- 1Key Laboratory of Plant Ecology, Northeast Forestry University, Harbin, China
- 2Botany Department, Faculty of Science, Menoufia University, Shibin El Kom, Egypt
- 3The College of Landscape, Northeast Forestry University, Harbin, China
- 4Guizhou Academy of Tobacco Research, Guiyang, China
This work aims at identifying the effects of ethylene on the response of Arabidopsis thaliana root system to cadmium chloride (CdCl2) stress. Two ethylene-insensitive mutants, ein2-5 and ein3-1eil1-1, were subjected to (25, 50, 75, and 100 μM) CdCl2 concentrations, from which 75 μM concentration decreased root growth by 40% compared with wild type Col-0 as a control. Ethylene biosynthesis increased in response to CdCl2 treatment. The length of primary root and root tip in ein2-5 and ein3-1eil1-1 decreased compared with wild type after CdCl2 treatment, suggesting that ethylene play a role in root system response to Cd stress. The superoxide concentration in roots of ein2-5 and ein3-1eil1-1 was greater than in wild type seedlings under Cd stress. Application of exogenous 1-aminocyclopropane-1-carboxylic acid (ACC) (a precursor of ethylene biosynthesis) in different concentrations (0.01, 0.05 and 0.5 μM) decreased superoxide accumulation in Col-0 root tips and increased the activities of superoxide dismutase (SOD) isoenzymes under Cd stress. This result was reversed with 5 μM of aminoisobutyric acid AIB (an inhibitor of ethylene biosynthesis). Moreover, it was accompanied by increase in lateral roots number and root hairs length, indicating the essential role of ethylene in modulating root system development by controlling superoxide accumulation through SOD isoenzymes activities. The suppressed Cd-induced superoxide accumulation in wild type plants decreased the occurrence of cells death while programmed cell death (PCD) was initiated in the root tip zone, altering root morphogenesis (decreased primary root length, more lateral roots and root hairs) to minimize the damage caused by Cd stress, whereas this response was absent in the ein2-5 and ein3-1eil1-1 seedlings. Hence, ethylene has a role in modulating root morphogenesis during CdCl2 stress in A. thaliana by increasing the activity of SOD isoenzymes to control superoxide accumulation.
Introduction
Heavy metal toxicity in soil was once limited to localities affected by mining, heavy industry or derived from natural mineral outcrops. Metal toxicity not only reduces crop productivity but also threatens the food chain (Poschenrieder and Barceló, 1999). Among the common heavy metal pollutants, cadmium (Cd) is perhaps one of the most aggressive and persistent and is also the most susceptible to accumulation through inappropriate agricultural practices (Choppala et al., 2014; Asgher et al., 2015; Wahid and Khaliq, 2015). The major Cd input into agricultural soils is the phosphorus fertilizers (Singh and Agrawal, 2007). Plants affected by Cd showed impaired photosynthesis (Burzynski and Klobus, 2004; Andresen et al., 2016), altered mineral nutrition and carbohydrate metabolism (Azevedo et al., 2005; Santos et al., 2010) and water imbalance (Sandalio et al., 2001). Cadmium is a non-essential element but is readily absorbed by roots and transported to the aerial parts of plants. Cadmium concentrations of 1–5 μM in the soil solution are sufficient to retard root growth: presumably such toxicity occurs because Cd can replace essential elements that play key roles at the active sites of enzymes, thus affecting many aspects of plant growth and development (Sofo et al., 2013; Vitti et al., 2013).
The plant hormone ethylene is involved in many aspects of the plant life cycle, including seed germination and root hair development (Johnson and Ecker, 1998). It is considered as a (stress hormone) for modulating a diverse array of defense responses (Montero-Palmero et al., 2014; Pan et al., 2016). Thus, ethylene is highly expected to play a role in plant response to Cd stress.
Ethylene acts as a regulator of stress-related morphological responses such as primary and secondary root growth (Parlanti et al., 2011). Interestingly, ethylene remodels the root system architecture to increase Pi uptake in responses to low phosphate (Pi) (Nagarajan and Smith, 2012). This indicates the role of ethylene in root development in relations to heavy metal stress.
Ethylene is derived from methionine, and the first committed step in this pathway is the conversion of S-adenosyl methionine to 1-aminocyclopropane-1-carboxylate (ACC), which is performed by the enzyme ACC synthase (ACS). ACC is then oxidized by ACC oxidase to form ethylene, CO2 and cyanide. During these processes, ACS-catalyzed ACC synthesis is generally the rate-limiting step for ethylene production (Skottke et al., 2011). The ACS isozymes are encoded for by a large gene family, among which phosphorylation of the type 1 isozymes ACS2 and ACS6 by stress-responsive MAPKs results in increased ethylene synthesis (Han et al., 2010; Skottke et al., 2011).
In Arabidopsis, ethylene signaling is first perceived by a family of five receptors that are located at the Golgi and endoplasmic reticulum (ER) membranes (Dong et al., 2010). Ethylene binding is proposed to inhibit receptor function, and CONSTITUTIVE TRIPLE RESPONSE 1 (CTR1) is proposed to be activated by the unoccupied receptor via a physiological interaction (Guo and Ecker, 2003), resulting in the activation of ETHYLENE INSENSITIVE 2 (EIN2). EIN2 plays a central role in the ethylene signaling transduction pathway, and the ein2 mutant, is completely insensitive to ethylene at the morphological, physiological and molecule levels in Arabidopsis (Shibuya et al., 2004). The ethylene signaling pathway then continues to the nuclear transcriptional factors EIN3 and EIN3-like (EIL). In Arabidopsis, there are six members in the EIN3 family, of which EIN3 and its close homolog EIL1 have been widely studied (An et al., 2010; Lingam et al., 2011; Zhu et al., 2011). Mutants in EIN3 and EIL1 have weak ethylene insensitivity (Chao et al., 1997; Binder et al., 2007). Interestingly, ein3 eil1 double mutants display complete ethylene insensitivity in all known ethylene responses, including the triple response and pathogen resistance (Guo and Ecker, 2003). Subsequently, these primary transcription factors activate other secondary transcription factors, such as the ETHYLENE RESPONSE FACTORs (ERFs), thereby regulating the expression of genes that are involved in the response to ethylene (Guo and Ecker, 2004; Kendrick and Chang, 2008). It has been demonstrated that the expression of ERF1 can be activated rapidly by ethylene (Lorenzo et al., 2003).
The production of ethylene is tightly regulated by internal signals during development and by responses to environmental stimuli from biotic and abiotic stresses, such as wounding, ozone, chilling or freezing (Wang et al., 2002; Yoo et al., 2009). Cd induces ethylene biosynthesis in the wild-type of Arabidopsis thaliana (Schellingen et al., 2014). Under Cd stress, the production of ethylene increases drastically in the root tip (Arteca and Arteca, 2007), the region where superoxide is predominantly localized (Dunand et al., 2007), which led us to investigate the connections between ethylene biosynthesis and superoxide production in the root tip. Cd stress is also related to the increase of ROS (Xu et al., 2010a), the oxidative damage of ROS requires the action of antioxidative enzymes, including superoxide dismutase (SOD), which can convert superoxide radicals into hydrogen peroxide, water and oxygen. SOD activity increased by the application of exogenous ACC and ethylene decrease the concentration of hydrogen peroxide under abiotic stress (Lin et al., 2012, 2013a,b). This better support our hypothesis that ethylene regulate superoxide accumulation through SOD activity.
Programmed cell death (PCD) is a functional term used to describe cell death that eliminates harmful cells during the life cycle of multicellular organisms. Increasing evidence suggests that diverse abiotic stresses, such as salt, drought, nutrient deficiency and heavy metal toxicity, can induce PCD in plant root tips (Liu et al., 2009; Duan et al., 2010; Xu et al., 2010b; Demidchik et al., 2014). Under drought and long-term zinc exposure, the PCD program is activated in the root apical meristematic zone, so apical root dominance is removed. Thus, the root system architecture may be remodel allowing adaptation to the stressful environment (Duan et al., 2010; Xu et al., 2010b). This led us to expect that PCD might be the mechanism by which the plant responds to the damage caused by Cd stress especially because reactive oxygen species (ROS) accumulation may trigger the PCD process in stressed plants (Harding and Roberts, 1998; Chae and Lee, 2001). However, the relation between production of superoxide and the progress of PCD in root tips under Cd stress is still unknown.
So our hypothesis suggests that ethylene modulates root system by decreasing superoxide accumulations through decreasing NADPH oxidase activity and increasing SOD enzymes activity. Ethylene-induced suppression of superoxide accumulation modulates the root development through decreasing cell death (damaging effect of superoxide accumulation) and initiating PCD.
The aim of this study is to highlight the role of ethylene in response to the damage effect caused by Cd stress through its effects on superoxide accumulation by increasing the SOD activity, and to show the relation between the progression of cell death and PCD in root tips and the mechanism by which ethylene in minimize the damage effect caused by Cd stress, to propose a schematic model elucidating this mechanism by which the plant responses to Cd stress through inducing ethylene biosynthesis to modulate the root system development.
Materials and Methods
Plant Material and Growth Conditions
Seedlings of the following lines were used in this study: Arabidopsis thaliana ecotype Columbia-0 (Col-0) and the ethylene-insensitive mutants ein2-5 (Alonso et al., 1999) and ein3-1eil1-1 (Alonso et al., 2003) in the Col-0 background. All seeds were surface-sterilized by incubation in 70% ethanol containing 0.05% Triton X-100 (Solar Bio, Beijing, China) for 10 min, rinsed thoroughly with ethanol for 1 min and then washed with sterilized water. The sterilized seeds were sown on agar plates containing a MS basal salt mixture (Sigma–Aldrich, St. Louis, MO, USA), 1% sucrose and 0.8% agar (pH 5.7). The plates were maintained at 4°C in the dark for 48 h to enhance the germination process and then placed vertically at 23°C under a light intensity of 200 μml m-2 s-1 (16/8 h light cycle).
Treatments
To investigate the effects of Cd stress on root growth, 4-day-old seedlings cultivated in control agar plates were transferred to freshly prepared medium supplemented by various concentration of CdCl2 (25, 50, 75, and 100 μM) as indicated (Cao et al., 2009; Radeva et al., 2010) for 2 and 4 days. Preliminary experiments with seedling grown with various CdCl2 concentrations showed that 75 μM CdCl2 decreased root growth by about 40% and so this concentration was used to compare the effects of Cd stress between the wild type and the ethylene-insensitive mutants.
To investigate the effects of exogenous ACC on the accumulation of superoxide in the root tips of the Col-0 plants under Cd stress. Four-day-old seedlings were transferred to various pretreated agar plates with 75 μM CdCl2 or 75 μM CdCl2 plus various concentrations of ACC (0.01, 0.05, and 0.5 μM, a precursor of ethylene biosynthesis) with or without 5 μM AIB (an inhibitor of ethylene biosynthesis), as indicated, for 4 days.
All the experiments were repeated three times with three replicates of each treatment unless otherwise noted.
Quantitative RT-PCR Analysis of Gene Expression
To investigate whether ethylene biosynthesis and signaling were involved in the plant response to Cd stress, the relative expression of genes encoding for ethylene biosynthesis, and of perception and signaling proteins were measured in seedlings that were treated with different concentrations of CdCl2 for 4 days. Total RNA was isolated from 100 seedling roots using TRIzol solution (Invitrogen, Carlsbad, CA, USA). Two micrograms of total RNA was used for first-strand cDNA synthesis using RevertAid Reverse Transcriptase and Oligo d(T)primers (Takara, Dalian, China). The cDNA yield was measured according to the PCR signal generated from the internal standard, with the housekeeping gene Actin 2 used as the internal control. The primers that were used in the quantitative RT-PCR are listed in Table 1 (Liu et al., 2010; Lin et al., 2013b; Li et al., 2014).
Quantitative RT-PCR was performed using a RealMasterMix kit (Tiangen, Beijing, China) with 25 cycles as follows: 94°C for 30 s, 58°C for 30 s and 72°C for 30 s, followed by 72°C for 10 min gene expression quantifications was performed using the relative 2-ΔΔCt method (Livak and Schmittgen, 2001). All experiments were performed in triplicate for each treatment.
GUS Staining
To investigate the mechanism by which CdCl2 affects the synthesis and distribution of ethylene, the relative expression of the ethylene reporter construct, EBS::GUS, in which the GUS reporter gene is driven by a synthetic EIN3-responsive promoter, was tested. GUS staining was as described by He et al. (2011). Four-day-old seedlings were transferred to agar plates supplemented with 75 μM CdCl2 for 4 days, then were collected and washed with staining buffer without X-Gluc and stained with GUS staining buffer (50 mM sodium phosphate buffer, pH 7.0, 10 mM Na2EDTA, 0.5 mM K4[Fe(CN)6]⋅3H2O, 0.5 mM K3[Fe(CN)6], 0.1% Triton X-100, and 1 mg/mL X-Gluc). Ethanol (70%) was used to terminate the staining reaction, and the seedlings were mounted on slides in 50 μL Hoyer’s solution (chloral hydrate: water: glycerol; 8:3:1; w/v/v) and examined and photographed with a DM 4000B stereomicroscope (Leica, Germany).
Morphometric Analysis
To further test the involvement of ethylene signaling in regulating the plant response to Cd stress, two ethylene-insensitive mutants (ein2-5 and ein3-1eil1-1) together with the wild type plant were subjected to Cd stress with or without ACC. The length of the whole primary root of the seedlings were measured either directly with a ruler or using NIS Elements software (Nikon, Japan) from digital images captured with a Nikon camera. The root tip length was also determined in the same way as the primary root. The number of the lateral roots was counted. The number of root hairs in a 2 mm section at the appropriate midpoint of the root was counted under a dissecting microscope. Root hairs from the photographs were measured with a ruler, and their length in μm was determined by comparison with an ocular micrometer photograph at the same magnification. All experiments for phenotypic analysis were performed at least three times, and the data represent one independent experiment.
Cadmium Analysis
The samples were rinsed twice with tap water then with de-ionized water before air drying is used to remove dirt and salt from root surface. High purity deionized water from a Millipore water purification system (Bedford, NY, USA) was used throughout our study. The root materials were dried at 80°C to a constant weight. The dried tissues were weighed and digested in HNO3 (100%) using the heat block (Shah et al., 2014; Olowu et al., 2015). The metal concentration was determined by inductively coupled plasma mass spectrometry (ICP-MS). Blanks (only HNO3) were analyzed for reference purposes. The cadmium standard solutions for ICP-MS (TraceCERT®, Sigma–Aldrich) were produced according to the ISO Guide 34 in the analysis of Cd concentrations. External calibration was performed using a five-point analytical curve, prepared by diluting the individual cadmium standards with 5.0% (v/v) HNO3.
Bioaccumulation factor (Bf) was calculated to measure plant uptake of Cd:
Measurement of Superoxide Accumulation in Root Tips by (NBT) Staining
Four-days-old wild-type seedlings cultivated on MS agar plates were transferred to agar plates supplemented with 75 μM CdCl2 or 75 μM CdCl2 plus various concentrations of ACC (0.01, 0.05, and 0.5 μM, a precursor of ethylene biosynthesis) with or without 5 μM AIB (an inhibitor of ethylene biosynthesis). Superoxide within the root tip was detected by nitroblue tetrazolium (NBT) staining which is used to detect O2- production, as well as other compounds (such as ascorbate) as described by Hernández et al. (2001). Seedlings were collected, and the roots were immersed in a 0.1% solution of NBT in 50 mM K–phosphate buffer (pH 6.4), containing 10 mM Na-azide in the absence of light. Seedlings were then transferred to distilled water to stop the reaction. The roots were observed and photographed using a DM 4000B Leica stereomicroscope equipped with a DC300F Nikon camera. The NIH ImageJ software was used to assess the mean staining intensity of the elongation and meristematic zones (0, white; 255, black) following Dunand et al. (2007). At least 20 individual roots were analyzed for each genotype and treatment, and one representative image was selected for the figure.
NADPH-Dependent O2⋅- Determination
The determination of the NADPH dependent O2- generating activity in isolated plasma membrane vesicles was carried out according to Quartacci et al. (2001) and Van Gestelen et al. (1997), by measuring the rate of SOD inhibitable reduction of NBT using NADPH as electron donor. The reaction mixture consisted of 50 mM TRIS HCl buffer, pH 7.5, 0.25 M sucrose, 0.1 mM NBT, and 50–100 μg proteins. After 1 min preincubation the reaction started by the addition of 0.1 mM NADPH and the absorbance changes at 530 nm were followed for 5 min. Rates of O2- generation were calculated using an extinction coefficient of 12.8 mM-1cm-1.
Measurement of SOD and Its Three Isoenzymes Activities
To investigate whether ethylene reduced Cd stress-induced superoxide accumulation through the SOD pathway, 4-day-old seedlings were transferred to various agar plates for 4 days, and the activities of SOD and its three isoenzymes (Cu/Zn-SOD, Fe-SOD and Mn-SOD) were measured according to the method of Yu and Rengel (1999) with minor modifications. Frozen root samples (0.05 g) were weighed and homogenized on ice for 2 min in 5 ml of homogenizing solution containing 50 mm HEPES buffer and 0.1 mm Na2EDTA (pH 7.6). The homogenate was centrifuged at 15000 g for 15 min at 4°C to produce the crude extract for SOD assays. SOD activity was assayed by monitoring the inhibition of the photochemical reduction of NBT. For the total SOD assay, a 5-ml reaction mixture containing 50 mM HEPES (pH 7.6), 0.1 mM EDTA, 50 mM Na2CO3 (pH 10.4), 13 mM methionine, 0.025% (w/v) Triton X-100, 75 μM NBT, 2 μM riboflavin and an appropriate aliquot of enzyme extract was utilized. The reaction mixtures were illuminated for 15 min at a light intensity of 350 μmol⋅m-2⋅s-1. One unit of SOD activity was defined as the amount of enzyme required to cause a 50% inhibition in the reduction of NBT, as monitored at 560 nm. The activities of different SOD forms were identified by adding KCN and/or H2O2 to the reaction mixture (Giannopolitis and Ries, 1977). KCN inhibits Cu/Zn-SOD but does not affect Mn-SOD or Fe-SOD, whereas H2O2 inactivates Cu/Zn-SOD and Fe-SOD but not Mn-SOD. In addition, peroxidases might interfere with the SOD assay in the presence of exogenous H2O2 (Yu et al., 1998). After the extensive preliminary testing of a range of concentrations, KCN (at final concentration of 3 mM) was added to the reaction mixture before the addition of H2O2 (5 mM final concentration) to eliminate the interference of peroxidase and catalase enzymes (Chen and Asada, 1989). Mn-SOD activity was determined in the presence of both 3 mM KCN and 5 mM H2O2. Fe-SOD activity was obtained by subtracting the Mn-SOD activity from the activity in the presence of 3 mM KCN, and Cu/Zn-SOD activity was calculated from the differences between the total activity and that of Mn-SOD and Fe-SOD. Identical reaction mixtures that had not been illuminated were used to correct for background absorbance.
Detection of Cell Death in Meristimatic and Elongation Zones under Cd Stress with Trypan Blue and Propidium Iodide (PI) Staining
To assess cell death in the root tips under Cd stress, the roots were immersed in 4 mg ml-1 Trypan blue solution (Sigma–Aldrich, Saint Louis, MO, USA) for 15 min at room temperature and then washed with distilled water three times. The samples were then observed by light microscopy, and pictures were taken as detailed earlier.
For PI staining, the seedlings were immersed in the PI (Sigma–Aldrich, St. Louis, MO, USA) solution (final concentration of 1 μg ml-1) for 10 min at room temperature in the dark and then washed with phosphate buffer solution (PBS) (pH 7.4) three times. The samples were then examined using a DM 4000B fluorescence microscope (Leica, Germany) with an excitation wavelength of 546 nm. Both experiments were repeated three times. For each treatment and genotype, at least 20 roots were analyzed for both stains, and one representative image was selected for the figure.
Terminal Deoxynucleotidyl Transferase-Mediated dUTP Nick End Labeling (TUNEL) and 4, 6-Diamidino -2-Phenylindole (DAPI) Assay
Cadmium-induced cell death in root tips can occur through either necrosis or PCD. To examine whether PCD is involved in the cell death at the root tips, we investigated the chromatin condensation and the internucleosomal fragmentation of the DNA by DAPI staining and the TUNEL assay, respectively. To detect cell apoptosis caused by Cd stress, an In Situ Cell Death Detection Kit, AP (Roche, Germany) was used. The TUNEL assay was performed according to the manufacturer’s instructions with a few modifications. In brief, whole seedlings were fixed in 4% paraformaldehyde in PBS (pH 7.4) for 20 min (20°C). After washing the samples for 30 min with PBS, the samples were incubated in permeabilization solution (0.1% Triton X-100, 0.1% sodium citrate) for 2 min (4°C), followed by two washes with PBS. Fifty microliters of TUNEL reaction mixture was then added to the sample, and it was incubated for 60 min (37°C) in a humidified atmosphere in the dark. After being rinsed three times with PBS, the samples were analyzed under a DM 4000B fluorescence microscope (Leica, Germany) with a 488 nm excitation line and a 530 nm emission filter.
DAPI staining was performed by fixing roots in a solution of ethanol and acetic acid (3:1) for 1 h. The roots were then washed twice with PBS (pH 7.4) and stained in PBS containing 1 μg/ml of DAPI for 20 min at room temperature. After two PBS washes, the samples were analyzed under a DM 4000B fluorescence microscope (Leica, Germany) with a UV light filter, and the images were captured immediately. Both experiments were repeated three times. For each treatment and genotype, at least 20 roots were analyzed for each experiment, and one representative image was selected for the figure.
Statistical Analyses
For all experiments, the data were statistically analyzed using SPSS 17.0 (SPSS, Chicago, IL, USA). One-way analysis of variance with a Duncan post hoc test was used to test the differences at a 0.05 level for the primary root length and distance to the first root hair. The data presented here are the means with standard error (SE).
NADPH oxidase and SOD enzymes activities values were used for the calculation of abundance ratios between groups and for statistical evaluation by Student’s t-test (P = 0.05).
Results
Traits Indicating Involvement of Ethylene in Plant Responses to Cd Stress
To investigate whether ethylene biosynthesis and signaling were involved in the plant response to Cd stress, quantitative RT-PCR was performed using a Real Master Mix kit (Tiangen, Beijing, China) to measure the relative expression of genes encoding for ethylene biosynthesis, and of perception and signaling proteins. The relative expression of ACS2, ACS6, ERF1, and EIN3 increased slightly under small concentrations of Cd but increased drastically after increasing Cd concentrations to 75 μM but decreased with further increase to 100 μM although expression still higher than the control (Figure 1A).
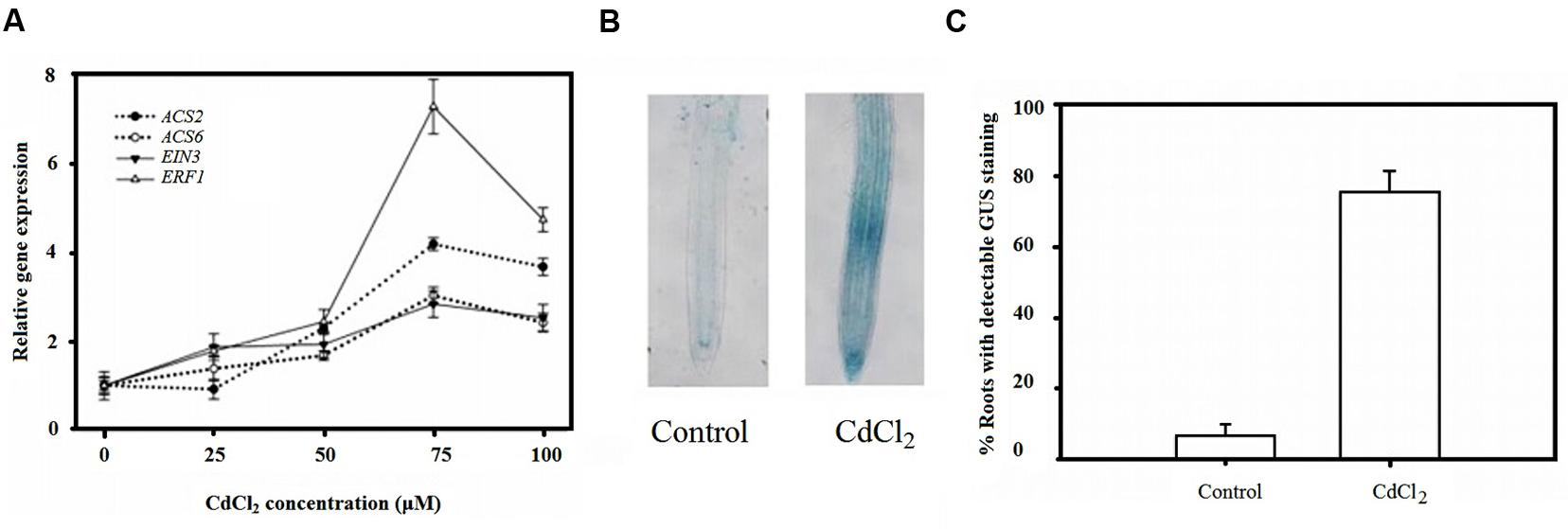
FIGURE 1. Ethylene signaling involved in plant root response to Cd stress. (A) QRT-PCR analysis of relative gene expression of ACS2, ACS6, EIN3 and ERF2 in Col-0 plants under Cd stress. (B) Increased ethylene response after Cd treatment visualized by EBS::GUS staining. (C) Frequency of stained root tip as (B) indicated. Seeds were germinated and grown on MS agar plates for 4 days and then transferred to CdCl2 (μM) pretreated plates, as indicated, for another 4 days.
To investigate the mechanism by which CdCl2 affects the synthesis and distribution of ethylene, the relative expression of the ethylene reporter construct, EBS::GUS, in which the GUS reporter gene is driven by a synthetic EIN3-responsive promoter, was tested. Remarkably, less than 10% of root tips in unstressed seedlings exhibited GUS staining while nearly 80% of them in stressed seedlings were stained (Figures 1B,C). Taken these results together, suggested the involvement of ethylene signaling in plant responses to Cd stress.
Effect of Cd Stress on Root System Development
The primary root lengths of the wild-type (Col-0) and the ein2-5 and ein3-1eil1-1 plants were severely inhibited by 75 μM CdCl2 (Supplementary Figure S1).
However, Figure 2A showed that the primary roots lengths of both the ein2-5 and ein3-1eil1-1 plants were slightly longer than those of the wild-type (Col-0) plants after 2 days of Cd stress. This phenomenon was reversed after 4 days treatment as the primary roots of the ein2-5 and ein3-1eil1-1 plants were significantly shorter than the wild-type plants.
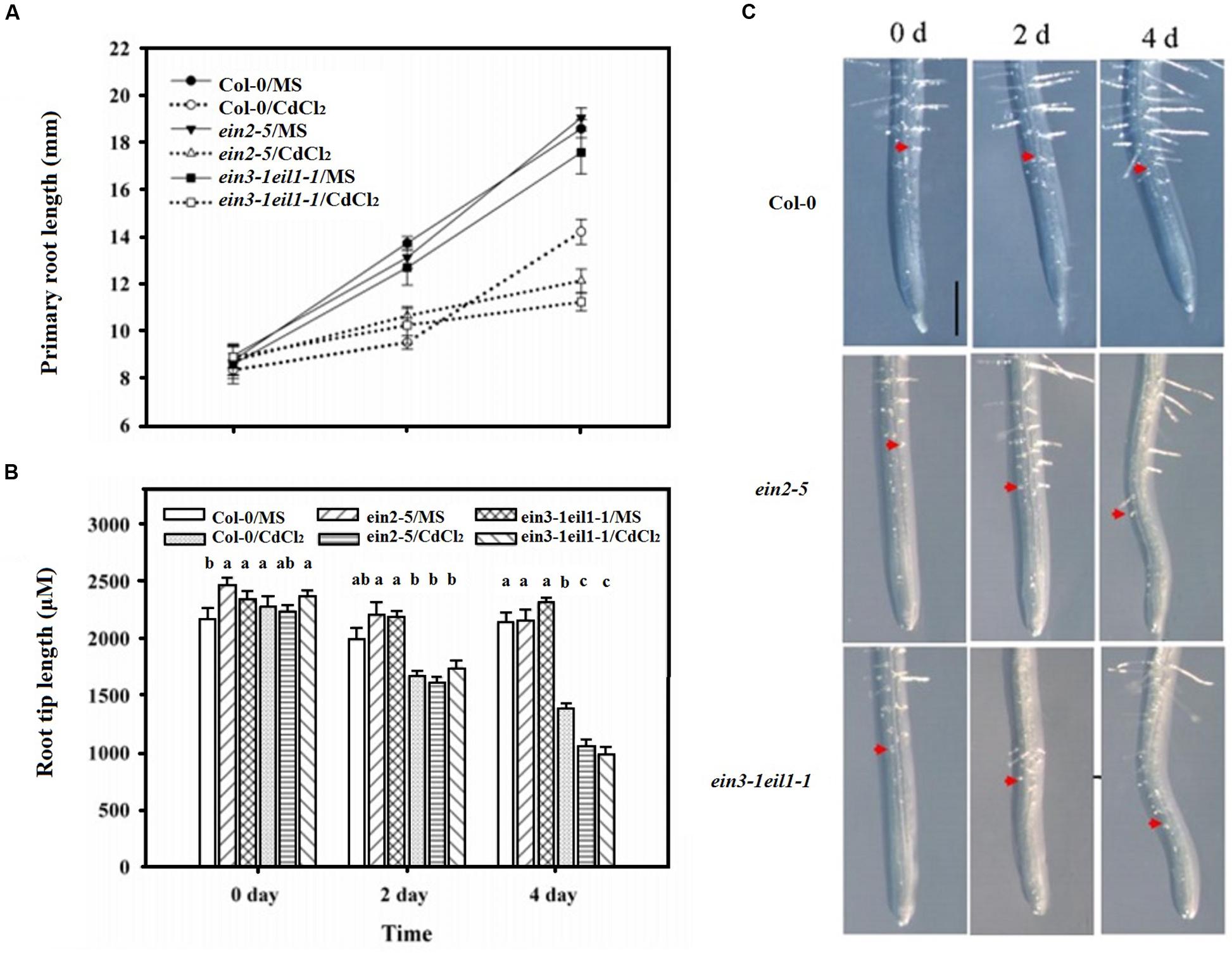
FIGURE 2. The inhibitory effects of increased duration of exposure to Cd on the elongation of the primary roots and root tips of the Col-0, ein2-5, and ein3-1eil1-1 plants. Four-day-old seedlings were transferred to CdCl2 (75 μM)-treated agar plates for 2 and 4 days, as indicated. (A) The primary root lengths of the Col-0, ein2-5, and ein3-1eil1-1 plants after being transferred to the Cd-treated agar plates for different periods, (B) and (C) the root tip length. Values represent the mean ± SE of 20 individual plants, and the letters indicate significant differences (P < 0.05). Scale bars = 50 μm.
The root tip length (distance to the first root hair) was also determined after 4 days of Cd treatment (Figures 2B,C). Similar to the results for the primary root length, the length of the meristematic and elongation/transition zone was less inhibited in the ein2-5 and ein3-1eil1-1 mutants than in the wild-type plants after 2 days of Cd treatment. The length of the zones in the wild-type plant Col-0, ein2-5, and ein3-1eil1-1 plants was 1433.1, 1614.6, and 1741.7 μm, respectively. In contrast, after 4 days treatment, the root tip lengths of both the ein2-5 (1056.6 μm) and ein3-1eil1-1 (983.8 μm) plants were significantly shorter compared with the Col-0 (1393.1 μm) plants.
The lateral root number of the wild-type plants also significantly increased, whereas it was reduced in the ein2-5 and ein3-1eil1-1 mutants in response to Cd stress (Figure 3A). This result was expected since the primary root of wild-type plants was longer than that of the ein2-5 and ein3-1eil1-1 mutants under of Cd stress.
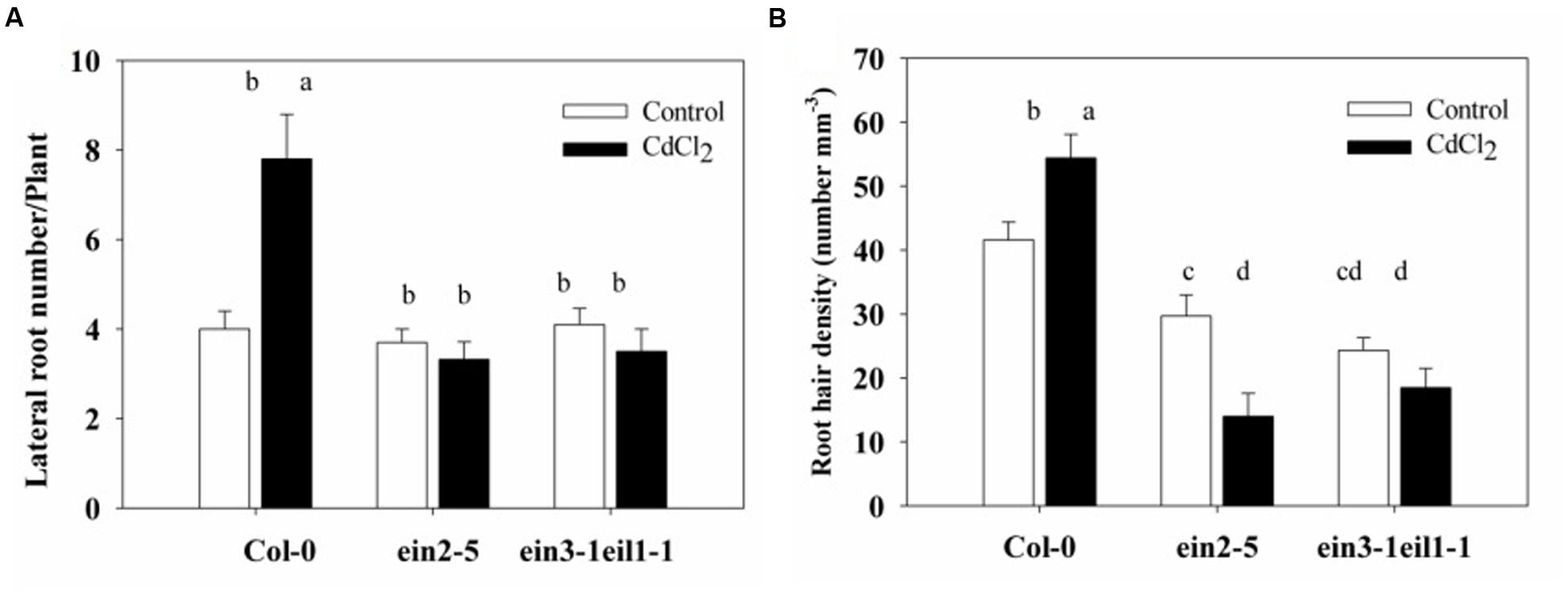
FIGURE 3. Effects of Cd stress on the root system architecture of the Col-0, ein2-5, and ein3-1eil1-1 plants. (A) Lateral root number and (B) Root hair density of the 4-days-old seedlings transferred to agar plates with or without CdCl2 (75 μM) for 7 days. Values represent the mean ± SE of more than 20 individual seedlings, and the bars indicate the standard error. The letters indicate significant differences (P < 0.05).
Moreover, the root hair density also significantly increased in the Col-0 plants, whereas it severely decreased in both the ein2-5 and ein3-1eil1-1 plants under Cd stress (Figure 3B). In addition, it is worth noting that very few root hairs emerged from mutants under Cd stress and also the length of these hairs decreased (Supplementary Figure S2).
These results show that the wild type plants improve their root system development under Cd stress in a better way than ein2-5 and ein3-1eil1-1 plants, this led us to suggest the involvement of ethylene in modulating the root system development in response to Cd stress.
Cd Contents
Three-week-old A. thaliana seedlings were exposed to 0, 10, 20, and 40 μM CdCl2 for 8 days. (Figure 4) showed that a significant accumulation of Cd content in roots treated with Cd. In the Col-0 root, the Cd content was highest-level after exposure to 20 μM CdCl2. At the 20 μM CdCl2 treatment, the Cd contents of ein2-5 and ein3-1eil1-1 were even higher than the Cd content of Col-0. Bioaccumulation factors (Bfs) in the Col-0 got their maximum at 20 μM CdCl2. Furthermore, the Bfs of ein2-5 and ein3-1eil1-1 were even bigger than Col-0 under 20 μM CdCl2 treatment. Ein2-5 and ein3-1eil1-1 are both ethylene insensitive mutants. Moreover, application of ACC with different concentrations of Cd (50, 60, and 75 μM) decreased the Cd content in roots compared with roots treated with Cd alone (Supplementary Figure S3).
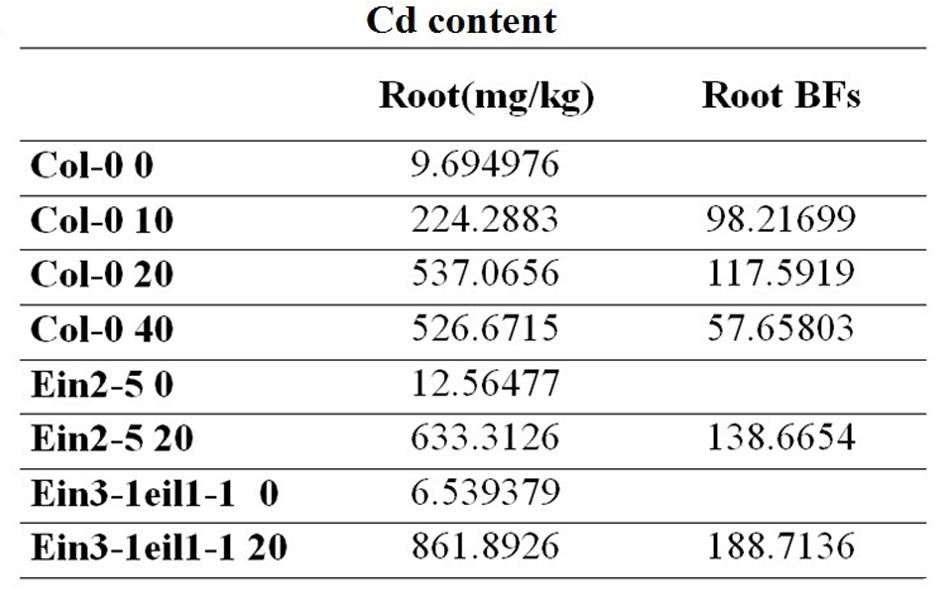
FIGURE 4. The Cd content of Arabidopsis seedlings roots. Three-week-old Arabidopsis thaliana seedlings were exposed to 0, 10, 20, and 40 μM CdCl2 for 8 days. At the 20 μM CdCl2 treatment, the Cd contents of ein2-5 and ein3-1eil1-1 were even higher than the Cd content of Col-0. Furthermore, the Bfs of ein2-5 and ein3-1eil1-1 were even bigger than Col-0 under 20 μM CdCl2 treatment.
These results showed that ethylene could play a role in plant response to Cd stress supporting the above mentioned results and led us to wonder about the mechanism by which ethylene modulates root system in response to Cd stress.
Accumulation of Superoxide in Root Tips under Cd Stress
To understand the mechanism of ethylene response to Cd stress, we determined the superoxide in the root tips of Col-0, ein2-5, and ein3-1eil1-1 under Cd stress. As the results in Figure 5 indicate, superoxide was predominantly localized at the meristematic and elongation zones. Treatment of the 4-days-old seedlings with 75 μM Cd for 2 days significantly increased the production of superoxide in all plants. However, the accumulation of superoxide in both the ein2-5 and ein3-1eil1-1 mutants was significantly greater than in the wild-type plants. Furthermore, after 4 days of Cd treatment, the concentration of superoxide in the root tips was still higher in the two ethylene-insensitive mutants than in the wild-type plants.

FIGURE 5. The accumulation of superoxide in the root tips of the Col-0, ein2-5, and ein3-1eil1-1 plants. Four-day-old seedlings were transferred to CdCl2 (75 μM) -treated agar plates for different periods, as indicated. 2 days treatment significantly increased the production of superoxide in all plants. In addition, the accumulation of superoxide in both the ein2-5 and ein3-1eil1-1 mutants was significantly greater than in the wild-type plants even after 4 days of Cd treatment. Values (mean ± SE) show the staining intensity, and the letters indicate significant differences (P < 0.05). At least 20 individual roots were analyzed for each genotype and treatment, and one representative image was selected for the figure.
The altered response pattern of superoxide accumulation in ein2-5 and ein3-1eil1-1 suggested that ethylene signaling regulates superoxide accumulation in the Cd-stressed root tips.
Effect of ACC (an Ethylene Precursor) on Superoxide Accumulation and Root System Development under Cd Stress
To investigate whether regulating superoxide is the mechanism by which ethylene modulate root system under Cd stress, effect of exogenous ACC (a precursor of ethylene biosynthesis) on the accumulation of superoxide in the root tips of the Col-0 plants under Cd stress was determined. Generally, Cd stress-induced increases in superoxide accumulation were detected after 4 days of treatment in the Col-0 root tips as shown in Figure 6. Although the supplementation with ACC slightly increased superoxide accumulation, treatment with ACC together with Cd markedly reduced the production of superoxide compared with the Cd treatment alone in the root tips, especially with 0.01 μM concentration of ACC. Conversely, the ACC-induced suppression of the production of superoxide was reversed by AIB (an inhibitor of ethylene biosynthesis) application with ACC and Cd, confirming the results in Figure 5 and supporting the involvement of ethylene in regulation of superoxide accumulation in the Cd-stressed root tips.

FIGURE 6. Effects of exogenous ACC on the accumulation of superoxide in the root tips of the Col-0 plants under Cd stress. Four-day-old seedlings were transferred to various pretreated agar plates with CdCl2 (75 μM) or CdCl2 (75 μM) plus various concentrations of ACC (0.01, 0.05, and 0.5 μM, a precursor of ethylene biosynthesis) with or without 5 μM AIB (an inhibitor of ethylene biosynthesis), as indicated, for 4 days. Values (mean ± SE) show the staining intensity, and the letters indicate significant differences (P < 0.05). At least 20 individual roots were analyzed for each genotype and treatment, and one representative image was selected for the figure.
It was well established that PMs NADPH oxidase transfers electrons from cytoplasmic NADPH to O2 to form O2- thus we subsequently determined activity of NADPH in seedlings roots. Same results were also obtained in determining NADPH oxidase activities when exogenous ACC were applied to CdCl2 stressed seedlings and also with the addition of AIB (Figure 7).
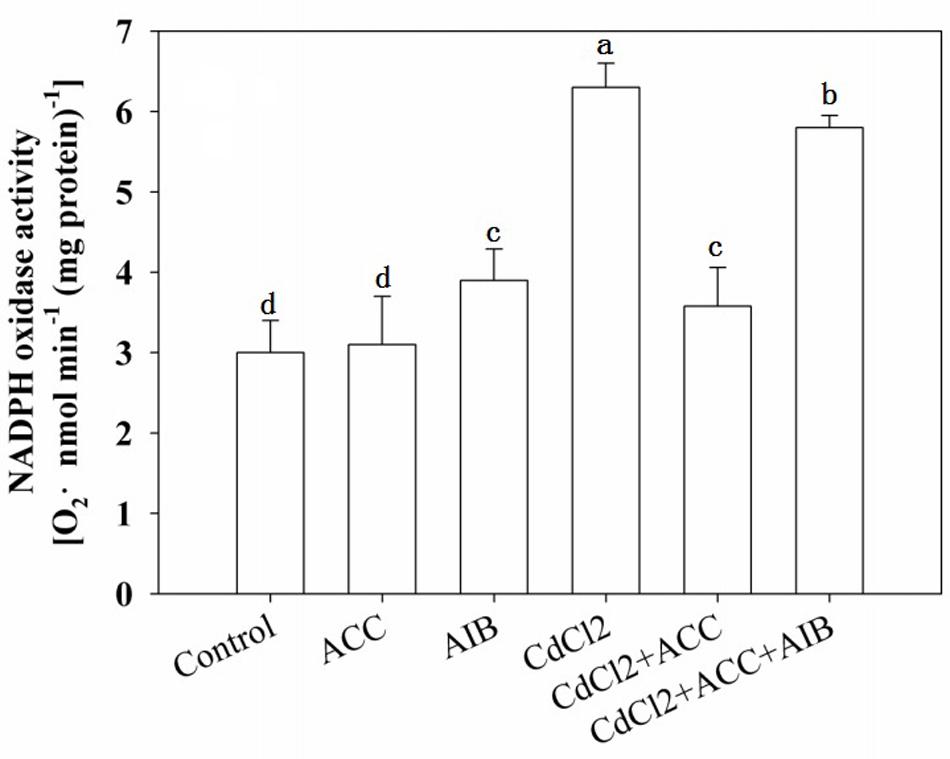
FIGURE 7. Effects of various pretreatments (ACC, 0.01 μM; AIB, 5 μM) on the production of O2- in roots of Arabidopsis Col-0 seedlings under Cd (75 μM) stress. Four-day-old seedlings were transferred to various pretreated agar plates for 4 days. Values represent the mean ± SE, and the bars indicate the standard error.
The effect of ACC on the root system development in Col-0 plants under Cd stress was also determined. Application of exogenous ACC significantly increased lateral roots number and the length of root hairs. The effect of ACC was reversed by AIB that decreased the root hairs density and root hairs length (Supplementary Figure S4).
Taken these results together, we suggested that ethylene modulates root system development under Cd stress by regulating superoxide concentration.
Effect of Ethylene on SOD and Its Three Isoenzymes Activities in Root under Cd Stress
To investigate whether ethylene reduces Cd stress-induced superoxide accumulation through the SOD pathway, the activities of SOD and its three isoenzymes (Cu/Zn-SOD, Fe-SOD, and Mn-SOD) were measured in Col-0, ein2-5, and ein3-1eil1-1 under Cd stress.
As the results in Figure 8 illustrate, the SOD activities were increased after Cd treatment in the Col-0 but decreased in the ein2-5 and ein3-1eil1-1 plants. In addition, the application of exogenous ACC significantly increased these activities compared with the Cd stress in both Col-0 and the ein3-1eil1-1 mutants. However, ACC displayed no effects on the SOD activities of the ein2-5 plants. Furthermore, the Cu/Zn-SOD and Fe-SOD activities significantly increased while the Mn-SOD activity did not change after 5 days of Cd treatment in the Col-0. Conversely, the activities of all three isoenzymes decreased in the ein2-5 and ein3-1eil1-1 roots. The application of ACC to the Cd-stressed seedlings increased the activities of all three isoenzymes in both the wild-type and ein3-1eil1-1 plants, whereas ACC only increased the activity of Fe-SOD in the ein2-5 mutant.
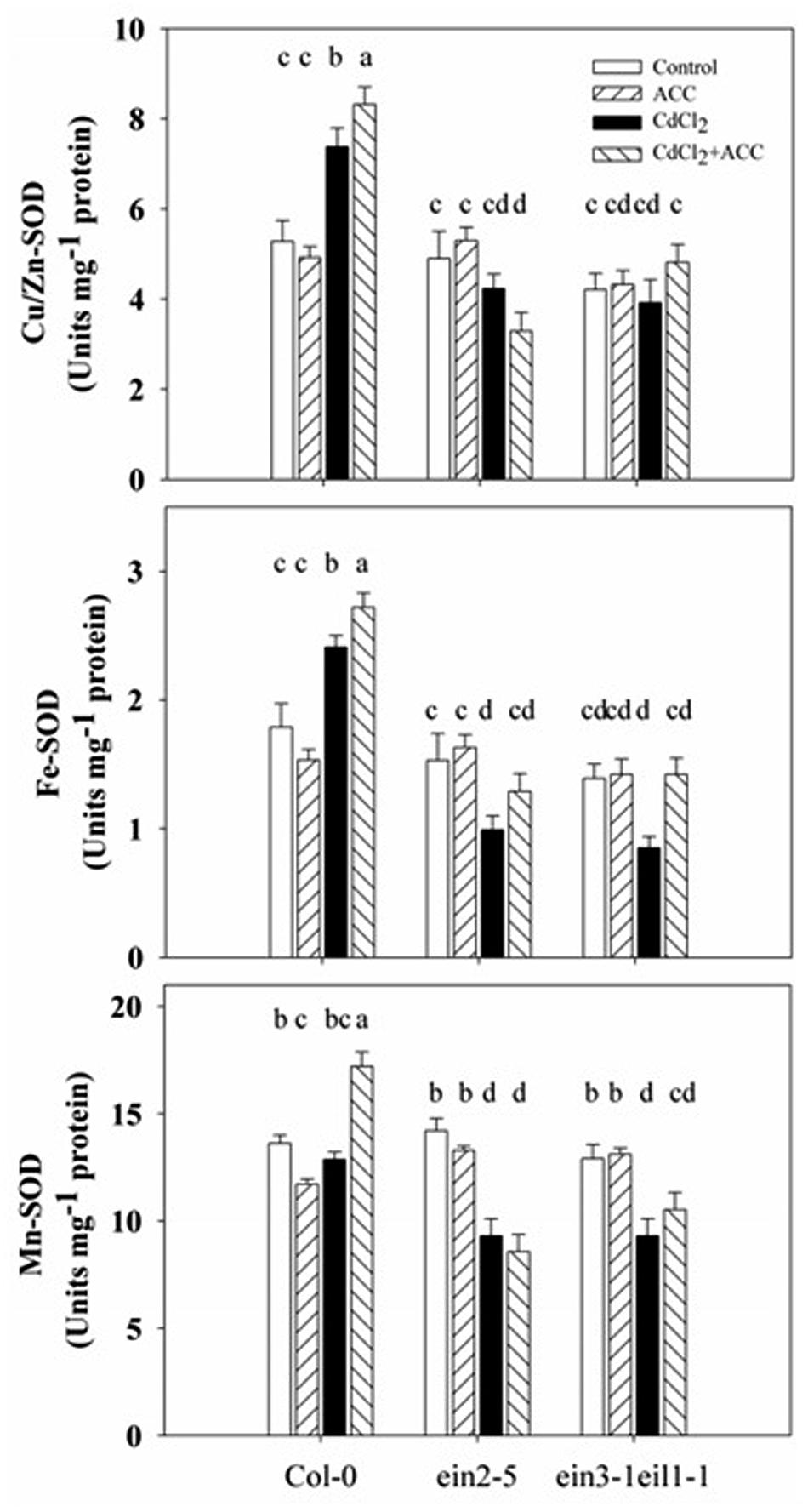
FIGURE 8. Activities of SOD and its Cu/Zn-SOD, Fe-SOD, and Mn-SOD isoenzymes in the roots of the Col-0, ein2-5, and ein3-1eil1-1 plants under Cd stress. Four-day-old seedlings were transferred for 4 days to agar plates, or agar plates with CdCl2 (75 μM), or with ACC (0.01 μM, a precursor of ethylene biosynthesis), or with CdCl2 (75 μM) plus ACC (0.01 μM), as indicated. Values represent the mean ± SE, and the bars indicate the standard error. The letters indicate significant differences (P < 0.05).
The increased SOD activities in the Col-0 compared with the ein2-5 and ein3-1eil1-1 plants under Cd stress suggests that ethylene reduces Cd stress-induced superoxide accumulation through the SOD pathway.
Effect of Cd Stress on Cell Death in the Meristematic and Elongation Zones
High concentrations of superoxide induce oxidative stress, which ultimately leads to cell death. We evaluated the progression of cell death in the root tips using PI and Trypan blue. As the results of the PI staining show, exposing the seedlings to Cd stress generally led to significant cell death in the primary root tips after treatment, especially from the end of the meristematic zone to the elongation zone. However, the PI-positive cells staining intensity were significantly increased in the ein2-5 and ein3-1eil1-1 mutant root tips compared with the wild-type plants after 2 days of Cd treatment. Furthermore, after 4 days of Cd treatment, the entire meristematic and most of the elongation zone of the stressed root tips were strongly stained in the ein2-5 and ein3-1eil1-1 mutants, whereas only the end of the elongation zone was affected in the wild-type plants. Staining analyses using Trypan blue showed a similar progression of cell death, confirming the above mentioned results (Figure 9).
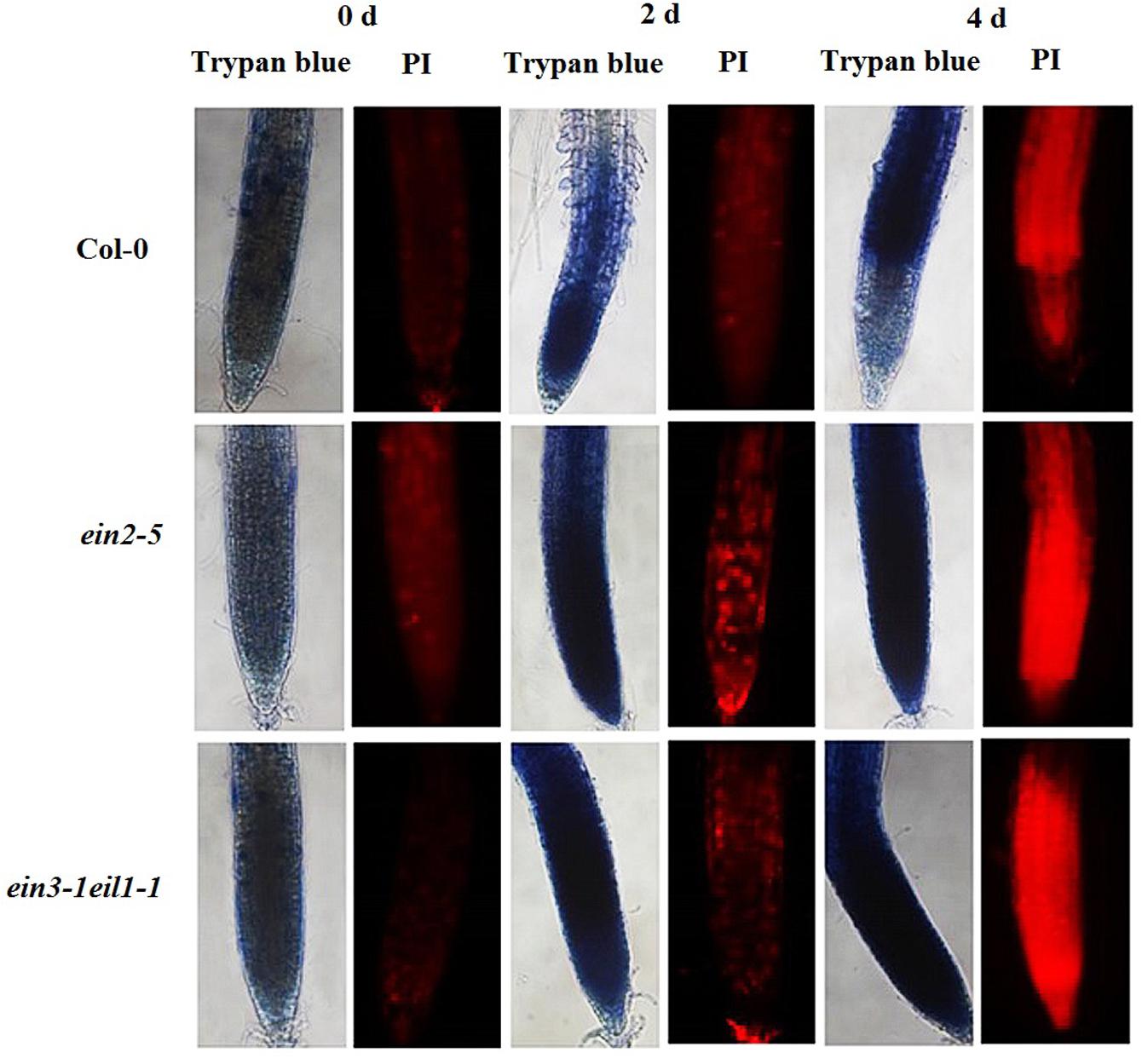
FIGURE 9. Progression of cell death/necrosis in the root tips of the Col-0, ein2-5, and ein3-1eil1-1 plants during Cd stress. Four-day-old seedlings were transferred to CdCl2 (75 μM)-treated agar plates for different periods, as indicated. The seedlings were then collected and incubated in propidium iodide (PI) or Trypan blue. At least 20 individual roots were analyzed for each genotype and treatment, and one representative image was selected for the figure.
TUNEL and DAPI Assay
As shown in Figure 10, only weak DAPI (weak fluorescence and round, homogenously stained nuclei) and TUNEL signals were detected in the untreated seedling roots. However, a marked increase in DAPI fluorescence and condensed and granular nuclear staining and TUNEL-positive signals were detected in the meristematic zone to the elongation zone from root tips after Cd treatment. However, higher intensity of DAPI fluorescence and TUNEL-positive signals were also detected in the wild-type root tips compared with the ein2-5 and ein3-1eil1-1 mutants.
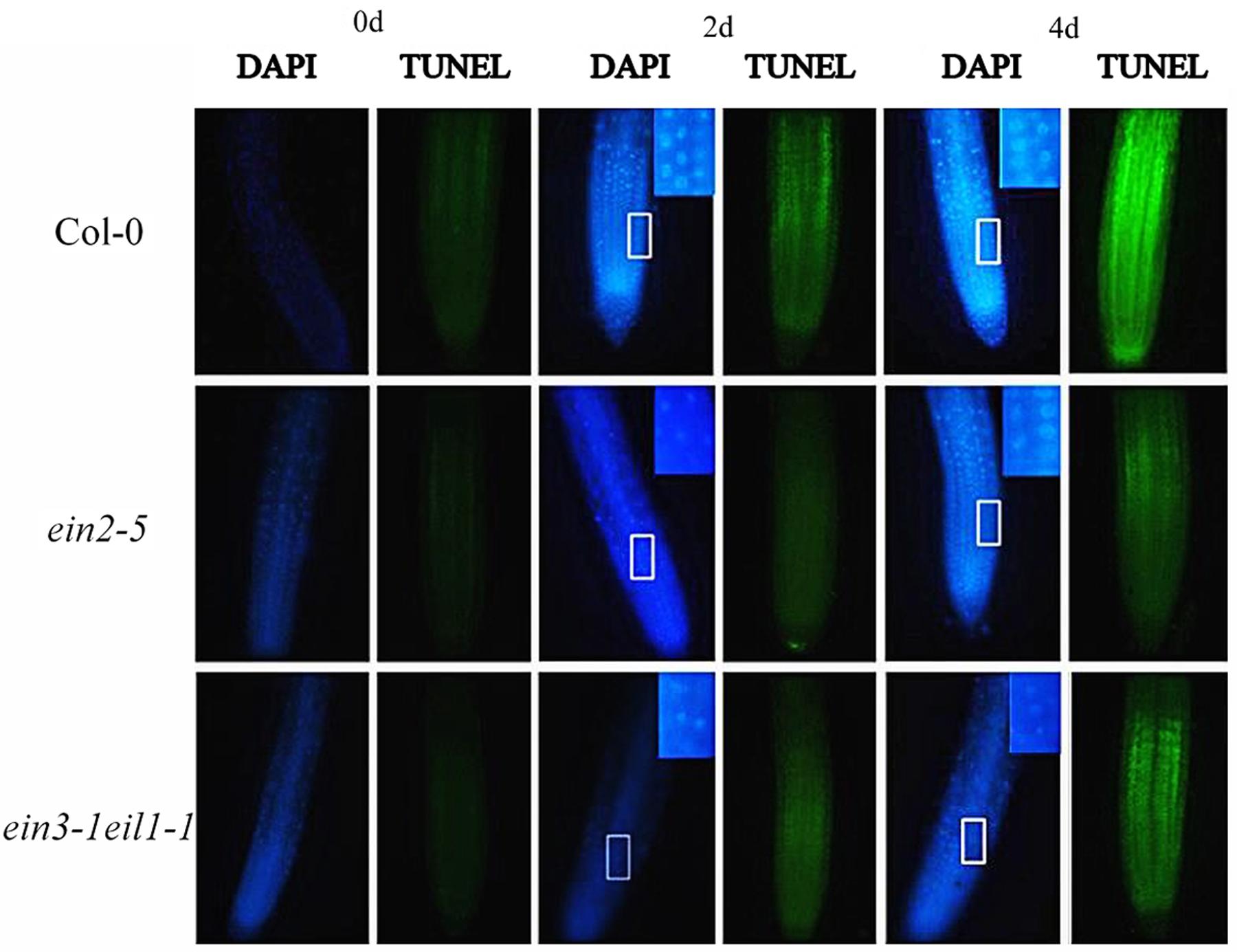
FIGURE 10. Progression of programmed cell death, indicated by DAPI and TUNEL staining in the root tips of the Col-0, ein2-5, and ein3-1eil1-1 plants during Cd stress. Four-day-old seedlings were transferred to CdCl2 (75 μM)-treated agar plates for different time points, as indicated. Insets: close up observations enlargement of chromatin condensation. At least 20 individual roots were analyzed for each genotype and treatment, and one representative image was selected for the figure.
Discussion
In plants, the root system is the first part of plant to suffer from heavy metal toxicity and respond to it (Li et al., 2012; Duan et al., 2015). Thus, the root system is of significant importance and relevance when investigating the responsive and adaptive patterns of plants to environmental stress. This study is particularly to understand the mechanisms by which Cd affects the root system growth and how plants response to this kind of stress. A key feature of the effects of Cd is the increased biosynthesis of ethylene and production of superoxide in roots of A. thaliana seedlings. Ethylene was demonstrated to control superoxide concentrations by modulating the activities of SOD isoenzymes.
Figure 1A indicated that the expression of both ACS2 and ACS6, which encode two ACS isoforms (ACS2 and ACS6) can be induced by Cd, as observed for other abiotic factors (Smith and Arteca, 2000; Wang et al., 2002; Schellingen et al., 2014). Furthermore, under Cd stress, the expression of EIN3, whose expression increases in the presence of ethylene, and the ethylene-responsive gene ERF1, which is an immediate target of EIN3 (Solano et al., 1998) were increased, indicating the presence of ethylene in the seedling roots. This confirms that ethylene signaling was involved in the plant response to Cd stress in the roots. However, expression of genes related to ethylene biosynthesis and perception reaches its maximum value at a certain dose of CdCl2 (75 μM) but is decreased with a higher concentration (100 μM), indicating that at 100 μM, cells suffered from extensive damage and many cellular components are affected. This would disrupt signaling networks. Moreover, the enhanced expression of these genes might account for the observed Cd-induced stimulation of EBS::GUS activity in the root apex (Figures 1B,C). The present results are consistent with a previous study indicating that Cd stress induces rapid ethylene production in root tips (Arteca and Arteca, 2007), suggesting that ethylene is involved in regulating plant responses to heavy metals.
The primary root lengths of the wild-type (Col-0) and the ein2-5 and ein3-1eil1-1 plants were severely inhibited by 75 μM CdCl2 (Supplementary Figure S1), consistent with a previous report that primary root length decreases in a Cd dose-dependent manner (Schellingen et al., 2015).
However, Figure 2 showed that the primary roots and the root tip lengths of both the ein2-5 and ein3-1eil1-1 plants were slightly longer than those of the wild-type (Col-0) plants after 2 days of Cd stress. This phenomenon was reversed after 4 days treatment as ein2-5 and ein3-1eil1-1 plants primary roots and root tips were significantly shorter than those of the wild-type plants.
The lateral root number and the root hairs density of the wild-type plants also significantly increased, consistent with the stress-induced morphogenic response (SIMR) reported before (Bochicchio et al., 2015). Whereas they were reduced in the ein2-5 and ein3-1eil1-1 mutants in response to Cd stress (Figure 3). Several studies indicate that morphological alterations that result in increased root surface area, such as the formation of root hairs and lateral roots, could be functionally related to stress avoidance mechanisms (Sofo et al., 2013; Vitti et al., 2013).
All of these facts highlighted the essential role of ethylene in the process. It is known that ethylene upregulates auxin biosynthesis in Arabidopsis seedlings root tips (Swarup et al., 2007; He et al., 2011), while auxin is involved in lateral root initiation and emergence in a number of plants including Arabidopsis (Aloni et al., 2006; Zolla et al., 2010; Sofo et al., 2013). Thus, the crosstalk between ethylene and axuin in regulating lateral root initiation and emergence may also contribute to root SIMR under Cd stress.
Figure 4 in consistent with a previous study; showed a significant accumulation of Cd content in root treated with Cd (Khan et al., 2016). The Cd contents of ein2-5 and ein3-1eil1-1 were even higher than the Cd content of the wild-type. Moreover, the bioaccumulation factors (Bfs) were even bigger in ein2-5 and ein3-1eil1-1 than Col-0. Application of ACC with different concentrations of Cd (50, 60, and 75 μM) decreased the Cd content in roots compared with roots treated with Cd alone (Supplementary Figure S3). These results suggested that ethylene could play a role in plant response to Cd stress.
The tip of roots is a zone of active ROS production (Liszkay et al., 2004). ROS such as O2-, H2O2 and HO∙ are considered as key factors in the oxidative burst, and play important physiological roles in plants (Mittler et al., 2004). Cd stress is related to the increase of ethylene and ROS (Xu et al., 2010a). The oxidative damage of ROS requires the action of antioxidative enzymes, including SOD, which can convert superoxide radicals into hydrogen peroxide, water and oxygen. As the results indicate, Cd stimulated superoxide production in the root tips (Figure 5), consistent with the results of Liptáková et al. (2012). However, the accumulation of superoxide in both the ein2-5 and ein3-1eil1-1 mutants was significantly greater than in the wild-type plants. The altered response pattern of superoxide accumulation in ein2-5 and ein3-1eil1-1 suggested that ethylene signaling regulates superoxide accumulation in the Cd-stressed root tips.
Application of exogenous ACC suppressed Cd stress-induced production of superoxide in the root tips of wild type plants (Figure 6), consistent with the salinity stress results (Lin et al., 2013a). On the other side, application of AIB, the inhibitor of ethylene biosynthesis, together with ACC and Cd reversed the ACC-induced suppression of superoxide production, consistent with the reverse effect of AIB to ACC effect on seed germination under stress (Lin et al., 2013b) and depending on the fact that AIB inhibits the endogenous as well as the ACC-dependent ethylene production (Satoh and Esashi, 1982), these results supports the involvement of ethylene in regulation of superoxide accumulation in the Cd-stressed root tips.
In agreement with former results, CdCl2 treatment increased NADPH oxidase activity while application of exogenous ACC decreased CdCl2 induced NADPH oxidase activity. Moreover, application of AIB with ACC and Cd reversed the ACC effect and increased NADPH oxidase activity (Figure 7), indicating ethylene regulation of O2∙- production under Cd stress, consistent with pervious result that an oxidative burst induction by NADPH oxidases under stress is connected to ethylene (Montero-Palmero et al., 2014).
In accompanied to the suppressed Cd stress-induced production of superoxide and the decreased CdCl2 induced NADPH oxidase activity, roots hairs density and root hairs length increased under CdCl2 stress by application of exogenous ACC (Supplementary Figure S4). The effect of ACC (a precursor of ethylene biosynthesis) was reversed by AIB (an inhibitor of ethylene biosynthesis). Taken these results together, we suggested that ethylene modulates root system development under Cd stress by regulating superoxide concentration.
Interestingly, under Cd stress, the activities of all three SOD isoenzymes increased in the wild-type plants compared with ein2-5 and ein2-5eil1-1. Moreover, application of exogenous ACC to the Cd-treated seedlings increased all three SOD isoenzymes activities in the wild-type plants (Figure 8), consistent with the increased SOD activity in Col-0 plants that was accompanied by a significant up-regulation of the genes FeSOD compared with ein2-5 mutant under salt stress (Lin et al., 2013a). Results in Supplementary Figure S5 show up-regulation of the three SOD genes in Col-0 plants compared with ein2-5 mutant under oxidative stress induced by salinity. This observation that suggests that ethylene may directly increase SOD activity under Cd stress, may be the underlying mechanism through which ethylene controls the amount of superoxide, which then initiates distinct forms of responses and acclimations depending upon the levels of superoxide in Arabidopsis root tips under Cd stress.
High concentrations of superoxide lead to oxidative stress, which directly inhibits or modifies some proteins and ultimately induces cell death (Miller et al., 2010). There are two types of cell death, necrosis and apoptosis, which is a subset of PCD. PI, a nucleic acid dye that intercalates into double-stranded nucleic acids, is excluded from viable cells but can penetrate the cell membranes of dead cells (Ning et al., 2002; Duan et al., 2010). Generally, Cd treatment induced the onset of necrosis at the meristematic and elongation zones of all plants compared with the control (Figure 9). However, greater PI-positive staining was observed after (4 days) Cd treatment in both the ein2-5 and ein3-1eil1-1 mutant root tips compared with the Col-0 plants. In addition, these results were confirmed by Trypan blue staining, another molecular prove for cell necrosis. These results suggest that ethylene may play crucial roles in preventing cells of the meristematic and elongation zones from undergoing necrosis during stress and recovery periods when plants are facing stresses.
Programmed cell death is an activate process of cellular suicide that is essential for development and stress responses in plants. To examine whether PCD is involved in cell death at root tips, DAPI staining and the TUNEL assay, were used, respectively, which are typically used as diagnostic markers for PCD (Duan et al., 2010; Xu et al., 2010b). In contrast to the progression of necrosis, the initiation of PCD was more rapid and intensive in the wild-type plant root tips compared with the ein2-5 and ein3-1eil1-1 plants, confirming the critical role of ethylene in the progression of PCD under Cd stress (Figure 10).
Based on our combined results, a schematic model (Figure 11) elucidating the interaction of ethylene, SOD, superoxide and PCD in the root meristematic and elongation zones under Cd stress, is presented. In this model, Cd treatment induces the rapid production of ethylene and superoxide by the gene up-regulation of ethylene synthesis and the Cd-caused oxidative damage, respectively. Under Cd stress, the presence of ethylene maintains or increases the activity of SOD, which maintains the level of superoxide lower than what expected without ethylene and consequently prevent cells of the meristematic and elongation zones from undergoing necrosis. Superoxide could function as a signaling molecule that initiates the occurrence of PCD in root tips at the early stage of Cd. PCD occurs in the region of root apical meristem in undifferentiated cells, where it alters root apical dominance and remodels the root system architecture, inhibiting primary root elongation and increasing lateral roots number and root hairs density, perhaps to minimize the damage caused by stress conditions. This response is an adaptive and acclimation strategy of plants to adverse environments.
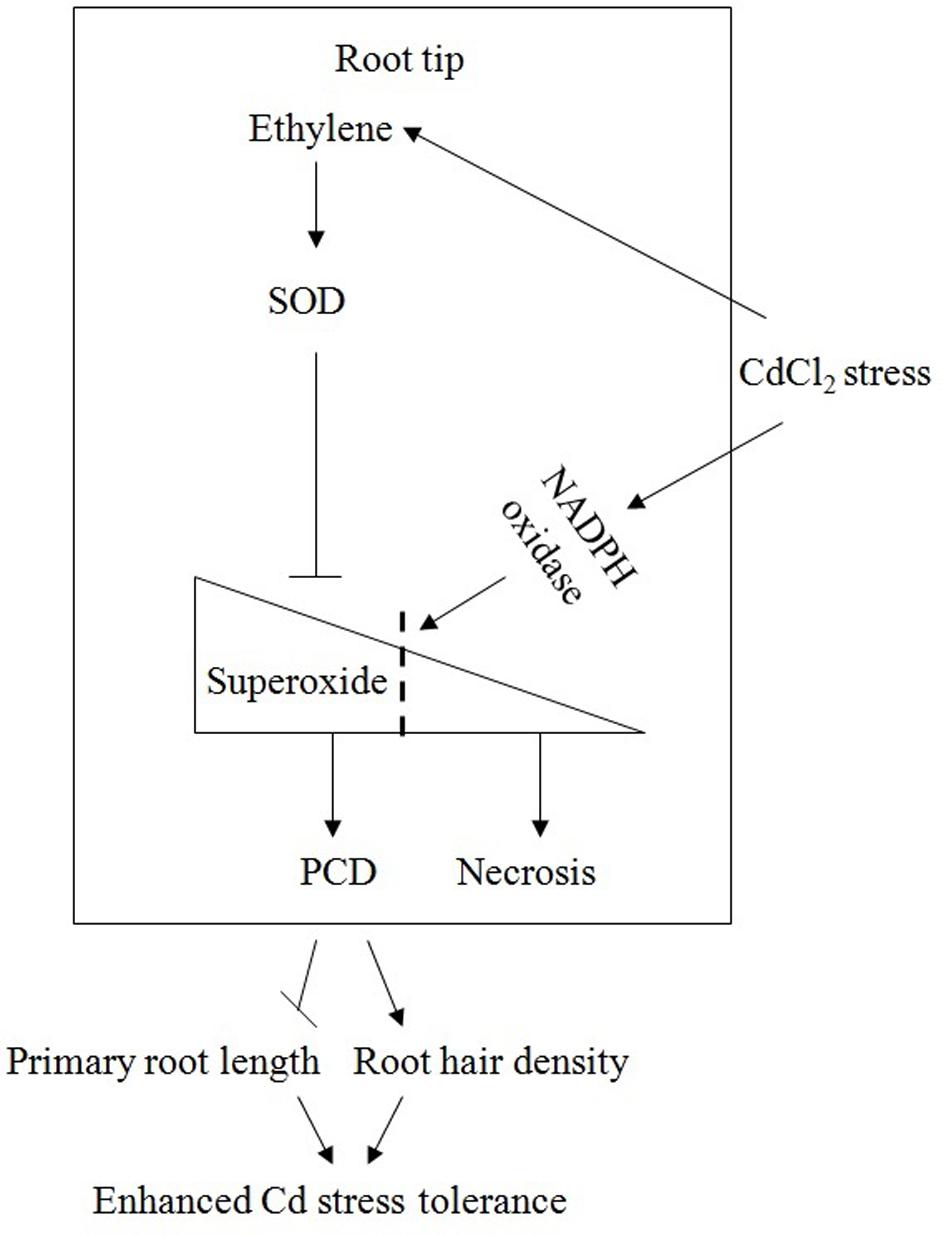
FIGURE 11. A schematic model elucidating the interaction of ethylene, SOD, superoxide and PCD in the root meristematic and elongation zones under Cd stress.
Conclusion
The presence of ethylene enables plants to perceive adverse stimuli and to respond effectively, thus enhancing their acclimation and adaptation to stress. In ein2-5 and ein3-1eil1-1 seedlings, the increased superoxide concentration induced by Cd stress resulted in cell death and defects in root growth and development in the root meristem zone, by which root elongation was inhibited. In contrast, ethylene reduced the concentration of superoxide maintained in wild type plants. Moreover, superoxide concentration was reduced by ACC application and reversely increased by application of AIB that inhibits the endogenous as well as the ACC-dependent ethylene production. The ethylene-induced suppression of superoxide decreased the occurrence of cells death and initiated PCD to minimize the damage caused by Cd stress. Hence, this is a general adaptive mechanism in A. thaliana through ethylene induction to improve root system development by modulating superoxide anion concentration under Cd stress.
Author Contributions
ZT conceived and designed the experiments. AA, ZY, and YL performed the experiments and wrote the manuscript. JL and ZZ revised the manuscript. All authors reviewed the manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The reviewer MA and handling Editor declared their shared affiliation, and the handling Editor states that the process nevertheless met the standards of a fair and objective review.
Acknowledgments
The authors thank Prof. Hongwei Guo (Peking University) for providing Arabidopsis thaliana seeds. We express our appreciation to Prof. David Lawlor for critical reading of the manuscript. This study was supported by the National Natural Science foundation of China (Grant No. 31370007).
Supplementary Material
The Supplementary Material for this article can be found online at: http://journal.frontiersin.org/article/10.3389/fpls.2017.00253/full#supplementary-material
References
Aloni, R., Aloni, E., Langhans, M., and Ullrich, C. I. (2006). Role of cytokinin and auxin in shaping root architecture: regulating vascular differentiation, lateral root initiation, root apical dominance and root gravitropism. Ann. Bot. 97, 883–893. doi: 10.1093/aob/mcl027
Alonso, J. M., Hirayama, T., Roman, G., Nourizadeh, S., and Ecker, J. R. (1999). EIN2, a bifunctional transducer of ethylene and stress responses in Arabidopsis. Science 284, 2148–2152. doi: 10.1126/science.284.5423.2148
Alonso, J. M., Stepanova, A. N., Solano, R., Wisman, E., Ferrari, S., Ausubel, F. M., et al. (2003). Five components of the ethylene-response pathway identified in a screen for weak ethylene-insensitive mutants in Arabidopsis. Proc. Natl. Acad. Sci. U.S.A. 100, 2992–2997. doi: 10.1073/pnas.0438070100
An, F., Zhao, Q., Ji, Y., Li, W., Jiang, Z., Yu, X., et al. (2010). Ethylene-induced stabilization of ETHYLENE INSENSITIVE3 and EIN3-LIKE1 is mediated by proteasomal degradation of EIN3 binding F-box 1 and 2 that requires EIN2 in Arabidopsis. Plant Cell 22, 2384–2401. doi: 10.1105/tpc.110.076588
Andresen, E., Kappel, S., Stärk, H. J., Riegger, U., Borovec, J., Mattusch, J., et al. (2016). Cadmium toxicity investigated at the physiological and biophysical levels under environmentally relevant conditions using the aquatic model plant Ceratophyllum demersum. New Phytol. 210, 1244–1258. doi: 10.1111/nph.13840
Arteca, R. N., and Arteca, J. M. (2007). Heavy-metal-induced ethylene production in Arabidopsis thaliana. J. Plant Physiol. 164, 1480–1488. doi: 10.1016/j.jplph.2006.09.006
Asgher, M., Khan, M. I. R., Anjum, N. A., and Khan, N. A. (2015). Minimising toxicity of cadmium in plants—role of plant growth regulators. Protoplasma 252, 399–413. doi: 10.1007/s00709-014-0710-4
Azevedo, H., Pinto, G., Fernandes, J., Loureiro, S., and Santos, C. (2005). Cadmium effects on sunflower: growth and photosynthesis. J. Plant Nutr. 28, 2211–2220. doi: 10.1080/01904160500324782
Binder, B. M., Walker, J. M., Gagne, J. M., Emborg, T. J., Hemmann, G., Bleecker, A. B., et al. (2007). The Arabidopsis EIN3 binding F-Box proteins EBF1 and EBF2 have distinct but overlapping roles in ethylene signaling. Plant Cell 19, 509–523. doi: 10.1105/tpc.106.048140
Bochicchio, R., Sofo, A., Terzano, R., Gattullo, C. E., Amato, M., and Scopa, A. (2015). Root architecture and morphometric analysis of Arabidopsis thaliana grown in Cd/Cu/Zn-gradient agar dishes: a new screening technique for studying plant response to metals. Plant Physiol. Biochem. 91, 20–27. doi: 10.1016/j.plaphy.2015.03.010
Burzynski, M., and Klobus, G. (2004). Changes of photosynthetic parameters in cucumber leaves under Cu, Cd, and Pb stress. Photosynth 42, 505–510. doi: 10.1007/S11099-005-0005-2
Cao, S. Q., Ren, G., Jiang, L., Yuan, H. B., and Ma, G. H. (2009). The role of β-aminobutyric acid in enhancing cadmium tolerance in Arabidopsis thaliana. Russ. J. Plant Physiol. 56, 575–579. doi: 10.1134/S1021443709040190
Chae, H., and Lee, W. (2001). Ethylene-and enzyme-mediated superoxide production and cell death in carrot cells grown under carbon starvation. Plant Cell Rep. 20, 256–261. doi: 10.1007/s002990000307
Chao, Q., Rothenberg, M., Solano, R., Roman, G., Terzaghi, W., and Ecker, J. R. (1997). Activation of the ethylene gas response pathway in Arabidopsis by the nuclear protein ETHYLENE-INSENSITIVE3 and related proteins. Cell 89, 1133–1144. doi: 10.1016/S0092-8674(00)80300-1
Chen, G. X., and Asada, K. (1989). Ascorbate peroxidase in tea leaves: occurrence of two isozymes and the differences in their enzymatic and molecular properties. Plant Cell Physiol. 30, 987–998. doi: 10.1093/oxfordjournals.pcp.a077844
Choppala, G., Saifullah Bolan, N., Bibi, S., Iqbal, M., Rengel, Z., Kunhikrishinan, A., et al. (2014). Cellular mechanisms in higher plants governing tolerance to cadmium toxicity. Crit. Rev. Plant Sci. 33, 374–391. doi: 10.1080/07352689.2014.903747
Demidchik, V., Straltsova, D., Medvedev, S. S., Pozhvanov, G. A., Sokolik, A., and Yurin, V. (2014). Stress-induced electrolyte leakage: the role of K+-permeable channels and involvement in programmed cell death and metabolic adjustment. J. Exp. Bot. 65, 1259–1270. doi: 10.1093/jxb/eru004
Dong, C. H., Jang, M., Scharein, B., Malach, A., Rivarola, M., Liesch, J., et al. (2010). Molecular association of the Arabidopsis ETR1 ethylene receptor and a regulator of ethylene signaling, RTE1. J. Biol. Chem. 285, 40706–40713. doi: 10.1074/jbc.M110.146605
Duan, X., Li, X., Ding, F., Zhao, J., Guo, A., Zhang, L., et al. (2015). Interaction of nitric oxide and reactive oxygen species and associated regulation of root growth in wheat seedlings under zinc stress. Ecotoxicol. Environ. Saf. 113, 95–102. doi: 10.1016/j.ecoenv.2014.11.030
Duan, Y., Zhang, W., Li, B., Wang, Y., Li, K., Han, C., et al. (2010). An endoplasmic reticulum response pathway mediates programmed cell death of root tip induced by water stress in Arabidopsis. New Phytol. 186, 681–695. doi: 10.1111/j.1469-8137.2010.03207.x
Dunand, C., Crèvecoreur, M., and Penel, C. (2007). Distribution of superoxide and hydrogen peroxide in Arabidopsis root and their influence on root development: possible interaction with peroxidases. New Phytol. 174, 332–341. doi: 10.1111/j.1469-8137.2007.01995.x
Giannopolitis, C. N., and Ries, S. K. (1977). Superoxide dismutases I. Occurrence in higher plants. Plant physiol. 59, 309–314. doi: 10.1104/pp.59.2.309
Guo, H., and Ecker, J. R. (2003). Plant responses to ethylene gas are mediated by SCF EBF1/EBF2-dependent proteolysis of EIN3 transcription factor. Cell 115, 667–677. doi: 10.1016/S0092-8674(03)00969-3
Guo, H., and Ecker, J. R. (2004). The ethylene signaling pathway: new insights. Curr. Opin. Plant Biol. 7, 40–49. doi: 10.1016/j.pbi.2003.11.011
Han, L., Li, G. J., Yang, K. Y., Mao, G., Wang, R., Liu, Y., et al. (2010). Mitogen-activated protein kinase 3 and 6 regulate Botrytis cinerea-induced ethylene production in Arabidopsis. Plant J. 64, 114–127. doi: 10.1111/j.1365-313X.2010.04318.x
Harding, S. A., and Roberts, D. M. (1998). Incompatible pathogen infection results in enhanced reactive oxygen and cell death responses in transgenic tobacco expressing a hyperactive mutant calmodulin. Planta 206, 253–258. doi: 10.1007/s004250050397
He, W., Brumos, J., Li, H., Ji, Y., Ke, M., Gong, X., et al. (2011). A small-molecule screen identifies L-kynurenine as a competitive inhibitor of TAA1/TAR activity in ethylene-directed auxin biosynthesis and root growth in Arabidopsis. Plant Cell 23, 3944–3960. doi: 10.1105/tpc.111.089029
Hernández, J. A., Ferrer, M. A., Jiménez, A., Barceló, A. R., and Sevilla, F. (2001). Antioxidant systems and O2.-/H2O2 production in the apoplast of pea leaves. its relation with salt-induced necrotic lesions in minor veins. Plant Physiol. 127, 817–831. doi: 10.1104/pp.010188
Johnson, P. R., and Ecker, J. R. (1998). The ethylene gas signal transduction pathway: a molecular perspective. Annu. Rev. Genet. 32, 227–254. doi: 10.1146/annurev.genet.32.1.227
Kendrick, M. D., and Chang, C. (2008). Ethylene signaling: new levels of complexity and regulation. Curr. Opin. Plant Biol. 11, 479–485. doi: 10.1016/j.pbi.2008.06.011
Khan, N. A., Asgher, M., Per, T. S., Masood, A., Fatma, M., and Khan, M. I. R. (2016). Ethylene potentiates sulfur-mediated reversal of cadmium inhibited photosynthetic responses in mustard. Front. Plant Sci. 7:1628. doi: 10.3389/fpls.2016.01628
Li, J., Jia, H., and Wang, J. (2014). cGMP and ethylene are involved in maintaining ion homeostasis under salt stress in Arabidopsis roots. Plant Cell Rep. 33, 447–459. doi: 10.1007/s00299-013-1545-8
Li, X., Yang, Y., Zhang, J., Jia, L., Li, Q., Zhang, T., et al. (2012). Zinc induced phytotoxicity mechanism involved in root growth of Triticum aestivum L. Ecotoxicol. Environ. Saf. 86, 198–203. doi: 10.1016/j.ecoenv.2012.09.021
Lin, Y., Chen, D., Paul, M., Zu, Y., and Tang, Z. (2013a). Loss-of-function mutation of EIN2 in Arabidopsis exaggerates oxidative stress induced by salinity. Acta Physiol Plant. 35, 1319–1328. doi: 10.1007/s11738-012-1172-y
Lin, Y., Yang, L., Paul, M., Zu, Y., and Tang, Z. (2013b). Ethylene promotes germination of Arabidopsis seed under salinity by decreasing reactive oxygen species: evidence for the involvement of nitric oxide simulated by sodium nitroprusside. Plant Physiol. Biochem. 73, 211–218. doi: 10.1016/j.plaphy.2013.10.003
Lin, Y. C., Wang, J. J., Zu, Y. G., and Tang, Z. H. (2012). Ethylene antagonizes the inhibition of germination in Arabidopsis induced by salinity by modulating the concentration of hydrogen peroxide. Acta Physiol. Plant. 34, 1895–1904. doi: 10.1007/s11738-012-0989-8
Lingam, S., Mohrbacher, J., Brumbarova, T., Potuschak, T., Fink-Straube, C., Blondet, E., et al. (2011). Interaction between the bHLH transcription factor FIT and ETHYLENE INSENSITIVE3/ETHYLENE INSENSITIVE3-LIKE1 reveals molecular linkage between the regulation of iron acquisition and ethylene signaling in Arabidopsis. Plant Cell 23, 1815–1829. doi: 10.1105/tpc.111.084715
Liptáková, L. U., Boèová, B., Huttová, J., Mistrík, I., and Tamás, L. (2012). Superoxide production induced by short-term exposure of barley roots to cadmium, auxin, alloxan and sodium dodecyl sulfate. Plant Cell Rep. 31, 2189–2197. doi: 10.1007/s00299-012-1329-6
Liszkay, A., van der Zalm, E., and Schopfer, P. (2004). Production of reactive oxygen intermediates (O2-, H2O2, and OH) by maize roots and their role in wall loosening and elongation growth. Plant Physiol. 136, 3114–3123. doi: 10.1104/pp.104.044784
Liu, Y., Xiong, Y., and Bassham, D. C. (2009). Autophagy is required for tolerance of drought and salt stress in plants. Autophagy 5, 954–963. doi: 10.4161/auto.5.7.9290
Liu, Y., Ye, N., Liu, R., Chen, M., and Zhang, J. (2010). H2O2 mediates the regulation of ABA catabolism and GA biosynthesis in Arabidopsis seed dormancy and germination. J. Exp. Bot. 61, 1–12. doi: 10.1093/jxb/erq125
Livak, K. J., and Schmittgen, T. D. (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method. Methods 25, 402–408. doi: 10.1006/meth.2001.1262
Lorenzo, O., Piqueras, R., Sánchez-Serrano, J. J., and Solano, R. (2003). ETHYLENE RESPONSE FACTOR1 integrates signals from ethylene and jasmonate pathways in plant defense. Plant Cell 15, 165–178. doi: 10.1105/tpc.007468
Miller, G. A. D., Suzuki, N., Ciftci-Yilmaz, S., and Mittler, R. O. N. (2010). Reactive oxygen species homeostasis and signalling during drought and salinity stresses. Plant Cell Environ. 33, 453–467. doi: 10.1111/j.1365-3040.2009.02041.x
Mittler, R., Vanderauwera, S., Gollery, M., and Van Breusegem, F. (2004). Reactive oxygen gene network of plants. Trends Plant Sci. 9, 490–498. doi: 10.1016/j.tplants.2004.08.009
Montero-Palmero, M. B., Martín-Barranco, A., Escobar, C., and Hernández, L. E. (2014). Early transcriptional responses to mercury: a role for ethylene in mercury-induced stress. New Phytol. 201, 116–130. doi: 10.1111/nph.12486
Nagarajan, V. K., and Smith, A. P. (2012). Ethylene’s role in phosphate starvation signaling: more than just a root growth regulator. Plant Cell Physiol. 53, 277–286. doi: 10.1093/pcp/pcr186
Ning, S. B., Wang, L., and Song, Y. C. (2002). Identification of programmed cell death in situ in individual plant cells in vivo using a chromosome preparation technique. J. Exp. Bot. 53, 651–658. doi: 10.1093/jexbot/53.369.651
Olowu, R. A., Adewuyi, G. O., Onipede, O. J., Lawal, O. A., and Sunday, O. M. (2015). Concentration of heavy metals in root, stem and leaves of Acalypha indica and Panicum maximum jacq from three major dumpsites in ibadan metropolis, South West Nigeria. Am. J. Chem. 5, 40–48. doi: 10.5923/j.chemistry.20150501.06
Pan, Y. J., Liu, L., Lin, Y. C., Zu, Y. G., Li, L. P., and Tang, Z. H. (2016). Ethylene antagonizes salt-induced growth retardation and cell death process via transcriptional controlling of ethylene-, BAG-and senescence-associated genes in Arabidopsis. Front. Plant Sci. 7:696. doi: 10.3389/fpls.2016.00696
Parlanti, S., Kudahettige, N. P., Lombardi, L., Mensuali-Sodi, A., Alpi, A., Perata, P., et al. (2011). Distinct mechanisms for aerenchyma formation in leaf sheaths of rice genotypes displaying a quiescence or escape strategy for flooding tolerance. Ann. Bot. 107, 1335–1343. doi: 10.1093/aob/mcr086
Poschenrieder, C. H., and Barceló, J. (1999). “Water relations in heavy metal stressed plants,” in Heavy Metal Stress in Plants, ed. M. N. V. Prasad (Berlin: Springer), 207–229. doi: 10.1007/978-3-662-07745-0_10
Quartacci, M. F., Cosi, E., and Navari-Izzo, F. (2001). Lipids and NADPH-dependent superoxide production in plasma membrane vesicles from roots of wheat grown under copper deficiency or excess. J. Exp. Bot. 52, 77–84. doi: 10.1093/jxb/52.354.77
Radeva, V., Petrov, V., Minkov, I., Toneva, V., and Gechev, T. (2010). Effect of cadmium on Arabidopsis thaliana mutants tolerant to oxidative stress. Biotechnol. Biotechnol. Equip. 24, 113–118. doi: 10.1080/13102818.2010.10817823
Sandalio, L., Dalurzo, H., Gomez, M., Romero-Puertas, M., and Del Rio, L. (2001). Cadmium-induced changes in the growth and oxidative metabolism of pea plants. J. Exp. Bot. 52, 2115–2126. doi: 10.1093/jexbot/52.364.2115
Santos, C., Monteiro, M., and Dias, M. (2010). Cadmium Toxicity in Crops: A Review. Environmental Science, Engineering and Technology. Novinka: Nova Publishers.
Satoh, S., and Esashi, Y. (1982). Effects of α-aminoisobutyric acid and D-and L-amino acids on ethylene production and content of 1-aminocyclopropane-1-carboxylic acid in cotyledonary segments of cocklebur seeds. Physiol. Plant. 54, 147–152. doi: 10.1111/j.1399-3054.1982.tb06318.x
Schellingen, K., Van Der Straeten, D., Remans, T., Loix, C., Vangronsveld, J., and Cuypers, A. (2015). Ethylene biosynthesis is involved in the early oxidative challenge induced by moderate Cd exposure in Arabidopsis thaliana. Environ. Exp. Bot. 117, 1–11. doi: 10.1016/j.envexpbot.2015.04.005
Schellingen, K., Van Der Straeten, D., Vandenbussche, F., Prinsen, E., Remans, T., Vangronsveld, J., et al. (2014). Cadmium-induced ethylene production and responses in Arabidopsis thaliana rely on ACS2 and ACS6 gene expression. BMC Plant Biol. 14:1. doi: 10.1186/s12870-014-0214-6
Shah, M. T., Ara, J., Muhammad, S., Khan, S., Asad, S. A., and Ali, L. (2014). Potential heavy metals accumulation of indigenous plant species along the mafic and ultramafic terrain in the Mohmand Agency, Pakistan. Clean 42, 339–346. doi: 10.1002/clen.201200632
Shibuya, K., Barry, K. G., Ciardi, J. A., Loucas, H. M., Underwood, B. A., Nourizadeh, S., et al. (2004). The central role of PhEIN2 in ethylene responses throughout plant development in petunia. Plant Physiol. 136, 2900–2912. doi: 10.1104/pp.104.046979
Singh, R., and Agrawal, M. (2007). Effects of sewage sludge amendment on heavy metal accumulation and consequent responses of Beta vulgaris plants. Chemosphere 67, 2229–2240. doi: 10.1016/j.chemosphere.2006.12.019
Skottke, K. R., Yoon, G. M., Kieber, J. J., and DeLong, A. (2011). Correction: protein phosphatase 2A controls ethylene biosynthesis by differentially regulating the turnover of ACC synthase isoforms. PLoS Genet. 7:e1001370. doi: 10.1371/annotation/b4fc15d6-b3ae-4fbb-8d88-b7d674a79697
Smith, J. M., and Arteca, R. N. (2000). Molecular control of ethylene production by cyanide in Arabidopsis thaliana. Physiol. Plant. 109, 180–187. doi: 10.1034/j.1399-3054.2000.100210.x
Sofo, A., Vitti, A., Nuzzaci, M., Tataranni, G., Scopa, A., Vangronsveld, J., et al. (2013). Correlation between hormonal homeostasis and morphogenic responses in Arabidopsis thaliana seedlings growing in a Cd/Cu/Zn multi-pollution context. Physiol. Plant. 149, 487–498. doi: 10.1111/ppl.12050
Solano, R., Stepanova, A., Chao, Q., and Ecker, J. R. (1998). Nuclear events in ethylene signaling: a transcriptional cascade mediated by ETHYLENE-INSENSITIVE3 and ETHYLENE-RESPONSE-FACTOR1. Genes Dev. 12, 3703–3714. doi: 10.1101/gad.12.23.3703
Swarup, R., Perry, P., Hagenbeek, D., Van Der Straeten, D., Beemster, G. T., Sandberg, G., et al. (2007). Ethylene upregulates auxin biosynthesis in Arabidopsis seedlings to enhance inhibition of root cell elongation. Plant Cell 19, 2186–2196. doi: 10.1105/tpc.107.052100
Van Gestelen, P., Asard, H., and Caubergs, R. J. (1997). Solubilization and separation of a plant plasma membrane NADPH-O2-synthase from other NAD (P) H oxidoreductases. Plant Physiol. 115, 543–550. doi: 10.1104/pp.115.2.543
Vitti, A., Nuzzaci, M., Scopa, A., Tataranni, G., Remans, T., Vangronsveld, J., et al. (2013). Auxin and cytokinin metabolism and root morphological modifications in Arabidopsis thaliana seedlings infected with Cucumber mosaic virus (CMV) or exposed to cadmium. Int. J. Mol. Sci. 14, 6889–6902. doi: 10.3390/ijms14046889
Wahid, A., and Khaliq, S. (2015). Architectural and biochemical changes in embryonic tissues of maize under cadmium toxicity. Plant Biol. 17, 1005–1012. doi: 10.1111/plb.12326
Wang, K. L.-C., Li, H., and Ecker, J. R. (2002). Ethylene biosynthesis and signaling networks. Plant Cell 14(Suppl.), S131–S151. doi: 10.1105/tpc.001768
Xu, J., Wang, W. Y., Yin, H. X., Sun, H., and Mi, Q. (2010a). Exogenous nitricoxide improves antioxidative capacity and reduces auxin degradation in roots of Medicago truncatula seedlings under cadmium stress. Plant Soil 326, 321. doi: 10.1007/s11104-009-0011-4
Xu, J., Yin, H., Li, Y., and Liu, X. (2010b). Nitric oxide is associated with long-term zinc tolerance in Solanum nigrum. Plant Physiol. 154, 1319–1334. doi: 10.1104/pp.110.162982
Yoo, S. D., Cho, Y., and Sheen, J. (2009). Emerging connections in the ethylene signaling network. Trends Plant Sci. 14, 270–279. doi: 10.1016/j.tplants.2009.02.007
Yu, Q., Osborne, L., and Rengel, Z. (1998). Micronutrient deficiency changes activities of superoxide dismutase and ascorbate peroxidase in tobacco plants. J. Plant Nutr. 21, 1427–1437. doi: 10.1080/01904169809365493
Yu, Q., and Rengel, Z. (1999). Micronutrient deficiency influences plant growth and activities of superoxide dismutases in narrow-leafed lupins. Ann. Bot. 83, 175–182. doi: 10.1006/anbo.1998.0811
Zhu, Z., An, F., Feng, Y., Li, P., Xue, L., Mu, A., et al. (2011). Derepression of ethylene-stabilized transcription factors (EIN3/EIL1) mediates jasmonate and ethylene signaling synergy in Arabidopsis. Proc. Natl. Acad. Sci. U.S.A. 108, 12539–12544. doi: 10.1073/pnas.1103959108
Keywords: ethylene, Cd stress, superoxide, programmed cell death, root system, Arabidopsis thaliana
Citation: Abozeid A, Ying Z, Lin Y, Liu J, Zhang Z and Tang Z (2017) Ethylene Improves Root System Development under Cadmium Stress by Modulating Superoxide Anion Concentration in Arabidopsis thaliana. Front. Plant Sci. 8:253. doi: 10.3389/fpls.2017.00253
Received: 04 November 2016; Accepted: 09 February 2017;
Published: 24 February 2017.
Edited by:
Nafees A. Khan, Aligarh Muslim University, IndiaReviewed by:
Naser A. Anjum, University of Aveiro, PortugalMohd Asgher, Aligarh Muslim University, India
Copyright © 2017 Abozeid, Ying, Lin, Liu, Zhang and Tang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Zhonghua Tang, dGFuZ3poQG5lZnUuZWR1LmNu Ann Abozeid, YW5uYWJvemVpZEB5YWhvby5jb20=
†These authors have contributed equally to this work.
 Ann Abozeid
Ann Abozeid Zuojia Ying
Zuojia Ying Yingchao Lin
Yingchao Lin Jia Liu
Jia Liu Zhonghua Zhang1
Zhonghua Zhang1