Abstract
The identification and phylogenetic relationships of potato cyst nematodes (PCN) were studied to assess the potential value of geographical distribution information for integrated pest management of potato production in Portugal. This research focused on PCN species, Globodera pallida and Globodera rostochiensis. From 2013 until 2019, 748 soil samples from the rhizosphere of different potato cultivars were surveyed in the Portuguese mainland to detect and identify both species and track their location. PCN are widespread invasive species throughout Portugal. In fact, during the survey period an incidence of 22.5% was estimated for the tested samples. The patterns of infestation vary among regions, increasing from south to north, where PCN were first detected. Currently, both species are present in all potato producing regions of the country, with a greater incidence of G. pallida. Phytosanitary control measures are influencing to the observed results. The use of potato cultivars resistant to G. rostochiensis led to a decrease of this species but had no influence on G. pallida detections, which continues its reproduction freely since there are no effective resistant cultivars for this species. The relationship between the presence, infestation rate, spread and geographical distribution of PCN is discussed in terms of behavioral responses of the potato cultivars and the implications for developing new integrated crop protection measures.
Introduction
Potato crop (Solanum tuberosum) has great social and economic importance in Portugal since it is grown throughout the country. The most representative production regions are the North and West Regions (Figure 1), with a total potato growing area of approximately 20,000 hectares and a total production of 430,000 tons. Several nematode species have been reported associated with potato. Among those, the potato cyst nematodes (PCN), Globodera rostochiensis (Wollenweber, 1923; Skarbilovich, 1959) and Globodera pallida (Stone, 1973), are two of the major species limiting potato yield. These two species are sedentary endoparasites of the potato root system, deteriorate the quality and commercial value of tubers and contribute to infection of potatoes by other opportunistic plant pathogens, such as fungi (Lavrova et al., 2017).
FIGURE 1
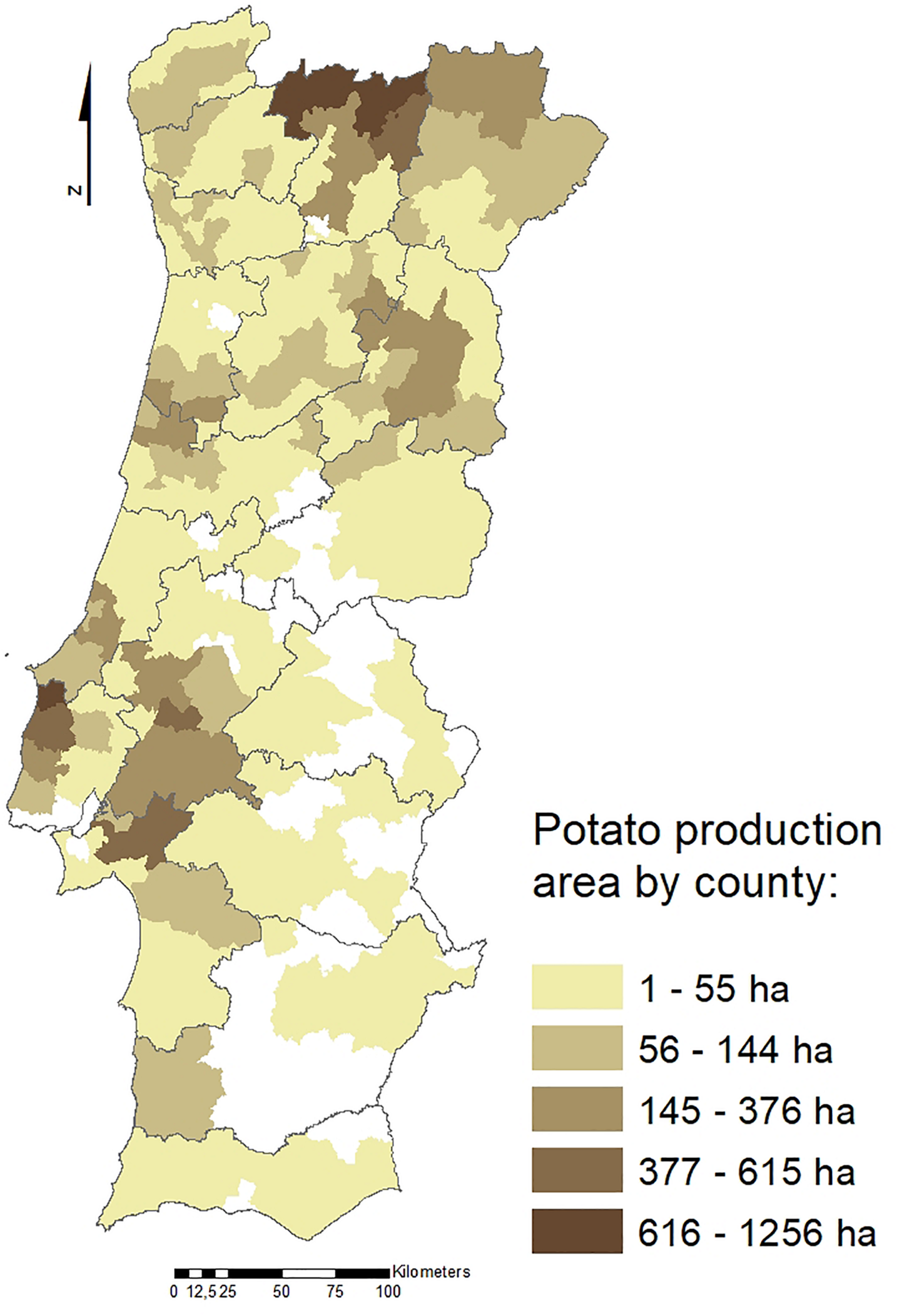
Potato production areas (ha) in Portugal by county (data from INE, 2011).
Yield losses due to the presence of PCN, estimated at €220 million/year in Europe (Viaene, 2016), can vary from slight losses to crop failure depending on the infestation level (Lima et al., 2018).
Both PCN species are considered harmful quarantine organisms and are subject to stringent regulatory measures when detected singly or in combination (EPPO, 2017).
The golden potato cyst nematode, G. rostochiensis, and the pale potato cyst nematode, G. pallida, originated from the Andes region in southern Peru and have spread as the result of anthropogenic activity into many regions of the world (Grenier et al., 2010). They are thought to have been introduced to Europe in the 16–17th century by means of potato tubers carrying infested soil and nowadays have worldwide distribution. PCN have been reported throughout Europe, South America and parts of Asia, North America, Oceania and Africa where potatoes are grown (EPPO, 2020). However, new Globodera sp. detections continue to be reported (Hafez et al., 2007; Mburu et al., 2018; Niragire et al., 2019; Inácio et al., 2020).
In Portugal, G. rostochiensis was first reported in 1956 (Macara, 1963) in a field of seed potatoes near Bragança (Trás-os-Montes district, North of Portugal) and is currently present in all potato producing regions of the country (DGAV, 2015; Camacho et al., 2017), including the Madeira and Azores islands (DGAV, 2015; Inácio et al., 2020). Globodera pallida was first identified in 1988 (Santos and Fernandes, 1988), also in Trás-os-Montes, but its current national distribution has not yet been reported.
The knowledge on the geographical distribution, density and spatial dynamics of pest populations is indispensable in integrated pest management (IPM) systems, as it raises considerable interest among plant breeders and plant pathologists for the need to better understand the interaction between pest or pathogen and host and to estimate the risk of crop damage. Therefore, information of PCN distribution and potato cultivars used is essential to understand the Globodera spp. regional range of expansion since their first report. As human activity is the most probable means of spreading PCN, there is a specific interest in the evaluation of the implemented control measures and their consequences to adopt more effective management practices.
Controlling PCN is a difficult task due to their high level of adaptation to the environment, the prolonged viability of cysts in the absence of the host plant for more than 20 years, either quiescent or diapause in the form of encysted eggs (Christoforou et al., 2014), and the risk of appearance of aggressive pathotypes in the monoculture of nematode-tolerant potato cultivars. To assess the prevalence and distribution of PCN species across the territory, a country-wide survey was established in 2010, outlining a new framework for phytosanitary protection measures against these harmful organisms to avoid dispersion in national and European Community territories and to ensure potato production of a guaranteed quality for consumers. The main potato growing regions of Portugal have been surveyed for the presence of G. rostochiensis and G. pallida since 2013.
Before the national survey started, infestations were almost entirely due to G. rostochiensis (Santos and Fernandes, 1988; Santos et al., 1995; Martins et al., 1996; Conceição et al., 2003, Cunha et al., 2004, 2006, 2012). The few G. pallida populations found in Portugal may suggest that it was introduced after G. rostochiensis or there were only few introductions that were kept confined by their low natural mobility. Recently, the analysis of soils sampled in Portuguese potato fields revealed a spread of G. pallida (Camacho et al., 2017). In case of PCN positive detection, growers have to choose one of the following options as a phytosanitary measure: (a) culture with a PCN-resistant potato cultivar for a 3-year quarantine period, (b) culture with non-host species or (c) uncultivated land for a 6-year quarantine period. The use of resistant cultivars must be done carefully, in order to prevent the increase of G. pallida populations, which are more difficult to control as there are only a few available resistant cultivars.
Currently, in Portugal, there is a lack of detailed information on the geographical distribution of potato cyst nematodes, the correlation between their pattern, the potato cultivars and the near future implications for potato production. Therefore, this study aims to: (i) gather all PCN detections data in Portugal; (ii) carry out a molecular characterization of Portuguese Globodera isolates based on sequences of the ITS-rRNA region; (iii) study the phylogenetic relationships of Globodera spp. isolates from Portugal; and (iv) correlate cyst infestations with potato cultivars used.
The research reported herein includes PCN isolates collected from Portuguese potato fields for the national PCN surveys from 2013 to 2019, which made it possible to obtain an accurate assessment of the incidence and phylogenetic relationship of the two PCN species in the territory and their spread in different PCN-resistant cultivars fields.
Materials and Methods
Sampling
Soil was collected during the surveys between 2013 and 2019. Sampling was conducted by official inspectors of the National Plant Protection Organization (DGAV, Portugal). According to Annex II of DL 87/2010, sampling consists of a randomized collection of a soil volume with 1500 ml of soil/ha, harvested at least 100 subsamples/ha, preferably in a rectangular mesh, not less than 5 m wide and no more than 20 m long between sampling points, covering the entire field. Soil samples were stored in plastic bags and individually coded by the official services to ensure the anonymity of the samples during the analysis period. Potato field location at the county level and potato cultivars used in these fields were accessed only after analysis results.
The detection, identification and infestation rate of the PCN species were related to their sample location, given by DGAV, and species positive detection maps were made using the ArcMap 10.6 software (ESRI, United States), CAOP2017_PORTUGAL and CAOPP2017_DISTRITOS shapefiles (DGT, 2017).
Globodera spp. Molecular Identification
Cysts were extracted from soil samples using the Fenwick’s can method (Fenwick, 1940), according to the EPPO PM7/40 (3) protocol, isolated and counted under a binocular microscope (Leica MZ6, Germany). Cysts (1 to 20 depending on the sample infestation) containing eggs and juveniles were used for DNA extraction by means of the DNeasy Blood & Tissue Kit (Qiagen, Valencia, CA, United States) following the manufacturer’s instructions. The internal transcribed spacer region (ITS) of the ribosomal DNA repeat unit was amplified by duplex PCR for species identification. PCR reactions were performed in a 25 μL final volume using the Promega GoTaq Flexi DNA Polymerase Kit (Promega, Madison, United States), containing 1 μL template DNA, 5 μL GoTaq Flexi PCR buffer (2x), 1.5 mM MgCl2, 0.20 mM each dNTPs, 1.25 U GoTaq Flexi DNA Polymerase (Promega, Madison, United States) and 0.4 μM of each primer in a Biometra TGradient thermocycler (Biometra, Gottingen, Germany). The set of primers was composed of the forward primer ITS5 (5′-GGA AGT AAA AGT CGT AAC AAG G-3′) and the reverse PITSr3 (5′-AGC GCA GAC ATG CCG CAA-3′) for G. rostochiensis and PITSp4 (5′-ACA ACA GCA ATC GTC GAG-3′) for G. pallida (Bulman and Marshall, 1997). The amplification profile for ITS-rDNA consisted of an initial denaturation of 94°C for 2 min followed by 35 cycles of 94°C for 30 s, 55°C for 30 s, and 72°C for 30 s and a final extension of 72°C for 7 min (EPPO, 2017). The amplified products were loaded onto a 1.5% agarose gel containing 0.5 μg.mL–1 ethidium bromide and 0.5× Tris-borate-EDTA (TBE) running buffer and electrophoresed at 5 V/cm. Amplifications were visualized using the VersaDoc Gel Imaging System (Bio-Rad, United States). The expected length of the PCR products was 265 bp for G. pallida and 434 bp for G. rostochiensis. Possible contaminations were checked by including negative controls (no template control – NTC) in all amplifications.
Globodera spp. Phylogenetic Analysis
The ITS-rDNA region of 36 samples was amplified and sequenced using the primers 5′-CGT AAC AAG GTA GCT GTA G-3′ and 5′-TCC TCC GCT AAA TGA TAT G-3′ (Ferris et al., 1993). The expected length of PCR fragments is 1040 bp and corresponds to the 3′ end of 18S rDNA-ITS1-5.8S-ITS2-5′ of 28S rDNA. The thermal cycling conditions performed consisted of an initial denaturation of 95°C for 5 min followed by 40 cycles of 94°C for 30 s, 55°C for 30 s, and 72°C for 33 s and a final extension of 72°C for 7 min. Nucleotide sequences were edited and analyzed using BioEdit v7.2.0 (Hall, 2007). The resulting ITS-rDNA sequences were used as query at BLAST from NCBI GenBank to retrieve the most similar sequences within Globodera species for phylogenetic reconstruction, and they were deposited in the GenBank database (NCBI). Sequences from Globodera artemisiae, Globodera tabacum, and Globodera hypolysi were selected as outgroup taxa. All sequences were aligned by CLUSTAW (Thompson et al., 1994) with default parameters, trimmed manually and evaluated by Maximum Likelihood phylogeny using the best AIC (Akaike Information Criteria) nucleotide substitution model determined, namely Hasegawa-Kishino-Yano with Gamma Distribution (HKY + G). A bootstrap analysis with 1000 replications was also conducted to infer robustness of the phylogenetic tree. The CLC Main Workbench software package 8.11 was used for phylogenetic analysis.
Statistical Analysis
The differences obtained in the detection of the two PCN species in Portugal were achieved through a Z-test for the equality of two proportions using the software R2. Only soil samples with one or more cysts were used. The hypothesis tests were performed with a significance level α = 0.05.
Subsequently, the same test was used, with the same level of significance, to infer differences between PCN detections in north, center and south producing regions and between G. pallida and G. rostochiensis detections in fields with PCN susceptible and G. rostochiensis resistant potato cultivars.
Results and Discussion
During the survey period (2013–2019), 748 soil samples were collected throughout the country by the official services and tested in the plant health national reference laboratory (INIAV). Potato cyst nematodes were identified in 168 samples, representing 22.5% of the tested samples. Forty-eight samples tested positive for G. rostochiensis populations alone (28.6%) and 83 for G. pallida populations alone (49.4%). Mixed populations were found in 37 samples (22%) (Table 1). Statistics revealed that two species detections are significantly different (p-value = 0.00014, α = 0.05), G. pallida detection being greater than G. rostochiensis detection (p-value = 0.999, α = 0.05, which allows us to accept the null hypothesis that G. pallida detections are significantly greater to G. rostochiensis detections) between 2013 and 2019. These results contrast with those reported by Cunha et al. (2004) in which out of 423 tested populations (samples collected from various districts of continental Portugal), 83% were G. rostochiensis populations alone, 8% were G. pallida populations alone and 9% consisted of a mixture of the two species. This reverse situation can be explained due to the use of G. rostochiensis resistant potato cultivars, which has been considered the most widespread PCN species in Portugal.
TABLE 1
| Region | Positive detections | Negative detections | Total | ||||||||
| G. rostochiensis | G. pallida | Gr + Gp | Total | ||||||||
| Value | % | Value | % | Value | % | Value | % | Value | % | ||
| North | 30 | 40.5 | 39 | 52.7 | 5 | 06.8 | 74 | 42.5 | 100 | 57.5 | 174 |
| Center | 11 | 18.0 | 32 | 52.5 | 18 | 29.5 | 61 | 25.5 | 178 | 74.5 | 239 |
| South | 7 | 21.2 | 12 | 36.4 | 14 | 42.4 | 33 | 9.9 | 302 | 90.1 | 335 |
| Total | 48 | 28.6 | 83 | 49.4 | 37 | 22.0 | 168 | 22.5 | 580 | 77.5 | 748 |
Samples tested for Globodera rostochiensis and Globodera pallida in Portuguese regions between 2013 and 2019 (absolute values and %).
The use of G. rostochiensis resistant potato cultivars (Table 2), effective only against certain races of G. rostochiensis and with no resistance to G. pallida, is leading to the predominance in Portugal of the more difficult species to control, G. pallida. The obtained p-value (p-value = 0.996, α = 0.05) supported the null hypothesis, confirming that G. rostochiensis detection in potato fields with G. rostochiensis resistant cultivars is significantly smaller than G. rostochiensis detection in potato fields with PCN susceptible cultivars. With this result it is possible to infer that resistant cultivars are more efficient in reducing cyst infestations in potato production fields compared with susceptible cultivars fields. However, G. pallida detection in potato fields with G. rostochiensis resistant cultivars is not different to G. pallida detection (p-value = 0.2048, α = 0.05, which allows us to accept the null hypothesis that G. pallida detections in G. rostochiensis resistant cultivars are significantly similar to G. pallida detections in PCN susceptible cultivars) and G. rostochiensis detection in PCN susceptible potato cultivars fields (p-value = 0.5415, α = 0.05, which allows us to accept the null hypothesis that G. pallida detections in G. rostochiensis resistant cultivars are significantly similar to G. rostochiensis detections in PCN susceptible cultivars). With this result it is possible to infer that resistant cultivars used in Portugal allow us to reduce G. rostochiensis cysts infestation but has no influence on G. pallida cysts infestations in potato production fields. Therefore, the use of G. rostochiensis resistant potato cultivars has led to a decrease in G. rostochiensis detection but has no influence on G. pallida detection. These results agree with the published literature (Minnis et al., 2002; Pickup et al., 2019).
TABLE 2
| Cultivar | Resistance status | Resistance status | |||
| G. rostochiensis | G. pallida | Cultivar | G. rostochiensis | G. pallida | |
| Agria | R | S | Jelly | R | S |
| Alcander | R | R | Kenebeck | S | S |
| Allison | R | R | Lady rosetta | R | S |
| Asterix | R | S | Manitou | R | S |
| Aurea | R | S | Monalisa | S | S |
| Baraka | R | S | Monte carlo | R | R |
| Bellarosa | R | Olho de perdiz | R | ||
| Camberra | R | Picasso | R | ||
| Carlita | R | Red Lady | R | ||
| Colomba | R | S | Red scarlet | R | |
| Daifla | R | S | Romano | S | S |
| Delila | S | S | Rudolph | S | S |
| Désirée | S | S | Soleny | S | S |
| Evolution | R | S | Stemster | R | |
| Evora | S | S | Taurus | R | S |
| Hermes | S | S | Yona | R | S |
Potato cultivars grown in Portuguese sampled fields (2013–2019) and their resistance status toward Globodera rostochiensis and Globodera pallida.
R, resistant; S, susceptible.
There is no available data to infer about the use of G. pallida resistant potato cultivars. This raises the question of whether phytosanitary measures are effective or whether they are contributing to the increase of G. pallida, as also reported in the United Kingdom (Minnis et al., 2002). On the other hand, the market has caused potato growers to predominantly use G. rostochiensis resistant potato cultivars (i.e., Aurea, Agria, Lady rosetta, Taurus), and this is the main cause of G. pallida detections increase.
The geographical distribution of PCN infestations in Portugal is illustrated in Figures 2, 3, which present the infestation rate in counties with positive detections of G. rostochiensis and G. pallida between 2013 and 2019. This information completes a picture of the PCN situation in Portugal to date.
FIGURE 2
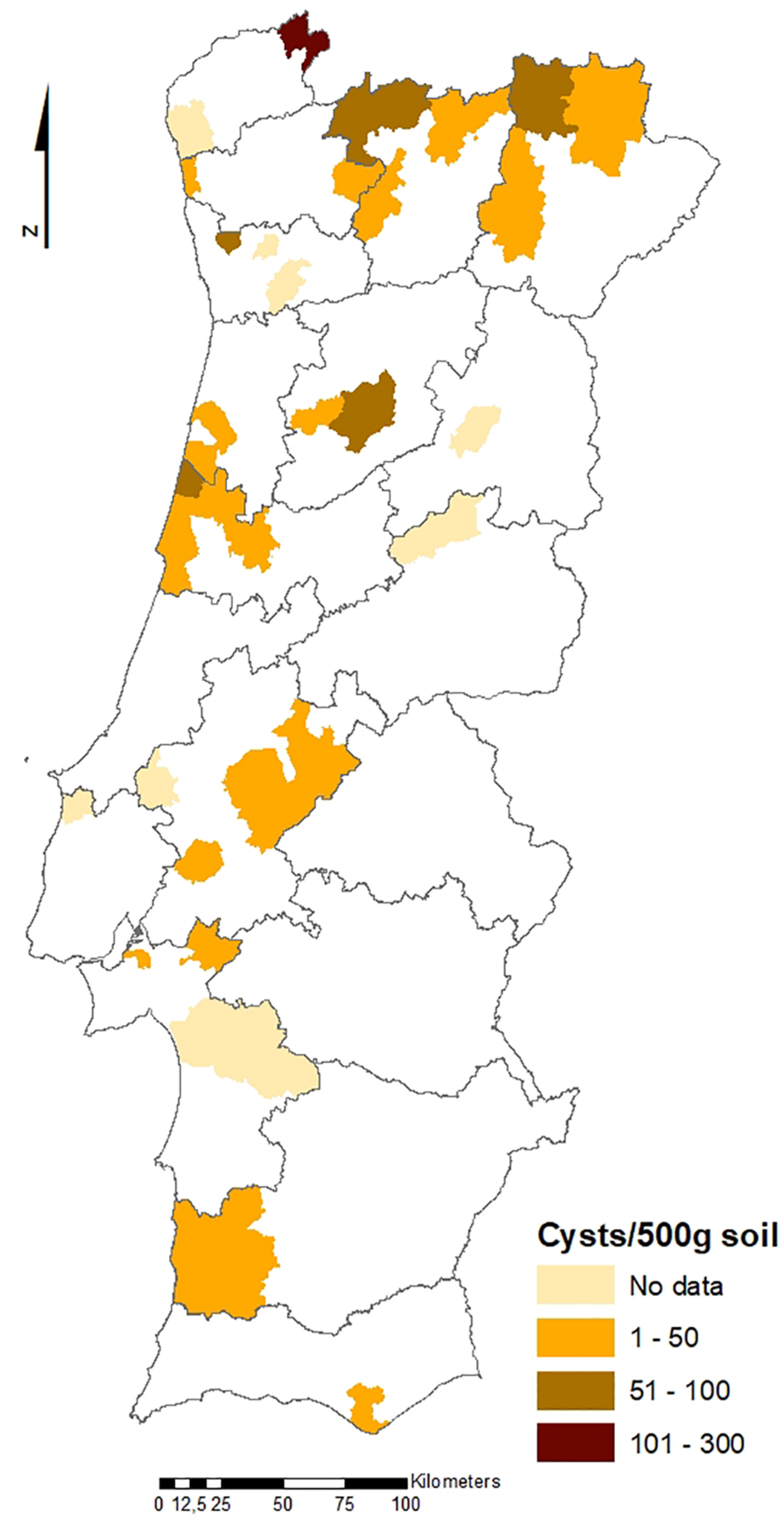
Counties with positive detections of Globodera rostochiensis in Portugal between 2013 and 2019.
FIGURE 3
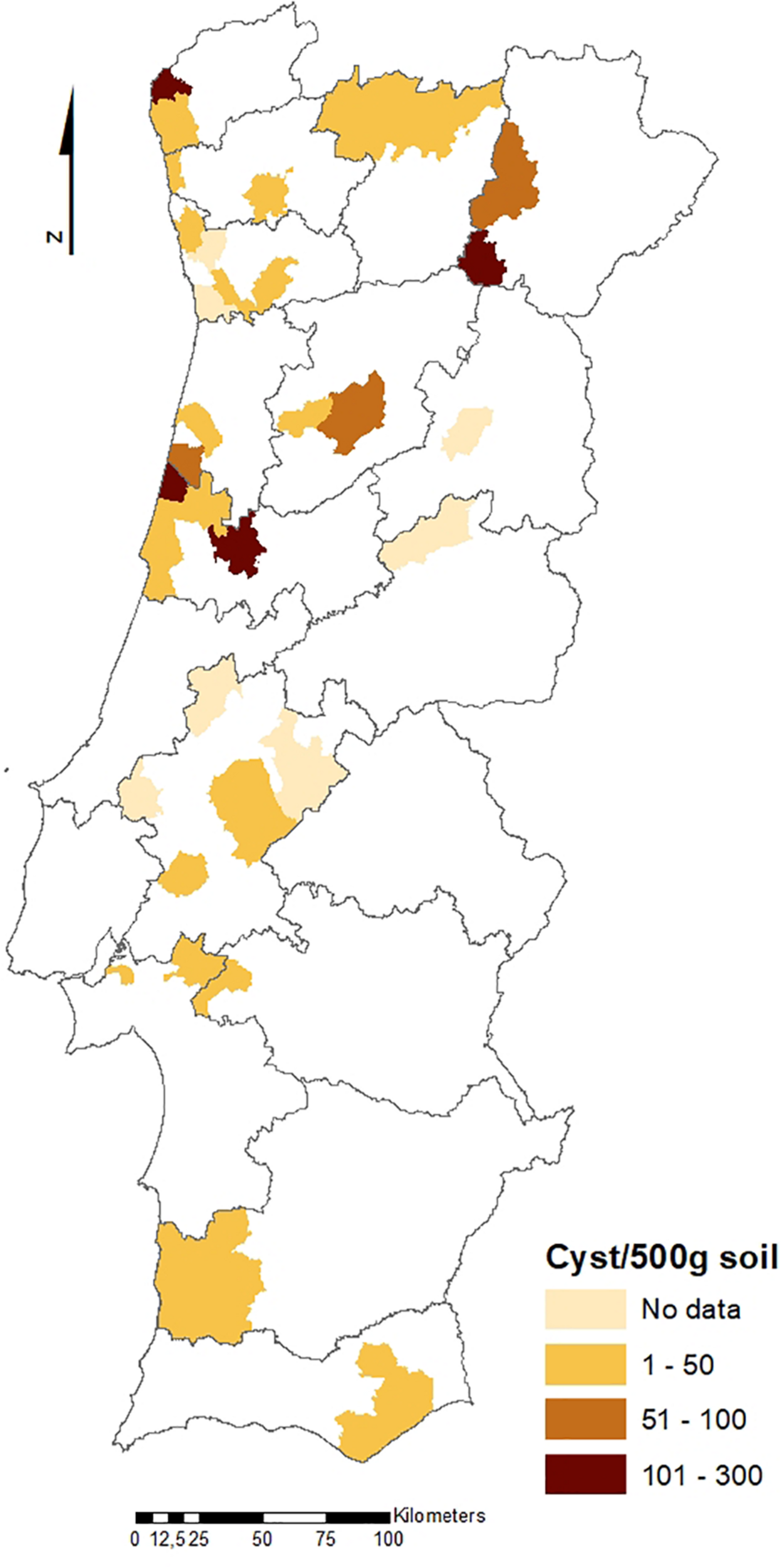
Counties with positive detections of Globodera pallida in Portugal between 2013 and 2019.
According to these results, the incidence of PCN in Portugal is quite high, and both species are currently present in all potato producing regions of the country. PCN detections in the different regions are significantly different. Statistics revealed that the Northern PCN detection is greater than the Center PCN detection (p-value = 0.998, α = 0.05, which allows us to accept the null hypothesis that PCN detection in northern fields is significantly greater than PCN detection in central fields) and the Center PCN detection is greater than the Southern (Lisbon and Tagus Valley, Alentejo and Algarve regions) PCN detection (p-value = 1, α = 0.05, which allows us to accept the null hypothesis that PCN detection in central fields is significantly greater than the PCN detection in southern fields), meaning that PCN detection increases from south to north (see Figures 2, 3), where PCN were first detected and nematode reproduction are happening for a longer period. These results are also in line with previous reports, which state that the cysts are adapted to higher altitudes (Jones et al., 2017) since the altitude grows from south to northern regions in Portugal.
To infer the phylogenetic relationship of Globodera isolates, ML analyses were performed (Figure 4). Two major clades, highly supported, can be observed: clade (I) with sub-clades G. rostochiensis and G. pallida and clade (II) with the sub-clades Globodera sp. recently re-detected. Within the first clade, two sub-clades were formed with G. rostochiensis and the related species G. tabacum and G. pallida. The second clade groups a Portuguese Globodera sp., discovered in 1997 (Reis, 1997; Sabo et al., 2002) and not re-detected until recently (data not shown), and their most closely related Globodera species, G. hypolysi and G. artemisiae. As can be clearly seen, no spatial-temporal relation can be redrawn evidencing the co-existence between the two major species of Globodera in Portugal. These results are in accordance with those reported by Cunha et al. (2012), who reported that no relationship could be found between the two-dimensional electrophoresis protein patterns or virulence behavior of the isolates and their geographic origin within Portugal.
FIGURE 4
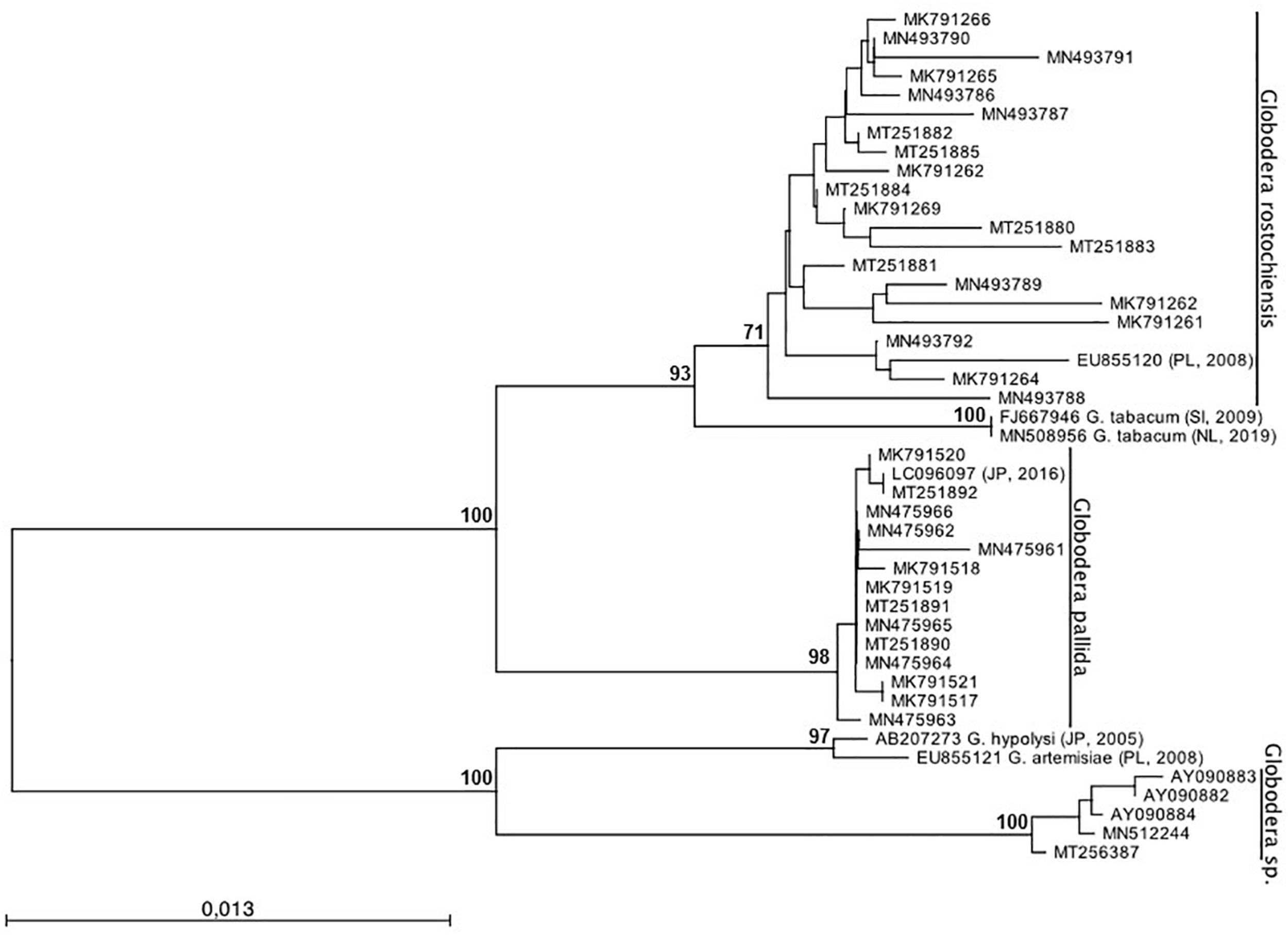
Phylogenetic relationships of 36 Globodera sp. isolates collected from Portugal based on the sequence alignment of the ITS-rDNA loci. The condensed phylogenetic tree was generated using the Maximum Likelihood method based on the HKY + G model with 1,000 bootstrap replications. Bootstrap values are indicated at the nodes (bootstrap higher than 70%). The analysis involved a total of 45 nucleotide sequences. All positions containing gaps and missing data were eliminated. Globodera tabacum, G. hypolysi, and G. artemisiae were used as outgroup.
It is also worth noting that the topology differs between G. rostochiensis and G. pallida sub-clades. The first is more branched, with 96–100% of similarity, showing more genetic variability due to being present for a longer period in Portugal, while the second is flatter, with 99–100% of similarity, showing more identical sequences (Supplementary Table 1).
Concerning the new species Globodera sp. (Reis, 1997; Sabo et al., 2002), re-detected recently in Portugal, it is out of the scope of this work, but additional research is being carried out to determine its pathogenicity and impact on potato.
The nucleotide sequences obtained in this study were deposited in the GenBank database (NCBI) under the accession numbers given in Table 3.
TABLE 3
| Globodera species | GenBank accession number | Locality | Collection code/year | Sequence length (bp) | NCBI BLAST homology (%) | |
| Globodera rostochiensis | EU855120 | Poland | * | 2008 | 4064 | 100.00 |
| MK791260 | Coimbra | 650P | 2014 | 893 | 100.00 | |
| MK791261 | Montalegre | 5244 | 2015 | 888 | 100.00 | |
| MK791262 | Montalegre | 5245 | 2015 | 909 | 100.00 | |
| MK791263 | Viseu | 9996 | 2018 | 871 | 98.62 | |
| MK791264 | Mirandela | 14598 | 2018 | 969 | 99.79 | |
| MK791265 | Mirandela | 14600 | 2018 | 871 | 99.89 | |
| MK791266 | Bragança | 14601 | 2018 | 909 | 99.89 | |
| MN493786 | Montalegre | 13486 | 2017 | 937 | 99.25 | |
| MN493787 | Chaves | 8850 | 2016 | 937 | 98.50 | |
| MN493788 | Viseu | 9610 | 2017 | 920 | 98.58 | |
| MN493789 | Viseu | 5967 | 2016 | 936 | 98.82 | |
| MN493790 | Viseu | 7047 | 2017 | 973 | 100.00 | |
| MN493791 | Odemira | 3663 | 2018 | 915 | 99.13 | |
| MN493792 | Aveiro | 7913 | 2018 | 897 | 99.78 | |
| MT251880 | Coimbra | 1252 | 2019 | 929 | 99.14 | |
| MT251881 | Montalegre | 1681-2 | 2019 | 909 | 99.34 | |
| MT251882 | Montalegre | 1681-6 | 2019 | 924 | 99.89 | |
| MT251883 | Chaves | 1681-7 | 2019 | 933 | 98.71 | |
| MT251884 | Mirandela | 1681-10 | 2019 | 928 | 99.35 | |
| MT251885 | Melgaço | 1249-1 | 2019 | 946 | 98.94 | |
| Globodera tabacum | FJ667946 | Slovenia | * | 2009 | 923 | 99.46 |
| MN508956 | Netherlands | NL:c6876 | 2018 | 953 | 99.89 | |
| Globodera pallida | LC096097 | Japan | * | 2016 | 964 | 100.00 |
| MN475961 | Viseu | 3876 | 2014 | 898 | 99.33 | |
| MN475962 | S. Magos | 4261 | 2016 | 970 | 99.90 | |
| MN475963 | S. Magos | 15731 | 2018 | 933 | 99.03 | |
| MN475964 | Vagos | 9993 | 2018 | 977 | 98.89 | |
| MN475965 | Montalegre | 14002 | 2017 | 914 | 99.89 | |
| MN475966 | Esposende | 5087 | 2016 | 926 | 99.56 | |
| MK791517 | Penafiel | 4694 | 2015 | 873 | 100.00 | |
| MK791518 | Viseu | 5961 | 2016 | 890 | 99.22 | |
| MK791519 | Guimarães | 11309 | 2018 | 901 | 99.78 | |
| MK791520 | Mirandela | 14593 | 2018 | 878 | 100.00 | |
| MK791521 | Mirandela | 14599 | 2018 | 873 | 100.00 | |
| MT251890 | Vagos | 1223-7 | 2019 | 938 | 100.00 | |
| MT251891 | Aveiro | 1223-8 | 2019 | 915 | 99.89 | |
| MT251892 | Mira | 1086-3 | 2019 | 913 | 99.67 | |
| Globodera sp. | AY090883 | Bouro | * | 1997 | 908 | 99.89 |
| AY090882 | Canha | * | 1997 | 908 | 99.89 | |
| AY090884 | Ladoeiro | * | 1997 | 908 | 99.78 | |
| MN512244 | Montijo | 12031 | 2018 | 953 | 99.45 | |
| MT256387 | Lagameças | 1479-2 | 2019 | 913 | 99.67 | |
| Globodera artemisiae | EU855121 | Poland | * | 2008 | 4092 | 100.00 |
| Globodera hypolysi | AB207273 | Japan | * | 2005 | 909 | 99.45 |
Globodera spp. isolates sequenced in the present study (E-value = 0.0).
*Sequences available from GenBank, NCBI.
Phytosanitary measures have been taken to prevent further spread of Globodera spp. in recent years. In the case of G. rostochiensis, up until now the dominant species, measures include non-host crop rotation (for 6 years), fallow (for 6 years) or growing of resistant potato cultivars (for 3 years). The use of resistant cultivars containing the H1 gene (single dominant resistance gene for G. rostochiensis) (Gebhardt et al., 1993), as already shown, is effective against many populations of G. rostochiensis and is likely to be an advantageous management tactic to reduce population densities and thereby yield losses. However, the deployment of resistance in such cultivars may have caused the predominance of G. pallida in Portugal, as already predicted by Cunha et al. (2004) and statistically verified in this study.
Therefore, it is urgent to follow a new approach for the management of PCN, mainly G. pallida. Non-infested areas need to be managed to minimize the opportunities for the introduction of Globodera species. On the other hand, and in infested soils, a greater use of integrated control strategies (such as crop rotation, solarization, trap cropping, biofumigation and selected nematicides) (Evans and Haydock, 2000; Alptekin, 2011; Davie et al., 2019), in addition to PCN-resistant potato cultivars, should be a priority. These interactions require careful research into the effects of one or another strategy under a specific set of environmental conditions and a specific nematode infestation level. The efficacy of the integrated program will be determined by the interaction, overlap and complementarity of the various components. Despite the difficulties associated with G. pallida resistance being quantitatively inherited, the breeding of more resistance with different R-genes to avoid PCN capacity to overcome the plant resistance and commercially attractive cultivars is highly important. As G. pallida field populations tend to show increased virulence toward a particular partially resistant cultivar each time that it is grown (Trudgill et al., 2003; Pickup et al., 2019), potato growers would need a choice of different cultivars to allow effectiveness to be maintained. Currently, there are insufficient alternatives to partially resistant cultivars for growers to meet the requirements of markets.
Statements
Data availability statement
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found in the article/Supplementary Material.
Author contributions
MC, MI, EA, and MM: conceptualization. MC: investigation and writing – original draft. MC, MI, EA, FN, CV, and LR: methodology. MI, EA, and MM: supervision. MC, MI, EA, MM, and CV: writing – review and editing. All authors contributed to the article and approved the submitted version.
Funding
This work was funded by National Funds through FCT – Foundation for Science and Technology under the Ph.D. fellowships BD 138724/2018 and through the R&D Unit, UIDB/04551/2020 (GREEN-IT – Bioresources for Sustainability).
Acknowledgments
Thanks to DGAV (The National Plant Protection Authority) for the information to support this study.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fpls.2020.606178/full#supplementary-material
Supplementary Table 1Sequence percentage of similarity.
References
1
AlptekinY. (2011). Integrated pest management of potatoes.Agric. Sci.2297–300. 10.4236/as.2011.23039
2
BulmanS.MarshallJ. (1997). Differentiation of Australasian potato cyst nematode (PCN) populations using the polymerase chain reaction (PCR).N. Z. J. Crop Hortic. Sci.25123–129. 10.1080/01140671.1997.9513998
3
CamachoM. J.NóbregaF.LimaA.MotaM.InácioM. L. (2017). Morphological and molecular identification of the potato cyst nematodes (Globodera rostochiensis and G. pallida) in Portuguese potato fields.Nematology19883–889. 10.1163/15685411-00003094
4
ChristoforouM.PantelidesI. S.KanetisL.IoannouN.TsaltasD. (2014). Rapid detection and quantification of viable potato cyst nematodes using qPCR in combination with propidium monoazide.Plant Pathol.631185–1192. 10.1111/ppa.12193
5
ConceiçãoI.SantosM.AbrantesI.SantosS. (2003). Using RAPD markers to analyse genetic diversity in Portuguese potato cyst nematode populations.Nematology5137–143. 10.1163/156854102765216759
6
CunhaM.ConceiçãoI.AbrantesI.EvansK.SantosS. (2004). Characterisation of potato cyst nematode populations from Portugal.Nematology655–58. 10.1163/156854104323072928
7
CunhaM.ConceiçãoI.AbrantesI.SantosS. (2006). Assessment of the use of high-performance capillary gel electrophoresis to differentiate isolates of Globodera spp.Nematology8139–146. 10.1163/156854106776179980
8
CunhaM.CunhaM.ConceiçãoI.AbrantesI.SantosS. (2012). Virulence assessment of Portuguese isolates of potato cyst nematodes (Globodera spp.).Phytopathol. Mediterr.5151–68. 10.14601/Phytopathol_Mediterr-9517
9
DavieK.PickupJ.BlockV.KuhfussL.KettleH.EwingD.et al (2019). Potato cyst nematodes in Scotland – management challenges and options to overcome these.Asp. Appl. Biol.14225–31.
10
DGAV (2015). Organismos de Quarentena. Acções Realizadas. Relatórios de 2014.Lisbon: Direção-Geral de Alimentação e Veterinária.
11
DGT (2017). Carta Administrativa Oficial de Portugal - CAOP2017.Direção-Geral do Território. Available online at: https://www.dgterritorio.gov.pt/cartografia/cartografia-tematica/caop#tab-24b1t-1(accessed December 2018).
12
EPPO (2017). Globodera rostochiensis and Globodera pallida PM 7/40 (4). European and mediterranean plant protection organization.EPPO Bull.47174–197. 10.1111/epp.12391
13
EPPO (2020). EPPO Global Database: Globodera pallida.European and Mediterranean Plant Protection Organization. Available online at: https://gd.eppo.int/taxon/HETDPA/distribution(accessed October 30, 2020).
14
EvansK.HaydockP. (2000). Potato cyst nematode management – present and future.Asp. Appl. Biol.5991–97.
15
FenwickD. (1940). Methods for the recovery and counting of cysts of Heterodera schachtii from soil.J. Helminthol.18155–172. 10.1017/S0022149X00031485
16
FerrisV. R.FerrisJ. M.FaghihiJ. (1993). Variation in spacer ribosomal DNA in some cyst-forming species of plant parasitic nematodes.Fundam. Appl. Nematol.16177–184.
17
GebhardtC.MugnieryD.RitterE.SalaminiF.BonnelE. (1993). Identification of RFLP markers closely linked to the H1 gene conferring resistance to Globodera rostochiensis in potato.Theor. Appl. Genet.85541–544. 10.1007/BF00220911
18
GrenierE.FournetS.PetitE.AnthoineG. (2010). A cyst nematode ‘species factory’ called the Andes.Nematology12163–169. 10.1163/138855409X12573393054942
19
HafezS. L.SundararajP.HandooZ. A.SkantarA. M.CartaL. K.ChitwoodD. J. (2007). First report of the pale cyst nematode, Globodera pallida, in the United States.Plant Dis.91:325. 10.1094/PDIS-91-3-0325B
20
HallT. (2007). BioEdit: Biological Sequence Alignment Editor for Win95/98/NT/2K/XP. Available online at: http://www.mbio.ncsu.edu/BioEdit/bioedit.html
21
InácioM. L.CamachoM. J.SerraC.CordevileC.CordeiroL.AndradeE. (2020). First report of the potato cyst nematode, Globodera rostochiensis, on potato in the Azores, Portugal.Plant Dis.104:1874. 10.1094/PDIS-07-19-1429-PDN
22
INE (2011). Superfície das Culturas Temporárias (ha) por Localização Geográfica (NUTS - 2001) e Tipo (Culturas temporárias); Decenal – INE, Recenseamento Agrícola – Séries Históricas. Instituto Nacional de Estatística. Available online at: https://www.ine.pt/xportal/xmain?xpid=RA2009&xpgid=ine_ra2009_indicador&contexto=ind&indOcorrCod=0004356&selTab=tab10&xlang=pt(accessed November 3, 2019).
23
JonesL. M.KoehlerA. K.TrnkaM.BalekJ.ChallinorA. J.AtkinsonH. J.et al (2017). Climate change is predicted to alter the current pest status of Globodera pallida and G. rostochiensis in the United Kingdom.Glob. Chang. Biol.234497–4507. 10.1111/gcb.13676
24
LavrovaV. V.MatveevaE. M.ZinovievaS. V. (2017). Expression of genes, encoded defense proteins, in potato plants infected with the cyst-forming nematode Globodera rostochiensis (Wollenweber, 1923) Behrens, 1975 and modulation of their activity during short-term exposure to low temperatures.Biol. Bull. Russ. Acad. Sci.44128–136. 10.1134/S1062359017020108
25
LimaF. S. O.MattosV. S.SilvaE. S.CarvalhoM. A. S.TeixeiraR. A.SilvaJ. C.et al (2018). “Nematodes affecting potato and sustainable practices for their management,” in Potato: From Incas to All Over the World, ed.YildizM. (London: IntechOpen), 107. 10.5772/intechopen.73056
26
MacaraA. (1963). Aspectos sobre a importância dos nemátodos de interesse agrícola em Portugal e no ultramar português.Agros46367–384.
27
MartinsF.SantosS.AbreuC.AbrantesI.EvansK. (1996). “Potato cyst nematodes – the situation in the north east part of Portugal,” in Proceedings of the Abstracts Book of the 13th Triennial Conference of EAPR, Veldhoven, 369–370.
28
MburuH.CortadaL.MwangiG.GitauK.KirigaA.KinyuaZ.et al (2018). First report of potato cyst nematode Globodera pallida infecting potato (Solanum tuberosum) in Kenya.Plant Dis.12:1671. 10.1094/PDIS-11-17-1777-PDN
29
MinnisS. T.HaydockP. P.IbrahimS. K.GroveI. G. (2002). Potato cyst nematodes in England and Wales occurrence and distribution.Ann. Appl. Biol.140187–195. 10.1111/j.1744-7348.2002.tb00172.x
30
NiragireI.CouvreurM.KarssenG.UwumukizaB.BertW. (2019). First report of potato cyst nematode (Globodera rostochiensis) infecting potato (Solanum tuberosum L.) in Rwanda.Plant Dis.104:293. 10.1094/PDIS-04-19-0891-PDN
31
PickupJ.DavieK.BlockV.KuhfussL.KettleH.EwingD.et al (2019). Potato cyst nematodes in Scotland – where are we now and where are we heading.Asp. Appl. Biol.14211–17.
32
ReisL. G. (1997). Some morphological characters of a remarkable Globodera species (Nematoda: Heteroderidae) occurring in Portugal.Parasitol. Port.4126–127.
33
SaboA.ReisL. G.KrallE.Mundo-OcampoM.FerrisV. R. (2002). Phylogenetic relationships of a distinct species of Globodera from Portugal and two Punctodera species.J. Nematol.34263–266.
34
SantosS.EvansK.AbreuC.MartinsF.AbrantesI. (1995). A review of potato cyst nematodes in Portugal.Nematol. Mediterr.2335–42.
35
SantosS.FernandesM. (1988). The occurrence of Globodera rostochiensis and G. pallida in Portugal.Nematol. Mediterr.16:145.
36
SkarbilovichT. S. (1959). On the structure and systematics of the nematode order Tylenchida Thorne, 1949.Acta Pol.7117–132.
37
StoneA. (1973). Heterodera pallida n.sp. (Nematoda: Heteroderidae), a second species of potato cyst nematode.Nematologica18:591. 10.1163/187529272X00179
38
ThompsonJ. D.HigginsD. G.GibsonT. J. (1994). CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice.Nucleic Acids Res.224673–4680. 10.1093/nar/22.22.4673
39
TrudgillD. L.ElliottM. J.EvansK.PhillipsM. S. (2003). The white potato cyst nematode (Globodera pallida) – a critical analysis of the threat in Britain.Ann. Appl. Biol.14373–80. 10.1111/j.1744-7348.2003.00073.x
40
ViaeneN. (2016). “Ring testing of diagnostic methods for the identification of potato cyst nematodes and assessing resistance of potato cultivars,” in Poster at the Report of the Joint EEC-EPPO Workshop on Euphresco, (Moscow: Eurasian Economic Commission (EEC) and the European and Mediterranean Plant Protection Organisation (EPPO)).
41
WollenweberH. (1923). Krankheiten und BeschSdigungen der Kartoffel.Berlin: Arb. Forsch. Inst. Kartof, 56.
Summary
Keywords
Globodera pallida, Globodera rostochiensis, Solanum tuberosum, disease, Heteroderidae
Citation
Camacho MJ, de Andrade E, Mota M, Nobrega F, Vicente C, Rusinque L and Inácio ML (2020) Potato Cyst Nematodes: Geographical Distribution, Phylogenetic Relationships and Integrated Pest Management Outcomes in Portugal. Front. Plant Sci. 11:606178. doi: 10.3389/fpls.2020.606178
Received
14 September 2020
Accepted
16 November 2020
Published
17 December 2020
Volume
11 - 2020
Edited by
Christina Cowger, Plant Science Research Unit (USDA-ARS), United States
Reviewed by
Aarti Bairwa, Indian Council of Agricultural Research (ICAR), India; Zafar Ahmad Handoo, United States Department of Agriculture, United States; Julio Carlos Pereira Silva, Federal University of Santa Maria, Brazil; Valdir Correia, Instituto Federal de Educação, Ciência e Tecnologia do Tocantins (IFTO), Brazil
Updates
Copyright
© 2020 Camacho, de Andrade, Mota, Nobrega, Vicente, Rusinque and Inácio.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Maria Lurdes Inácio, lurdes.inacio@iniav.pt
This article was submitted to Plant Pathogen Interactions, a section of the journal Frontiers in Plant Science
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.