Abstract
Plant associated bacteria with plant growth promotion (PGP) properties have been proposed for use as environmentally friendly biofertilizers for sustainable agriculture; however, analysis of their efficacy in the field is often limited. In this study, greenhouse and field trials were carried out using individual endophytic Pseudomonas fluorescens strains, the well characterized rhizospheric P. fluorescens F113 and an endophytic microbial consortium of 10 different strains. These bacteria had been previously characterized with respect to their PGP properties in vitro and had been shown to harbor a range of traits associated with PGP including siderophore production, 1-aminocyclopropane-1-carboxylic acid (ACC) deaminase activity, and inorganic phosphate solubilization. In greenhouse experiments individual strains tagged with gfp and Kmr were applied to Brassica napus as a seed coat and were shown to effectively colonize the rhizosphere and root of B. napus and in addition they demonstrated a significant increase in plant biomass compared with the non-inoculated control. In the field experiment, the bacteria (individual and consortium) were spray inoculated to winter oilseed rape B. napus var. Compass which was grown under standard North Western European agronomic conditions. Analysis of the data provides evidence that the application of the live bacterial biofertilizers can enhance aspects of crop development in B. napus at field scale. The field data demonstrated statistically significant increases in crop height, stem/leaf, and pod biomass, particularly, in the case of the consortium inoculated treatment. However, although seed and oil yield were increased in the field in response to inoculation, these data were not statistically significant under the experimental conditions tested. Future field trials will investigate the effectiveness of the inoculants under different agronomic conditions.
Introduction
The world’s population is expected to increase to 9 billion by 2050 (Tilman et al., 2011) and will require greater levels of crop production to meet the increase in demand for food (Godfray et al., 2010). In addition, world energy supply remains highly dependent on non-renewable resources. The potential of bioenergy crops, in conjunction with other renewable energy sources, has received a great deal of interest over the last decade (Hill et al., 2006). However, one of the main concerns regarding bioenergy crops is the intensive agricultural practices associated with their production (Germaine et al., 2010) and the associated negative effect on the environment. Impacts include a loss in biodiversity (Geiger et al., 2010), the contamination of water ways causing eutrophication, loss of aquatic biodiversity and drinking water sources (Camargo and Alonso, 2006), and causing destruction to soil through degradation over time (Oldeman, 1992). Brassica napus L. (oilseed rape) is an important global crop that is a major source of vegetable oil for human consumption (USDA-FAS, 2015) as well as a source of oil for producing biodiesel. In addition, winter oilseed rape is used as a break crop in cereal production and has been shown to reduce “take-all” fungal disease in wheat when used as part of a crop rotation (Angus et al., 1991; Hilton et al., 2013).
Microorganisms play a key role in the health and development of crops (Tikhonovich and Provorov, 2011; Cory and Franklin, 2012) and the relationship between rhizobacteria and endophytes with their plant hosts has been reviewed extensively (Ryan et al., 2008; Hayat et al., 2010; Mercado-Blanco and Lugtenberg, 2014). Such plant growth promoting rhizobacteria (PGPR) have considerable potential as biological inoculants in sustainable agriculture (Saharan and Nehra, 2011; Glick, 2012; Sivasakthi et al., 2014). Plants that are inoculated with PGPR benefit from the resulting plant–microbe interaction as the bacteria contribute to plant growth and health by multiple mechanisms including nitrogen fixation, synthesis of phytohormones, modulation of plant ethylene levels, solubilization of unavailable soil phosphate and suppression of pathogens through niche exclusion, and the production of anti-microbial metabolites (Fuentes-Ramirez and Caballero-Mellado, 2005; Franche et al., 2009; Babalola, 2010; Compant et al., 2010).
A collection of plant associated bacteria from various plant hosts (Miscanthus × giganteus, B. napus, and Iris pseudacorus) have been isolated and partially characterized (Otieno et al., 2013). Strains were isolated following standard procedures from rhizosphere and internal plant tissues including root, leaves, and stem (Germaine et al., 2004, 2009; Otieno et al., 2013). Many of these strains have multiple plant growth promotion (PGP) characteristics including siderophore production, 1-aminocyclopropane-1-carboxylic acid (ACC) deaminase activity (for the regulation of plant stress hormone ethylene caused by abiotic stress conditions), phosphate solubilization, and in some cases biocontrol against fungal plant pathogens.
Of particular interest are three bacterial strains isolated from Miscanthus × giganteus and a “Mastermix consortium” (MM consortium) of these strains combined with seven other isolated PGP microorganisms. These three strains are P. fluorescens L228, L111, and L321 which have been shown to have PGP potential in vitro (Brennan, 2010; Otieno et al., 2013, 2015) and in silico following genome sequence analysis (Moreira et al., 2016). We also included in this study P. fluorescens F113 a well-characterized biocontrol and PGPR strain that was originally isolated in Ireland from Beta vulgaris (L) (sugar beet) (Shanahan et al., 1992; Redondo-Nieto et al., 2013). This serves as a useful reference strain in our study. P. fluorescens are members of the Class gammaproteobacteria and are a very diverse complex (Garrido-Sanz et al., 2016). Pseudomonas spp. are important bacteria in agriculture and have been shown to promote growth, protect plants from pathogens and herbivores (Adesemoye and Egamberdieva, 2013; Sivasakthi et al., 2014), play a role in phytoremediation (Germaine et al., 2009; Glick, 2012), and are a part of the core microbiome of many plants.
The aim of this study is to investigate the potential of these bacteria as biofertilizers in greenhouse and field conditions to determine if they induce PGP of oilseed rape, an important biofuel, food, and break crop in Europe and globally. The colonization potential of each of these strains was examined on oilseed rape using gfp marked derivatives under greenhouse conditions to determine the interaction of each strain in the rhizosphere, root, leaf, and PGP effects. Finally, a field trial was undertaken to investigate the impact of these inoculants under standard Irish agronomic conditions for oilseed rape on crop growth and biomass parameters.
Materials and Methods
Greenhouse Experiments
Green fluorescent protein (gfp) expressing; kanamycin-resistant marker (Kmr) derivatives of P. fluorescens L321, L228, L111, and F113 (Table 1) were constructed as described in Germaine et al. (2004) through biparental mating with E. coli S17.1 λpir pUTmTn5gusA-pgfp21 (Xi et al., 1999). The greenhouse experiment consisted of four bacterial treatments (F113gfp:kmr, L321gfp:kmr, L111gfp:kmr, and L228gfp:kmr) and a control (no bacterial inoculation). B. napus var Compass seeds were surface sterilized with 95% ethanol and dried aseptically, in a laminar cabinet. The seeds were then coated with an alginate preparation containing the PGP strains essentially as described by Power et al. (2011). Alginate coatings were prepared using a solution of 5% alginate, 10% skimmed milk and bacterial culture, made to a final concentration of 106 CFUs ml-1 to give a concentration of bacteria of approximately 105 to 106 g-1 seed. The alginate solution was dropped over the seeds, using a Pasteur pipette, to coat the seeds and the seeds were added to a 2% calcium chloride solution. The coated solidified seeds were washed twice in sterile water to remove the excess salt and then transferred to a sterile petri dish. The negative control was seed coated with alginate that had no cultures present. Seeds were transferred into plastic horticultural pots containing 500 g of topsoil (clay loam, pH 5.5–6.0, 5% soil organic matter). All soil was mixed prior to the experiment and randomly divided into pots. This ensured nutrient uniformity across the experiment. Plants were watered equally two to three times weekly (typically 50–100 ml of water was added to each pot). There were no added nutrients applied to the pots during the greenhouse experiment.
Table 1
| Strain | Identification | Plant of origin | Isolated from | Origin | Plant growth promotion characteristicsa | Reference |
|---|---|---|---|---|---|---|
| L111∗ | Pseudomonas fluorescens | Miscanthus × giganteus | Leaf | Oak Park, Co. Carlow, Ireland | Sid, PS, ACC | Otieno et al., 2013; Moreira et al., 2016 |
| L117 | Pseudomonas sp. | Miscanthus × giganteus | Leaf | Oak Park, Co. Carlow, Ireland | Sid, BC, PS, ACC | Otieno et al., 2013 |
| L130 | Pseudomonas sp. | Miscanthus × giganteus | Leaf | Oak Park, Co. Carlow, Ireland | BC, PS | Otieno et al., 2013 |
| L132 | Pseudomonas sp. | Miscanthus × giganteus | Leaf | Oak Park, Co. Carlow, Ireland | Sid, BC, PS | Otieno et al., 2013 |
| L228∗ | Pseudomonas fluorescens | Miscanthus × giganteus | Leaf | Oak Park, Co. Carlow, Ireland | Sid, BC, PS | Otieno et al., 2013; Moreira et al., 2016 |
| L321∗ | Pseudomonas fluorescens | Miscanthus × giganteus | Leaf | Oak Park, Co. Carlow, Ireland | PS, ACC | Otieno et al., 2013; Moreira et al., 2016 |
| R324 | Serratia sp. | Miscanthus × giganteus | Rhizosphere | Oak Park, Co. Carlow, Ireland | PS | Otieno et al., 2013 |
| R232 | Enterobacter sp. | Miscanthus × giganteus | Rhizosphere | Oak Park, Co. Carlow, Ireland | PS | Otieno et al., 2013 |
| S120 | Serratia sp. | Miscanthus × giganteus | Stem | Oak Park, Co. Carlow, Ireland | PS | Otieno et al., 2013 |
| Rt03 | Pseudomonas sp. | Brassica napus | Root | Oak Park, Co. Carlow, Ireland | PS | Otieno et al., 2013 |
| F113∗ | Pseudomonas fluorescens | Beta vulgaris | Rhizosphere | Fota, Co. Cork, Ireland | Sid, PS, BC, ACC | Shanahan et al., 1992; Redondo-Nieto et al., 2013 |
List of strains used in this study; origin and plant growth promoting characteristics.
aSid, siderophore production; PS, phosphate solubilization capability; BC, biocontrol activity against Pythium ultimum or Fusarium oxysporum; and ACC, 1-aminocyclopropane-1-carboxylic acid deaminase activity. ∗Strains used as single inoculants. All strains with the exception of F113 were used as the MM consortia application.
Greenhouse growing conditions were as follows: light cycle of 16 h light and 8 h dark phase, with a mean light phase temperature of 30°C and dark phase temperature of 24°C. Per treatment there were 15 pots at the start of the experiment. Three pots were randomly selected for sampling at each time point for CFU analysis (n = 3), there was one seed planted per pot.
Greenhouse plant samples were harvested from each treatment (n = 3) after 2, 5, 13, 18, and 24 weeks to assess the colonization of the strains on the plants. Rhizosphere, root, and surface sterilized leaf samples were assessed to determine the CFUs in each plant compartment. Surface leaf sterilization was carried out to determine the strains endophytic capability by the method described in Germaine et al. (2004). This was carried out by washing the plant sample with sterilized deionized water for 30 s, followed by a wash in 10% sodium hypochlorite for 1 min, then a final wash in 95% ethanol for 20 s. Samples were then rinsed in sterile deionized H2O and 100 μl of the rinse water was spread plated in triplicate on Nutrient Agar “E” (Lab M), to validate the surface sterilization procedure.
Microbial Counts in Root, Rhizosphere, and Leaf Tissue
Colony forming units per gram of sample were determined using a sucrose glutamine agar (SGA) (Lally, 2016) which promotes the production of fluorescent siderophore, supplemented with kanamycin to select for kanamycin resistance. This media was prepared with H2O in 1 l bottles using 20 g sucrose, 2 g glutamine, 1 g K2HPO4, and 15 g Technical Agar No. 1. After autoclaving, 5 ml of 10% MgSO4 was added per liter of media. Kanamycin was added to a final concentration of 50 μg ml-1 and cycloheximide to 32.5 μg ml-1. Plant tissue was homogenized aseptically with a pestle and mortar and bacterial CFUs were enumerated by dilution and plate counts (Miles and Misra, 1938). Homogenized samples (0.1 g per replicate) of rhizosphere, root, and leaves were added to 900 μl of 14 strength Ringer’s solution. Samples were vortexed for 1 min and serially diluted 1:10 until a 10-7 dilution was reached. Aliquots of 20 μl were plated from each dilution onto agar plates in quintuplicate. Plates were then dried in a laminar airflow cabinet, inverted, and then incubated at 30°C. Plates were monitored for 3 days and P. fluorescens CFUs were recorded. A number of colonies were randomly selected to examine for gfp expression under UV light, using an epifluorescence microscope.
Dry Plant Weight Following Greenhouse Harvest
At week 24, all plant samples were harvested from the remaining pots. The plant samples were weighed to determine fresh weight (FW). The samples were cut and dried in foil containers for 48 h at 70°C. The samples were then weighed to determine the dry weight (DW) and moisture content. Plant length was determined by measuring the length of the above ground plant material.
Epifluorescence Microscopy
Epifluorescent microscopy was conducted at the final time point of the experiment. Three plants were used for each treatment (n = 3). Root samples were taken at week 24 and mounted for cryostat sectioning. Samples were submerged in PolyFreeze (Sigma-Aldrich), frozen using a cryostat instrument (Leica, CM 1510 S) at -20°C, and sliced to 3 μm longitudinally. Specimens were mounted on glass slides, stained with acridine orange to aid the visualization of the green fluorescent protein expressing isolates, and covered with glass cover slides. Each sample was examined at ×400 magnification and ×1000 using immersion oil under blue light (395 nm) using a Nikon eclipse 8i epi-fluorescent microscope equipped with a 100-W mercury short arc photo-optic lamp. The image was then exported as a PDF and converted to a tiff image using GIMP image software (Germaine et al., 2009).
Field Trial Setup
The field trial was carried out at Teagasc Crop Research Centre, Oak Park, Carlow, Ireland. The variety of B. napus “Compass” was chosen for the use in this trial. It was sown in Oak Park Carlow (coordinates N 52° 51′ 59.41″, W 6° 55′ 25.306″). The soil was a loam (20% clay, 37% silt, and 43% sand) with 4.8% organic matter. The previous crop was winter barley, following which the straw was baled the land ploughed, power harrowed, drilled, and rolled.
The field trial was sown in September 2012 and was monitored until harvest in August 2013. The trial was set up as a randomized complete block design and included six blocks. Each block contained six individual treatments (F113, L228, L321, L111, “MM,” and a negative control). There were six replicates per treatment in the field investigation. Each block contained one replicate. These treatments were replicated throughout each block using randomized allocation for each treatment. The treatment plots were 4 m in width × 23 m in length, a total plot area of 92 m2, with 0.5 m spacing between each treatment plot and 1 m spacing between each block. The seed was sown at a rate of 60 seeds/m2. Post-germination, the trial received three separate spray applications of the bacteria during the period of early crop development. Crops received normal nutrient fertilizer application of nitrogen, phosphorous, and potassium [240 kg/ha N, 35 kg/ha P, and 65 kg/ha K applied in two applications (30% in late February 70% in late March)]. One spray application of Proline fungicide (Bayer Crop Science Ltd., Cambridge, United Kingdom) was applied as a foliar application to control seasonal disease pressures, it was applied during pod development post flowering 40 weeks into the trial and was applied equally on all blocks. Supplementary Figures 1–3 illustrates the field trial and sampling.
Preparation and Application of Treatments
There were five live inoculant and one control treatments used in the field trial. These were nutrient broth (LabM Limited, Lancashire, United Kingdom) grown inocula of P. fluorescens F113, L228, L321, L111, respectively, and the MM consortium of 10 strains S120, Rt03, R324, L232, L132, L117, L130 (Table 1) which also included L228, L321, and L111. For individual strain treatments 100 μl of stock culture was added to the sterile media prepared in two 1 l Duran bottles (1 l in each) and incubated by shaking at 100 rpm at a temperature of 30°C for 24°h. For MM consortium preparation, 100 μl of each strain was taken from culture stocks and added to 200 ml of sterile media, incubated as above and then pooled into a 2 l volume. Two liters of inoculum was sprayed directly onto the soil in each treatment plot using portable manual pump pressurized sprayers (equivalent to 217 l/ha). The concentration of the inoculum was approximately 106 CFU ml-1 equivalent to an application rate of approximately 2 × 107 CFU m-2. It was applied at this concentration three times; 3 weeks after sowing, 12 weeks after sowing, and 28 weeks after sowing. A control dead inoculum (F113 culture autoclaved at 121°C for 15 min) was applied at the same rate (2 l/plot) to each control plot as a negative control.
Sampling of the Field Trial for Plant Biomass Analysis
The crop was sampled using a set square (0.5 m2). Each plot was split randomly into sub-samples, eight plants per plot were used to measure each parameter. Samples were analyzed by block. Destructive samples from the trial were collected on three separate occasions, early development (3 months), mid-development (7 months), and pre-harvest (9 months). All seed and yield analysis were conducted post-harvest. Seed samples were subsamples of the total harvested seed, collected from the central 2.5 m strip of each plot, this was collected by a mechanical harvester. Bags of seed were stored at room temperature until analyzed. Four plots were removed from the analysis due to weed influx at the border strip of four blocks. Sample size is presented in Table 2.
Table 2
| Treatment | Sample size (n) | Average plant biomass (g) ± standard deviation | P-value | % Greater than the control | Average plant length (cm) ± standard deviation | P-value | % Greater than the control |
|---|---|---|---|---|---|---|---|
| F113 | 6 | 41.53 ± 2.23 | 0.245 | 14.12 | 154.04 ± 6.29 | 0.454 | 3.27 |
| L228 | 5 | 41.59 ± 5.48 | 0.279 | 14.27 | 154.76 ± 10.74 | 0.347 | 3.74 |
| L111 | 6 | 40.37 ± 6.33 | 0.514 | 11.3 | 153.00 ± 3.89 | 0.703 | 2.6 |
| L321 | 5 | 40.61 ± 5.74 | 0.502 | 11.89 | 158.23 ± 4.09∗ | 0.018∗ | 5.99∗ |
| MM | 5 | 45.76 ± 5.66∗∗ | 0.003∗∗ | 23.73∗∗ | 158.28 ± 5.05∗ | 0.017∗ | 5.96∗ |
| Control | 5 | 36.05 ± 3.63 | – | – | 149.07 ± 6.81 | – | – |
Summary statistics from the field trial of B. napus inoculated with plant growth promoting Pseudomonas and a bacterial consortium (MM).
∗P < 0.05; ∗∗P < 0.01.
Dry Weight, Plant Length, and Yield
At the time of the third sampling date the pods were at the late stages of development resulting in the early deciduous phase of the leaf cycle; therefore, no green leaf area measurements were taken at this time point. Plants were gently extracted from the soil to obtain the root (root length refers to the length of the tap root). Plants with incomplete roots were not measured. The plants were individually measured to determine plant root and stem length. The samples were separated into roots, stems/leaves, and pods. Plant characteristics were counted (stems, leaves, and pods), the plants were weighed fresh and then were dried for a minimum of 24 h at 70°C in a forced air oven until DW stabilized. If further drying was required samples were exposed to the same conditions for an additional period of 16–24 h. DW was then recorded.
The thousand seed weights were measured using a Contador Pfeuffer (Pfeuffer Kitzingen, Germany). When 1000 counted seeds had passed into the drop compartment the total weight of the seeds was taken. This was repeated in triplicate for each sample. Total harvested seed was determined by the plot measurement given by the combine harvester at the time of the harvest. Seed oil content was analyzed by adding samples from the seed harvest to a seed oil analyzer (CropScan 1000B NIR, Technology Systems, Australia). Ten random subsamples were removed from the harvest sample and measured for oil and moisture content.
Statistics
Greenhouse and field experiments were carried out once. Greenhouse data were analyzed using univariate analysis with one-way ANOVA and post hoc tested using the Bonferroni test. Field trial data were analyzed as a randomized complete block design using a univariate ANOVA. Significant data were subjected to post hoc testing using Tukey’s HSD test. In both cases, results with a P-value lower than P = 0.05 were considered significant following post hoc testing. All statistical analysis was carried out using SPSS statistical software version 20.0 (IBM, 2012).
Results
Colonization Dynamics of gfp-Tagged P. fluorescens Strains on B. napus (Oilseed Rape) in Greenhouse Experiments
Chromosomal mini-Tn::gfp km-tagged derivatives of P. fluorescens F113, L111, L228, and L321 were used to investigate the colonization dynamics in different plant compartments of B. napus. The Kmr allows the direct selection of the inoculant strain and the gfp marker allows the strains to be visualized in the plant by Epifluorescence Microscopy (see Supplementary Figures 4A,C). Under these experimental conditions, no Kmr CFUs were detected in the uninoculated control treatments (data not shown).
Pseudomonas fluorescens L111, L228, and L321 were found to be effective colonizers of the rhizosphere and they displayed capabilities similar to that of P. fluorescens F113gfp:kmr, a proficient rhizosphere colonizer (Villacieros et al., 2005; Rivilla et al., 2013). The strains were capable of colonizing the rhizosphere at levels of 103–104 CFUs g-1 (Figure 1A) up to 24 weeks post-inoculation. In the rhizosphere at week 2 each strain colonized the rhizosphere at similar levels. At week 5, L111gfp:kmr and L228gfp:kmr had increased in numbers whereas F113gfp:kmr and L321gfp:kmr were seen to decrease from 105 to 103 and 102 CFU g-1, respectively. Each of the strains then stabilized at levels of 103 CFU g-1. L111 maintained this colonization level at week 18, while F113gfp:kmr showed a decline in numbers. L228gfp:kmr and L321gfp:kmr were undetected at week 18 suggesting an issue with the sampling of these replicates as analysis at week 24 indicated that L111gfp:kmr, F113gfp:kmr, and L321gfp:kmr were present in the rhizosphere at concentrations of 104 CFU g-1 and L228gfp:kmr at 103 g-1.
FIGURE 1
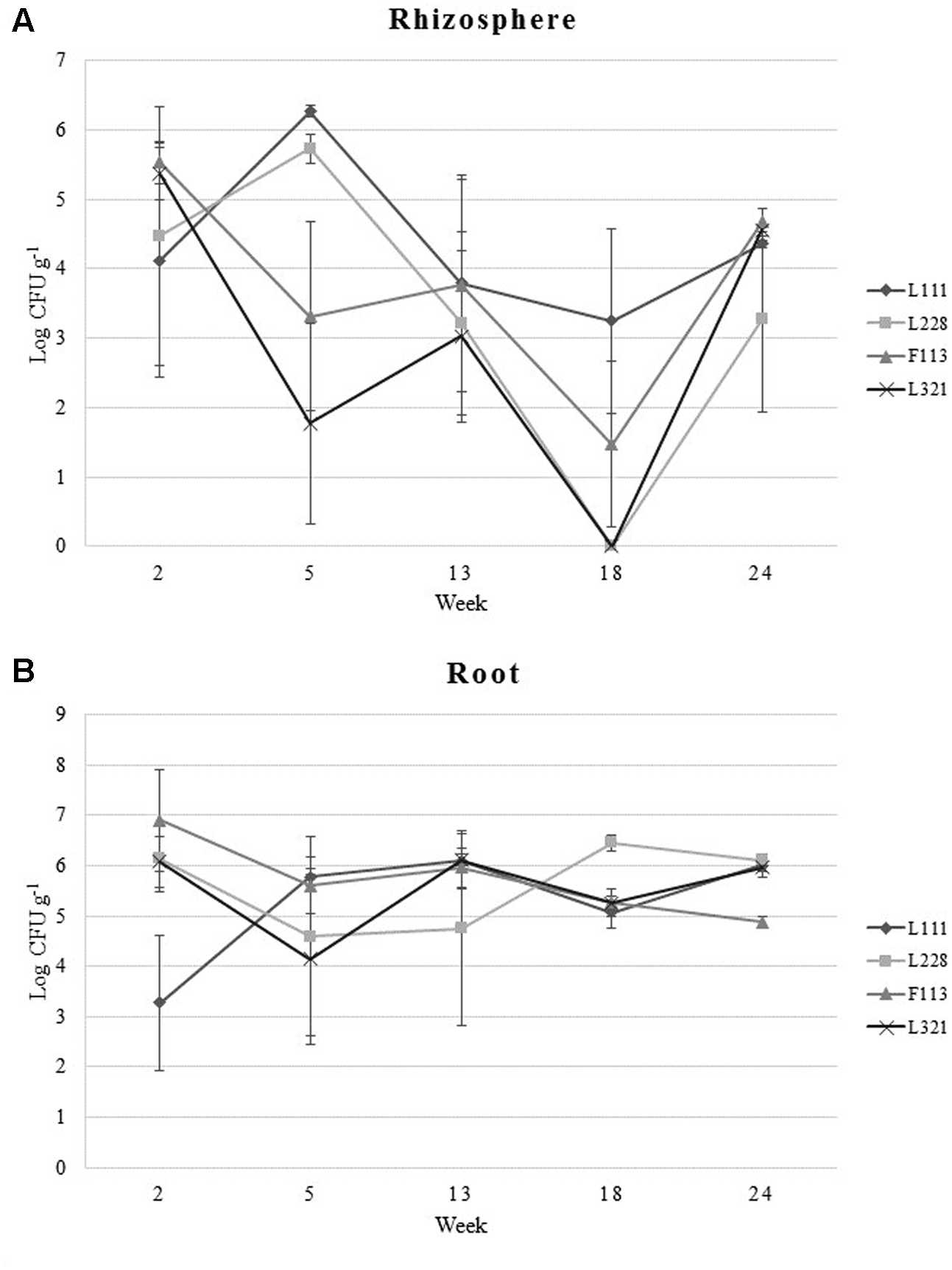
Bacterial colonization of Brassica napus by PGP Pseudomonas and a bacterial consortium during a 24-week greenhouse trial. (A) Bacterial colonization levels of plant rhizosphere. (B) Colonization of plant root samples. Data represent the average number of CFUs at each sample time. Error bars represent standard error (±SE).
Compared with colonization dynamics in the rhizosphere, the root samples had less fluctuations (Figure 1B). At week 2, L111gfp:kmr had the lowest population number at 103 CFU g-1. The other three strains were present at 106 CFU g-1. At week 5 each of the strains maintained uniform population numbers between 104 and 105 CFU g-1. At week 13, L111gfp:kmr and L321gfp:kmr reached numbers as high as 106 CFU g-1 while L228gfp:kmr remained at the same colonization level as week 5. At week 18, L228gfp:kmr numbers increased to 106 CFU g-1 while L111gfp:kmr, F113gfp:kmr, and L321gfp:kmr decreased to 105 CFU g-1. At the final sample date, week 24, L321gfp:kmr, L228gfp:kmr, and L111gfp:kmr colonized the roots at levels of 106 CFU g-1 and F113gfp:kmr colonized at 105 CFU g-1.
Each of the Pseudomonas strains were detected in leaf samples after week 2. Isolate L111gfp:kmr was present at levels as low as 101 CFU g-1. Strains F113gfp:kmr and L321gfp:kmr were detected at 103 CFU g-1 and L228gfp:kmr was observed in the highest numbers at 106 CFU g-1. However, there was no further detection of bacterial colonization following subsequent sampling at weeks 5, 13, 18, and 24 (data not presented), indicating that the strains did not colonize the leaf under these experimental conditions.
Oilseed Rape Inoculated with P. fluorescens Strains Show Significant Increases in Plant Biomass in Greenhouse Experiments
Above ground plant biomass was investigated as an indicator of PGP due to inoculant treatment in the greenhouse experiments. The final date of sampling (week 24) was used to obtain samples for above ground biomass analysis (n = 3). Statistical analysis was carried out using univariate analysis with one-way ANOVA and post hoc tested using the Bonferroni test. There were no differences detected in the moisture levels of the plant samples under the treatment application (data not shown). Supplementary Figures 1–3, 4B show PGP effects due to inoculation with the bacterial treatments.
Fresh weight biomass (above ground) was significantly increased (Figure 2) in each bacterial treatment except for L111 under the Bonferroni test (F113gfp:kmr P = 0.003, L321gfp:kmr P = 0.013, and L228gfp:kmr P = 0.024). L321gfp:kmr showed an average increase of 1.10 g above the control, L228gfp:kmr showed an average of 1.13 g greater than the control and F113gfp:kmr showed 1.39 g greater than the control. DW biomass analysis also displayed significant increases under Bonferroni test in each treatment except the L111gfp:kmr treatment (F113gfp:kmr P = 0.005, L321gfp:kmr P = 0.021, L228gfp:kmr P = 0.018) (Figure 2).
FIGURE 2
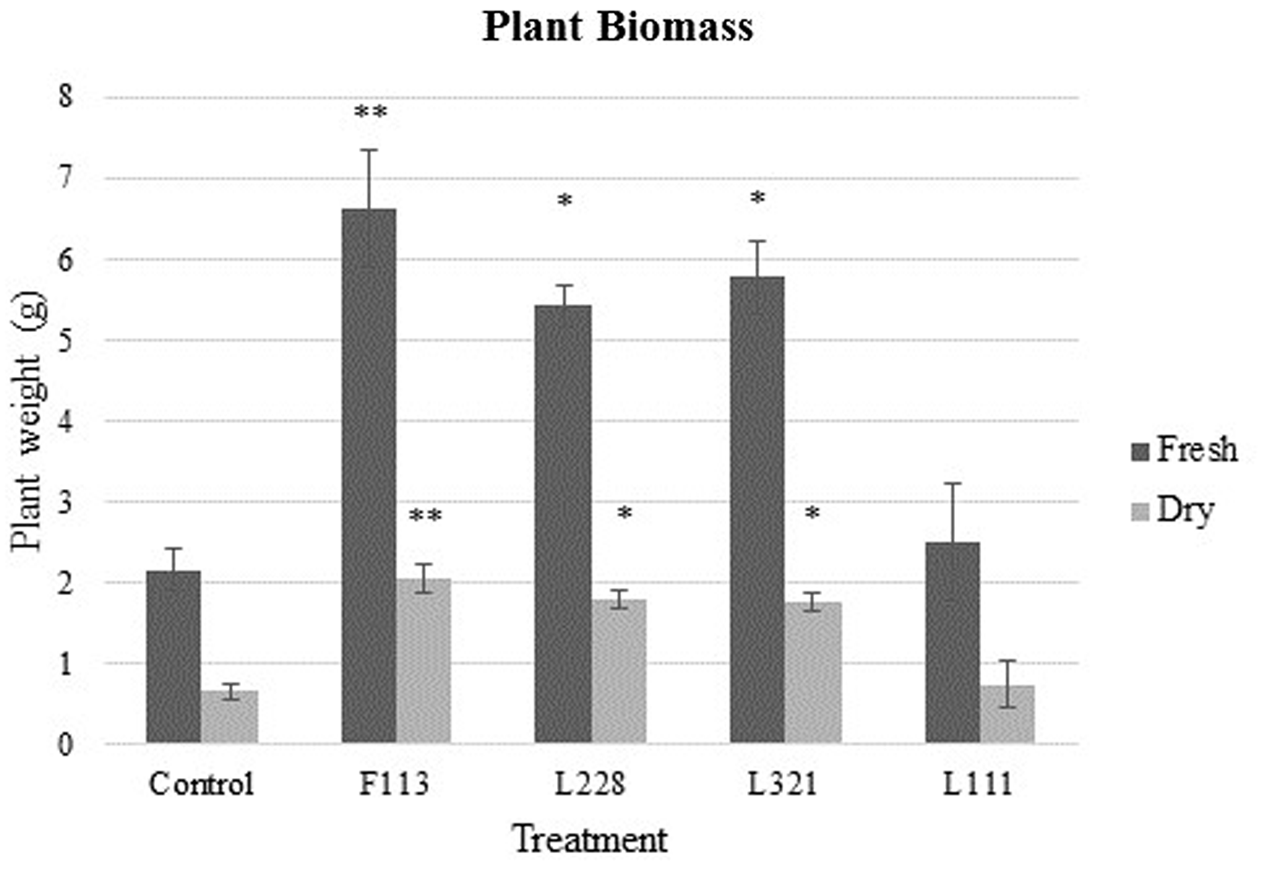
Biomass analysis of B. napus in a greenhouse experiment following inoculation by PGP Pseudomonas and a bacterial consortium after 24 weeks. Data display above ground (shoots/leaves) plant biomass after 24 weeks colonization with the bacterial strains L111, F113, L321, and L228. Bars represent average value for each data series (n = 3). Error bars represent standard error (±SE). ∗∗P < 0.01; ∗P < 0.05.
Oilseed Rape Inoculated with P. fluorescens L321 and a Bacterial Consortium Show Significant Increases in Plant Length and Biomass in the Field
Following spray inoculation in the field trial, samples were taken at three time points early development (3 months), mid-development (7 months), and pre-harvest (9 months), and analyzed for a number of key agronomic parameters. Data were analyzed statistically. For the first two sampling times, the data obtained showed that there were no statistical differences between any of the treatments and the control (data not shown).
The third time point (9 months) showed that all inoculated treatments had an increase in biomass compared to the control (Figure 3); however, only the MM consortium and P. fluorescens L321 were statistically significantly different with respect to the total plant length compared to the control plants. The MM consortium treatment showed a mean difference of 9.20 cm (5.99% increase; P-value = 0.012) and P. fluorescens L321 showed a mean difference of 9.26 cm (5.96% increase; P-value = 0.012) greater than the control (Figure 3). Statistically significant results were also observed with respect to dry plant biomass at the third sample date (pre-harvest stage). The control treatment had statistically significantly less biomass in three aspects of plant biomass: stem/leaf, pod, and total plant biomass compared to the MM consortium treatment (Figure 3). In the MM treatment, stem/leaf biomass had a mean difference of 4.49 g (22.43% increase; P-value = 0.005) greater than the control, the pod weight biomass, a mean difference of 4.93 g (25.11% increase; P-value = 0.008), and a total dry biomass mean difference of 9.70 g (23.97% increase; P-value = 0.003) compared to the control treatment (Table 2). The results gave no indication of negative effects on crop growth as a result of the biofertilizer treatments.
FIGURE 3
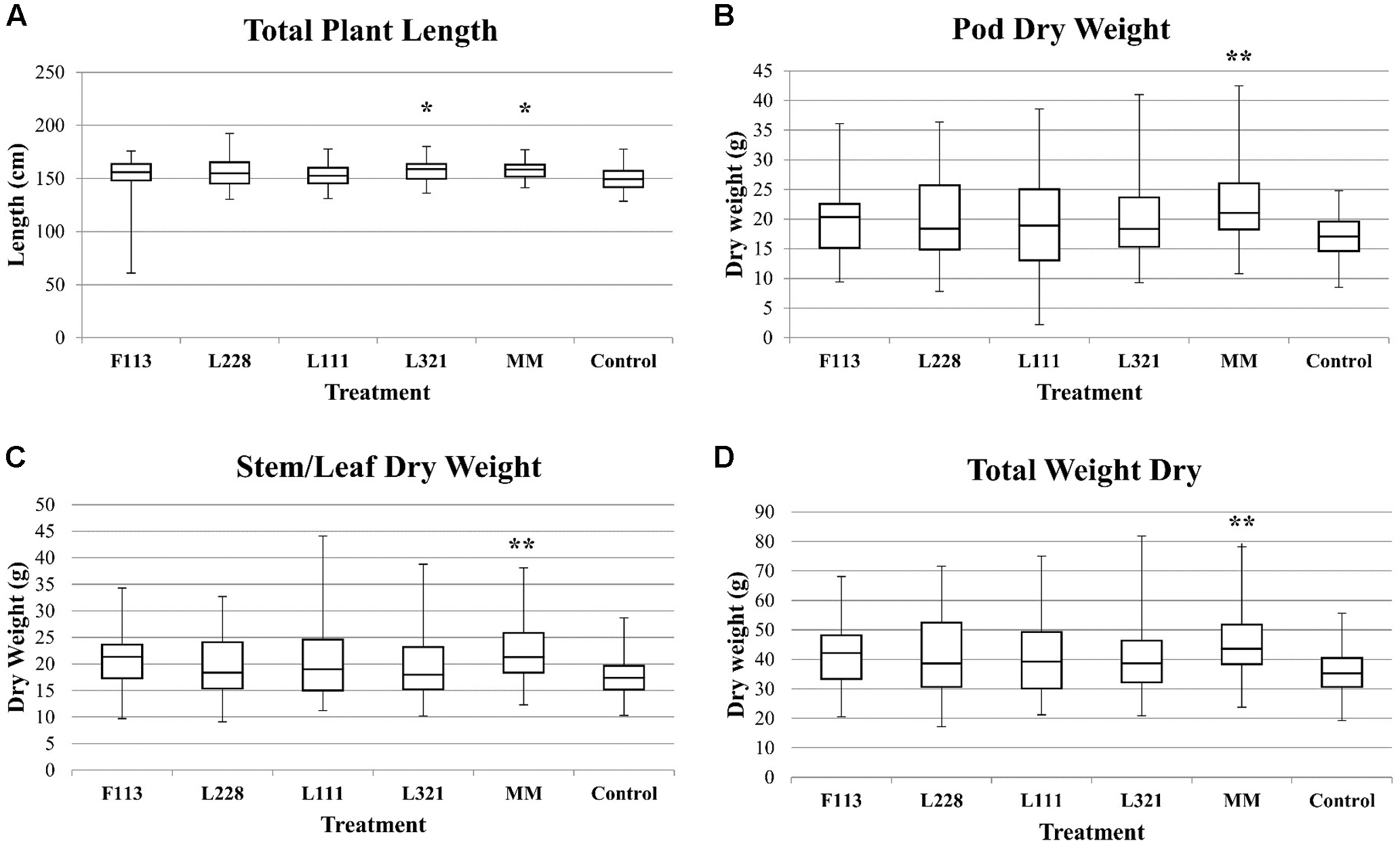
Biomass analysis of B. napus in a field experiment following inoculation by PGP Pseudomonas and a bacterial consortium at 9 months. Box plots provide a summary of the significant field trial data. These graphs represent the variation within the sample populations. (A) The total plant length from the third sample date. (B–D) The DW data from the third sampling date. ∗∗P < 0.01; ∗P < 0.05.
With respect to the (Figure 4) seed weights and oil yield, there was an observable increase in the case of P. fluorescens F113 and L228 compared to other treatments and the non-inoculated control; however, these increases were not statistically significant.
FIGURE 4
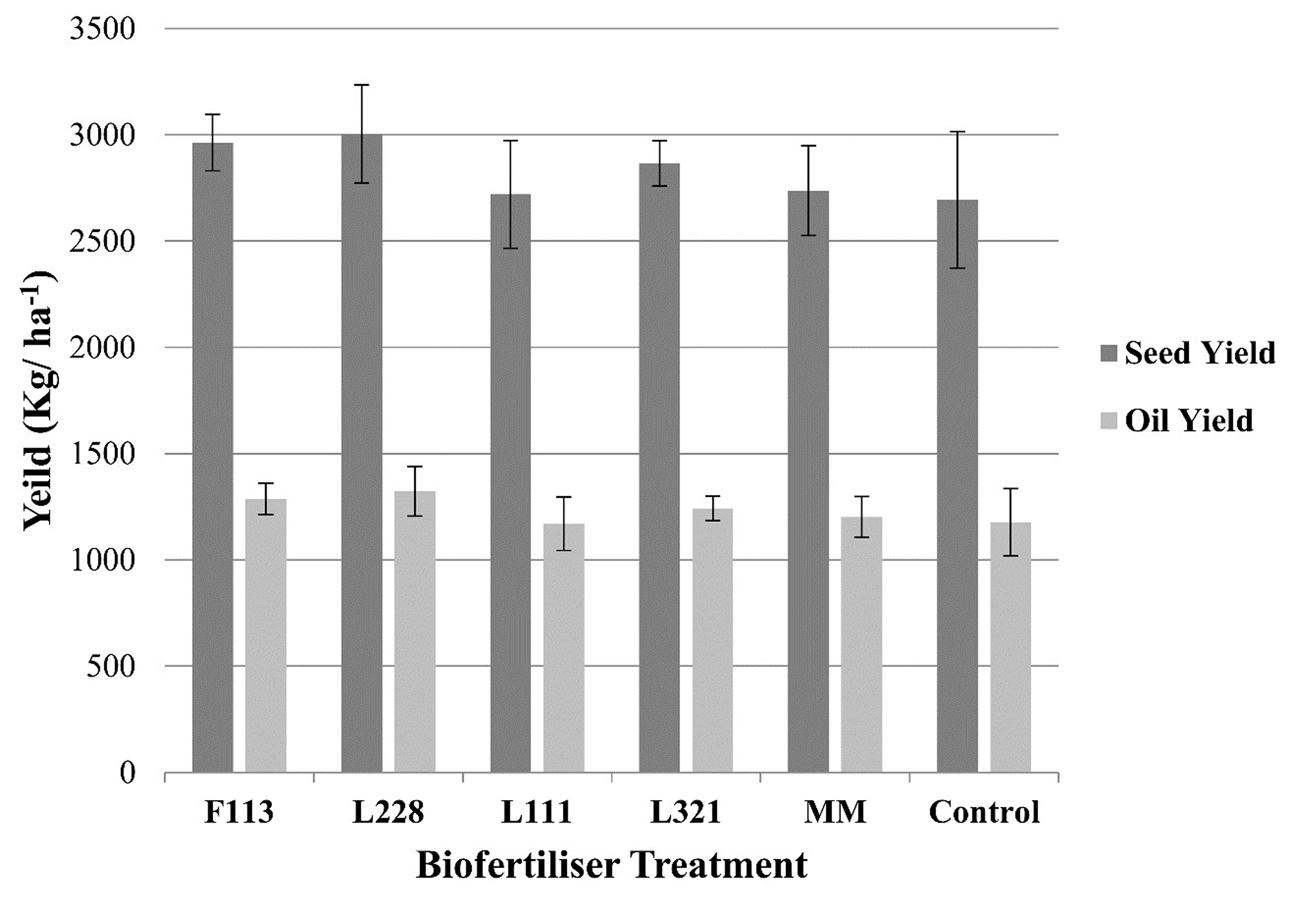
Seed and oil yield output from B. napus in a field experiment following inoculation by PGP Pseudomonas and a bacterial consortium at harvest. Error bars represent the standard error of the mean (±SE). There were no significant differences found in seed or oil yields.
Discussion
In this study, the use of PGP isolates was assessed for their application in sustainable agriculture. Two of the strains P. fluorescens L321 and P. fluorescens L228 were observed to significantly increase plant biomass in a greenhouse experiment and were efficient colonizers of the rhizosphere and root compartment of B. napus, achieving populations as high as the well-characterized rhizobacteria P. fluorescens F113 strain.
Each of the strains used in this study are considered plant growth promoting bacteria based on their phenotypic qualities, having been screened for various PGP mechanisms. Their ability to colonize B. napus was observed in previous greenhouse trials (Otieno et al., 2013). Strains F113, L228, and L321 produce high levels of gluconic acid and demonstrate high levels of inorganic P solubilization compared to L111 (Otieno et al., 2015) when measured in vitro and L321 has previously been shown to increase the growth of pea plants in low phosphate soil (Otieno et al., 2015). This trait may be an important factor in the growth promotion effects observed in the greenhouse study.
Of the five treatments tested in the field trial, two displayed the ability to increase plant growth. Treatments L321 and MM showed the ability to increase plant length by 5.96 and 5.99%, respectively, the MM increased stem/leaf biomass and pod biomass by 23.97% when compared to the control treatment in the trial. In the field experiment two treatments, L321 and the MM, resulted in significantly taller plants, suggesting that direct length promotion may have been due to cell elongation as a result in ACC deaminase activity reducing ethylene levels in the plants and maximizing plant growth (Abeles et al., 1992; Glick et al., 1997; Penrose and Glick, 2001) by ACC deaminase producing strains L111, L117, and L321. This mechanism of PGP usually affects root hair development, resulting in structurally improved rooting systems. In addition there is evidence that strains L111, L228, and L321 can produce the phytohormone indole acetic acid (IAA) (Otieno et al., 2013). As these strains encode genes implicated in the IAA biosynthesis pathway (Moreira et al., 2016) and IAA has been detected by HPLC analysis of culture filtrates (Obermeier and Dowling, Unpublished data). IAA production by these strains may also have contributed to the growth promotion effects observed in both the greenhouse and field experiments.
In the case of the MM treatment multiple strains with a range of PGP traits could improve the chance of exhibiting positive PGP under field conditions. This has been previously suggested as a solution to the inconsistency in results obtained when using single strains as plant growth promoters (Van Veen et al., 1997).
Bacteria exhibiting production of IAA (Idris et al., 2007; Naveed et al., 2014), siderophores (Kloepper et al., 1980; Katiyar and Goel, 2004), phosphate solubilization (Rodríguez and Fraga, 1999; Peix et al., 2001), atmospheric nitrogen fixers (Çakmakç et al., 2001), and ACC deaminase (Mayak et al., 2004; Shaharoona et al., 2006) resulted in plant growth enhancement. In the current study each of the strains used in the MM treatment contained at least three of the PGP traits mentioned above. The data show that the MM consortia treatment was capable of increasing aspects of plant biomass at a field scale, suggesting that multiple mechanisms may have been the key contributors to this finding.
Evidence of the ability of phosphate solubilizing and nitrogen fixing bacterial strains to increase the nutrient uptake and the yield of Zea mays, Triticum aestivum, Cicer arietinum, Glycine max, Lactuca sativa, Oryza sativa, and Cucumis sativus has been reported (Adesemoye and Kloepper, 2009; Adesemoye and Egamberdieva, 2013). However, evidence to support the positive effect of microbial biofertilizers on crop yield increase in oilseeds such as B. napus is not widely available. However, Kumar et al. (2009) report a significant oil yield increase in a trial of Sesamum indicum after inoculation with Pseudomonas aeruginosa LES4 and growth promotion of Maize (Z. mays L) following inoculation with Azospirillum and Azotobacter PGPR has also been reported under field conditions (Gholami et al., 2012).
The result of the field trial showed that while, on average, there was an increase in the seed yield due to plant inoculation with these strains (L321:2.65, L228:3.05, and F113:2.65 t/ha) there was no statistically significant difference compared to the seed yields from the control plots (control: 2.45 t/ha). Although these increased yields were not statistically significant, they do in fact represent a significant economic increase for the farmer. In 2016, the market price of rape seed was €357/t (IFA, 2016). The increased yields due to the biofertilizer application would correspond to an average increase of €71.4/ha (with strain L321), €178/ha (with strain F113), and €200/ha (with strain L228). With the cost of application of these biofertilizers ranging from €50 to 100/ha, our results show that the use of these microbes could result in economic benefits for the farmer.
The applications of chemical fertilizers in this field study may have masked our ability to observe significant growth impacts on crop yields. Previous investigations suggest that strategies combining both reduced rates of agriculture fertilizers and biofertilizers can benefit plant development and nutrient uptake (Adesemoye et al., 2010; Duarah et al., 2011; Mäder et al., 2011). PGP Pseudomonas strains have been shown to have the ability to induce disease resistance in pearl millet as well as increase biomass under greenhouse and laboratory conditions (Jogaiah et al., 2010). In a study by Babu et al. (2015), rhizobacteria were reported to improve growth and fruit weight in tomato as well as protection against early blight disease (Alternaria solani). Some of our strains have biocontrol properties in vitro but they were not evaluated for biocontrol activity as part of this study, however, disease pressure was not observed during the timeline of the field experiment (note a commercial fungicide was applied at week 40).
Conclusion
Greenhouse and field trials were carried out using individual endophytic P. fluorescens strains and an endophytic microbial consortium. In greenhouse experiments, individual strains tagged with gfp and Kmr were shown to effectively colonize the rhizosphere and root of B. napus (but not the leaves) and they demonstrated a significant increase in plant biomass compared to the non-inoculated control. In the field experiment analysis, the data provide evidence that the application of the live bacterial biofertilizers could enhance aspects of crop development in B. napus at field scale. The field data demonstrated statistically significant increases in crop height, stem/leaf, and pod biomass, particularly, in the case of the consortium inoculated treatment. Due to the agronomic conditions pertaining to the experiment (normal fertilizer, pesticide, and fungicide application) the most likely traits implicated in the observed effect were ACC deaminase activity and IAA synthesis. Seed and oil yield were increased in the field in response to inoculation, and although these data were not statistically significant, a real economic value to the grower could be envisaged.
Future work will address the optimization of their application with reduced synthetic fertilizer use with a view to the maximization of crop growth and yield potential. Overall focus, based on this work, should be directed at combining PGP strains with efficient plant colonization ability and that express a range of PGP traits, for use as consortia applications on globally important crops.
Dedication
We would like to dedicate this work to the family and friends of Dr. Paul Galbally who sadly passed away during the preparation of this paper.
Statements
Author contributions
DR, KG, JS, and DD conceived of and designed the study. RL, PG, and AM designed and performed the greenhouse and field experiments. RL, KG, and DD wrote the paper.
Acknowledgments
The authors would like to acknowledge the Science Foundation Ireland Technology Innovation Development Award (SFI, 11/TIDA/B2018), Institutes of Technology Ireland (IoTI) and the Institute of Technology Carlow (ITC), and Irish Research Council (IRC) postgraduate scholarship (to RL) funding to support this research in part.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fpls.2017.02193/full#supplementary-material
References
1
Abeles F. B. Morgan P. W. Saltveit M. E. (1992). Ethylene in Plant Biology.New York, NY: Academic Press.
2
Adesemoye A. O. Egamberdieva D. (2013). “Beneficial effects of plant growth-promoting rhizobacteria on improved crop production: prospects for developing economies,” inBacteria in Agrobiology: Crop Productivity, edsMaheshwariD. K.AeronM.SarafA. (Berlin: Springer-Verlag), 45–63. 10.1007/978-3-642-37241-4_2
3
Adesemoye A. O. Kloepper J. W. (2009). Plant-microbes interactions in enhanced fertilizer-use efficiency.Appl. Microbiol. Biotechnol.51–12. 10.1007/s00253-009-2196-0
4
Adesemoye A. O. Torbert H. A. Kloepper J. W. (2010). Increased plant uptake of nitrogen from 15N-depleted fertilizer using plant growth-promoting rhizobacteria.Appl. Soil Ecol.4654–58. 10.1016/j.apsoil.2010.06.010
5
Angus J. Herwaarden A. Howe G. Van H. A. (1991). Productivity and break crop effects of winter-growing oilseeds.Aust. J. Exp. Agric.31669–677. 10.1071/EA9910669
6
Babalola O. O. (2010). Beneficial bacteria of agricultural importance.Biotechnol. Lett.321559–1570. 10.1007/s10529-010-0347-0
7
Babu A. N. Jogaiah S. Ito S.-I. Nagaraj A. K. Tran L.-S. P. (2015). Improvement of growth, fruit weight and early blight disease protection of tomato plants by rhizosphere bacteria is correlated with their beneficial traits and induced biosynthesis of antioxidant peroxidase and polyphenol oxidase.Plant Sci.23162–73. 10.1016/j.plantsci.2014.11.006
8
Brennan E. (2010). Purification and Kinetic Analysis of a Specific Glutathione Transferase (BphKLB400) Involved in the Detoxification of PCBs.Ph.D. thesis, Institute of Technology Carlow, Carlow.
9
Çakmakç R. Kantar F. A. Sahin F. (2001). Effect of N2-fixing bacterial inoculations on yield of sugar beet and barley.J. Plant Nutr. Soil Sci.164527–531. 10.1002/1522-2624(200110)164:5<527::AID-JPLN527>3.0.CO;2-1
10
Camargo J. A. Alonso A. (2006). Ecological and toxicological effects of inorganic nitrogen pollution in aquatic ecosystems: a global assessment.Environ. Int.32831–849. 10.1016/j.envint.2006.05.002
11
Compant S. Clément C. Sessitsch A. (2010). Plant growth-promoting bacteria in the rhizo-and endosphere of plants: their role, colonization, mechanisms involved and prospects for utilization.Soil Biol. Biochem.42669–678. 10.1016/j.soilbio.2009.11.024
12
Cory J. S. Franklin M. T. (2012). Evolution and the microbial control of insects.Evol. Appl.5455–469. 10.1111/j.1752-4571.2012.00269.x
13
Duarah I. Deka M. Saikia N. Deka Boruah H. P. (2011). Phosphate solubilizers enhance NPK fertilizer use efficiency in rice and legume cultivation.3 Biotech1227–238. 10.1007/s13205-011-0028-2
14
Franche C. Lindström K. Elmerich C. (2009). Nitrogen-fixing bacteria associated with leguminous and non-leguminous plants.Plant Soil32135–59.
15
Fuentes-Ramirez L. E. A. Caballero-Mellado J. (2005). “Bacterial biofertilizers,” inPGPR: Biocontrol and Biofertilization, ed.SiddiquiZ. A. (Dordrecht: Springer), 143–172.
16
Garrido-Sanz D. Meier-Kolthoff J. P. Göker M. Martín M. Rivilla R. Redondo-Nieto M. (2016). Genomic and genetic diversity within the Pseudomonas fluorescens complex.PLOS ONE11:e0150183. 10.1371/journal.pone.0150183
17
Geiger F. Bengtsson J. Berendse F. Weisser W. W. Emmerson M. Morales M. B. et al (2010). Persistent negative effects of pesticides on biodiversity and biological control potential on European farmland.Basic Appl. Ecol.1197–105. 10.1016/j.baae.2009.12.001
18
Germaine K. J. Chhabra S. Song B. Brazil D. Dowling D. N. (2010). Microbes and sustainable production of biofuel crops: a nitrogen perspective.Biofuels1877–888. 10.4155/bfs.10.67
19
Germaine K. J. Keogh E. Garcia-Cabellos G. Borremans B. van der Lelie D. Barac T. et al (2004). Colonisation of poplar trees by gfp expressing bacterial endophytes.FEMS Microbiol. Ecol.48109–118. 10.1016/j.femsec.2003.12.009
20
Germaine K. J. Keogh E. Ryan D. Dowling D. N. (2009). Bacterial endophyte-mediated naphthalene phytoprotection and phytoremediation.FEMS Microbiol. Lett.296226–234. 10.1111/j.1574-6968.2009.01637.x
21
Gholami A. Biyari A. Gholipoor M. Rahmani H. A. (2012). Growth promotion of maize (Zea mays L.) by plant-growth-promoting rhizobacteria under field conditions.Commun. Soil Sci. Plant Anal.431263–1272. 10.1080/00103624.2012.666302
22
Glick B. R. (2012). Plant growth-promoting bacteria: mechanisms and applications.Scientifica2012:963401. 10.6064/2012/963401
23
Glick B. R. Penrose D. M. Li J. (1997). A model for the lowering of plant ethylene concentrations by plant growth-promoting bacteria.J. Theor. Biol.19063–68. 10.1006/jtbi.1997.0532
24
Godfray H. C. Beddington J. R. Crute I. R. Haddad L. Lawrence D. Muir J. F. et al (2010). Food security: the challenge of feeding 9 billion people.Science327812–818. 10.1126/science.1185383
25
Hayat R. Ali S. Amara U. Khalid R. Ahmed I. (2010). Soil beneficial bacteria and their role in plant growth promotion: a review.Ann. Microbiol.60579–598. 10.1007/s13213-010-0117-1
26
Hill J. Nelson E. Tilman D. Polasky S. Tiffany D. (2006). Environmental, economic, and energetic costs and benefits of biodiesel and ethanol biofuels.Proc. Natl. Acad. Sci. U.S.A.10311206–11210. 10.1073/pnas.0604600103
27
Hilton S. Bennett A. J. Keane G. Bending G. D. Chandler D. Stobart R. et al (2013). Impact of shortened crop rotation of oilseed rape on soil and rhizosphere microbial diversity in relation to yield decline.PLOS ONE8:e59859. 10.1371/journal.pone.0059859
28
IBM (2012). IBM SPSS Statistics for Windows, Version 21.0.Armonk, NY: IBM.
29
Idris E. E. Iglesias D. J. Talon M. A. Borriss R. (2007). Tryptophan-dependent production of indole-3-acetic acid (IAA) affects level of plant growth promotion by Bacillus amyloliquefaciens FZB42.Am. Phytopathol. Soc.20619–626.
30
IFA (2016). Market Reports. Available at: http://www.ifarm.ie/grain/inputs-market-commentary/[accessed December 8, 2016].
31
Jogaiah S. Shivanna R. K. Gnanaprakash P. H. Hunthrike S. S. (2010). Evaluation of plant growth-promoting Rhizobacteria for their efficiency to promote growth and systemic resistance in pearl millet against Downey mildew disease.Arch. Phytopathol. Plant Prot.43368–378. 10.1080/03235400701806377
32
Katiyar V. A. Goel R. (2004). Siderophore mediated plant growth promotion at low temperature by mutant of fluorescent pseudomonad.Plant Growth Regul.42239–244. 10.1023/B:GROW.0000026477.10681.d2
33
Kloepper J. W. Leong J. Teintze M. Schroth M. N. (1980). Enhanced plant growth by siderophores produced by plant growth-promoting rhizobacteria.Nature286885–886. 10.1038/286885a0
34
Kumar S. Pandey P. Maheshwari D. K. (2009). Reduction in dose of chemical fertilizers and growth enhancement of sesame (Sesamum indicum L.) with application of rhizospheric competent Pseudomonas aeruginosa LES4.Eur. J. Soil Biol.45334–340. 10.1016/j.ejsobi.2009.04.002
35
Lally R. (2016). Investigating the Application, Plant Growth Promotion and Genetic Potential of Three Endophytic Pseudomonas fluorescens Isolates for Use in Sustainable Agriculture.Ph.D. thesis, Institute of Technology Carlow, Carlow.
36
Mäder P. Kaiser F. Adholeya A. Singh R. Uppal H. S. Sharma A. K. et al (2011). Inoculation of root microorganisms for sustainable wheat–rice and wheat–black gram rotations in India.Soil Biol. Biochem.43609–619. 10.1016/j.soilbio.2010.11.031
37
Mayak S. Tirosh T. Glick B. R. (2004). Plant growth-promoting bacteria confer resistance in tomato plants to salt stress.Plant Physiol. Biochem.42565–572. 10.1016/j.plaphy.2004.05.009
38
Mercado-Blanco J. A. Lugtenberg B. J. J. (2014). Biotechnological applications of bacterial endophytes.Curr. Biotechnol.360–75. 10.2174/22115501113026660038
39
Miles A. A. Misra S. S. (1938). The estimation of the bactericidal power of the blood.J. Hyg.38732–749. 10.1017/S002217240001158X
40
Moreira A. S. Germaine K. J. Lloyd A. Lally R. D. Galbally P. T. Ryan D. et al (2016). Draft genome sequence of three endophyte strains of Pseudomonas fluorescens isolated from Miscanthus giganteus.Genome Announc.4:e00965-16. 10.1128/genomeA.00965-16
41
Naveed M. Qureshi M. A. Zahir Z. A. Hussain M. B. Sessitsch A. Mitter B. (2014). L-Tryptophan-dependent biosynthesis of indole-3-acetic acid (IAA) improves plant growth and colonization of maize by Burkholderia phytofirmans PsJN.Ann. Microbiol.20619–626.
42
Oldeman L. R. (1992). Global Extent of Soil Degradation.ISRIC Bi-Annual Report 18-36. Wageningen: International Soil Reference and Information Centre.
43
Otieno N. Culhane J. Germaine K. J. Brazil D. Ryan D. A. Dowling D. N. (2013). Screening of large collections of plant associated bacteria for effective plant growth promotion and colonisation.Asp. Appl. Biol.12023–28.
44
Otieno N. Lally R. D. Kiwanuka S. Lloyd A. Ryan D. Germaine K. J. et al (2015). Plant growth promotion induced by phosphate solubilizing endophytic Pseudomonas isolates.Front. Microbiol.6:745. 10.3389/fmicb.2015.00745
45
Peix A. Rivas-Boyero A. Mateos P. F. Rodriguez-Barrueco C. Martłnez-Molina E. Velazquez E. (2001). Growth promotion of chickpea and barley by a phosphate solubilizing strain of Mesorhizobium mediterraneum under growth chamber conditions.Soil Biol. Biochem.33103–110. 10.1016/S0038-0717(00)00120-6
46
Penrose D. M. Glick B. R. (2001). Levels of ACC and related compounds in exudate and extracts of canola seeds treated with ACC deaminase-containing plant growth-promoting bacteria.Can. J. Microbiol.47368–372. 10.1139/w01-014
47
Power B. Liu X. Germaine K. J. Ryan D. Brazil D. Dowling D. N. (2011). Alginate beads as a storage, delivery and containment system for genetically modified PCB degrader and PCB biosensor derivatives of Pseudomonas fluorescens F113.J. Appl. Microbiol.1101351–1358. 10.1111/j.1365-2672.2011.04993.x
48
Redondo-Nieto R. Barret M. Morrisey J. Germaine K. Martínez-Granero F. Barahona E. et al (2013). Genome sequence reveals that Pseudomonas fluorescens F113 possesses a large and diverse array of systems for rhizosphere function and host interaction.BMC Genomics14:54. 10.1186/1471-2164-14-54
49
Rivilla R. Martínez-Granero F. Martín M. (2013). “Motility, biofilm formation and rhizosphere colonization by Pseudomonas fluorescens F113,” inMolecular Microbial Ecology of the Rhizosphere, Vol. 1 and 2ed.deBruijnF. J. (Hoboken, NJ: John Wiley & Sons Inc.), 723–732. 10.1002/9781118297674.ch68
50
Rodríguez H. Fraga R. (1999). Phosphate solubilizing bacteria and their role in plant growth promotion.Biotechnol. Adv.17319–339. 10.1016/S0734-9750(99)00014-2
51
Ryan R. P. Germaine K. Franks A. Ryan D. J. Dowling D. N. (2008). Bacterial endophytes: recent developments and applications.FEMS Microbiol. Lett.2781–9. 10.1111/j.1574-6968.2007.00918.x
52
Saharan B. S. A. Nehra V. (2011). Plant growth promoting rhizobacteria: a critical review.Life Sci. Med. Res.211–30.
53
Shaharoona B. Arshad M. Zahir Z. A. (2006). Effect of plant growth promoting rhizobacteria containing ACC-deaminase on maize (Zea mays L.) growth under axenic conditions and on nodulation in mung bean (Vigna radiata L.).Lett. Appl. Microbiol.42155–159. 10.1111/j.1472-765X.2005.01827.x
54
Shanahan P. O’Sullivan D. J. Simpson P. Gennon J. D. O’Gara F. (1992). Isolation of 2,4-diacetylphloroglucinol from a fluorescent pseudomonad and investigation of the ecological parameters influencing its production.Appl. Environ. Microbiol.58353–358.
55
Sivasakthi S. Usharani G. A. Saranraj P. (2014). Biocontrol potentiality of plant growth promoting bacteria (PGPR) - Pseudomonas fluorescens and Bacillus subtilis: a review.Afr. J. Agric. Res.91265–1277.
56
Tikhonovich I. A. Provorov N. A. (2011). Microbiology is the basis of sustainable agriculture: an opinion.Ann. Appl. Biol.159155–168. 10.1111/j.1744-7348.2011.00489.x
57
Tilman D. Balzer C. Hill J. A. Beforta B. L. (2011). Global food demand and the sustainable intensification of agriculture.Proc. Natl. Acad. Sci. U.S.A.10820260–20264. 10.1073/pnas.1116437108
58
USDA-FAS (2015). USDA-FAS Oilseeds: World Markets and Trade. Available at: apps.fas.usda.gov/psdonline/circular/oilseds.pdf [accessed September 2017].
59
Van Veen J. A. Van Overbeek L. S. A. Van Elsas J. D. (1997). Fate and activity of microorganisms introduced into soil.Microbiol. Mol. Biol. Rev.61121–135.
60
Villacieros M. Whelan C. Mackova M. Molgaard J. Sánchez-Contreras M. Lloret J. et al (2005). Polychlorinated biphenyl rhizoremediation by Pseudomonas fluorescens F113 derivatives, using a Sinorhizobium meliloti nod system to drive bph gene expression.Appl. Environ. Microbiol.712687–2694. 10.1128/AEM.71.5.2687-2694.2005
61
Xi C. Lambrecht M. Vanderleyden J. Michiels J. (1999). Bi-functional gfp-and gusA-containing mini-Tn5 transposon derivatives for combined gene expression and bacterial localization studies.J. Microbiol. Methods355–92. 10.1016/S0167-7012(98)00103-1
Summary
Keywords
endophytes, oilseed rape, PGPR bacteria, biomass, yield, field experiments
Citation
Lally RD, Galbally P, Moreira AS, Spink J, Ryan D, Germaine KJ and Dowling DN (2017) Application of Endophytic Pseudomonas fluorescens and a Bacterial Consortium to Brassica napus Can Increase Plant Height and Biomass under Greenhouse and Field Conditions. Front. Plant Sci. 8:2193. doi: 10.3389/fpls.2017.02193
Received
02 August 2017
Accepted
12 December 2017
Published
22 December 2017
Volume
8 - 2017
Edited by
Essaid Ait Barka, University of Reims Champagne-Ardenne, France
Reviewed by
Sudisha Jogaiah, Karnatak University, India; Samina Mehnaz, Forman Christian College, Pakistan
Updates
Copyright
© 2017 Lally, Galbally, Moreira, Spink, Ryan, Germaine and Dowling.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: David N. Dowling, david.dowling@itcarlow.ie; dowlingd@itcarlow.ie
This article was submitted to Plant Microbe Interactions, a section of the journal Frontiers in Plant Science
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.