- 1Laboratory for Plant Physiology, Faculty of Science, University of Sarajevo, Sarajevo, Bosnia and Herzegovina
- 2Centre of the Region Haná for Biotechnological and Agricultural Research, Czech Advanced Technology and Research Institute, Palacký University, Olomouc, Czechia
- 3Centre of the Region Haná for Biotechnological and Agricultural Research, Department of Genetic Resources for Vegetables, Medicinal and Special Plants, Crop Research Institute, Olomouc, Czechia
Salvia L. is the largest genus in the family Lamiaceae, with about 1,000 species and a nearly cosmopolitan distribution. Salvia species are used in both traditional and conventional medicines, and other numerous industries, such as spices and perfumes. The number of papers dealing with Salvia exceeds 12,000 and mostly investigates their chemical composition and bioactive properties. A smaller proportion of papers however consider environmental factors, mostly on the effects of microclimate conditions on its geographic distribution along an altitudinal or longitudinal gradient, and very few studies can be found on the effects of emerging stressors on the commercial production of sages of medicinal and economical importance. Here, we summarize available data on the essential oil composition of three economically important sages from the Mediterranean area, that is, Salvia officinalis, Salvia officinalis subsp. lavandulifolia, and Salvia fruticosa, and the effects of climate-related environmental stressors on their chemical profiles. Environmental stress factors, such as an increase in soil salinity and aridity, and changes in annual average temperatures, are going to impose a serious risk on the commercial production of sage essential oils, which are commercially produced in many European countries. This review highlights the already confirmed effects of these stressors on three selected Salvia species and consequently the importance of mitigating the effects of climate change on the commercial production of these essential oils.
Introduction
The genus Salvia L. includes close to 1,000 species from all around the globe, and it is considered one of the largest genera in the Lamiaceae family. The diversification within the genus Salvia is considered to belong to biogeographical lines including three distinguished Salvia clades: clade I mostly distributed in the Old World and one lineage from the New World; clade II exclusively comprised of plants distributed in the New World; and, clade III which only includes the Asian lineage Salvia species (Walker et al., 2004). There are 36 Salvia species listed on European soil, including many endemic species mostly found in the Iberian and Balkan Peninsula with significant importance for perfumery, pharmaceutical, and other industries (Ardestani and Ghahfarrokhi, 2021). Some of the widely used Salvia species in traditional medicine are Salvia officinalis L., Salvia fruticosa Mill. (syn. Salvia triloba L.), Salvia miltiorrhiza Bunge, Salvia hispanica L., Salvia sclarea L., Salvia triloba L., Salvia stenophylla Burch. ex Benth., and Salvia repens Burch. ex Benth (Lopresti, 2016; Llurba-Montesino and Schmidt, 2018). However, in this review, we cover the selected Salvia species of European origin that possess the highest economical values and which are thought to be under the influence of climate-related environmental stresses.
Although species of Salvia L. are known since ancient times, plants belonging to this genus are still in scientific focus. The literature overview performed in Scopus® search engine shows over 12,000 reports on this genus, and more than half were published in the last 10 years. Most of the published reports are in the fields of medicine, pharmacology, and agricultural sciences (Figure 1). A large proportion of these studies (more than 1,000) was on essential oil composition, confirming that research on these complex mixtures is still important for their use in the pharmaceutical and food industries. The most investigated European species are S. officinalis, S. officinalis subsp. lavandulifolia, and S. fruticosa. Hence, we have decided to narrow this review to these three species.
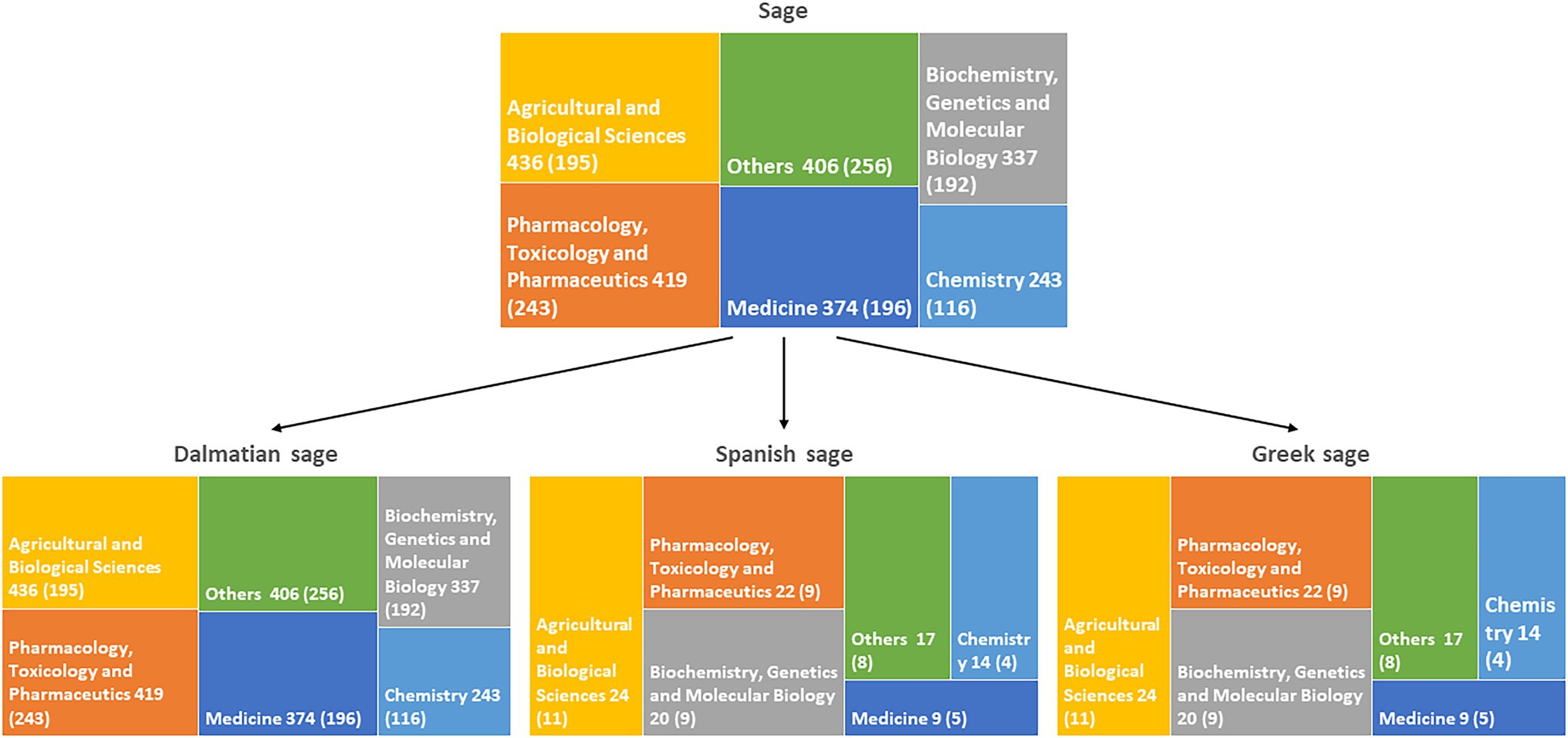
Figure 1. Numbers of studies by subject area on Salvia genus and species reviewed here. Numbers of studies since 2012 are indicated in the brackets.
There is a significant number of papers on the effects of environmental factors on Salvia species. Research shows that the composition and concentration of volatile compounds in Salvia are dependent not only on genetic and seasonal factors but also on environmental factors (Ramezani et al., 2020). Climate change is, with no doubt, one of the concerning, human-induced, environmental threats for plant biodiversity. The European Union is leading the way toward becoming the first climate-neutral zone by 2050 through the limitation of global warming, which was confirmed by the European Commission’s launch of the Green Deal in December 2019 (European Commission, 2019). Changes in the worlds’ climate could have serious impacts on the future distribution and industrial value of plants since an increase in global temperatures, disruption, and changes in hydroclimatic factors could lead to loss of habitats (Pressey et al., 2007).
Essential Oils of Salvia L.
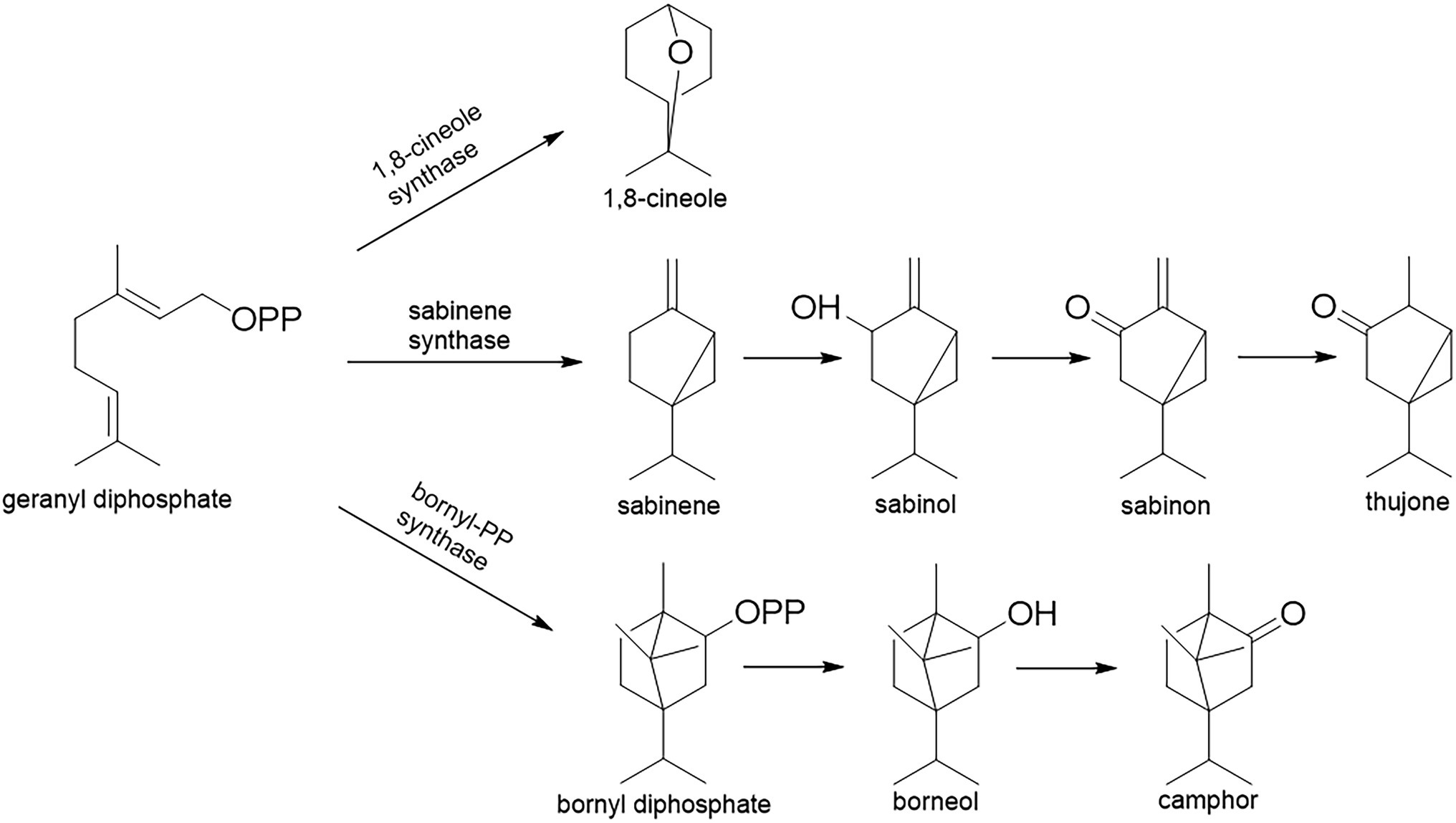
The majority of Salvia species have a long tradition of use in folk medicine, but also as flavoring agents and ornamental plants [Code of Federal Regulation (21CFR172.510), 2021]. For example, Salvia officinalis L. and S. fruticosa Mill. are used as a mouth wash against the inflammations of the oral cavity. Terpenes from essential oils are the main carriers of the bioactivity of Salvia sp. They are synthesized in glandular hairs (trichomes), and they play a crucial role in the plants’ defense against phytophagous insects or pests (Giuliani et al., 2017). The essential oils of the Salvia species reviewed here mainly consist of cyclic oxygenated monoterpenes, that is, 1,8-cineole (eucalyptol), thujones (α- and β-), and camphor (Li et al., 2015). These compounds are synthesized via three different biosynthetic pathways, but all of them include cyclization of geranyl diphosphate (GPP; Figure 2): (i) 1,8-cineole via 1,8-cineole synthase (geranyl-diphosphate diphosphate-lyase; EC 4.2.3.108); (ii) thujones via sabinene synthase (geranyl-diphosphate diphosphate-lyase; EC 4.2.3.110), which firstly forms sabinene that is transformed to thujone via several steps which include the oxidative enzyme cytochrome P450, but also reductase; (iii) camphor via bornyl-PP-synthase [(+)-bornyl-diphosphate lyase; EC 5.5.1.8] that forms bornyl diphosphate which is then hydrolyzed and oxidized to camphor (Selmar and Kleinwächter, 2013; Radwan et al., 2017).
However, it is well known that the composition of essential oils strongly depends on environmental conditions, although with the accelerating climate change this can potentially be problematic for the future production and use of these oils in the pharmaceutical and food industry. The majority of studies on the essential oil composition of Salvia species are merely descriptive, that is, only dealing with the content percentage of the components (see references in Supplementary Table 1), but solid explanations about the differences in composition are lacking. Yet, few studies about the impact of abiotic stressors on terpene production can be found in the literature. According to Selmar and Kleinwächter (2013), drought stress induces stomatal closure and decreases CO2 uptake. As a result, plants respond by moving biosynthesis toward compounds that can reduce reactive oxygen species. In addition, Radwan et al. (2017) studied the metabolism of monoterpenes in S. officinalis grown under drought stress, and they found that enzymes (Figure 2) of stressed plants are upregulated, especially bornyl-PP synthase, resulting in higher levels of camphor produced (Figure 3). A common problem found in the Salvia literature lies in different methodologies used by the authors, for example, there is no consensus on the selection of the plant organ to be studied, its proper drying technique, homogenization, and even on the method used for identification of the constituents. Similar obstacles were pinpointed for the phytochemical analysis of selected Mentha species, also an economically important genus of the Lamiaceae family (Ćavar Zeljković et al., 2021b). Turek and Stintzing (2013) reviewed possible changes in terpene composition and factors affecting their stability, both in dried plant material or already isolated essential oil. In addition, Ćavar Zeljković et al. (2021a) showed that the content percentage of terpenes, calculated from the relative area of chromatographic peaks, cannot be a sufficient method for the chemical characterization of essential oils, especially if they are used in the food and pharmaceutical industries. They pointed out that levels of at least the major constituents should be presented in exact concentrations.
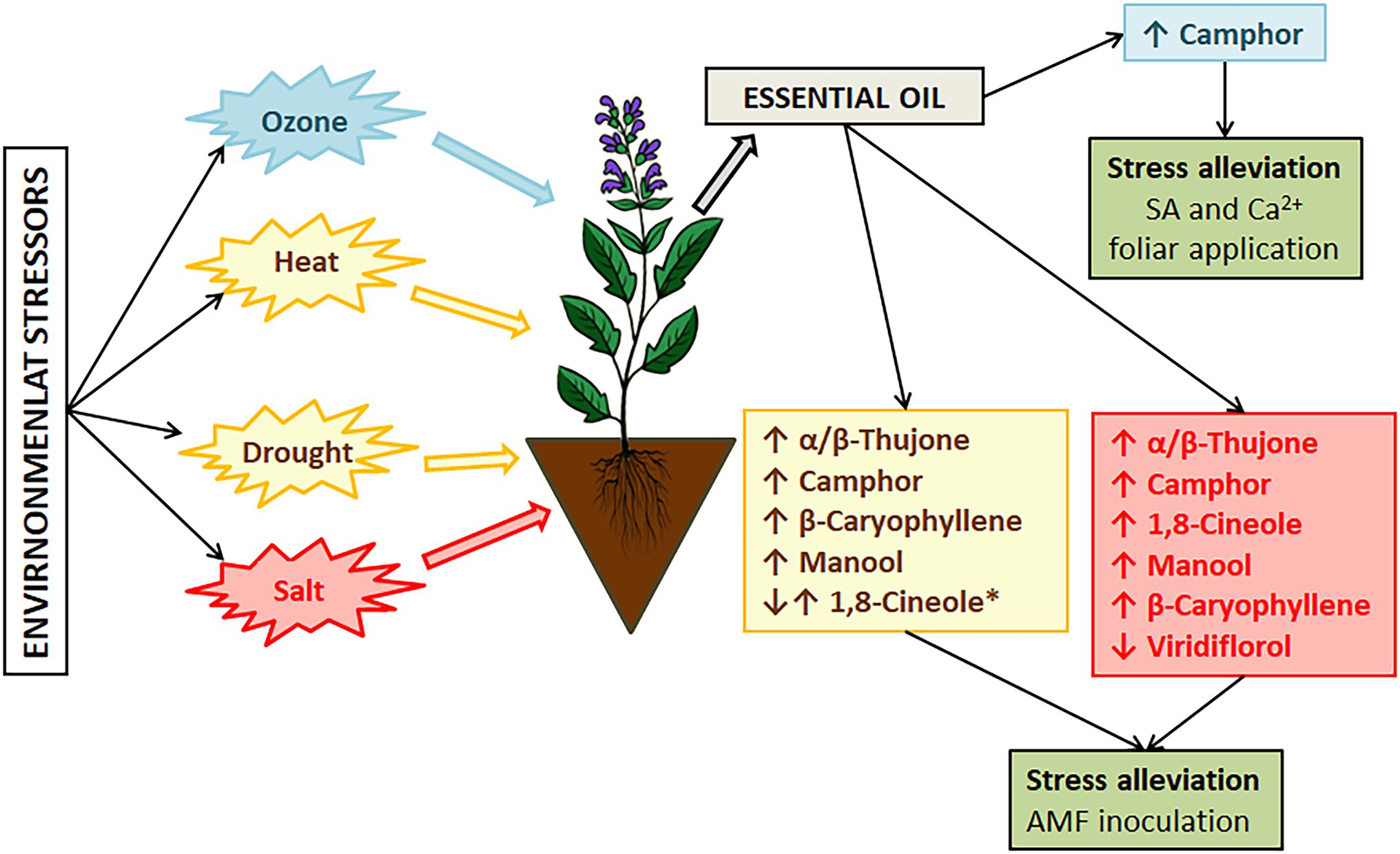
Figure 3. The major changes in sage essential oil composition induced by environmental stressors. *Changes of 1,8-cineole levels also depends on sage species.
Despite the medicinal potential of Salvia essential oils being well established, precautions must be made since some of the major compounds of the oils are toxic to humans. For example, 1,8-cineole, essential oils of Salvia are often an ingredient in mouthwash solutions. This monoterpene epoxide has however low toxicity (De Vincenzi et al., 2002) and it is approved by the Food and Drug Administration for food use (CFR 172.510). But the toxicity of thujone has been extensively studied (Pelkonen et al., 2013). Thujone has antagonistic properties to γ-aminobutyric acid (GABA), which results in muscle spasms and convulsions (Höld et al., 2002). According to the European regulations, its levels should not exceed 25 mg/kg of food prepared from any of the sage species (Regulation EC, 2008). Although camphor is often an ingredient of topical medications used for muscle pain relief and a cold remedy for the relief of chest congestion (Valdez et al., 1999), its inhalation is very toxic. In high doses, this monoterpene ketone produced causes irritability, disorientation, lethargy, muscle spasms, vomiting, abdominal cramps, convulsions, and seizures, especially in children (Uc et al., 2000).
Plant bioactive properties can be highly affected by environmental changes, such as changes in ozone concentrations, increased temperatures, changes in water regimes (drought and flooding), and an increase in soil salinity (Bettaieb et al., 2011). Therefore, the chemical composition of the oils of the mostly used Salvia species, Dalmatian sage (Salvia officinalis), and Spanish sage (Salvia officinalis subsp. lavandulifolia) are under the regulation of the International Standardization Organization (ISO). According to the ISO regulation, S. officinalis essential oil should contain 5.5%–13% of 1,8-cineole, 18%–43% of α-thujone, and 4.5%–45% of camphor (ISO 9909, 1997), while the oil of S. officinalis subsp. lavandulifolia should contain 10%–30% of 1,8-cineole, and 11%–36% of camphor (ISO 3526, 2005). Supplementary Table 1 summarizes the composition of the essential oils of the four Salvia species reviewed in this paper. The literature on Salvia species covers mainly the European continent (Figure 3). But wherever possible, we have reviewed whether these oils meet the ISO regulation. The heatmap in Figure 4A summarizes the literature data on levels of 1,8-cineole, camphor, and α-thujene (the main constituents of the oils of S. officinalis, S. officinalis susp. lavandulifolia, and S. fruticosa) from all three oils in different European countries. To construct the heatmap, the compounds with >10% were selected and their percentage content was log-transformed. Although it seems that the composition of these species is similar (Supplementary Table 1), they are separated into clusters. Monoterpenes from S. officinalis make one cluster, while monoterpenes from S. offcinalis subsp. lavanulifolia and S. fruticosa are in another cluster but grouped into two different subclusters.
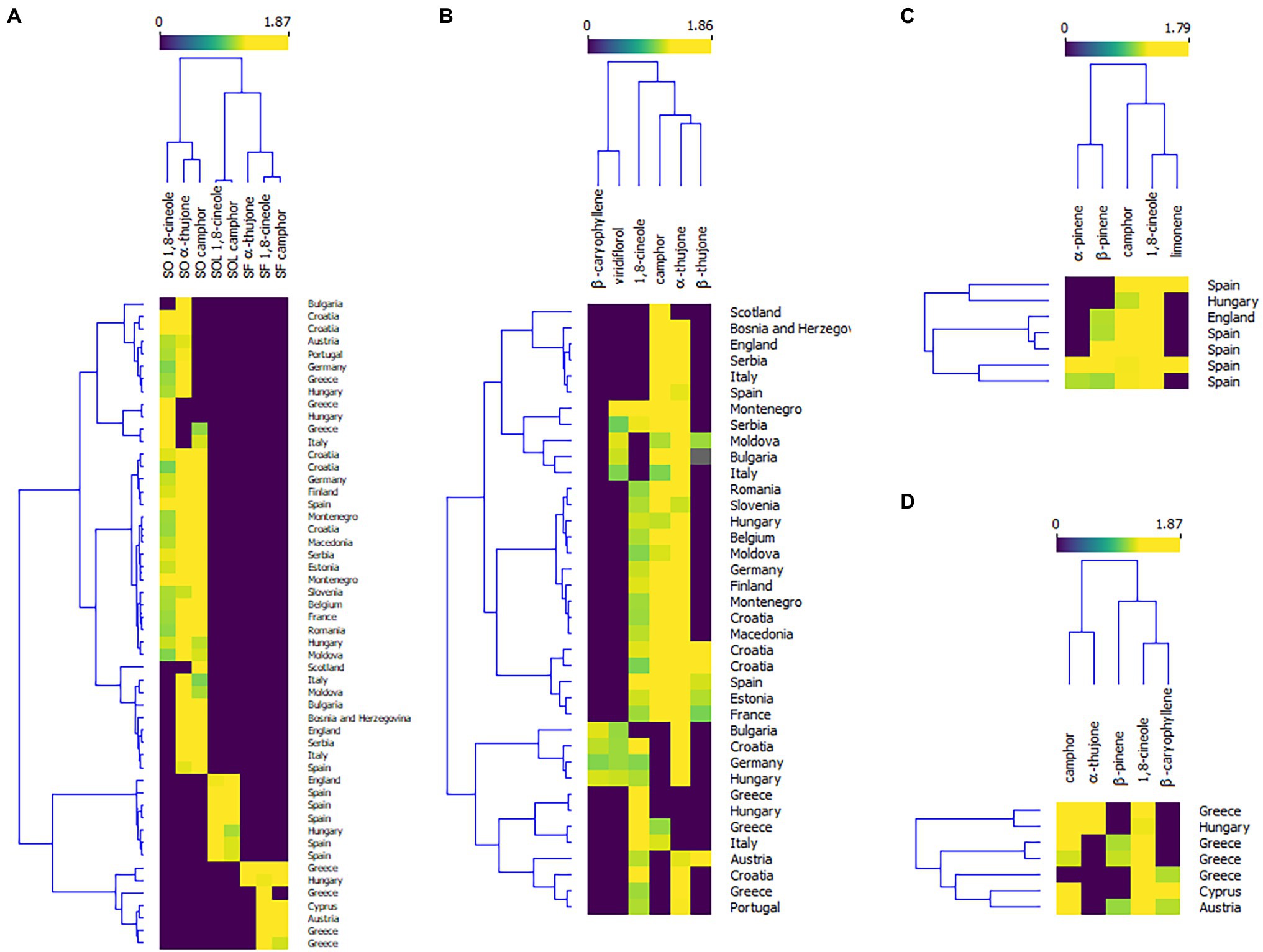
Figure 4. Heatmaps of the levels of the essential oil constituents in of Salvia officinalis—SO, S. officinalis subsp. lavanulifolia—SOL, and S. fruticosa—SF. (A) Country of origin in relation to the percentage content of the major monoterpenes in all three species; (B) Country of origin in relation to the percentage content of the major monoterpenes in S. officinalis; (C) Country of origin in relation to the percentage content of the major monoterpenes in, S. officinalis subsp. lavanulifolia; and (D) Country of origin in relation to the percentage content of the major monoterpenes in S. fruticosa.
Salvia officinalis L.
Dalmatian sage or common sage (Salvia officinalis L.) from the Lamiaceae family is a well-known aromatic and medicinal Mediterranean plant (Avato et al., 2005; Raal et al., 2007). Salvia officinalis is native to the coastal regions of the western Balkan and southern Apennine Peninsulas with a habitat reaching south into northwest Greece (di Pietro, 2011). It is a perennial subshrub cultivated in temperate regions worldwide (Ghorbani and Esmaeilizadeh, 2017). Species of Salvia usually grow 30–70 cm tall, with a woody stem, whitish beneath and greyish-green above leaves, and with purple-blue flowers up to 3 cm long appearing from early summer to early autumn.
The use of the Dalmatian sage in traditional medicine has a long history with many studies even in recent years providing evidence for beneficial properties of this plant. Experimental studies on S. officinalis extracts and essential oils have revealed health-beneficial properties, such as antioxidant (Hohmann et al., 1999), antibacterial, hypoglycemic (Alarcon-Aguilar et al., 2002), anti-inflammatory (Ehrnhöfer-Ressler et al., 2013; Schröder et al., 2013), fungistatic, astringent, eupeptic, and anti-hydrolytic activity (Martins et al., 2015), hypotensive properties, central nervous system depressant actions, and anti-spasmodic activity. Additionally, the essential oil demonstrated bactericidal and bacteriostatic effects against both Gram-positive and Gram-negative bacteria (Mitić-Ćulafić et al., 2005) and reduced UV-induced mutations in Escherichia coli and Saccharomyces cerevisiae (Vuković-Gačić et al., 2006).
This review paper covers studies on S. officinalis (Dalmatian sage) essential oils from the European continent, with Croatian species as the most investigated (Lamien-Meda et al., 2010; Jug-Dujaković et al., 2012; Cvetkovikj et al., 2015). For better visualization, we have constructed a heatmap of the levels of the main essential oil constituents found in the available literature (Figure 4B). When we compared the profile of the major compounds, there was no meaningful clustering on the plants’ geographical origin. Although the essential oil composition varies even within a single country and it is often correlated with the microclimate conditions suggesting that the composition and yield are sensitive to even small disruptions in environmental conditions, emphasizing the significance of tracking the climate-related environmental stressors and their effect on plants.
The essential oil composition of Dalmatian sage from Croatia significantly differs from study to study. This plant naturally grows in a relatively small area (12,158 km2) but with different climatic conditions within the area. It is well known that the average temperatures and humidity significantly differ between the coastal area and continental area of Dalmatia. Generally, the contents of 1,8-cineole and camphor in the oil of Croatian sage are relatively similar in all published studies, while the levels of thujones are significantly different within the species (Supplementary Table 1), which is the reason why some of the oils from Croatian S. officinalis do meet the ISO legislation (ISO 9909, 1997). Again, the composition of the Dalmatian sage oil from Montenegro showed high variability in thujone content, while 1,8-cineole remains quite similar (Perry et al., 1999; Couladis et al., 2002; Damjanovic-Vratnica et al., 2008; Steševic et al., 2014). Also, studies of S. officinalis from Greece show significant intraspecific variability in the essential oil composition (Lamien-Meda et al., 2010). Interestingly, the authors detected significantly high levels of 1,8-cineole in Greek S. officinalis (Raal et al., 2007; Figure 4B).
Salvia officinalis is a drought-susceptible species (Munné-Bosch et al., 2001). In Mediterranean areas, the crop is subjected to summer drought stress, high temperature, soil salinity, and enhanced ozone levels (Pellegrini et al., 2018), which affect its composition and potential use. An increase in global temperatures is imposing additional stress on sage affecting essential oil composition and affecting sage cultivation and production due to leaf etiolation, wilting, and reduced photosynthetic capacity of plants (Lin et al., 2021). Under hot and arid conditions, accumulation of thujone has been recorded as a response to heat and water stress, while under optimal conditions camphor is mainly accumulated (Nevkrytaya et al., 2021). However, the results of Radwan et al. (2017) suggest that bornyl and sabinene synthases are upregulated by drought stress, while cineole synthase is downregulated by drought stress (Figure 3). Therefore, as also suggested by these authors, further research is needed.
The search for protective mechanisms to keep the oil composition stable and plants’ health has been ongoing during the past few years. Salicylic acid and calcium are recognized as signaling molecules in plants’ responses to oxidative stress induced by different environmental factors (Laanemets et al., 2013; Janda and Ruelland, 2015). Growth inhibition can be effectively alleviated by external application of salicylic acid (SA) or calcium (Ca2+) alone or in combination, as reported by Lin et al. (2021). They recorded an increase in new leaves formation under high-temperature stress when plants were treated with SA and Ca2+ (Figure 3).
Soil salinity is increasing due to progressive climate aridity (Taarit et al., 2009). Just in Tunisia alone, more than 10% of soil is affected by salinity consequently affecting sage growth and oil composition. In their research, they investigated how salinity levels can affect the chemical profile of S. officinalis and demonstrated that changes in the composition of essential oils correlate with the salinity level. Viridiflorol was the main component in the essential oils of plants that were not subjected to salinity stress. When plants were subjected to low salinity (25 mM NaCl) the main compound was the same, but it was represented in higher percentage (21.8%), moderate salt stress (50 and 100 mM NaCl) induced changes in compound synthesis resulting in 1,8-cineole (21.6 and 23.8% respectively) as the main oil compound followed by α-thujone (21.4 and 22.2%, respectively; Figure 3). Further increase in soil salinity induced higher synthesis of manool (65.7%). Moderate and severe salt stress induced a significant decrease of viridiflorol (13.4, 6.0, and 7.0% respectively). The accumulation of manool, labdane type diterpene, at high salinity levels can be exploited as a precursor of ambergris fragrant products (Taarit et al., 2010), but the severity of salt stress affects the physiological traits and yield, thus this can be considered more as a marker of severe salt stress in S. offciinalis. Alleviation of salt stress in sage has been confirmed after foliar application of SA with a variety of changes in the chemical composition of essential oils (Zohra Es-Sbihi et al., 2021). Sage essential oil from aerial plant parts is mainly composed of 1,8-cineole, α/β-thujone, and camphor, and salt stress can induce a significant increase in these compounds with some new compounds recorded, such as viridiflorol, β-caryophyllene, myrentol, and pulegone. When SA spray was applied water stress effects on major compounds were alleviated (Zohra Es-Sbihi et al., 2021). The application of SA can stimulate the conversion of thymol to carvacrol, and it can be correlated further with the synthesis of phytoalexins affecting the plants’ c potential (Sirvent and Gibson, 2002).
The occurrence of ozone in the troposphere because of NOx and volatile organic compounds emissions is considered to become an even bigger problem with climatic changes and anthropogenic activities. Ozone-induced injuries to plants include reduction of growth, photosynthetic rate, cell dehydration, and leaf necrosis (Sirvent and Gibson, 2002). Short exposure to ozone can be used for the enhancement of secondary metabolite production, especially to utilize excess excitation energy created by ozone exposure (Marchica et al., 2019). The main source of oxidative stress in plants exposed to drought lies in outbursts of toxic O2− radicals because of electron overflow from the photosynthetic electron transport chains (Wilhelm and Selmar, 2011). Metabolic changes under oxidative stress result in large consumption of NADPH+ through biosynthesis of specialized metabolites, such as phenols, terpenoids, alkaloids, glucosinolates, and others affecting essential oil composition (Selmar and Kleinwächter, 2013). Changes in monoterpene content can be correlated to upregulated expression of monoterpene synthase and bornyl diphosphate synthase (Radwan et al., 2017). Drought stress affects different growing stages of sage and the associated increase in production of monoterpenes under water deficit is a result of upregulation of 1,8-cineole synthase and bornyl diphosphate synthase in different growth stages of sage resulting in increased production of 1,8-cineole and camphor (Ramezani et al., 2020; Figure 3). In the context of reoccurring stress in S. officinalis increase in α-pinene has been recorded under repetitive drought stress (Kulak, 2020). Contrasting results have been recorded as well where reduction of α-pinene is recorded under water deficit conditions (Rioba et al., 2015; Chrysargyris et al., 2018) but in these cases water deficit was acute and no further investigation regarding stress memory was performed. Such results suggest that stress memory in S. officinalis can be reflected in essential oil composition and environmental changes can have long-lasting effects that right now cannot be comprehended.
Salvia officinalis subsp. lavandulifolia (Vahl) Gams
Salvia officinalis subsp. lavandulifolia Vahl., known as “Spanish sage,” is a native of the Iberian Peninsula. It is a small woody herbaceous perennial shrub up to 17–100 cm with mauve-blue flowers. It occurs preferably on the sandy-calcareous soils (at 350–2,000 m) of Spain, southeast of France, and northwest of Africa (Sáez, 2010). This plant is characterized by a small subshrub or herb up to 17–100 cm, with branched stems, and tector hairs. The leaves are elliptic to linear-lanceolate. The inflorescence is simple or branched, with 2–8 flowers. The flowers are regular, tubular, or campanulate, usually green or violet-purple, pubescent. The blooming develops for about 1 month in late spring and early summer (Sáez, 2010). The leaf presents seasonal dimorphism (Palacio and Montserrat-Marti, 2006). Salvia lavandulifolia found its use in Mediterranean folk medicine as spasmolytic, antiseptic, analgesic, sedative, and antioxidant (Porres-Martínez et al., 2013). Studies have shown the potential value of Spanish sage in dementia therapy attributed to its sedative, antioxidant, anti-inflammatory, estrogenic, and antimicrobial and anti-cholinesterase activities (Perry et al., 2002; Cutillas et al., 2017). The essential oil of this species has been described as a safe memory enhancer, suggesting it could be used in Alzheimer’s disease therapy (Kennedy et al., 2011).
The majority of essential oil compositions of Spanish sage published to date (Supplementary Table 1) meet the ISO legislative (ISO 3526, 2005). Most of the studies are performed in Spain, where this plant originates from Figure 4C. However, the oil of this species also shows high intraspecific variability, which depends on locality (Herraiz-Peñalver et al., 2010; Usano-Alemany et al., 2014), but also phenological stage (Porres-Martínez et al., 2014; Usano-Alemany et al., 2014; Méndez-Tovar et al., 2016). Generally, wild Spanish sages grown in southern and central Spain contain high levels of 1,8-cineole, while those grown on the north contain more camphor and its biosynthetic precursor borneol (Herraiz-Peñalver et al., 2010; Usano-Alemany et al., 2014). Also, it is described that the levels of 1,8-cineole in the oil are the highest after the flowering period, with contents remaining high until the formation of new young leaves (Usano-Alemany et al., 2014). In addition, Porres-Martínez et al. (2014) found the levels of both 1,8-cineole and camphor increase from the vegetative stage to the full flowering stage.
Temperature stress in S. officinalis subsp. lavandulifolia can affect the ratio of monoterpene and sesquiterpene fraction in essential oil (Usano-Alemany et al., 2014) resulting in different quality and effectiveness, and oils of different biological properties. Under elevated growing temperature above 24°C (up to 32°C), there is an observed significant decrease in monoterpenes and an increase in sesquiterpenes with changes in levels of β-caryophyllene, caryophyllene oxide, and manool (Figure 3). The recorded changes resulted in changes in chemotype. In the context of drought stress, Spanish sage is tolerant to moderate drought stress with consistent essential oil quality and yield as demonstrated in recent studies (García-Caparrós et al., 2019). During cultivation, significant deviation/change in the chemical composition of the oils can arise because of changing environmental conditions, such as an increase in temperature and decreased precipitation. In cases of mild arid conditions with higher temperatures, this species can produce more essential oils with variable proportions of compounds as recorded through a 4-year analysis of cultivated plants, that is, 1,8-cineole become a prominent compound through the years (Usano-Alemany et al., 2016).
Salvia fruticosa Mill.
This shrub, with the former name Salvia triloba L., is also known as Greek sage. It is native to the Mediterranean including Southern Italy, southern parts of the Balkan Peninsula (Greece) to West Syria. Three-lobed sage is found on dry rocky limestone soils or the edges of pine forests, riverbeds, and roadsides, at altitudes from 100 to 800 m a.s.l. (Radosavljević et al., 2012). This plant is also cultivated as an ornamental plant in other Mediterranean countries (Papafotiou et al., 2021). It has been used in folk medicine since ancient times with many studies demonstrating antioxidant (Triantaphyllou et al., 2001), anti-inflammatory (El-Sayed et al., 2006), anti-cholinesterase (Şenol et al., 2011), and antifungal activity (Exarchou et al., 2002). Traditionally, it is used as a medicinal (herbal tea), culinary, and as a melliferous plant (Clebsch, 2003). Antibacterial and antifungal activity has been confirmed against food contaminants (Delamare et al., 2007) and soilborne pathogens (Pitarokili et al., 2002). In nature, this plant is very drought resistant but in cultivation, such as cultivation on green roofs for its melliferous properties it cannot tolerate drought (Papafotiou et al., 2021).
The essential oil of Greek sage has a long tradition of medicinal uses in Greece (Clebsch, 2003), but still is not included in ISO legislation. According to the studies summarized in Supplementary Table 1, this species showed even higher intraspecific variability than S. officinalis. As presented in Figure 4D, clustering is very weak, that is, there is no grouping of this oil according to its geographical origin. In most of the studies, the oil of this species does not contain significant levels of thujones, except in the studies of Karousou et al. (1998) and Máthé et al. (2010). On the other hand, the amounts of 1,8-cineole are around 50%–70% (Bellomaria et al., 1992; Giweli et al., 2013; Sarrou et al., 2016), but can be up to 83.5% (Zgheib et al., 2019). But Karioti et al. (2003) and Arikat et al. (2004) have shown that this species produces lower amounts of 1,8-cineole when plants are grown in vitro.
Improvement of stress tolerance and bioactive properties has been tested through arbuscular mycorrhizal fungi (AMF) inoculation of S. fruticosa and improvement of plant growth mass and chlorophyll content has been recorded. Significant improvement (33%) in the utilization of adsorbed energy in photochemistry was observed for plants with inoculation with Rhizophagus irregularis (Moustakas et al., 2020). AMF beneficial effects on Salvia sp. stress tolerance have been recorded in other cases as well, such as Salvia hispanica when inoculated with an inoculum containing Glomus mosseae spores, hyphae, and colonized maize roots (Ouzounidou et al., 2015; Figure 3).
Conclusion
Plants are plastic organisms and respond to changes in the environment by adapting their metabolism to offset stress-induced damages. Salvia sp. as economically important plants are under direct environmental stress due to climate change, which affects their chemical composition influencing the quality and yield of essential oils and therefore can have serious economic consequences. This review emphasizes the already confirmed effects of environmental stressors on three selected Salvia sp. and at the same time highlights the importance of such research to mitigate the effects of stress factors in the commercial production of these essential oils. The data from the literature indicate that the chemical composition of the Salvia officinalis essential oils is mostly affected by the soil salinity, while the increase in temperature affects the ratio between monoterpenes and sesquiterpenes in the essential oil of S. officinalis subs. lavandulifolia. Although there are no papers investigating the direct influence of environmental stressors on the oil composition of S. fruticosa, it was previously found that inoculation of AMF has a beneficial effect on the plant yield. This strategy might be useful for other Salvia species too, to alleviate the effects of environmental stressors. The fact that out of 12,000 results related to Salvia only a few could be found directly investigating climate-related environmental stressors emphasizes the need for further research to explain how these stressors affect economically produced Salvia essential oils. Evaluating data available on Salvia officinalis species essential oils it is clear that their commercial production will be affected by the changing climate. The presented review stresses the importance of studies oriented toward the investigation of how complex climate-related stressors can affect the commercial production of essential oils, and what are our next steps in the alleviation of these effects. In addition, in vitro culture or vertical farming under controlled conditions could be prospective solutions that can ensure sustainable production of plants and enable constant levels of metabolites of interest.
Author Contributions
EK and SC: conceptualization, writing, and editing. SD: writing. PT: writing, editing, and reviewing. All authors contributed to the article and approved the submitted version.
Funding
This work was funded by project No. RO0418 (Sustainable systems and technologies, improving crop production for a higher quality of production of food, feed, and raw materials, under conditions of changing climate), the Ministry of Agriculture, Czechia, and the project “Plants as a tool for sustainable global development” (registration number: CZ.02.1.01/0.0/0.0/16_019/0000827) within the program Research, Development, and Education (OP RDE).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fpls.2022.864807/full#supplementary-material
References
Alarcon-Aguilar, F. J., Roman-Ramos, R., Flores-Saenz, J. L., and Aguirre-Garcia, F. (2002). Investigation on the hypoglycaemic effects of extracts of four Mexican medicinal plants in normal and Alloxan-diabetic mice. Phytother. Res. 16, 383–386. doi: 10.1002/ptr.914
Ardestani, E. G., and Ghahfarrokhi, Z. H. (2021). Ensembpecies distribution modeling of Salvia hydrangea under future climate change scenarios in Central Zagros Mountains. Iran. Glob. Ecol. Conserv. 26:e01488. doi: 10.1016/j.gecco.2021.e01488
Arikat, N. A., Jawad, F. M., Karam, N. S., and Shibli, R. A. (2004). Micropropagation and accumulation of essential oils in wild sage (Salvia fruticosa Mill.). Sci. Hortic. 100, 193–202. doi: 10.1016/j.scienta.2003.07.006
Avato, P., Fortunato, I. M., Ruta, C., and D’Elia, R. (2005). Glandular hairs and essential oils in micropropagated plants of Salvia officinalis L. Plant Sci. 169, 29–36. doi: 10.1016/j.plantsci.2005.02.004
Bellomaria, B., Arnold, N., Valentini, G., and Arnold, H. J. (1992). Contribution to the study of the essential oils from three species of Salvia growing wild in the eastern mediterranean region. J. Essent. Oil Res. 4, 607–614. doi: 10.1080/10412905.1992.9698143
Bettaieb, I., Hamrouni-Sellami, I., Bourgou, S., Limam, F., and Marzouk, B. (2011). Drought effects on polyphenol composition and antioxidant activities in aerial parts of Salvia officinalis L. Acta Physiol. Plant. 33, 1103–1111. doi: 10.1007/s11738-010-0638-z
Ćavar Zeljković, S., Šišková, J., Komzáková, K., De Diego, N., Kaffková, K., and Tarkowski, P. (2021b). Phenolic compounds and biological activity of selected Mentha species. Plan. Theory 10:550. doi: 10.3390/plants10030550
Ćavar Zeljković, S., Smekalová, K., Kaffková, K., and Štefelová, N. (2021a). Influence of post-harvesting period on quality of thyme and spearmint essential oils. J. Appl. Res. Med. Aromat. Plants 25:100335. doi: 10.1016/j.jarmap.2021.100335
Chrysargyris, A., Michailidi, E., and Tzortzakis, N. (2018). Physiological and biochemical responses of Lavandula angustifolia to salinity under mineral foliar application. Front. Plant Sci. 9:489. doi: 10.3389/fpls.2018.00489
Code of Federal Regulation (21CFR172.510) (2021). Food Additives Permitted for Direct Addition to Food for Human Consumption. Available at: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?CFRPart=172 (Accessed January 14, 2022).
Couladis, M., Tzakou, O., Mimica-Dukić, N., Jančić, R., and Stojanović, D. (2002). Essential oil of Salvia officinalis L. from Serbia and Montenegro. Flavour Fragr. J. 17, 119–126. doi: 10.1002/ffj.1065
Cutillas, A. B., Carrasco, A., Martinez-Gutierrez, R., Tomas, V., and Tudela, J. (2017). Composition and antioxidant, antienzymatic and antimicrobial activities of volatile molecules from Spanish Salvia lavandulifolia (Vahl) essential oils. Molecules 22:1382. doi: 10.3390/molecules22081382
Cvetkovikj, I., Stefkov, G., Karapandzova, M., Kulevanova, S., and Satovic, Z. (2015). Essential oils and chemical diversity of southeast european populations of Salvia officinalis L. Chem. Biodivers. 12, 1025–1039. doi: 10.1002/cbdv.201400273
Damjanovic-Vratnica, B., Ðakov, T., Šukovic, D., and Damjanovic, J. (2008). Chemical composition and antimicrobial activity of essential oil of wild-growing Salvia officinalis L. from Montenegro. J. Essent. Oil-Bear. Plant. 11, 79–89. doi: 10.1080/0972060X.2008.10643602
Delamare, A. P. L., Moschen-Pistorello, I. T., Artico, L., Atti-Serafini, L., and Echeverrigaray, S. (2007). Antibacterial activity of the essential oils of Salvia officinalis L. and Salvia triloba L. cultivated in South Brazil. Food Chem. 100, 603–608. doi: 10.1016/j.foodchem.2005.09.078
De Vincenzi, M., Silano, M., De Vincenzi, A., Maialetti, F., and Bcazzocchio, B. (2002). Constituents of aromatic plants: eucalyptol. Fitoterapia 73, 269–275. doi: 10.1016/S0367-326X(02)00062-X
di Pietro, R. (2011). New dry grassland associations from the Ausoni-Aurunci mountains (Central Italy)–Syntaxonomical updating and discussion on the higher rank syntaxa. Hacquetia 10, 183–231. doi: 10.2478/v10028-011-0011-9
Ehrnhöfer-Ressler, M. M., Fricke, K., Pignitter, M., Walker, J. M., Walker, J., Rychlik, M., et al. (2013). Identification of 1, 8-cineole, borneol, camphor, and thujone as anti-inflammatory compounds in a Salvia officinalis L. infusion using human gingival fibroblasts. J. Agric. Food Chem. 61, 3451–3459. doi: 10.1021/jf305472t
El-Sayed, N. H., El-Eraky, W., Ibrahim, M. T., and Mabry, T. J. (2006). Antiinflammatory and ulcerogenic activities of Salvia triloba extracts. Fitoterapia 77, 333–335. doi: 10.1016/j.fitote.2006.04.002
European Commission (2019). “COM(2019) 640 Final. Green New Deal. Communication from the Commission to the European Parliament,” European Commission the European Council, the Council, the European Economic and Social Committee and the Committee of the Regions [Preprint].
Exarchou, V., Nenadis, N., Tsimidou, M., Gerothanassis, I. P., Troganis, A., and Boskou, D. (2002). Antioxidant activities and phenolic composition of extracts from Greek oregano, Greek sage, and summer savory. J. Agric. Food Chem. 50, 5294–5299. doi: 10.1021/jf020408a
García-Caparrós, P., Romero, M. J., Llanderal, A., Cermeño, P., Lao, M. T., and Segura, M. L. (2019). Effects of drought stress on biomass, essential oil content, nutritional parameters, and costs of production in six Lamiaceae species. WaterSA 11:573. doi: 10.3390/w11030573
Ghorbani, A., and Esmaeilizadeh, M. (2017). Pharmacological properties of Salvia officinalis and its components. J. Tradit. Complement. Med. 7, 433–440. doi: 10.1016/J.JTCME.2016.12.014
Giuliani, C., Ascrizzi, R., Tani, C., Bottoni, M., Bini, L. M., Flamini, G., et al. (2017). Salvia uliginosa Benth.: glandular trichomes as bio-factories of volatiles and essential oil. Flora 233, 12–21. doi: 10.1016/j.flora.2017.05.002
Giweli, A. A., Džamić, A. M., Soković, M., Ristić, M. S., Janackovic, P., and Marin, P. D. (2013). The chemical composition, antimicrobial and antioxidant activities of the essential oil of Salvia fruticosa growing wild in Libya. Arch. Biol. Sci. 65, 321–329. doi: 10.2298/ABS1301321G
Herraiz-Peñalver, D., Usano-Alemany, J., Cuadrado, J., Jordan, M. J., Lax, V., Sotomayor, J. A., et al. (2010). Essential oil composition of wild populations of Salvia lavandulifolia Vahl. From Castilla-La Mancha (Spain). Biochem. Syst. Ecol. 38, 1224–1230. doi: 10.1016/j.bse.2010.10.015
Hohmann, J., Zupkó, I., Rédei, D., Csányi, M., Falkay, G., Máthé, I., et al. (1999). Protective effects of the aerial parts of Salvia officinalis, Melissa officinalis and Lavandula angustifolia and their constituents against enzyme-dependent and enzyme-independent lipid peroxidation. Planta Med. 65, 576–578. doi: 10.1055/s-2006-960830
Höld, K. M., Sirisoma, N. S., Sparks, S. E., and Casida, J. E. (2002). Metabolism and mode of action of cis-and trans-3-pinanones (the active ingredients of hyssop oil). Xenobiotica 32, 251–265. doi: 10.1080/00498250110095745
Janda, M., and Ruelland, E. (2015). Magical mystery tour: salicylic acid signalling. Environ. Exp. Bot. 114, 117–128. doi: 10.1016/J.ENVEXPBOT.2014.07.003
Jug-Dujaković, M., Ristić, M., Pljevljakušić, D., Dajić-Stevanović, Z., Liber, Z., Hančević, K., et al. (2012). High diversity of indigenous populations of dalmatian sage (Salvia officinalis L.) in essential-oil composition. Chem. Biodivers. 9, 2309–2323. doi: 10.1002/cbdv.201200131
Karioti, A., Skaltsa, H., Demetzos, C., Perdetzoglou, D., Economakis, C. D., and Salem, A. B. (2003). Effect of nitrogen concentration of the nutrient solution on the volatile constituents of leaves of Salvia fruticosa Mill. in solution culture. J. Agric. Food Chem. 51, 6505–6508. doi: 10.1021/jf030308k
Karousou, R., Vokou, D., and Kokkini, S. (1998). Variation of Salvia fruticosa essential oils on the island of Crete (Greece). Bot. Acta 111, 250–254. doi: 10.1111/j.1438-8677.1998.tb00705.x
Kennedy, D. O., Dodd, F. L., Robertson, B. C., Okello, E. J., Reay, J. L., Scholey, A. B., et al. (2011). Monoterpenoid extract of sage (Salvia lavandulaefolia) with cholinesterase inhibiting properties improves cognitive performance and mood in healthy adults. J. Psychopharmacol. 25, 1088–1100. doi: 10.1177/0269881110385594
Kulak, M. (2020). Recurrent drought stress effects on essential oil profile of Lamiaceae plants: an approach regarding stress memory. Ind. Crop. Prod. 154:112695. doi: 10.1016/j.indcrop.2020.112695
Laanemets, K., Brandt, B., Li, J., Merilo, E., Wang, Y. F., Keshwani, M. M., et al. (2013). Calcium-dependent and-independent stomatal signaling network and compensatory feedback control of stomatal opening via Ca2+ sensitivity priming. Plant Physiol. 163, 504–513. doi: 10.1104/pp.113.220343
Lamien-Meda, A., Schmiderer, C., Lohwasser, U., Börner, A., Franz, C., and Novak, J. (2010). Variability of the essential oil composition in the sage collection of the Genebank Gatersleben: a new viridiflorol chemotype. Flavour Fragr. J. 25, 75–82. doi: 10.1002/ffj.1969
Li, B., Zhang, C., Peng, L., Liang, Z., Yan, X., Zhu, Y., et al. (2015). Comparison of essential oil composition and phenolic acid content of selected Salvia species measured by GC-MS and HPLC methods. Ind. Crop. Prod. 69, 329–334. doi: 10.1016/j.indcrop.2015.02.047
Lin, K.-H., Lin, T.-Y., Wu, C.-W., Chang, Y.-S., and Barceló, J. (2021). Protective effects of salicylic acid and calcium chloride on sage plants (Salvia officinalis L. and Salvia elegans Vahl) under high-temperature stress. Plan. Theory 10:2110. doi: 10.3390/plants10102110
Llurba-Montesino, N., and Schmidt, T. J. (2018). Salvia species as sources of natural products with antiprotozoal activity. Int. J. Mol. Sci. 19:264. doi: 10.3390/ijms19010264
Lopresti, A. L. (2016). Salvia (sage): a review of its potential cognitive-enhancing and protective effects. Drugs R. D. 17, 53–64. doi: 10.1007/s40268-016-0157-5
Marchica, A., Loré, S., Cotrozzi, L., Lorenzini, G., Nali, C., Pellegrini, E., et al. (2019). Early detection of sage (Salvia officinalis L.) responses to ozone using reflectance spectroscopy. Plan. Theory 8:346. doi: 10.3390/plants8090346
Martins, N., Barros, L., Santos-Buelga, C., Henriques, M., Silva, S., and Ferreira, I. C. (2015). Evaluation of bioactive properties and phenolic compounds in different extracts prepared from Salvia officinalis L. Food Chem. 170, 378–385. doi: 10.1016/j.foodchem.2014.08.096
Máthé, I., Máthé, Á., Hohmann, J., and Janicsák, G. (2010). Volatile and some non-volatile chemical constituents of Mediterranean Salvia species beyond their native area. Isr. J. Plant Sci. 58, 273–277. doi: 10.1560/IJPS.58.3-4.273
Méndez-Tovar, I., Novak, J., Sponza, S., Herrero, B., and Asensio-S-Manzanera, M. C. (2016). Variability in essential oil composition of wild populations of Labiatae species collected in Spain. Ind. Crop. Prod. 79, 18–28. doi: 10.1016/j.indcrop.2015.10.009
Mitić-Ćulafić, D., Vuković-Gačić, B. S., Knežević-Vukčević, J. B., Stanković, S., and Simić, D. M. (2005). Comparative study on the antibacterial activity of volatiles from sage (Salvia officinalis L.). Arch. Biol. Sci 57, 173–178. doi: 10.2298/ABS0503173M
Moustakas, M., Bayçu, G., Sperdouli, I., Eroğlu, H., and Eleftheriou, E. P. (2020). Arbuscular mycorrhizal symbiosis enhances photosynthesis in the medicinal herb Salvia fruticosa by improving photosystem ii photochemistry. Plan. Theory 9, 1–18. doi: 10.3390/plants9080962
Munné-Bosch, S., Jubany-Marí, T., and Alegre, L. (2001). Drought-induced senescence is characterized by a loss of antioxidant defences in chloroplasts. Plant Cell Environ. 24, 1319–1327. doi: 10.1046/j.1365-3040.2001.00794.x
Nevkrytaya, N., Novikov, I., Soboleva, E., Kashirina, N., and Radchenko, L. (2021). Manifestation features of the productivity potential of Salvia officinalis L. in the conditions of the Crimea foothills. E3S Web Conf. 254:e01006. doi: 10.1051/e3sconf/202125401006
Ouzounidou, G., Skiada, V., Papadopoulou, K. K., Stamatis, N., Kavvadias, V., Eleftheriadis, E., et al. (2015). Effects of soil pH and arbuscular mycorrhiza (AM) inoculation on growth and chemical composition of chia (Salvia hispanica L.) leaves. Rev. Bras. Bot. 38, 487–495. doi: 10.1007/s40415-015-0166-6
Palacio, S., and Montserrat-Marti, G. (2006). Comparison of the bud morphology and shoot growth dynamics of four species of Mediterranean subshrub growing along an altitudinal gradient. Bot. J. Linn. Soc. 151, 527–539. doi: 10.1111/j.1095-8339.2006.00542.x
Papafotiou, M., Martini, A. N., Papanikolaou, E., Stylias, E. G., and Kalantzis, A. (2021). Hybrids development between Greek salvia species and their drought resistance evaluation along with Salvia fruticosa, under Attapulgite-amended substrate. Agronomy 11:2401. doi: 10.3390/agronomy11122401
Pelkonen, O., Abass, K., and Wiesner, J. (2013). Thujone and thujone-containing herbal medicinal and botanical products: toxicological assessment. Regul. Toxicol. Pharmacol. 65, 100–107. doi: 10.1016/j.yrtph.2012.11.002
Pellegrini, M., Ricci, A., Serio, A., Chaves-López, C., Mazzarrino, G., D’Amato, S., et al. (2018). Characterization of essential oils obtained from Abruzzo autochthonous plants: antioxidant and antimicrobial activities assessment for food application. Foods 7:19. doi: 10.3390/foods7020019
Perry, N. B., Anderson, R. E., Brennan, N. J., Douglas, M. H., Heaney, A. J., McGimpsey, J. A., et al. (1999). Essential oils from Dalmatian sage (Salvia officinalis L.): variations among individuals, plant parts, seasons, and sites. J. Agric. Food Chem. 47, 2048–2054. doi: 10.1021/jf981170m
Perry, N. S. L., Houghton, P. J., Jenner, P., Keith, A., and Perry, E. K. (2002). Salvia lavandulaefolia essential oil inhibits cholinesterase in vivo. Phytomedicine 9, 48–51. doi: 10.1078/0944-7113-00082
Pitarokili, D., Couladis, M., Petsikos-Panayotarou, N., and Tzakou, O. (2002). Composition and antifungal activity on soil-borne pathogens of the essential oil of Salvia sclarea from Greece. J. Agric. Food Chem. 50, 6688–6691. doi: 10.1021/jf020422n
Porres-Martínez, M., González-Burgos, E., Accame, M. E. C., and Gómez-Serranillos, M. P. (2013). Phytochemical composition, antioxidant and cytoprotective activities of essential oil of Salvia lavandulifolia Vahl. Food Res. Int. 54, 523–531. doi: 10.1016/j.foodres.2013.07.029
Porres-Martínez, M., González-Burgos, E., Carretero, M. E., and Gómez-Serranillos, M. P. (2014). Influence of phenological stage on chemical composition and antioxidant activity of Salvia lavandulifolia Vahl. Essential oils. Ind. Crop. Prod. 53, 71–77. doi: 10.1016/j.indcrop.2013.12.024
Pressey, R. L., Cabeza, M., Watts, M. E., Cowling, R. M., and Wilson, K. A. (2007). Conservation planning in a changing world. Trends Ecol. Evol. 22, 583–592. doi: 10.1016/J.TREE.2007.10.001
Raal, A., Orav, A., and Arak, E. (2007). Composition of the essential oil of Salvia officinalis L. from various European countries. Nat. Prod. Res. 21, 406–411. doi: 10.1080/14786410500528478
Radosavljević, I., Satovic, Z., Jakse, J., Javornik, B., Greguraš, D., Jug-Dujaković, M., et al. (2012). Development of new microsatellite markers for Salvia officinalis L. and its potential use in conservation-genetic studies of narrow endemic salvia brachyodon Vandas. Int. J. Mol. Sci. 13, 12082–12093. doi: 10.3390/ijms130912082
Radwan, A., Kleinwächter, M., and Selmar, D. (2017). Impact of drought stress on specialised metabolism: biosynthesis and the expression of monoterpene synthases in sage (Salvia officinalis). Phytochemistry 141, 20–26. doi: 10.1016/j.phytochem.2017.05.005
Ramezani, S., Abbasi, A., Sobhanverdi, S., Shojaeiyan, A., and Ahmadi, N. (2020). The effects of water deficit on the expression of monoterpene synthases and essential oils composition in Salvia ecotypes. Physiol. Mol. Biol. Plants 26, 2199–2207. doi: 10.1007/s12298-020-00892-1
Regulation EC (2008). Regulation (EC) No 1334/2008 of the European Parliament and Council of 16 December 2008, European Commission.
Rioba, N. B., Itulya, F. M., Saidi, M., Dudai, N., and Bernstein, N. (2015). Effects of nitrogen, phosphorus and irrigation frequency on essential oil content and composition of sage (Salvia officinalis L.). J. Appl. Res. Med. Aromat. Plants 2, 21–29. doi: 10.1016/j.jarmap.2015.01.003
Sáez, L. (2010). “Salvia L.” in Flora iberica. Vol. XII. Verbenaceae-Labiatae-Callitrichaceae. eds. R. Morales, A. Quintanar, F. Cabezas, A. J. Pujadas, and S. Cirujano (Madrid: Real Jardín Botánico de Madrid (C.S.I.C.)), 298–326.
Sarrou, E., Martens, S., and Chatzopoulou, P. (2016). Metabolite profiling and antioxidative activity of sage (Salvia fruticosa Mill.) under the influence of genotype and harvesting period. Ind. Crop. Prod. 94, 240–250. doi: 10.1016/j.indcrop.2016.08.022
Schröder, S., Beckmann, K., Franconi, G., Meyer-Hamme, G., Friedemann, T., Greten, H. J., et al. (2013). Can medical herbs stimulate regeneration or neuroprotection and treat neuropathic pain in chemotherapy-induced peripheral neuropathy? Evid. Based Complement. Alternat. Med. 2013:423713. doi: 10.1155/2013/423713
Selmar, D., and Kleinwächter, M. (2013). Influencing the product quality by deliberately applying drought stress during the cultivation of medicinal plants. Ind. Crop. Prod. 42, 558–566. doi: 10.1016/j.indcrop.2012.06.020
Şenol, F. S., Orhan, I. E., Erdem, S. A., Kartal, M., Şener, B., Kan, Y., et al. (2011). Evaluation of cholinesterase inhibitory and antioxidant activities of wild and cultivated samples of sage (Salvia fruticosa) by activity-guided fractionation. J. Med. Food 14, 1476–1483. doi: 10.1089/jmf.2010.0158
Sirvent, T., and Gibson, D. (2002). Induction of hypericins and hyperforin in Hypericum perforatum L. in response to biotic and chemical elicitors. Physiol. Mol. Plant Pathol. 60, 311–320. doi: 10.1006/PMPP.2002.0410
Steševic, D., Ristic, M., Nikolic, V., Nedovic, M., Cakovic, D., and Šatovic, Z. (2014). Chemotype diversity of indigenous dalmatian sage (Salvia officinalis L.) populations in Montenegro. Chem. Biodivers. 11, 101–114. doi: 10.1002/cbdv.201300233
Taarit, M. B., Msaada, K., Hosni, K., Hammami, M., Kchouk, M. E., and Marzouk, B. (2009). Plant growth, essential oil yield and composition of sage (Salvia officinalis L.) fruits cultivated under salt stress conditions. Ind. Crop. Prod. 30, 333–337. doi: 10.1016/J.INDCROP.2009.06.001
Taarit, M. B., Msaada, K., Hosni, K., and Marzouk, B. (2010). Changes in fatty acid and essential oil composition of sage (Salvia officinalis L.) leaves under NaCl stress. Food Chem. 119, 951–956. doi: 10.1016/J.FOODCHEM.2009.07.055
Triantaphyllou, K., Blekas, G., and Boskou, D. (2001). Antioxidative properties of water extracts obtained from herbs of the species Lamiaceae. Int. J. Food Sci. Nutr. 52, 313–317. doi: 10.1080/09637480120057512
Turek, C., and Stintzing, F. C. (2013). Stability of essential oils. Compr. Rev. Food Sci. Food Saf. 12, 40–53. doi: 10.1111/1541-4337.12006
Uc, A., Bishop, W. P., and Sanders, K. D. (2000). Camphor hepatotoxicity. South. Med. J. 93, 596–598. doi: 10.1097/00007611-200093060-00011
Usano-Alemany, J., Palá-Paúl, J., and Herráiz-Peñalver, D. (2014). Comprehensive phenological description of essential-oil chemotypes of Salvia lavandulifolia VAHL grown under the same environmental conditions. Chem. Biodivers. 11, 1963–1977. doi: 10.1002/cbdv.201400090
Usano-Alemany, J., Palá-Paúl, J., and Herráiz-Peñalver, D. (2016). Essential oil yields and qualities of different clonal lines of Salvia lavandulifolia monitored in Spain over four years of cultivation. Ind. Crop. Prod. 80, 251–261. doi: 10.1016/J.INDCROP.2015.11.010
Valdez, J. S., Martin, D. K., and Mayersohn, M. (1999). Sensitive and selective gas chromatographic methods for the quantitation of camphor, menthol and methyl salicylate from human plasma. J. Chromatogr. B 729, 163–171. doi: 10.1016/s0378-4347(99)00161-9
Vuković-Gačić, B., Nikčević, S., Berić-Bjedov, T., Knežević-Vukčević, J., and Simić, D. (2006). Antimutagenic effect of essential oil of sage (Salvia officinalis L.) and its monoterpenes against UV-induced mutations in Escherichia coli and Saccharomyces cerevisiae. Food Chem. Toxicol. 44, 1730–1738. doi: 10.1016/j.fct.2006.05.011
Walker, J. B., Sytsma, K. J., Treutlein, J., and Wink, M. (2004). Salvia (Lamiaceae) is not monophyletic: implications for the systematics, radiation, and ecological specializations of Salvia and tribe Mentheae. Am. J. Bot. 91, 1115–1125. doi: 10.3732/ajb.91.7.1115
Wilhelm, C., and Selmar, D. (2011). Energy dissipation is an essential mechanism to sustain the viability of plants: the physiological limits of improved photosynthesis. J. Plant Physiol. 168, 79–87. doi: 10.1016/j.jplph.2010.07.012
Zgheib, R., Yassine, C., Azzi-Achkhouty, S., and Beyrouthy, M. E. (2019). Investigation of essential oil chemical polymorphism of Salvia fruticosa naturally growing in Lebanon. J. Essent. Oil-Bear. Plants 22, 408–430. doi: 10.1080/0972060X.2019.1623085
Keywords: Salvia L., essential oil, bioactivity, environmental stress, chemodiversity
Citation: Karalija E, Dahija S, Tarkowski P and Ćavar Zeljković S (2022) Influence of Climate-Related Environmental Stresses on Economically Important Essential Oils of Mediterranean Salvia sp. Front. Plant Sci. 13:864807. doi: 10.3389/fpls.2022.864807
Edited by:
Stefan Martens, Fondazione Edmund Mach, ItalyReviewed by:
Adam Matkowski, Wroclaw Medical University, PolandVasil Georgiev Georgiev, Bulgarian Academy of Sciences, Bulgaria
Copyright © 2022 Karalija, Dahija, Tarkowski and Ćavar Zeljković. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Sanja Ćavar Zeljković, c2FuamEuY2F2YXJAdXBvbC5jeg==; emVsamtvdmljQHZ1cnYuY3o=
 Erna Karalija
Erna Karalija Sabina Dahija
Sabina Dahija Petr Tarkowski
Petr Tarkowski Sanja Ćavar Zeljković
Sanja Ćavar Zeljković