- 1Key Laboratory for Plant Diversity and Biogeography of East Asia, Kunming Institute of Botany, Chinese Academy of Sciences, Yunnan, China
- 2Germplasm Bank of Wild Species, Kunming Institute of Botany, Chinese Academy of Sciences, Yunnan, China
- 3Kunming College of Life Sciences, University of Chinese Academy of Sciences, Beijing, China
- 4Institute for Evolution and Biodiversity, University of Münster, Münster, Germany
The subfamily Cercidoideae is an early-branching legume lineage, which consists of 13 genera distributed in the tropical and warm temperate Northern Hemisphere. A previous study detected two plastid genomic variations in this subfamily, but the limited taxon sampling left the overall plastid genome (plastome) diversification across the subfamily unaddressed, and phylogenetic relationships within this clade remained unresolved. Here, we assembled eight plastomes from seven Cercidoideae genera and conducted phylogenomic-comparative analyses in a broad evolutionary framework across legumes. The plastomes of Cercidoideae all exhibited a typical quadripartite structure with a conserved gene content typical of most angiosperm plastomes. Plastome size ranged from 151,705 to 165,416 bp, mainly due to the expansion and contraction of inverted repeat (IR) regions. The order of genes varied due to the occurrence of several inversions. In Tylosema species, a plastome with a 29-bp IR-mediated inversion was found to coexist with a canonical-type plastome, and the abundance of the two arrangements of isomeric molecules differed between individuals. Complete plastome data were much more efficient at resolving intergeneric relationships of Cercidoideae than the previously used selection of only a few plastid or nuclear loci. In sum, our study revealed novel insights into the structural diversification of plastomes in an early-branching legume lineage, and, thus, into the evolutionary trajectories of legume plastomes in general.
Introduction
Chloroplast genomes (plastomes) of photosynthetic angiosperms usually are highly conserved regarding their overall gene content (115–160 genes) and order and GC content (34–40%). They often present a quadripartite structure that consists of a pair of large inverted repeats (IRs; usually around 25 kb, but can vary from 7 to 88 kb) separated by large and small single copy regions (LSC of ca. 80 kb length and SSC of ca. 20 kb, respectively) (Jansen and Ruhlman, 2012; Ruhlman and Jansen, 2014). The large IRs of the plastome are hypothesized to contribute to plastome stabilization, because their absence often coincides with additional severe changes of gene order (Palmer and Thompson, 1982), although causation remains unclear. With the advent of next-generation sequencing, complete plastome sequencing has increased dramatically. We are becoming more and more aware of an increasing number of plastome rearrangements also in photosynthetic angiosperms that retain a quadripartite structure, like in Campanulaceae (Cosner et al., 2004; Haberle et al., 2008), Geraniaceae (Palmer et al., 1987; Chumley et al., 2006; Guisinger et al., 2011; Weng et al., 2014), or Oleaceae (Lee et al., 2007).
The legume family (Fabaceae) is notable for its departures from the typical plastome structure, of which several rearrangements are of phylogenetic relevance. Plastome size in legumes varies greatly because of either expansion, contraction or loss of the IR. Smaller plastomes characterize species of the inverted repeat-lacking clade (IRLC), which have lost the IR (Wojciechowski et al., 2000). In contrast, larger plastomes are typical of species in the inverted repeat-expanding clade (IREC) that have IRs expanding into the SSC by ca. 13 kb (Dugas et al., 2015; Wang et al., 2017a). The loss of two housekeeping genes, namely the translation initiation factor (infA) and the ribosomal protein L22 (rpl22), is shared among all legumes (Gantt et al., 1991; Magee et al., 2010). Other genes, such as accD, clpP, psaI, rpl33, rps16, and ycf4, have been functionally lost in various legume lineages (Keller et al., 2017). In addition, group IIA-introns have been lost from clpP, rpl2, and rps12 in various legume lineages (Doyle et al., 1995; Jansen et al., 2008; Dugas et al., 2015; Wang et al., 2017a). Many of these unusual plastome features of legumes known so far are restricted to papilionoids and mimosoids, and modifications of the “normal” angiosperm plastome structure (a unique 7.5-kb inversion and 5-kb IR expansion into SSC) have been reported only in Tylosema esculentum of Cercidoideae (Kim and Cullis, 2017).
Plastome inversions are common in papilionoids. Except for a few of early diverging lineages of papilionoids, members of this subfamily typically share a 50-kb inversion in the LSC (Doyle et al., 1996). A 78-kb inversion characterizes the subtribe Phaseolinae of Phaseoleae (Bruneau et al., 1990), whereas in Robinia a 39-kb inversion is known (Schwarz et al., 2015). Inversions of 23, 24, or 36 kb have been reported in different taxa of the Genistoid clade (Martin et al., 2014; Choi and Choi, 2017; Feng et al., 2017; Keller et al., 2017), and multiple inversions have been detected in IRLC-legumes (Milligan et al., 1989; Cai et al., 2008; Sabir et al., 2014; Sveinsson and Cronk, 2014; Lei et al., 2016). However, only two inversions have been reported in other legumes, including the aforementioned 7.5-kb inversion from T. esculentum (Kim and Cullis, 2017) and a 421-bp inversion from a mimosoid species (Wang et al., 2017a). The 36-kb and 39-kb inversions of some genistoids and Robinia mentioned above, respectively, are both located between a pair of 29-bp short inverted repeats situated in the 3′-ends of two trnS genes. Inverted repeats are thought to contribute to inversions by mediating intramolecular recombination that may result in the formation of isomers. The most typical example of such isomers is illustrated by the relative orientation of single copy (SC) regions existing in a single plant as demonstrated by Palmer (1983). Besides, Stein et al. (1986) predicted a universal existence of isomeric plastomes in all plastomes with typical IRs. Some isomeric plastomes caused by small inverted repeats other than the primary IRs have been reported in several conifers (Tsumura et al., 2000; Wu et al., 2011; Yi et al., 2013; Guo et al., 2014; Qu et al., 2017b). However, it remains unknown whether the 29-bp IRs in legume plastomes could also mediate isomers.
Cercidoideae is one of six recently recognized subfamilies of Fabaceae (LPWG, 2017) and probably represents the first-branching legume lineage (Bruneau et al., 2001; Herendeen et al., 2003). This subfamily consists of ca. 335 species in 13 genera that are distributed in tropical and warm temperate regions of the Northern Hemisphere (Clark et al., 2017; LPWG, 2017). Some of its species are of economic value to humans. For instance, several species of Barklya, Bauhinia, Cercis, Griffonia, Phanera, Piliostigma, and Tylosema are valued for the production of foods, timbers, dyes, or ropes, or they find application as medicinal and ornamental plants, or even as coffee substitutes (Lewis et al., 2005). Phylogenetically, intergeneric relationships of Cercidoideae remain unresolved in previous phylogenetic studies (Bruneau et al., 2008; Sinou et al., 2009; LPWG, 2013, 2017). Clarifying relationships in Cercidoideae will facilitate many aspects of studies on this economically important group, and contribute to elucidating the evolutionary trajectory of plastid genome evolution in legumes in general. Given plastome variations having been found in only four published species, it is likely that more divergent plastomes hide in Cercidoideae.
Here, we present an analysis of eight newly sequenced plastomes of Cercidoideae. We complement our dataset with four more species from three genera of this subfamily and 45 other legumes to reconstruct the phylogeny of Cercidoideae based on 77 protein-coding genes, 136 intergenic spacers, and 19 introns. Our comparative plastome analysis involving examinations of IR boundary shifts, inversions and locally collinear blocks (LCB), and the existence of isomeric plastomes uncovers unique plastome features and illustrates the variation of plastomes in this clade. Finally, a critical review of plastome structures across the legume family sheds further light on the mechanisms of plastome evolution in Fabaceae.
Materials and Methods
Plant Sampling
For the plastome analysis, we sampled fresh or silica gel-dried leaves of eight species representing seven genera of the subfamily Cercidoideae. Of these, plastomes of genera Barklya, Bauhinia, Griffonia, Lysiphyllum, Piliostigma, and Schnella were sequenced for the first time. To verify the existence of isomeric plastomes, we isolated total genomic DNA of three additional individuals of two Tylosema species. Supplementary Table S1 summarizes voucher information and material type for sampled plants.
Chloroplast DNA Extraction and Sequencing
Total genomic DNA was isolated with a modified CTAB (Cetyl Trimethyl Ammonium Bromide) method described in Yang et al. (2014). For species from which DNA was obtained from fresh leaves, chloroplast DNA (cpDNA) was amplified using long-range PCR (LPCR) with fifteen primer pairs described in Zhang et al. (2016). DNA extracts and cpDNA-amplicons were fragmented for short-insert, paired-end (PE) library construction and sequenced on either an Illumina HiSeq 2000/2500 or X-Ten instrument at Beijing Genomics Institute (BGI, Shenzhen, China), or at the Plant Germplasm and Genomics Center, Kunming Institute of Botany, Chinese Academy of Sciences (KIB, CAS, Kunming, China), respectively.
Plastome Assembly and Annotation
All raw sequence data from LPCR-based plastome enrichment was quality-checked using the NGS QC Tool Kit (Patel and Jain, 2012) with default parameters. High-quality PE reads were de novo assembled into contigs using CLC Genomics Workbench v.8.5.1 with a k-mer of 63 and an automatic bubble size. We retained only contigs with a minimum length of 1 kb and aligned these with Haematoxylum brasiletto (NC_026679) as reference employing nucleotide BLAST (Altschul et al., 1990) at default search parameters. Then, the most probable order of the aligned contigs was determined according to the reference plastome, and the gaps between the contigs were filled by mapping the raw reads to the reference plastome. For shotgun-sequenced genomic DNA, raw reads were filtered and assembled into contigs using GetOrganelle.py1 with the plastome of H. brasiletto as the reference. Contigs were then connected with the help of Bandage Ubuntu dynamic v.080 (Wick et al., 2015) and manual correction where necessary.
Annotation of the plastomes was performed in DOGMA (Wyman et al., 2004), coupled with manual corrections in Geneious v.9.0.2 (Biomatters, Inc.). Protein coding genes were double-checked by finding open reading frames using the Find ORFs function in Geneious v.9.0.2. We used the online tRNAscan-SE service (Schattner et al., 2005) to improve the identification of tRNA genes. Physical maps of all sequenced plastomes were prepared with OrganellarGenomeDRAW v.1.2 (Lohse et al., 2013), and are enclosed here as Supplementary Figure S1. To detect the number of matched reads and the depth of coverage, raw reads were remapped to the assembled plastomes with Bowtie2 (Langmead and Salzberg, 2012), implemented in Geneious v.9.0.2. We used the End-to-End alignment type and Medium Sensitivity/Fast preset, and adjusted the maximum insert size to 800 bp; sequences remained untrimmed before mapping. The final annotated plastomes are deposited in GenBank under accession numbers MF135594-MF135601 (Table 1).
Analysis of Plastome Rearrangements and Inversions
To detect the breakpoints of inversions in the plastomes of Griffonia simplicifolia, Piliostigma thonningii, Tylosema esculentum, and T. fassoglensis, and to identify locally collinear blocks (LCBs) in plastomes of all sampled legumes (see “Phylogeny reconstruction” in Section “Materials and Methods” and Supplementary Table S2), we performed a whole-plastome alignment using Mauve v.2.3.1 (Darling et al., 2010), implemented in Geneious v.9.0.2. To this end, we used the progressiveMauve algorithm with both the seed weight and minimum LCB score being calculated automatically. To detect the breakpoints of inversions in those four species, the plastome of Cercis glabra, which has a typical angiosperm plastome organization, was used as the reference in Mauve alignments. For the Mauve alignment of all sampled legumes, species were ordered alphabetically. Because IRLC legumes all lost the IRA in their plastomes, the IRA of plastomes was removed for each species outside the IRLC.
Analysis of Isomeric Plastomes
We found a 38-kb inversion between a pair of 29-bp IRs in the 3′-ends of trnSGCU and trnSGGA that was absent from the plastome of the previously published Tylosema species (Kim and Cullis, 2017). We pursued two approaches to explore whether a plastome with this inversion coexists with a canonical plastome in Tylosema fassoglensis and other Tylosema species: Firstly, we used Bowtie2 (as before) to map raw PE reads of T. fassoglensis to the four regions corresponding to the breakpoints of the inversion. For convenience, we here defined the plastome with the 38-kb inversion as IPWI (isomeric plastome with inverted arrangement), whereas the plastome with its reverse-complement (canonical) orientation as IPWC (isomeric plastome with canonical arrangement). Accordingly, psbI-trnSGGA-ycf3 and trnGUCC-trnSGCU-rps4 both were captured from IPWI, whereas psbI-trnSGCU-trnGUCC and ycf3-trnSGGA-rps4 were captured from IPWC. Secondly, we performed a PCR assay with specially designed primer pairs that target all four breakpoint regions of the isomeric plastomes. In so doing, we included three additional individuals of Tylosema (Supplementary Table S1) to investigate the universality of the isomeric plastomes in this genus. Each of the 25.5 μL PCR reaction mixture contained 1 μL total genomic DNA (ca. 100 ng/μL) as the template, 0.5 μL each of the forward and reverse primers (10 μmol/L), 12.5 μL Tiangen 2× Taq PCR MasterMix, and 11 μL double-distilled water. To account for potentially different qualities of the template DNAs as well as the possibility of the non-universality of the primer pairs, we used different primer pairs and PCR conditions in our screening, as detailed in Supplementary Table S3.
Phylogenetic Reconstructions
To reconstruct the phylogenetic relationships among Cercidoideae and other legumes, we complemented our data set of eight Cercidoideae species with 49 previously published legume plastomes (Kato et al., 2000; Saski et al., 2005; Guo et al., 2007; Jansen et al., 2008; Magee et al., 2010; Kazakoff et al., 2012; Sabir et al., 2014; Sveinsson and Cronk, 2014; Dugas et al., 2015; Lei et al., 2016; Choi and Choi, 2017; Donkpegan et al., 2017; Feng et al., 2017; Kim and Cullis, 2017; Wang et al., 2017a,b and Cadellia pentastylis (Surianaceae; Li et al., unpublished data) as outgroups (Supplementary Table S2). Protein coding (CDS) and non-genic (intergenic [IGS] and intron) sequences were extracted and aligned to generate original CDS, IGS and intron alignments, respectively, using MAFFT v.7.271 (Katoh and Standley, 2013) with default parameters. Ambiguously aligned sites in all these alignments were removed using GBLOCKS v.0.91b (Castresana, 2000; Talavera and Castresana, 2007) with default parameters, except that all gap positions were allowed. All original alignments and the GBLOCKS-edited versions were concatenated separately to generate supermatrices. Four more supermatrices of original and GBLOCKS-edited alignments for non-genic regions (IGS + intron, hereafter: NGS) and all plastome regions (CDS + IGS + intron) were then obtained by concatenating the corresponding supermatrices. These datasets were subjected to maximum likelihood (ML) phylogenetic reconstructions using RAxML-HPC v.8.2.4 (Stamatakis, 2014) with the GTR-CAT substitution model and 1,000 replicates of rapid bootstrap.
Results
Organization of Cercidoideae Plastomes
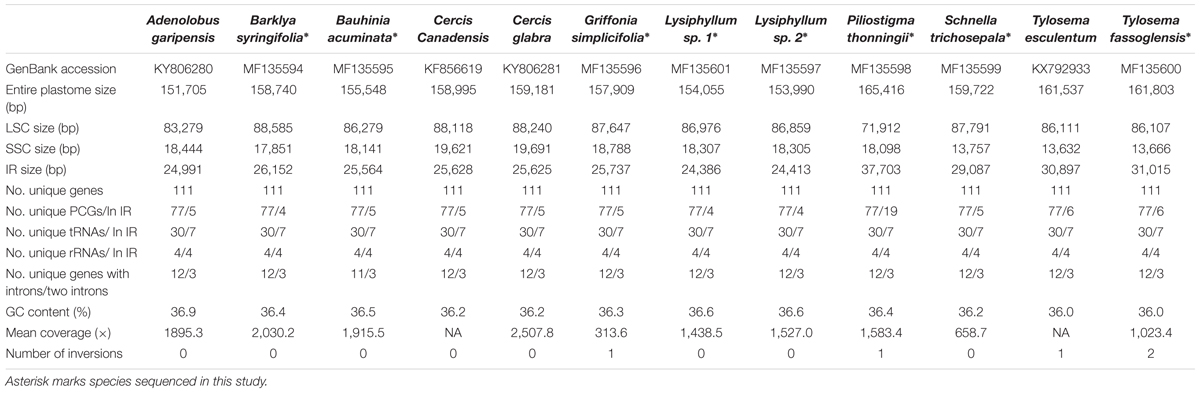
Due to differences regarding both the plant materials and the experimental procedures, the average plastome coverage varied significantly from 313.6× to 2030.2× (Table 1). While the total plastome sizes, including their respective LSC, SSC, and IR regions, differ considerably, we observed only marginal variation in GC contents (36.0 to 36.6%). Plastome size among the sampled Cercidoideae species ranges from 151,705 bp in Adenolobus garipensis to 165,416 bp in Piliostigma thonningii. The length of IR ranges from 24,386 bp in Lysiphyllum sp. 1 to 37,703 bp in P. thonningii. This followed the substantial length variation for LSC from 71,912 bp in P. thonningii to 88,585 bp in Barklya syringifolia. The length of the SSC also varies substantially, ranging from 13,632 bp in Tylosema esculentum to 19,691 bp in Cercis glabra.
Plastome Rearrangement
All sampled plastomes of Cercidoideae exhibit a typical quadripartite structure and a conserved gene content; only Bauhinia acuminata has lost the rpl2 intron (Supplementary Figure S1). The plastid accD genes in species of Barklya, Lysiphyllum, Schnella, and Tylosema apparently lack 260-714 bp at their respective 5′-ends, and the matK genes of Tylosema are 131-bp shorter at their 5′-ends compared with other Cercidoideae species. However, both genes retain intact open reading frames (ORFs).
Unlike gene content, gene order differs notably between species due to the occurrence of several inversions (Figure 1). The plastome of Griffonia simplicifolia has a 24-kb inversion from its rpl16 gene to psaI, resulting in the adjacencies of accD with psaI and rpl16 with rps3. In contrast, a 1.3-kb inversion from trnRUCU and trnGUCC, which results in trnSGCU neighboring trnRUCU and trnGUCC being adjacent to atpA, characterizes the plastome of P. thonningii. Tylosema fassoglensis has two inversions in its plastome, one of which is a 7.5-kb inversion spanning from rbcL to petA, thus resulting in the colocalizations of atpB with petA and rbcL with psbJ – a gene order seen in T. esculentum, too. The second inversion of 38 kb in size lies between the 29-bp IRs at the 3′ -ends of trnSGCU and trnSGGA genes, and, in consequence, psbI is positioned adjacent to trnSGGA and trnSGCU neighbors rps4.
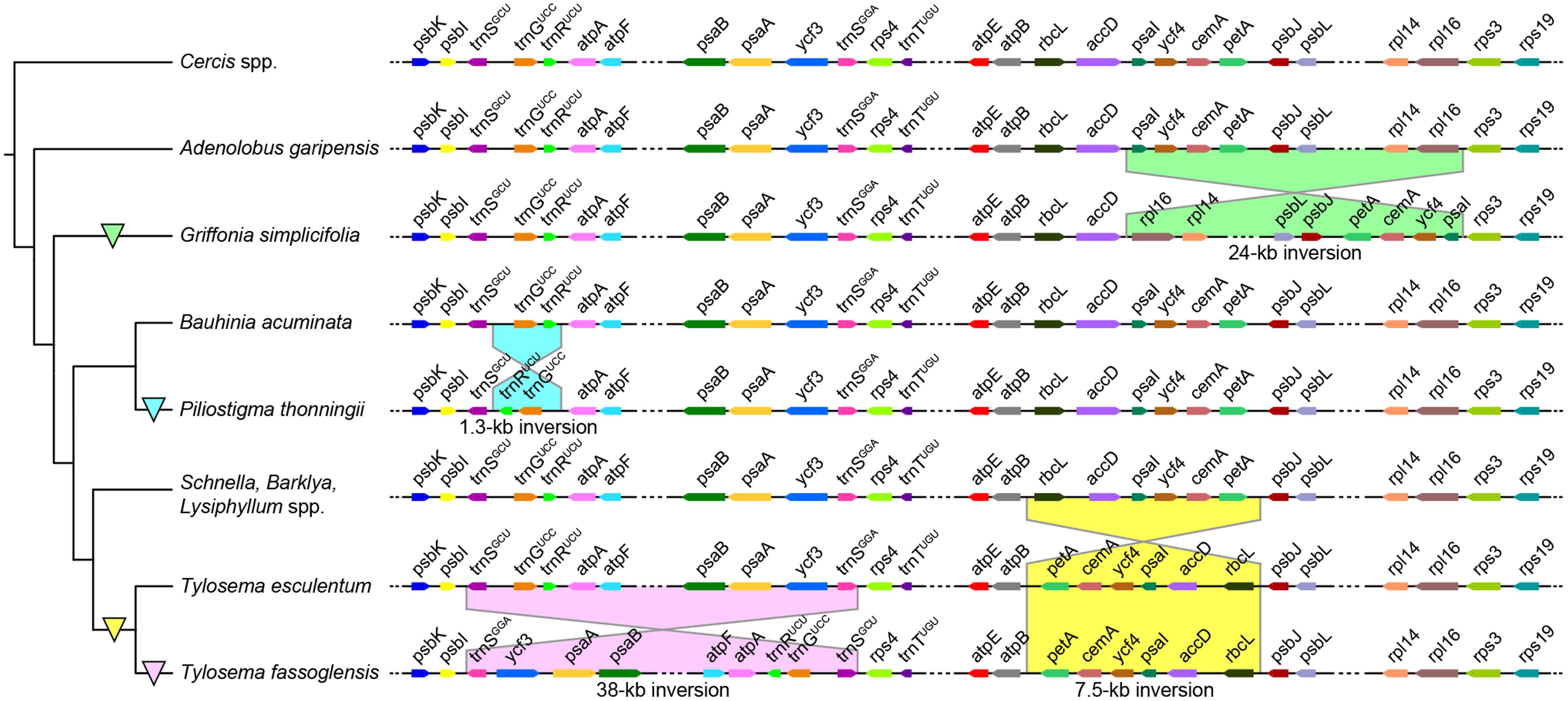
FIGURE 1. Plastome inversions in Cercidoideae. Griffonia, Piliostigma, and Tylosema show independent inversions in their LSCs compared with other closely related Cercidoideae. The partial plastome maps (right) are not drawn to scale, and the tree topology (left) is depicted according to our phylogenic reconstructions based on the combined plastid coding regions (see Results). Colored triangles on branches demarcate the evolutionary origin of the inversion shown to the right.
The plastomes of Adenolobus, Bauhinia, Cercis, and Griffonia have a conserved IR length, ranging from 24,991 (A. garipensis) to 25,737 bp (G. simplicifolia) (Figure 2 and Supplementary Figure S1). The locations of IR-SC junctions of these plastomes are also similar to other canonical angiosperm plastomes. Specifically, the LSC/IRB junction (JLB) lies within the rps19 gene, which leads to some length variation regarding the duplicated 3’-ends of this gene (from 151 bp in Cercis glabra to 178 bp in Bau. acuminate) at the border of the IRA/LSC junction (JLA) between rpl2 (IRA) and trnH (LSC). The position of the IRA/SSC junction (JSA) is within the ycf1. Hence, the duplicated 3′-ends of ycf1 range from 335 (G. simplicifolia) to 807 bp (Bau. acuminata) at the border of the IRB/SSC junction (JSB) between trnN (IRB) and ndhF (SSC). The plastomes of Bar. syringifolia, Lysiphyllum sp. 1, Lysiphyllum sp. 2, and Schnella trichosepala uniformly exhibit a contraction of their IRs. Their LSCs are narrowed by ca. 1.6 kb due to a shift of the JLB into the 3′-exon of rpl2 through which 82 (Bar. syringifolia) to 208 bp (S. trichosepala) of this gene’s 3′-ends is duplicated in the IRA. Among these taxa, S. trichosepala also displays an expansion of approximately 5 kb on the opposite end of its IR, which now contains the entire ycf1 gene, and the JSB lies in the 5′-end of ndhF (14 bp are duplicating in IRA). Tylosema esculentum and T. fassoglensis similarly expanded their IRs to contain intact ycf1; JSB and JSA are accordingly shifted into ndhF and rps15, respectively. The IRs of Bar. syringifolia expand by ca. 1.3 kb into the SSC but the IR/SSC junction remains within the ycf1 coding region. With a gain of 12 kb in size, P. thonningii shows the most extreme IR expansion, leading to the duplication of both rps19 and 13 other genes spanning from rps3 to psbB, and the JLB lies in the intergenic spacer of clpP and psbB.

FIGURE 2. IR/SC junctions in Cercidoideae plastomes. Junctions from IRs into single-copy regions are depicted for all Cercidoideae, which are arranged according to their phylogenetic placement. JLB, JSB, JSA, and JLA refer to the junctions of LSC/IRB, SSC/IRB, SSC/IRA, and LSC/IRA, respectively. Genes are indicated by colored boxes above (encoded on the plus strand) and below (minus strand) the horizontal lines. The tree topology (left) is modified from our reconstructed phylogeny (as in Figure 1; see Results).
Isomeric Plastomes in Tylosema Species
A schematic of the endpoint location of isomeric plastomes is depicted in Figures 3A,B. According to the read-mapping results of the putative T. fassoglensis isomers, 1,579 reads of the over 15 million PE reads obtained by sequencing span the 29-bp IRs in the IPWI orientation, whereas only 15 sequences cover the 29-bp IRs in the IPWC type. Consequently, the frequency of the IPWI and IPWC can be assumed as 99.06% and 0.94%, respectively (Figure 3C). Details regarding the statistics of the read-mapping results are presented in Supplementary Table S4. Using PCR validations, the coexistence of IPWI and IPWC was demonstrated to occur also in other three individuals of Tylosema (Figure 3D). In general, gel electrophoresis revealed fragments for IPWI of T. fassoglensis, T. fassoglensis 1, and T. esculentum to be much brighter, i.e., more abundant, than in T. fassoglensis 2, while that for IPWC normally are much fainter. In addition, in T. fassoglensis, T. fassoglensis 1, and T. esculentum, the band expected for IPWI appears much brighter than that for IPWC, except for its ycf3-trnSGGA-rps4 region, which may be a result of high primer specificity.
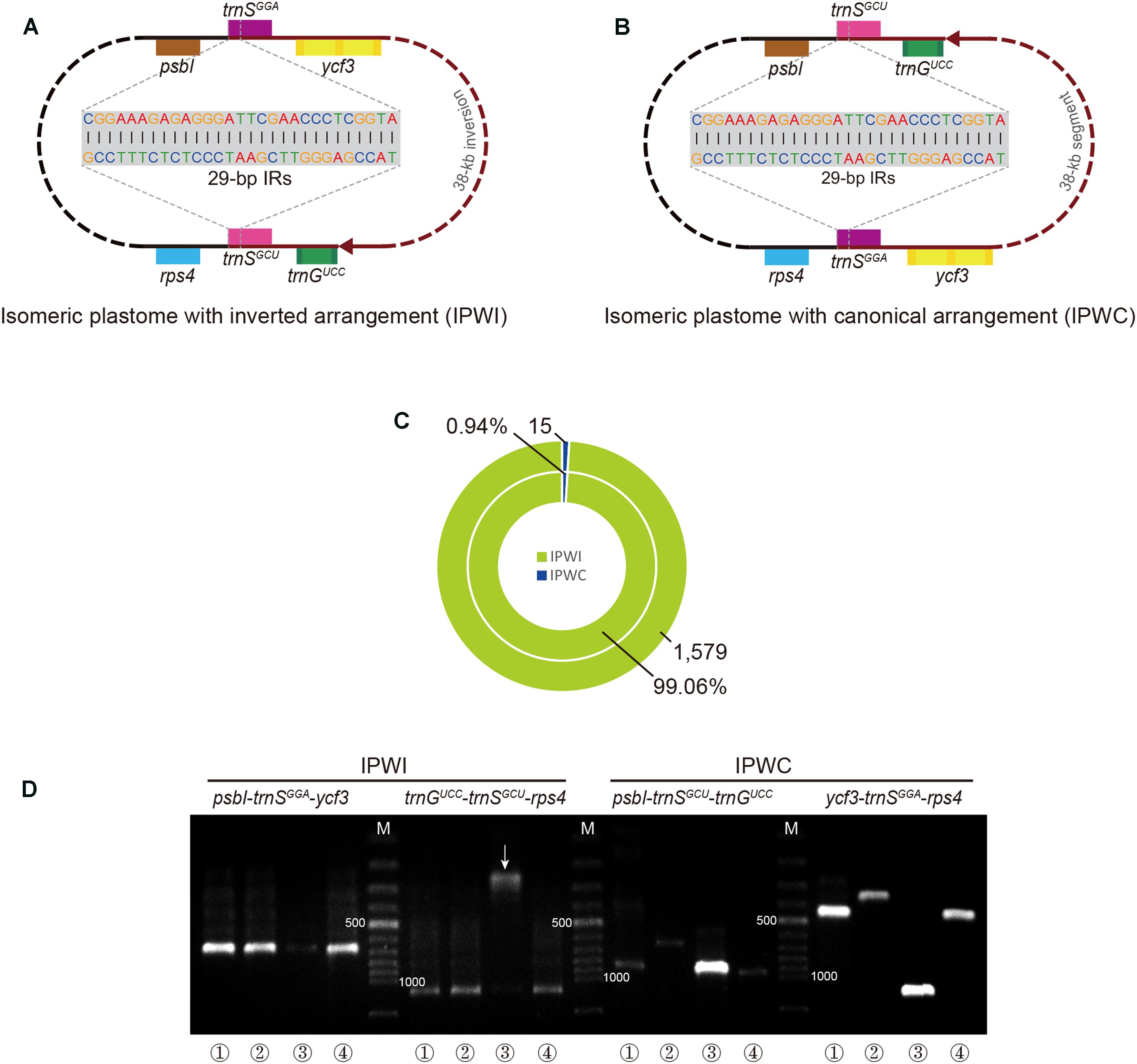
FIGURE 3. Isomeric plastomes in Tylosema species. (A) Gene adjacency at the inversion breakpoints in the IPWI-type plastome. (B) Gene adjacency at inversion breakpoints in the IPWC-type plastome. (C) The results of our read-mapping analysis are depicted as numbers of paired-end reads spanning the 29-bp IR in the outer (total number of reads) and inner circles (proportion of matched reads to total mapped reads). (D) PCR amplicons of four breakpoint regions in four individuals of Tylosema (1, T. fassoglensis; 2, T. fassoglensis 1; 3, T. fassoglensis 2; 4, T. esculentum). The white arrow highlights an abnormal result that may be caused by poor primer specificity. Red lines in A and B refer to the 38-kb inverted segments. “M” in D refers to a 100 bp plus DNA ladder, with “500” and “1000” indicating fragment lengths of 500 and 1 kb, respectively.
Plastid Phylogeny of Cercidoideae
Our phylogenetic analyses supported the monophyly of Cercidoideae with strong bootstrap support (BS) of 100% and contributed to clarifying intergeneric relationships (Supplementary Figure S2). Cercis is well-resolved as the first-branching lineage (BS = 100%), and Adenolobus is confidently placed as sister to the remaining Cercidoideae species (BS = 100%). Bauhinia and Piliostigma are strongly supported as sister clades (BS ≥ 94%). Schnella, Barklya, and Lysiphyllum form a clade with BS = 100%. However, our different data matrices produced conflicting relationships among Griffonia (G), the Bauhinia + Piliostigma (BP) clade, and the lineage containing the remaining species from a clade of Barklya + Lysiphyllum + Schnella + Tylosema, hereafter: BLST). The matrices of original and GBLOCKS-edited CDS and all markers support Griffonia as sister to BP + BLST clade, while other matrices resolve Griffonia together with Bauhinia and Piliostigma as sister to BLST. Incongruence was also found among Schnella, Barklya and Lysiphyllum in different matrices, but Schnella as sister to Barklya and Lysiphyllum was the most strongly supported relationship.
Discussion
Structural Diversity of Plastomes in Cercidoideae
Our plastid genome analyses reveal various structural variations in Cercidoideae, including several inversions, shifts of IR-SC junction, and intron losses. Inversions and IR boundary shifts represent essential mechanisms for plastome rearrangements, which contribute to the structural diversification of plant plastomes (Wicke et al., 2011; Jansen and Ruhlman, 2012). Our study thus adds new results, because legume plastomes outside the Papilionoideae subfamily have long been considered to be conserved regarding their structure and gene content (Schwarz et al., 2015). We now can show that the evolutionary stasis of angiosperm plastomes breaks up already in early-branching lineages of Fabaceae, even though the two early-diverging Cercidoideae genera show no departures from the typical angiosperm plastome organization (Wang et al., 2017b). Major IR expansions and contractions plus some other structural variations such as inversions, gene duplications, and intron losses were also reported for mimosoids recently (Dugas et al., 2015; Wang et al., 2017a).
Inversions of over 1 kb in length are typical for papilionoid plastomes but rarely encountered in other legumes. No large inversions occur in plastomes of Caesalpinioideae (including mimosoids) and Detarioideae, and the unique 7.5-kb inversion has been identified in only one Cercidoideae species, as reported recently (Kim and Cullis, 2017). By analyzing additional genera of Cercidoideae, we here discover that a 7.5-kb inversion is restricted to examined species of Tylosema instead of representing a synapomorphy for the Bauhinia s.l. group as speculated by Kim and Cullis (2017). We also discover three more inversions in Cercidoideae, one of which, the 38-kb inversion in Tylosema fassoglensis, appears to be directly mediated by a pair of 29-bp IRs, resembling the situation of the 36 and 39-kb inversions in Papilionoideae. The other inversions may be promoted by dispersed repeats in the breakpoint regions through intermolecular recombination (Jansen and Ruhlman, 2012). These newly discovered inversions considerably increase the complexity of plastome arrangements in the legume family, as indicated by a comparative analysis of genome structures across legumes (Supplementary Figure S3).
Large IR expansions and contractions are now known from numerous angiosperm lineages. In Fabaceae, the loss of the IRs in the IRLC papilionoids (including Cicereae, Fabeae, Galegeae s.l., Hedysareae, Millettieae p.p., Trifolieae, and a few allies like Callerya, Glycyrrhiza, and Ononis) represents the extreme end of the spectrum of plastome rearrangements in legumes (Wojciechowski et al., 2000). IR expansion in Fabaceae has been reported first from the IREC clade (including Ingeae and Acacia s.s.), which was named by a synapomorphic 13-kb IR expansion (Dugas et al., 2015; Wang et al., 2017a). In addition, IR-LSC junction shifts were found in two genera of IREC (Williams et al., 2015; Wang et al., 2017a). IR expansion into the SSC was also observed in the plastome of Tylosema esculentum, resulting in the duplication of the complete ycf1 gene (Kim and Cullis, 2017). Here, we show that IR-SC junction shifts also affect Bauhinia s.l., except for Bauhinia itself (Figure 2). A double-strand break model (Goulding et al., 1996) and illegitimate recombination (Downie and Jansen, 2015; Blazier et al., 2016) may be causal for IR expansions and contractions in mimosoid plastomes (Wang et al., 2017a). The same mechanism may also underpin IR boundary shifts in Cercidoideae plastomes.
Isomeric Plastome in Tylosema spp.
The 29-bp IR-associated inversion we observed in T. fassoglensis was also previously detected in some genistoids (Martin et al., 2014; Keller et al., 2017) as well as in a Robinia species (Schwarz et al., 2015), where it was thought to result from a flip-flop recombination event. As these 29-bp IRs, which lie at the 3′-ends of trnSGCU and trnSGGA, universally exist in almost all legume plastomes, it can be expected that the inversion between this pair of IRs might have occurred or may occur time and again in other legume plastomes through the same mechanism (Martin et al., 2014). Thus, flip-flop recombination may also explain the 29-bp IR-mediated 38-kb inversion in T. fassoglensis.
Interestingly, the plastome with this inversion appears to be an isomeric plastome (IPWI) that coexists with the canonical type (IPWC) in each individual of Tylosema (Figure 3). Isomeric plastomes, a result of flip-flop recombination, have been observed in several cupressophytes (Yi et al., 2013; Guo et al., 2014; Hsu et al., 2016; Qu et al., 2017b). Thus far, no isomeric plastomes have been reported in legumes, although two stable plastome configurations relating to a 45-kb inversion between a pair of imperfect repeats were found in different individuals of Medicago truncatula (Gurdon and Maliga, 2014). Here, we observed two arrangements of isomeric plastomes in four different Tylosema taxa. IPWI is the dominating conformation over IPWC in T. fassoglensis (the sequenced individual), based on both read-mapping and PCR results. PCR screens also confirmed the domination of IPWI in T. fassoglensis 1 as well as in T. esculentum. On the contrary, the IPWC dominates over IPWI in T. fassoglensis 2. In sum, our results illustrate that isomeric plastomes not only coexist in all of the examined taxa of Tylosema, but the proportions of their relative conformations may be individual-specific. These findings, therefore, provide essential, new insights into the complexity of plastomes in the legume family. Still, further study is needed to explore isomeric plastomes in other Tylosema species and legumes in general, and to clarify the molecular-evolutionary mechanisms and their relevance.
Phylogenetic Relationships in Cercidoideae
Several intergeneric relationships within Cercidoideae are hard to resolve (Bruneau et al., 2008; Sinou et al., 2009; LPWG, 2013, 2017). The sister relationship between Adenolobus and the remaining genera of Cercidoideae (except for the basal Cercis) were all weakly supported in these studies. Sinou et al. (2009) conducted a relatively dense-sampled phylogenetic study of this subfamily. In their work, the phylogenetic relationships of Griffonia, the Clade 1 of Bauhinia s.l. (represented by a clade of Barklya + Lysiphyllum + Schnella + Tylosema in our study), and the Clade 2 of Bauhinia s.l. (Bauhinia + Piliostigma herein) were unresolved. Our plastome data also failed to clarify the phylogenetic relationship of these three groups but allowed resolving most intergeneric relationships among the sampled genera (Figure 4 and Supplementary Figure S2). Plastid phylogenomics has been successfully applied to resolve difficult relationships at the generic level (Ma et al., 2014; Givnish et al., 2015; Qu et al., 2017a; Zhang et al., 2017). However, the tree topology might not hold up when the second largest genus Phanera and four other genera of this subfamily are included in a phylogenetic survey of Cercidoideae. Also, plastid phylogenomics might only resolve a uniparental evolutionary line, not necessarily reflecting the full coalescent history (Wicke and Schneeweiss, 2015). Future studies with an improved, denser taxon sampling in combination with data of other genomic compartments may provide enhanced resolution of the relationships among the genera of this subfamily.
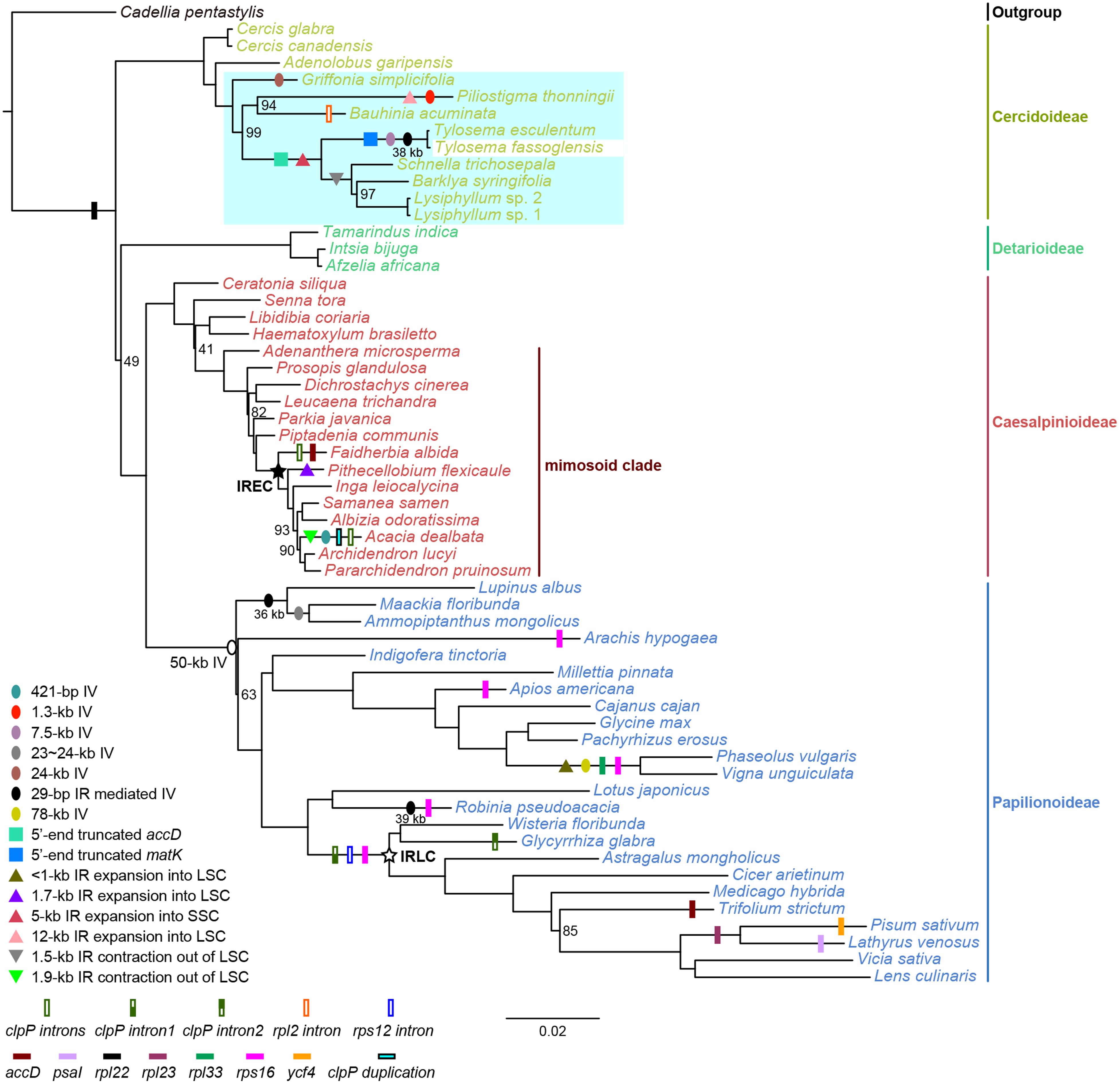
FIGURE 4. Maximum likelihood phylogeny reconstructed using a concatenated dataset of 77 plastid protein-coding genes and major plastome reconfigurations in legumes. The depicted ML tree was reconstructed from a concatenated dataset of 77 plastid protein-coding genes (CDS, Supplementary Figure S2). Species names are colored by their subfamily affiliation. Bootstrap support values of less than 100% are given at nodes. The scale bar indicates the mean number of nucleotide substitutions per site along a branch. As indicated in the legend at the bottom left, inversions (IV), functional, and physical gene losses, IR expansion/contraction, gene duplications, and intron losses are plotted onto branches using colored ovals, squares, triangles, and rectangles, respectively. Hollow circles and pentagons on nodes demarcate the most recent common ancestors of the 50-kb inversion clade, the IR-lacking clade (IRLC), and the IR expanding clade (IREC), respectively. Lengths of 29-bp IR mediated inversions are given below black ovals. A blue-shaded area highlights the plastome variations found in the species sequenced herein, while shown structural features other than these are summarized as reported by Bruneau et al. (1990); Schwarz et al. (2015), Choi and Choi (2017), and Wang et al. (2017a).
Evolutionary Pattern of Plastome Variations in Legumes
The plastid accD gene encodes the β-carboxyl transferase subunit of acetyl-CoA carboxylase (ACCase). It is essential for plants but has been lost independently in at least six photosynthetic angiosperm lineages (Gurdon and Maliga, 2014). In some legume species, the plastid accD was reported to have been functionally transferred to the nucleus (Magee et al., 2010; Sabir et al., 2014), allowing the non-functionalization of the plastid copy. The highly divergent 5′-end of accD in Pisum (Doyle et al., 1995) may be as much a result of the non-functionalization of plastid accD as the 5′-end truncation of Cercidoideae accD we have reported here. The matK gene encodes an intron maturase, which has never been found to be a pseudogene or even absent from the plastome of a photosynthetic land plant (Zoschke et al., 2010; Wicke et al., 2011). We found that in Tylosema, the matK gene lacks more than 100 bp at its 5′-end although it still constitutes an intact open reading frame. As in other legumes, Tylosema retains the same set of group IIA-introns, which are usually associated with the Matk protein during splicing (Zoschke et al., 2010). In some Orobanchaceae, a truncation at matK’s 5′-end results in the use of an alternative start codon that restores the maturase function (Wolfe et al., 1992; Wicke et al., 2013). More research is needed to experimentally validate whether accD and matK are still functioning in Tylosema.
As mentioned earlier, many other structural plastome features have been reported as useful characters to support phylogenetic relationships in legumes (Figure 4). Here, we have shown that a shorter accD may be a synapomorphy of the clade containing Barklya, Lysiphyllum, Schnella, and Tylosema. A 5′-truncation of matK a 7.5-kb and a 29-bp IR-mediated 38-kb inversions characterize Tylosema plastomes. Tylosema and Schnella share a 5-kb IR expansion, which might have been ancestral to the entire clade, but which has lost from Barklya and Lysiphyllum. On the other hand, the 1.5-kb IR contraction we reported herein represents a synapomorphic character of the Barklya+Lysiphyllum+Schnella clade.
There are also many independent or parallel losses of genes or introns, inversions, and IR boundary shifts in legumes (Figure 4). In Cercidoideae, the 1.3 and 24-kb inversions are probably autapomorphies of Piliostigma thonningii and Griffonia simplicifolia, respectively, and the 12-kb IR expansion into the LSC may be another putative autapomorphy of P. thonningii. A denser sampling is needed to verify if these unusual plastome rearrangements are synapomorphic for certain lineages.
The loss of the rpl2 intron has been reported in at least 18 angiosperm families and is thought to be a potentially useful phylogenetic character (Downie et al., 1991; Kelchner, 2002; Judd et al., 2008; Dong et al., 2016; Gu et al., 2016). In Fabaceae, the intron of rpl2 lost several times independently in papilionoids, some Bauhinia species, and P. thonningii (Doyle et al., 1995; Lai et al., 1997; Sinou et al., 2009). Our study revealed that Bauhinia acuminata has lost the rpl2 intron as well, thus corroborating the findings of Lai et al. (1997). However, we detected the rpl2 intron in P. thonningii, a finding inconsistent with earlier reports (Sinou et al., 2009). Therefore, we believe that further research with an expanded sampling is urgently needed to determine the number of rpl2 intron losses in legumes and to evaluate its phylogenetic relevance.
Author Contributions
Y-HW, T-SY, D-ZL, and HW designed this research. Y-HW conducted the experiments and analyses. S-DZ collected some species and extracted DNA. J-JJ and S-YC assembled the plastomes. Y-HW, T-SY, and SW wrote the manuscript, and all authors revised it.
Funding
This study was funded by the National Key Basic Research Programme of China (2014CB954100), the National Science and Technology on Basic Research Programme (2013FY112600), the Strategic Priority Research Program of the Chinese Academy of Sciences (No. XDPB0201), and the Large-scale Scientific Facilities of the Chinese Academy of Sciences (No. 2017-LSF-GBOWS-02).
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We gratefully acknowledge financial support from the abovementioned funding sources. We would like to thank the Brisbane Botanic Garden (Australia) and Kunming Botanic Garden (China) for providing fresh plant material, Prof. Michelle van der Bank (African Centre for DNA Barcoding, South Africa) and Luciano Paganucci de Queiroz (Universidade Estadual de Feira de Santana, Brazil), as well as Sina M. Omosowon (Imperial College London, United Kingdom), for their contribution of silica-dried plant materials. All experiments for this study were carried out in the Key Laboratory of the Southwest China Germplasm Bank of Wild Species at the Kunming Institute of Botany, Chinese Academy of Sciences. We would like to acknowledge the dedicated assistance of all staff members of this laboratory. We also thank the two reviewers for their valuable comments and suggestions, and their recognition of this study.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fpls.2018.00138/full#supplementary-material
Footnotes
References
Altschul, S. F., Gish, W., Miller, W., Myers, E. W., and Lipman, D. J. (1990). Basic local alignment search tool. J. Mol. Biol. 215, 403–410. doi: 10.1016/S0022-2836(05)80360-2
Blazier, J. C., Jansen, R. K., Mower, J. P., Govindu, M., Zhang, J., Weng, M. L., et al. (2016). Variable presence of the inverted repeat and plastome stability in Erodium. Ann. Bot. 117, 1209–1220. doi: 10.1093/aob/mcw065
Bruneau, A., Doyle, J. J., and Palmer, J. D. (1990). A chloroplast DNA inversion as a subtribal character in the Phaseoleae (Leguminosae). Syst. Bot. 15, 378–386. doi: 10.2307/2419351
Bruneau, A., Forest, F., Herendeen, P. S., Klitgaard, B. B., and Lewis, G. P. (2001). Phylogenetic relationships in the Caesalpinioideae (Leguminosae) as inferred from chloroplast trnL intron sequences. Syst. Bot. 26, 487–514.
Bruneau, A., Mercure, M., Lewis, G. P., and Herendeen, P. S. (2008). Phylogenetic patterns and diversification in the caesalpinioid legumes. Botany 86, 697–718. doi: 10.1139/B08-058
Cai, Z. Q., Guisinger, M., Kim, H. G., Ruck, E., Blazier, J. C., McMurtry, V., et al. (2008). Extensive reorganization of the plastid genome of Trifolium subterraneum (Fabaceae) is associated with numerous repeated sequences and novel DNA insertions. J. Mol. Evol. 67, 696–704. doi: 10.1007/s00239-008-9180-7
Castresana, J. (2000). Selection of conserved blocks from multiple alignments for their use in phylogenetic analysis. Mol. Biol. Evol. 17, 540–552. doi: 10.1093/oxfordjournals.molbev.a026334
Choi, I. S., and Choi, B. H. (2017). The distinct plastid genome structure of Maackia fauriei (Fabaceae: Papilionoideae) and its systematic implications for genistoids and tribe Sophoreae. PLOS ONE 12:e0173766. doi: 10.1371/journal.pone.0173766
Chumley, T. W., Palmer, J. D., Mower, J. P., Fourcade, H. M., Calie, P. J., Boore, J. L., et al. (2006). The complete chloroplast genome sequence of Pelargonium x hortorum: organization and evolution of the largest and most highly rearranged chloroplast genome of land plants. Mol. Biol. Evol. 23, 2175–2190. doi: 10.1093/molbev/msl089
Clark, R. P., Mackinder, B. A., and Banks, H. (2017). Cheniella gen. nov. (Leguminosae: Cercidoideae) from southern China, Indochina and Malesia. Eur. J. Taxon. 360, 1–37. doi: 10.5852/ejt.2017.360
Cosner, M. E., Raubeson, L. A., and Jansen, R. K. (2004). Chloroplast DNA rearrangements in Campanulaceae: Phylogenetic utility of highly rearranged genomes. BMC Evol. Biol. 4:27. doi: 10.1186/1471-2148-4-27
Darling, A. E., Mau, B., and Perna, N. T. (2010). progressiveMauve: multiple genome alignment with gene gain, loss and rearrangement. PLOS ONE 5:e11147. doi: 10.1371/journal.pone.0011147
Dong, W. P., Xu, C., Li, D. Z., Jin, X. B., Li, R. L., Lu, Q., et al. (2016). Comparative analysis of the complete chloroplast genome sequences in psammophytic Haloxylon species (Amaranthaceae). PeerJ 4:e2699. doi: 10.7717/peerj.2699
Donkpegan, A. S. L., Doucet, J.-L., Migliore, J., Duminil, J., Dainou, K., Piñeiro, R., et al. (2017). Evolution in African tropical trees displaying ploidy-habitat association: The genus Afzelia (Leguminosae). Mol. Phylogenet. Evol 107, 270–281. doi: 10.1016/j.ympev.2016.11.004
Downie, S. R., and Jansen, R. K. (2015). A comparative analysis of whole plastid genomes from the Apiales: expansion and contraction of the inverted repeat, mitochondrial to plastid transfer of DNA, and identification of highly divergent noncoding regions. Syst. Bot. 40, 336–351. doi: 10.1600/036364415X686620
Downie, S. R., Olmstead, R. G., Zurawski, G., Soltis, D. E., Soltis, P. S., Watson, J. C., et al. (1991). Six independent losses of the chloroplast DNA rpl2 intron in dicotyledons: molecular and phylogenetic implications. Evolution 45, 1245–1259. doi: 10.1111/j.1558-5646.1991.tb04390.x
Doyle, J. J., Doyle, J. L., Ballenger, J. A., and Palmer, J. D. (1996). The distribution and phylogenetic significance of a 50-kb chloroplast DNA inversion in the flowering plant family Leguminosae. Mol. Phylogen. Evol. 5, 429–438. doi: 10.1006/mpev.1996.0038
Doyle, J. J., Doyle, J. L., and Palmer, J. D. (1995). Multiple independent losses of two genes and one intron from legume. Syst. Bot. 20, 272–294. doi: 10.2307/2419496
Dugas, D. V., Hernandez, D., Koenen, E. J. M., Schwarz, E., Straub, S., Hughes, C. E., et al. (2015). Mimosoid legume plastome evolution: IR expansion, tandem repeat expansions, and accelerated rate of evolution in clpP. Sci. Rep. 5:16958. doi: 10.1038/srep16958
Feng, L., Gu, L. F., Luo, J., Fu, A. S., Ding, Q., Yiu, S. M., et al. (2017). Complete plastid genomes of the genus Ammopiptanthus and identification of a novel 23-kb rearrangement. Conserv. Genet. Resour. 9, 647–650. doi: 10.1007/s12686-017-0747-8
Gantt, J. S., Baldauf, S. L., Calie, P. J., Weeden, N. F., and Palmer, J. D. (1991). Transfer of rpl22 to the nucleus greatly preceded its loss from the chloroplast and involved the gain of an intron. EMBO J. 10, 3073–3078.
Givnish, T. J., Spalink, D., Ames, M., Lyon, S. P., Hunter, S. J., Zuluaga, A., et al. (2015). Orchid phylogenomics and multiple drivers of their extraordinary diversification. Proc. R. Soc. B. 282, 1–10. doi: 10.1098/rspb.2015.1553
Goulding, S. E., Olmstead, R. G., Morden, C. W., and Wolfe, K. H. (1996). Ebb and flow of the chloroplast inverted repeat. Mol. Gen. Genet. 252, 195–206. doi: 10.1007/BF02173220
Gu, C., Tembrock, L. R., Johnson, N. G., Simmons, M. P., and Wu, Z. (2016). The complete plastid genome of Lagerstroemia fauriei and loss of rpl2 intron from Lagerstroemia (Lythraceae). PLOS ONE 11:e0150752. doi: 10.1371/journal.pone.0150752
Guisinger, M. M., Kuehl, J. V., Boore, J. L., and Jansen, R. K. (2011). Extreme reconfiguration of plastid genomes in the angiosperm family Geraniaceae: Rearrangements, repeats, and codon usage. Mol. Biol. Evol. 28, 583–600. doi: 10.1093/molbev/msq229
Guo, W., Grewe, F., Cobo-Clark, A., Fan, W., Duan, Z., Adams, R. P., et al. (2014). Predominant and substoichiometric isomers of the plastid genome coexist within Juniperus plants and have shifted multiple times during cupressophyte evolution. Genome Biol. Evol. 6, 580–590. doi: 10.1093/gbe/evu046
Guo, X. W., Castillo-Ramirez, S., Gonzalez, V., Bustos, P., Fernandez-Vazquez, J. L., Santamaria, R. I., et al. (2007). Rapid evolutionary change of common bean (Phaseolus vulgaris L.) plastome, and the genomic diversification of legume chloroplasts. BMC Genomics 8:228. doi: 10.1186/1471-2164-8-228
Gurdon, C., and Maliga, P. (2014). Two distinct plastid genome configurations and unprecedented intraspecies length variation in the accD coding region in Medicago truncatula. DNA Res. 21, 417–427. doi: 10.1093/dnares/dsu007
Haberle, R. C., Fourcade, H. M., Boore, J. L., and Jansen, R. K. (2008). Extensive rearrangements in the chloroplast genome of Trachelium caeruleum are associated with repeats and tRNA genes. J. Mol. Evol. 66, 350–361. doi: 10.1007/s00239-008-9086-4
Herendeen, P., Bruneau, A., and Lewis, G. (2003). “Phylogenetic relationships in caesalpinioid legumes: a preliminary analysis based on morphological and molecular data,” in Advances in Legume Systematics, part 10, Higher Level Systematics, eds B. B. Klitgaard and A. Bruneau (Richmond: Royal Botanic Gardens, Kew), 37–62.
Hsu, C. Y., Wu, C. S., and Chaw, S. M. (2016). Birth of four chimeric plastid gene clusters in Japanese umbrella pine. Genome Biol. Evol. 8, 1776–1784. doi: 10.1093/gbe/evw109
Jansen, R. K., and Ruhlman, T. A. (2012). “Plastid genomes of seed plants,” in Genomics of Chloroplasts and Mitochondria, eds R. Bock and V. Knoop (Dordrecht: Springer), 103–126.
Jansen, R. K., Wojciechowski, M. F., Sanniyasi, E., Lee, S. B., and Daniell, H. (2008). Complete plastid genome sequence of the chickpea (Cicer arietinum) and the phylogenetic distribution of rps12 and clpP intron losses among legumes (Leguminosae). Mol. Phylogenet. Evol. 48, 1204–1217. doi: 10.1016/j.ympev.2008.06.013
Judd, W. S., Campbell, C. S., Kellogg, E. A., Stevens, P. F., and Donoghue, M. J. (2008). Plant Systematics: A Phylogenetic Approach, Third Edn. Sunderland: Sinauer Associates, Inc.
Kato, T., Kaneko, T., Sato, S., Nakamura, Y., and Tabata, S. (2000). Complete structure of the chloroplast genome of a legume, Lotus japonicus. DNA Res. 7, 323–330. doi: 10.1093/dnares/7.6.323
Katoh, K., and Standley, D. M. (2013). MAFFT multiple sequence alignment software version 7: Improvements in performance and usability. Mol. Biol. Evol. 30, 772–780. doi: 10.1093/molbev/mst010
Kazakoff, S. H., Imelfort, M., Edwards, D., Koehorst, J., Biswas, B., Batley, J., et al. (2012). Capturing the biofuel wellhead and powerhouse: the chloroplast and mitochondrial genomes of the leguminous feedstock tree Pongamia pinnata. PLOS ONE 7:e51687. doi: 10.1371/journal.pone.0051687
Kelchner, S. A. (2002). Group II introns as phylogenetic tools: structure function, and evolutionary constraints. Am. J. Bot. 89, 1651–1669. doi: 10.3732/ajb.89.10.1651
Keller, J., Rousseau-Gueutin, M., Martin, G. E., Morice, J., Boutte, J., Coissac, E., et al. (2017). The evolutionary fate of the chloroplast and nuclear rps16 genes as revealed through the sequencing and comparative analyses of four novel legume chloroplast genomes from Lupinus. DNA Res. 24, 343–358. doi: 10.1093/dnares/dsx006
Kim, Y., and Cullis, C. (2017). A novel inversion in the chloroplast genome of marama (Tylosema esculentum). J. Exp. Bot. 68, 2065–2072. doi: 10.1093/jxb/erw500
Lai, M., Sceppa, J., Ballenger, J. A., Doyle, J. J., and Wunderlin, R. P. (1997). Polymorphism for the presence of the rpL2 intron in chloroplast genomes of Bauhinia (Leguminosae). Syst. Bot. 22, 519–528. doi: 10.2307/2419825
Langmead, B., and Salzberg, S. L. (2012). Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359. doi: 10.1038/nmeth.1923
Lee, H. L., Jansen, R. K., Chumley, T. W., and Kim, K. J. (2007). Gene relocations within chloroplast genomes of Jasminum and Menodora (Oleaceae) are due to multiple, overlapping inversions. Mol. Biol. Evol. 24, 1161–1180. doi: 10.1093/molbev/msm036
Lei, W., Ni, D., Wang, Y., Shao, J., Wang, X., Yang, D., et al. (2016). Intraspecific and heteroplasmic variations, gene losses and inversions in the chloroplast genome of Astragalus membranaceus. Sci. Rep. 6:21669. doi: 10.1038/srep21669
Lewis, G. P., Schrire, B. D., Mackinder, B. A., and Lock, M. (2005). Legumes of the World. Richmond: Royal Botanic Gardens, Kew.
Lohse, M., Drechsel, O., Kahlau, S., and Bock, R. (2013). OrganellarGenomeDRAW-a suite of tools for generating physical maps of plastid and mitochondrial genomes and visualizing expression data sets. Nucleic Acids Res. 41, W575–W581. doi: 10.1093/nar/gkt289
LPWG (2013). Legume phylogeny and classification in the 21st century: progress, prospects and lessons for other species-rich clades. Taxon 62, 217–248. doi: 10.12705/622.8
LPWG (2017). A new subfamily classification of the Leguminosae based on a taxonomically comprehensive phylogeny. Taxon 66, 44–77. doi: 10.12705/661.3
Ma, P. F., Zhang, Y. X., Zeng, C. X., Guo, Z. H., and Li, D. Z. (2014). Chloroplast phylogenomic analyses resolve deep-level relationships of an intractable bamboo tribe Arundinarieae (poaceae). Syst. Biol. 63, 933–950. doi: 10.1093/sysbio/syu054
Magee, A. M., Aspinall, S., Rice, D. W., Cusack, B. P., Semon, M., Perry, A. S., et al. (2010). Localized hypermutation and associated gene losses in legume chloroplast genomes. Genome Res. 20, 1700–1710. doi: 10.1101/gr.111955.110
Martin, G. E., Rousseau-Gueutin, M., Cordonnier, S., Lima, O., Michon-Coudouel, S., Naquin, D., et al. (2014). The first complete chloroplast genome of the Genistoid legume Lupinus luteus: evidence for a novel major lineage-specific rearrangement and new insights regarding plastome evolution in the legume family. Ann. Bot. 113, 1197–1210. doi: 10.1093/aob/mcu050
Milligan, B. G., Hampton, J. N., and Palmer, J. D. (1989). Dispersed repeats and structural reorganization in subclover chloroplast DNA. Mol. Biol. Evol. 6, 355–368.
Palmer, J. D. (1983). Chloroplast DNA exists in two orientations. Nature 301, 92–93. doi: 10.1038/301092a0
Palmer, J. D., Osorio, B., Aldrich, J., and Thompson, W. F. (1987). Chloroplast DNA evolution among legumes - loss of a large inverted repeat occurred prior to other sequence rearrangements. Curr. Genet. 11, 275–286. doi: 10.1007/BF00355401
Palmer, J. D., and Thompson, W. F. (1982). Chloroplast DNA rearrangements are more frequent when a large inverted repeat sequence is lost. Cell 29, 537–550. doi: 10.1016/0092-8674(82)90170-2
Patel, R. K., and Jain, M. (2012). NGS QC toolkit: a toolkit for quality control of next generation sequencing data. PLOS ONE 7:e30619. doi: 10.1371/journal.pone.0030619
Qu, X. J., Jin, J. J., Chaw, S. M., Li, D. Z., and Yi, T. S. (2017a). Multiple measures could alleviate long-branch attraction in phylogenomic reconstruction of Cupressoideae (Cupressaceae). Sci. Rep. 7:41005. doi: 10.1038/srep41005
Qu, X. J., Wu, C. S., Chaw, S. M., and Yi, T. S. (2017b). Insights into the existence of isomeric plastomes in Cupressoideae (Cupressaceae). Genome Biol. Evol. 9, 1110–1119. doi: 10.1093/gbe/evx071
Ruhlman, T. A., and Jansen, R. K. (2014). “The plastid genomes of flowering plants,” in Chloroplast Biotechnology: Methods and Protocols, ed. M. Pal (Totowa, NJ: Humana Press), 3–38.
Sabir, J., Schwarz, E., Ellison, N., Zhang, J., Baeshen, N. A., Mutwakil, M., et al. (2014). Evolutionary and biotechnology implications of plastid genome variation in the inverted-repeat-lacking clade of legumes. Plant Biotechnol. J. 12, 743–754. doi: 10.1111/pbi.12179
Saski, C., Lee, S.-B., Daniell, H., Wood, T. C., Tomkins, J., Kim, H.-G., et al. (2005). Complete chloroplast genome sequence of Glycine max and comparative analyses with other legume genomes. Plant Mol. Biol. 59, 309–322. doi: 10.1007/s11103-005-8882-0
Schattner, P., Brooks, A. N., and Lowe, T. M. (2005). The tRNAscan-SE, snoscan and snoGPS web servers for the detection of tRNAs and snoRNAs. Nucleic Acids Res. 33, W686–W689. doi: 10.1093/nar/gki366
Schwarz, E. N., Ruhlman, T. A., Sabir, J. S. M., Hajarah, N. H., Alharbi, N. S., Al-Malki, A. L., et al. (2015). Plastid genome sequences of legumes reveal parallel inversions and multiple losses of rps16 in papilionoids. J. Syst. Evol. 53, 458–468. doi: 10.1111/jse.12179
Sinou, C., Forest, F., Lewis, G. P., and Bruneau, A. (2009). The genus Bauhinia s.l. (Leguminosae): a phylogeny based on the plastid trnL–trnF region. Botany 87, 947–960. doi: 10.1139/B09-065
Stamatakis, A. (2014). RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 30, 1312–1313. doi: 10.1093/bioinformatics/btu033
Stein, D. B., Palmer, J. D., and Thompson, W. F. (1986). Structural evolution and flip-flop recombination of chloroplast DNA in the fern genus Osmunda. Curr. Genet. 10, 835–841. doi: 10.1007/BF00418530
Sveinsson, S., and Cronk, Q. (2014). Evolutionary origin of highly repetitive plastid genomes within the clover genus (Trifolium). BMC Evol. Biol. 14:228. doi: 10.1186/s12862-014-0228-6
Talavera, G., and Castresana, J. (2007). Improvement of phylogenies after removing divergent and ambiguously aligned blocks from protein sequence alignments. Syst. Biol. 56, 564–577. doi: 10.1080/10635150701472164
Tsumura, Y., Suyama, Y., and Yoshimura, K. (2000). Chloroplast DNA inversion polymorphism in populations of Abies and Tsuga. Mol. Biol. Evol. 17, 1302–1312. doi: 10.1093/oxfordjournals.molbev.a026414
Wang, Y. H., Qu, X. J., Chen, S. Y., Li, D. Z., and Yi, T. S. (2017a). Plastomes of Mimosoideae: structural and size variation, sequence divergence, and phylogenetic implication. Tree Genet. Genomes 13:41. doi: 10.1007/s11295-017-1124-1
Wang, Y. H., Wang, H., Yi, T. S., and Wang, Y. H. (2017b). The complete chloroplast genomes of Adenolobus garipensis and Cercis glabra (Cercidoideae, Fabaceae). Conserv. Genet. Resour. 9, 635–638. doi: 10.1007/s12686-017-0744-y
Weng, M. L., Blazier, J. C., Govindu, M., and Jansen, R. K. (2014). Reconstruction of the ancestral plastid genome in Geraniaceae reveals a correlation between genome rearrangements, repeats, and nucleotide substitution rates. Mol. Biol. Evol. 31, 645–659. doi: 10.1093/molbev/mst257
Wick, R. R., Schultz, M. B., Zobel, J., and Holt, K. E. (2015). Bandage: interactive visualization of de novo genome assemblies. Bioinformatics 31, 3350–3352. doi: 10.1093/bioinformatics/btv383
Wicke, S., Muller, K. F., de Pamphilis, C. W., Quandt, D., Wickett, N. J., Zhang, Y., et al. (2013). Mechanisms of functional and physical genome reduction in photosynthetic and nonphotosynthetic parasitic plants of the broomrape family. Plant Cell 25, 3711–3725. doi: 10.1105/tpc.113.113373
Wicke, S., and Schneeweiss, G. M. (2015). “Next-generation organellar genomics: potentials and pitfalls of high-throughput technologies for molecular evolutionary studies and plant systematics,” in Next-Generation Sequencing in Plant Systematics, eds E. Hörandl and M. S. Appelhans (Königstein: Koeltz Scientific Books), 9–50.
Wicke, S., Schneeweiss, G. M., dePamphilis, C. W., Muller, K. F., and Quandt, D. (2011). The evolution of the plastid chromosome in land plants: Gene content, gene order, gene function. Plant Mol. Biol. 76, 273–297. doi: 10.1007/s11103-011-9762-4
Williams, A. V., Boykin, L. M., Howell, K. A., Nevill, P. G., and Small, I. (2015). The complete sequence of the Acacia ligulata chloroplast genome reveals a highly divergent clpP1 gene. PLOS ONE 10:e0125768. doi: 10.1371/journal.pone.0125768
Wojciechowski, M. F., Sanderson, M. J., Steele, K. P., and Liston, A. (2000). “Molecular phylogeny of the “Temperate Herbaceous Tribes” of papilionoid legumes: a supertree approach,” in Advances in Legume Systematics, part 9, eds P. S. Herendeen and A. Bruneau (Richmond: Royal Botanic Gardens, Kew), 277–298.
Wolfe, K. H., Morden, C. W., and Palmer, J. D. (1992). Function and evolution of a minimal plastid genome from a nonphotosynthetic parasitic plant. Proc. Natl. Acad. Sci. U.S.A. 89, 10648–10652. doi: 10.1073/pnas.89.22.10648
Wu, C. S., Lin, C. P., Hsu, C. Y., Wang, R. J., and Chaw, S. M. (2011). Comparative chloroplast genomes of Pinaceae: insights into the mechanism of diversified genomic organizations. Genome Biol. Evol. 3, 309–319. doi: 10.1093/gbe/evr026
Wyman, S. K., Jansen, R. K., and Boore, J. L. (2004). Automatic annotation of organellar genomes with DOGMA. Bioinformatics 20, 3252–3255. doi: 10.1093/bioinformatics/bth352
Yang, J. B., Li, D. Z., and Li, H. T. (2014). Highly effective sequencing whole chloroplast genomes of angiosperms by nine novel universal primer pairs. Mol. Ecol. Resour. 14, 1024–1031. doi: 10.1111/1755-0998.12251
Yi, X., Gao, L., Wang, B., Su, Y. J., and Wang, T. (2013). The complete chloroplast genome sequence of Cephalotaxus oliveri (Cephalotaxaceae): evolutionary comparison of Cephalotaxus chloroplast DNAs and insights into the loss of inverted repeat copies in Gymnosperms. Genome Biol. Evol. 5, 688–698. doi: 10.1093/gbe/evt042
Zhang, S. D., Jin, J. J., Chen, S. Y., Chase, M. W., Soltis, D. E., Li, H. T., et al. (2017). Diversification of Rosaceae since the late cretaceous based on plastid phylogenomics. New Phytol. 214, 1355–1367. doi: 10.1111/nph.14461
Zhang, T., Zeng, C. X., Yang, J. B., Li, H. T., and Li, D. Z. (2016). Fifteen novel universal primer pairs for sequencing whole chloroplast genomes and a primer pair for nuclear ribosomal DNAs. J. Syst. Evol. 54, 219–227. doi: 10.1111/jse.12197
Keywords: inversion, isomeric plastomes, IR expansion/contraction, plastome, variation, Cercidoideae, Fabaceae
Citation: Wang Y-H, Wicke S, Wang H, Jin J-J, Chen S-Y, Zhang S-D, Li D-Z and Yi T-S (2018) Plastid Genome Evolution in the Early-Diverging Legume Subfamily Cercidoideae (Fabaceae). Front. Plant Sci. 9:138. doi: 10.3389/fpls.2018.00138
Received: 17 November 2017; Accepted: 24 January 2018;
Published: 08 February 2018.
Edited by:
Federico Luebert, University of Bonn, GermanyReviewed by:
Anne Bruneau, Université de Montréal, CanadaMartin F. Wojciechowski, Arizona State University, United States
Copyright © 2018 Wang, Wicke, Wang, Jin, Chen, Zhang, Li and Yi. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Ting-Shuang Yi, dGluZ3NodWFuZ3lpQG1haWwua2liLmFjLmNu De-Zhu Li, ZHpsQG1haWwua2liLmFjLmNu
 Yin-Huan Wang
Yin-Huan Wang Susann Wicke
Susann Wicke Hong Wang1
Hong Wang1 Jian-Jun Jin
Jian-Jun Jin De-Zhu Li
De-Zhu Li Ting-Shuang Yi
Ting-Shuang Yi