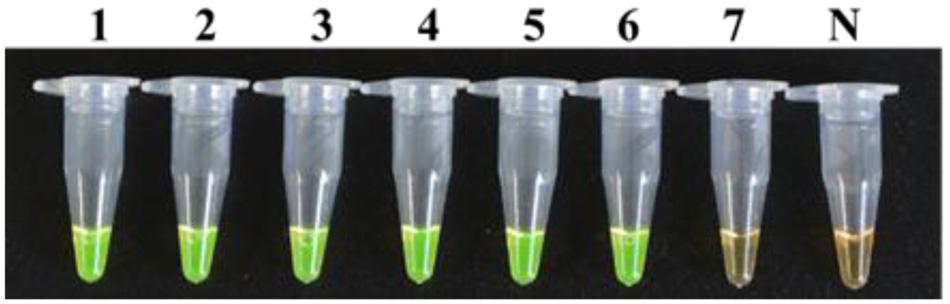
FIGURE 7. Detection limit of the LAMP method for a dilution series of strain M7 with mulberry extracts. Tubes 1–7, seven different dilutions made from a series of 10-fold bacterial suspensions of strain M7 with mulberry extracts: 2.2 × 107, 2.2 × 106, 2.2 × 105, 2.2 × 104, 2.2 × 103, 2.2 × 102, 2.2 × 101 CFU/ml. Tube N, negative control.
Application of LAMP for Plant Samples
The LAMP method and conventional PCR were performed on 22 mulberry samples collected from Zhejiang province and Beijing in China. LAMP and PCR were both positive for samples 1–20, for some of which obvious symptoms were observed. The non-infected samples (21–22) showed negative results (Figure 8). These results are indicative of the high detection capability of R. solanacearum phylotype I mulberry strains-specific LAMP.
‘
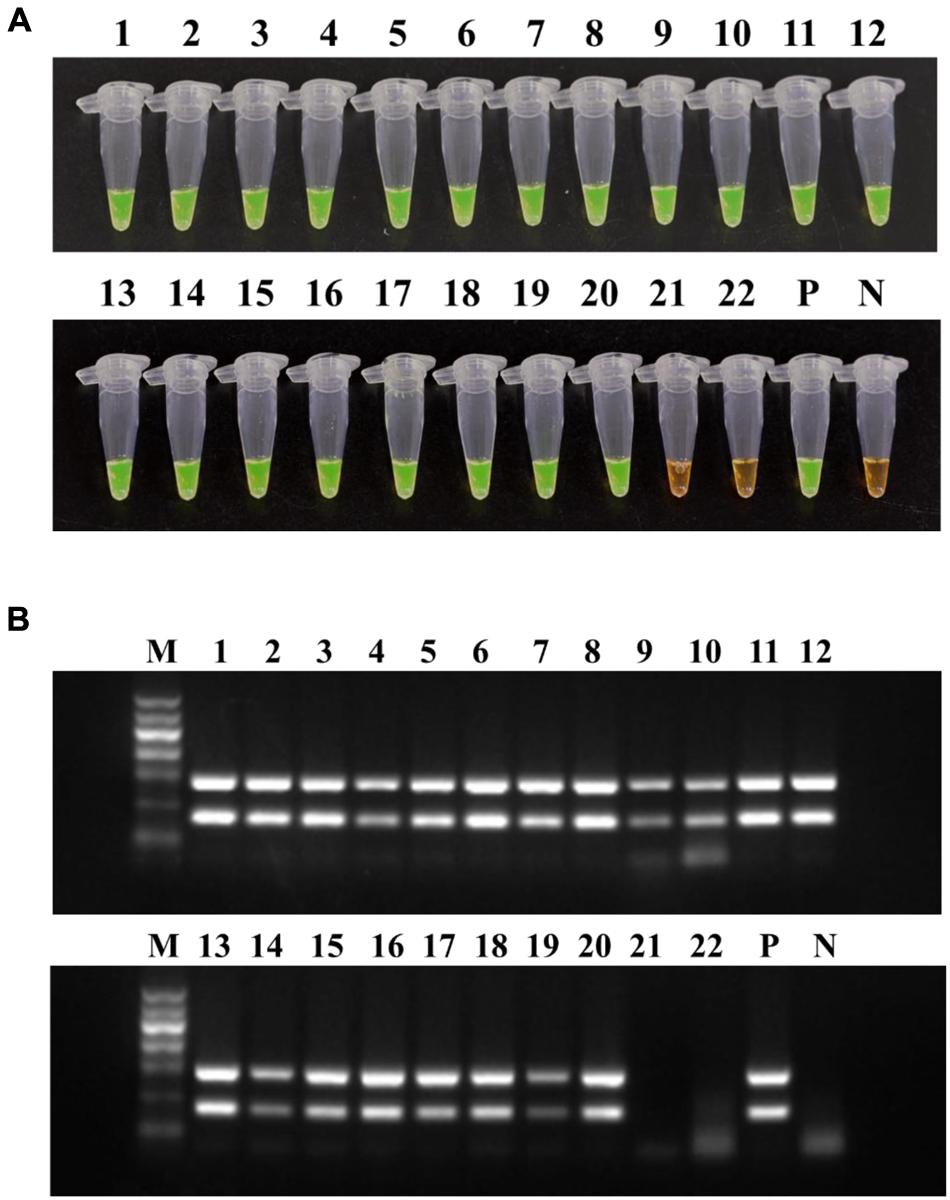
FIGURE 8. Application of LAMP (A) and conventional PCR (B) on mulberry samples. Tubes/lanes 1–20, mulberry samples collected from Zhejiang province. Tubes/lanes 21–22, mulberry samples collected from Beijing. Tube/lane P, positive control. Tube/lane N, negative control. Lane M, Marker I (700, 600, 500, 400, 300, 200, 100 kb) (BingDa Biotech).
Discussion
A plant disease early warning system based on rapid, sensitive and reliable detection of pathogens is the cornerstone of disease-management decision making. Up to now, numerous valuable cultures, serology, PCR and especially LAMP-based methods have been available for the detection of R. solanacearum at both the species and subspecies level (Kubota et al., 2008, 2011a,b; Lenarcic et al., 2014; Kubota and Jenkins, 2015). In contrast to the conventional PCR method, LAMP was suitable for on-site detection in the field because of its rapidity and robustness, and it could be applied even by laymen without the need of elaborate laboratory equipment (Kong et al., 2016).
Ralstonia solanacearum phylotype I mulberry strains, indigenous to China and undergoing lengthy coevolution with mulberry, constitutes a persistent menace to China’s sericulture industry. So far, however, no LAMP-based method for the detection of R. solanacearum phylotype I mulberry strains has been developed. Therefore, accurate identification of R. solanacearum at the infra-species level may be helpful to establish the most economically feasible crop rotation strategy.
In this study, to develop a highly practical and valid LAMP-based method for detecting R. solanacearum phylotype I mulberry strains, we designed a primer set consisting of six primers, in which F3, B3, FIP and BIP recognized six distinct sequences on the target DNA, MG67. LB and LF, the other two loop primers, which were designed for improving the detection efficiency, significantly shortened the reaction time from 70 to 18 min.
To evaluate the specificity of the LAMP-based method, 53 strains were used in this study, including 46 R. solanacearum strains covering Phylotypes I, II, III and seven other soil-borne bacteria strains. As expected, R. solanacearum phylotype I mulberry strains could be easily distinguished from the other strains, particularly the M5 and M6 that are atypical mulberry strains. The same results were observed with a multiplex PCR developed by Pan et al. (2013).
The LAMP method also showed high sensitivity over its PCR counterpart with the detection limit of 160 fg genomic DNA, which is 10 times higher than conventional PCR, and 2.2 × 102 CFU/ml of bacterial cells, which is 100 times higher than conventional PCR. In addition, the sensitivity of the phylotype I mulberry strains-specific LAMP was higher than those of the LAMP assays of Kubota et al. (2008) and Lenarcic et al. (2014).
Since the LAMP reaction could produce many bands of different sizes, forming a ladder of DNA fragments of 100 bp and larger (Kubota et al., 2008), it is common to verify the amplification results with gel electrophoresis. However, the reaction products would easily cause aerosol pollution under high temperature conditions, which results in higher false positive rates. In addition, taking more time and energy for the gel electrophoresis makes the LAMP assay not suitable for application in the field. Lenarcic et al. (2014) used Genie II equipment to monitor the process of amplification. Although amplification processes were visible by this, the detection costs were greatly increased. A relatively simple approach to detect LAMP amplicons is observing the turbidity-change due to the formation of white precipitate, magnesium pyrophosphate (Mori et al., 2001; Tomotada et al., 2003). For better visibility of the reaction result, DNA intercalating dyes such as propidium iodide, ethidium bromide, methylene blue, acridine orange, and SYBR Green I have been added to reaction tubes (Hill, 2008); among them, SYBR Green I produced the best visual discrimination. Kubota et al. (2011b) developed a non-instrumented nucleic acid amplification method which can be potentially used in field for detection of R. solanacearum race 3-biovar 2 strains.
In this study, we added 1 μl of 1/10 diluted original SYBR Green I onto the inner lid of every centrifuge tube before the reaction. Since the color change depended on the amount of DNA, the solutions would turn green with the presence of the LAMP amplicons after brief centrifugation. In this way, not only aerosol pollution caused by opening the lids could be avoided but visual effect of LAMP detection could be improved as well. In addition, we could save more time and money. Compared to conventional phylotype I mulberry strains-specific PCR, the established LAMP-based method can be performed well in a Veriti 96 well thermal cyclera water bath, a heating block and even an insulated mug (AIJIABAO®, China), thus makeing it possible to be applied in on-site detection.
The applicability of the described LAMP-based assay was verified with a total of 22 field samples, collected from both BWM-free and epidemic areas, by a parallel test using multiplex PCR, plating isolation, and the LAMP method. The LAMP-based method showed significant advantages over its counterparts. Firstly, it was more efficient, taking less than an hour as compared with 3 h for PCR and several days for isolation. Secondly, it was more user-friendly, convenient and inexpensive, since either planting isolation or PCR can only be conducted on dedicated equipment in laboratories, whereas the phylotype I mulberry strains-specific LAMP assay can be run in any simple constant-temperature device, water or metal bath, or even a vacuum insulation cup; thereby the LAMP method has great potential to be used in the field. Lastly, due to the high population of R. solanacearum present in diseased mulberry samples, no difference in detection results were observed by using phylotype I mulberry strains-specific LAMP and conventional PCR. However, in the case of latently infected mulberry propagation material, if occur, in which phylotype I mulberry strains occurred in low titer, the more sensitive LAMP method could substantially reduce the risk of false negative results. The plating isolation only gave 16 positive results as a result of the overgrowth of saprophytic bacteria (data not shown). Nevertheless, conventional culture-based methods cannot be completely replaced by DNA-based methods in the plant- and food-related fields.
This is the first report on the application of the LAMP assay, as an alternative method, for the specific detection of R. solanacearum phylotype I mulberry strains.
Conclusion
The LAMP-based method developed in this study allows for rapid and accurate identifications of R. solanacearum phylotype I mulberry strains. Due to its easy operation without sophisticated equipment, it could play an important role in the early diagnosis of BWM and provide technical support for disease-management decision making.
Author Contributions
JX and JF designed this experiment and helped to collecting the plant samples. WH performed this experiment and wrote this manuscript. HZ and JX provided assistance during the experiment. JF, JX, SW, XK, and WD revised this manuscript.
Funding
This work was supported by the Chinese National Natural Science Foundation (31272008, 31371908), the Special Fund for Agro-scientific Research in the Public Interest (201303015), and the National Key Technology Support Program (2015BAD08B03).
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgment
The authors gratefully acknowledge Professor Guanlin Xie, Zhejiang University (Hangzhou, Zhejiang, China), for help with plant sample collection.
Abbreviations
BIP, backward inner primer; BWM, bacterial wilt of mulberry; FIP, forward inner primer; LAMP, loop-mediated isothermal amplification.
Footnotes
References
Bentsink, L., Leone, G., Leone, G., van Beckhoven, J., van Schijndel, H., van Gemen, B., et al. (2002). Amplification of RNA by NASBA allows direct detection of viable cells of Ralstonia solanacearum in potato. J. Appl. Microbiol. 93, 647–655. doi: 10.1046/j.1365-2672.2002.01725.x
Cao, M. Q., Bao, Q., Yang, C. F., Wang, J., Sheng, S., and Wu, F. (2015). Establishment of a qPCR method for rapid detection of Ralstonia solanacearum race 5. Sci. Sericult. 41, 0807–0814.
Fegan, M., and Prior, P. (2005). “How complex is the Ralstonia solanacearum species complex,” in Bacterial Wilt Disease and the Ralstonia solanacearum Species Complex, eds C. Allen, P. Prior, and A. C. Hayward (St. Paul, MN: APS Press), 449–462.
Griep, R. A., van Twisk, C., van Beckhoven, J. R. C. M., van der Wolf, J. M., and Schots, A. (1998). Development of specific recombinant monoclonal antibodies against the lipopolysaccharide of Ralstonia solanacearum race 3. Phytopathology 88, 795–803. doi: 10.1094/PHYTO.1998.88.8.795
Ha, Y., Kim, J. S., Denny, T. P., and Schell, M. A. (2012). A rapid, sensitive assay for Ralstonia solanacearum race 3 biovar 2 in plant and soil samples using magnetic beads and real-time PCR. Plant Dis. 96, 258–264. doi: 10.1094/PDIS-05-11-0426
Hayward, A. C. (1991). Biology and epidemiology of bacterial wilt caused by Pseudomonas solanacearum. Annu. Rev. Phytopathol. 29, 67–87. doi: 10.1146/annurev.py.29.090191.000433
He, L. Y., Sequeira, L., and Kelman, A. (1983). Characteristics of strains of Pseudomonas solanacearum. Plant Dis. 67, 1357–1361. doi: 10.1094/PD-67-1357
Hill, J. (2008). Loop-mediated isothermal amplification assay for rapid detection of common strains of Escherichia coli. J. Clin. Microbiol. 46, 2800–2804. doi: 10.1128/JCM.00152-08
Horita, M., Yano, K., and Tsuchiya, K. (2004). PCR-based specific detection of Ralstonia solanacearum race 4 strains. J. Gen. Plant Pathol. 70, 278–283. doi: 10.1111/jam.12585
Kelman, A. (1954). The relationship of pathogenicity in Pseudomonas solanacearum to colony appearance on a tetrazolium medium. Phytopathology 44, 693–695.
Kong, X. J., Qin, W. T., Huang, X. Q., Kong, F. F., Schoen, C. D., Feng, J., et al. (2016). Development and application of loop-mediated isothermal amplification (LAMP) for detection of Plasmopara viticola. Sci. Rep. 6:28935. doi: 10.1038/srep28935
Kubota, R., and Jenkins, M. D. (2015). Real-time duplex applications of loop-mediated amplification (LAMP) by assimilating probes. Int. J. Mol. Sci. 16, 4786–4799. doi: 10.3390/ijms16034786
Kubota, R., LaBarre, P., Singleton, J., Beddoe, A., Weigl, H. B., Alvarez, M. A., et al. (2011b). Non-instrumented nucleic acid amplification (NINA) for rapid detection of Ralstonia solanacearum race 3 biovar 2. Biol. Eng. Trans. 4, 69–80. doi: 10.13031/2013.38508
Kubota, R., Schell, A. M., Peckham, D. G., Rue, J., Alvarez, M. A., Allen, C., et al. (2011a). In silico genomic subtraction guides development of highly accurate, DNA-based diagnostics for Ralstonia solanacearum race 3 biovar 2 and blood disease bacterium. J. Gen. Plant Pathol. 77, 182–193. doi: 10.1007/s10327-011-0305-2
Kubota, R., Vine, B. G., Alvarez, A. M., and Jenkins, D. M. (2008). Detection of Ralstonia solanacearum by loop-mediated isothermal amplification. Phytopathology 98, 1045–1051. doi: 10.1094/PHYTO-98-9-1045
Lenarcic, R., Morisset, D., Pirc, M., Llop, P., Ravnikar, M., and Dreo, T. (2014). Loop-mediated isothermal amplification of specific endoglucanase gene sequence for detection of the bacterial wilt pathogen Ralstonia solanacearum. PLoS ONE 9:e96027. doi: 10.1371/journal.pone.0096027
Mori, Y., Kitao, M., Tomita, N., and Notomi, T. (2004). Real-time turbidimetry of LAMP reaction for quantifying template DNA. J. Biochem. Biophys. Meth. 59, 145–157. doi: 10.1016/j.jbbm.2003.12.005
Mori, Y., Nagamine, K., Tomita, N., and Notomi, T. (2001). Detection of loop-mediated isothermal amplification reaction by turbidity derived from magnesium pyrophosphate formation. Biochem. Biophys. Res. Commun. 289, 150–154. doi: 10.1006/bbrc.2001.5921
Nagamine, K., Hase, T., and Notomi, T. (2002). Accelerated reaction by loop-mediated isothermal amplification using loop primers. Mol. Cell. Probes 16, 223–229. doi: 10.1006/mcpr.2002.0415
Notomi, T., Okayama, H., Masubuchi, H., Yonekawa, T., Watanabe, K., Amino, N., et al. (2000). Loop-mediated isothermal amplification of DNA. Nucleic Acids Res. 28:E63. doi: 10.1093/nar/28.12.e63
Pan, Z. C., Xu, J., Prior, P., Xu, J. S., Zhang, H., and Chen, K. Y. (2013). Development of a specific molecular tool for the detection of epidemiologically active mulberry causing-disease strains of Ralstonia solanacearum phylotype I (historically race 5-biovar 5) in China. Eur. J. Plant Pathol. 137, 377–391. doi: 10.1007/s10658-013-0249-9
Paret, M. L., Kubota, R., Jenkins, D. M., and Alvarez, A. M. (2010). Survival of Ralstonia solanacearum race 4 in drainage water and soil and detection with immunodiagnostic and DNA-based assays. Horttechnology 20, 539–548.
Prior, P., and Fegan, M. (2005). Recent development in the phylogeny and classification of Ralstonia solanacearum. Acta Hortic. 695, 127–136. doi: 10.1186/s12864-016-2413-z
Seal, S. E., Jackson, L. A., Young, J. P. W., and Daniels, M. J. (1993). Differentiation of Pseudomonas solanacearum, Pseudomonas syzygii, Pseudomonas pickettii and the blood disease bacterium by partial 16S rRNA sequencing: construction of oligonucleotide primers for sensitive detection by polymerase chain reaction. J. Gen. Microbiol. 139, 1587–1594. doi: 10.1099/00221287-139-7-1587
Smith, D. S., and De Boer, S. H. (2009). Implementation of an artificial reaction control in a TaqMan method for PCR detection of Ralstonia solanacearum race 3 biovar 2. Eur. J. Plant Pathol. 124, 405–412. doi: 10.1007/s10658-008-9427-6
Thammakijjawat, P., Thaveechai, N., Kositratana, W., Chunwongse, J., Frederick, R. D., and Schaad, N. W. (2006). Detection of Ralstonia solanacearum in ginger rhizomes by real-time PCR. Can. J. Plant Pathol. 28, 391–400. doi: 10.1080/07060660609507312
Tomlinson, J. A., Dickinson, M. J., and Boonham, N. (2010). Detection of Botrytis cinerea by loop-mediated isothermal amplification. Lett. Appl. Microbiol. 51, 650–657. doi: 10.1111/j.1472-765X.2010.02949.x
Tomotada, I., Toshiaki, S., and Kozaburo, H. (2003). Loop-mediated isothermal amplification for direct detection of Mycobacterium tuberculosis complex, M. avium, and M. intracellulare in sputum samples. J. Clin. Microbiol. 41, 2616–2622. doi: 10.1128/JCM.41.6.2616-2622.2003
Weller, S. A., Elphinstone, J. G., Smith, N. G., Boonham, N., and Stead, D. E. (2000). Detection of Ralstonia solanacearum strains with a quantitative, multiplex, real-time, fluorogenic PCR (TaqMan) assay. Appl. Environ. Microbiol. 66, 2853–2858. doi: 10.1128/AEM.66.7.2853-2858.2000
Xue, Q. Y., Yin, Y. N., Yang, W., Heuer, H., Prior, P., and Guo, J. H. (2011). Genetic diversity of Ralstonia solanacearum strains from China assessed by PCR-based fingerprints to unravel host plant- and site-dependent distribution patterns. FEMS Microbiol. Ecol. 75, 507–519. doi: 10.1111/j.1574-6941.2010.01026.x
Keywords: Ralstonia solanacearum, phylotype I mulberry strains, loop-mediated isothermal amplification, detection, bacterial wilt
Citation: Huang W, Zhang H, Xu J, Wang S, Kong X, Ding W, Xu J and Feng J (2017) Loop-Mediated Isothermal Amplification Method for the Rapid Detection of Ralstonia solanacearum Phylotype I Mulberry Strains in China. Front. Plant Sci. 8:76. doi: 10.3389/fpls.2017.00076
Received: 09 September 2016; Accepted: 13 January 2017;
Published: 31 January 2017.
Edited by:
Brigitte Mauch-Mani, University of Neuchâtel, SwitzerlandReviewed by:
Fanhong Meng, Texas A&M University, USADaolong Dou, Nanjing Agricultural University, China
Copyright © 2017 Huang, Zhang, Xu, Wang, Kong, Ding, Xu and Feng. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jie Feng, amZlbmdAaXBwY2Fhcy5jbg== Jin Xu, amlueHVAaXBwY2Fhcy5jbg==
 Wen Huang
Wen Huang Hao Zhang1
Hao Zhang1 Wei Ding
Wei Ding